Case Report Open Access
GI Mucosal Injury and Bleeding: Association with Sevelamer Crystals
Charumathi Raghu Subramanian1* and Andrew Roorda2
1Internal medicine, Guthrie clinic, Sayre, PA, USA
2El Camino GI Medical Associates, Mountain View, CA 94040, USA
- Corresponding Author:
- Charumathi Raghu Subramanian
Resident, Guthrie Robert packer Hospital
Internal medicine, 18840, USA
Tel: 8326719246
E-mail: mailto:charumathiraghus@gmail.com
Received Date: March 14, 2016; Accepted Date: March 28, 2016; Published Date: April 07, 2016
Citation: Subramanian CR, Roorda A (2016) GI Mucosal Injury and Bleeding: Association with Sevelamer Crystals. J Gastrointest Dig Syst 6:409. doi:10.4172/2161-069X.1000409
Copyright: © 2016 Subramanian CR, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Gastrointestinal & Digestive System
Abstract
Many medications have been listed as causes for GI mucosal injury. Sevelamer, a phosphate binder used in patients with CKD, has been studied recently as a possible cause. The first case report of lower gastrointestinal bleeding due to stercoral ulceration caused by sevelamer was described in 2008. Since then there have been a few case reports and studies regarding the same, although larger studies are required to prove causation. In this paper, we present a patient with lower GI bleed, whose colonoscopy revealed an ulcer, with biopsy report showing sevelamer crystals, and no atypia, granulomas, or evidence of acute inflammation. We discuss various reports of sevelamer and GI injury, its histological appearance and differentiation from other medications also known to cause GI damage.
Keywords
Mucosal ulcer; Sevelamer crystals
Case Presentation and Evolution
A 65 year old female, who lives in a skilled nursing facility, was admitted to the hospital in 2013, with complaints of bright blood per rectum for 2 days, and right sided abdominal pain, dull, not radiating, and not related to food intake. She also had episodes of greenish diarrhea and was hypotensive on the day of admission. No history of fever, dyspnea, nausea or vomiting. Her past medical history included multiple sclerosis, schizophrenia, bipolar disorder, supraventricular tachycardia, end stage renal disease on dialysis, generalized anxiety, cardiac tamponade. Her past surgical history comprises a pericardial window in 2012, percutaneous endoscopic gastrostomy tube placement for refusal to eat, and cholecystectomy. She is currently on klonopin, amiodarone, renvela, zyprexa, copaxone, folate and omeprazole. Family history is noncontributory. She lives in a skilled nursing facility, does not smoke or consume alcohol. On examination, the patient is non verbal, but able to track with her eyes. Abdomen soft, non tender, no organomegaly, bowel sounds heard. Labs showed a hemoglobin of 8 g/dl, which improved to 11 g/dl after 2 units of packed red blood cell transfusion; BUN 32, creatinine 2.1, sodium 140 and potassium 2.8. Coagulation studies normal, EKG sinus rhythm, and CXR showed B/L atelectasis. She was clostridium difficile positive and was treated for the same. Her vitals normalized and she was discharged.
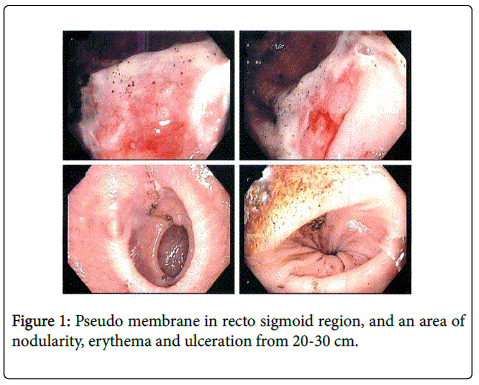
Two weeks later, the patient presented again with bright blood per rectum. This time she underwent an upper endoscopy and colonoscopy. The former showed mild gastritis and mild esophagitis distally, but otherwise normal. Colonoscopy revealed possible pseudomembrane in rectosigmoid region, and an area of nodularity, erythema and ulceration from 20-30 cm, which was biopsied (Figure 1). The differential diagnoses considered were clostridium difficile colitis, inflammatory bowel disease, and less likely malignancy. The pathology report revealed the presence of sevelamer crystals in the area of ulceration. Random biopsies of the colon showed no crypt abscesses, granulomas, atypia, or evidence of acute inflammation. She was discharged after stopping sevelamer, and has not reported GI bleeding since.
Discussion
Sevelamer is a non-calcium containing phosphate binder, which is used to treat hyperphosphatemia in patients with CKD. It is available as sevelamer hydrochloride (Renagel) and sevelamer carbonate (Renvela), which are cationic polymers that bind phosphate through ion exchange. The carbonate form is generally preferred over the hydrochloride preparation because the former does not lower serum bicarbonate [1]. The efficacy and safety of sevelamer in CKD patients, with or without dialysis has been demonstrated by several clinical trials. In an open label randomized control trial involving 46 incident hemodialysis patients, sevelamer treated patients experienced lower all cause mortality [2]. In another open label dosage-titration study, sevelamer was found to be effective and well tolerated by CKD patients, who were not on dialysis [3]. The adverse reactions include metabolic acidosis, nausea, vomiting, diarrhea, constipation, dyspepsia, abdominal pain, hypercalcemia, etc. Post marketing/case reports include fecal impaction, intestinal obstruction, intestinal perforation although rare. Its use is contraindicated in patients with intestinal obstruction. The most common reason for withdrawal so far is gastrointestinal adverse reactions [4-6].
Medications affecting the gastrointestinal tract have been studied widely and the list is ever expanding. Sevelamer as a cause for gastrointestinal mucosal injury has recently been considered. A recent study reported the first histologic description of sevelamer crystals in the GIT [7]. The study group consisted of 7 patients with chronic kidney disease who were on sevelamer, with dose ranging from 800 mg to 3200 mg thrice daily. Most patients presented with gastrointestinal symptoms - nausea and vomiting, dyspepsia, rectal bleeding, and an incarcerated hernia. 15 specimens (14 biopsies and 1 resection specimen) with histologically similar crystals were obtained prospectively over a one year period. Also, sevelamer (800 mg) tablets were crushed in the laboratory and sent for routine H&E staining and special staining similar to patient samples. Sevelamer crystals were identified in all the specimens. Associated mucosal findings included chronic mucosal injury, inflammatory polyps, ischemic injury and ulceration. These crystals and the crushed sevelamer tablets exhibited identical morphological features - non polarisable crystals with broad, curved and irregularly spaced ‘’fish scales’’ imparting a 2 - toned appearance. Also, an altered morphology was seen on those crystals embedded in ulcerated mucosa, ischemic mucosa or necrotic debris. The study could not prove the causative role of sevelamer in mucosal injury, and could not rule out the possibility that sevelamer could have been innocent bystanders while the actual injury was caused by other co morbidities. Nevertheless, some interesting observations were put forth. In cases of acute mucosal injury, the severity appeared to correlate with sevelamer dosage. The key contraindications for sevelamer use are intestinal obstruction followed by swallowing disorders. This, taken together with the known GI side effects of sevelamer, could suggest a possibility of sevelamer induced dysmotility leading to mucosal injury.
The first case report of lower gastrointestinal bleeding due to stercoral ulceration caused by sevelamer was described in 2008. Histological examination of the ulcer showed denuded mucosa with acute and chronic inflammation, though no crystals were reported. Sevelamer was stopped and the patient did not report any further episodes of bleeding [8].
In another case report in Japan, colon perforation was reported in two patients who were taking sevelamer. In one of the cases, a large volume of hard stool was found, thus suggesting dysmotility as a possible cause [9].
Another key consideration while encountering GIT crystals is to differentiate them from those of kayexalate and cholestyramine, which have also been shown to cause GI side effects. Kayexalate is a potassium binding resin whose crystals display a regularly spaced, narrowed rectangular “fish scale” pattern with perpendicular lines of intersection, a violet color on H&E, and magenta color on PAS/D. Cholestyramine is a bile acid resin that appears as uniform bright orange color on H&E, variable gray or hot pink color on PAS/D, and lack of an internal “fish scale” pattern [7]. The possibility of the crystals being of kayexalate arose in our case too until the morphological findings proved otherwise. Hence, knowledge of the difference might also result in reporting of more cases of sevelamer crystals.
Thus sevelamer should be considered in the differential for medication causing GI mucosal injury, though the above association warrants larger studies to prove causation.
References
- Delmez J, Block G, Robertson J, Chasan-Taber S, Blair A, et al. (2007) A Randomized, Double-blind, Crossover Design Study of Sevelamer Hydrochloride and Sevelamer Carbonate in Patients on Hemodialysis. Clinical Nephrology 6: 386-391.
- Di Iorio B, Molony D, Bell C, Cucciniello E, Bellizzi V, et al. (2013) Sevelamer Versus Calcium Carbonate in Incident Hemodialysis Patients: Results of an Open-label 24-month Randomized Clinical Trial. American Journal of Kidney Diseases: The Official Journal of the National Kidney Foundation 4: 771-778.
- Ketteler M, Rix M, Fan S, Pritchard N, Oestergaard O, et al. (2008) Efficacy and Tolerability of Sevelamer Carbonate in Hyperphosphatemic Patients Who Have Chronic Kidney Disease and Are Not on Dialysis. Clinical Journal of the American Society of Nephrology: CJASN 4: 1125-1130.
- (2014) Safety Information - Renagel (sevelamer Hydrochloride) Tablets and Renvela (sevelamer Carbonate) Powder for Oral Suspension and Tablets.
- (2014) Renagel (SevelamerHcl) Drug Information: Side Effects and Drug Interactions - Prescribing Information At. RxList.
- (2014) Renvela (Sevelamer Carbonate) Drug Information: Side Effects and Drug Interactions - Prescribing Information At.RxList.
- Swanson BJ, Limketkai BN, Liu TC, Montgomery E, Nazari K, et al. (2013) Sevelamer Crystals in the Gastrointestinal Tract (GIT): a New Entity Associated with Mucosal Injury.” The American Journal of Surgical Pathology 11: 1686-1693.
- Madan P, Bhayana S, Chandra P, Hughes JI (2008) Lower Gastrointestinal Bleeding: Association with Sevelamer Use.World Journal of Gastroenterology 16: 2615-2616.
- Nishiwada S, Osumi S, Yasuda S (2008) Colon Perforation in two hemodialysis patients on Sevelamer hydrochloride.Journal of Japan Surgical Association 69: 2601-2606.
Relevant Topics
- Constipation
- Digestive Enzymes
- Endoscopy
- Epigastric Pain
- Gall Bladder
- Gastric Cancer
- Gastrointestinal Bleeding
- Gastrointestinal Hormones
- Gastrointestinal Infections
- Gastrointestinal Inflammation
- Gastrointestinal Pathology
- Gastrointestinal Pharmacology
- Gastrointestinal Radiology
- Gastrointestinal Surgery
- Gastrointestinal Tuberculosis
- GIST Sarcoma
- Intestinal Blockage
- Pancreas
- Salivary Glands
- Stomach Bloating
- Stomach Cramps
- Stomach Disorders
- Stomach Ulcer
Recommended Journals
Article Tools
Article Usage
- Total views: 12345
- [From(publication date):
April-2016 - Aug 31, 2025] - Breakdown by view type
- HTML page views : 11331
- PDF downloads : 1014