Research Article Open Access
Methods of Analysis (Extraction, Separation, Identification and Quantification) of Carotenoids from Natural Products
Monica Butnariu*Banat’s University of Agricultural Sciences and Veterinary Medicine “Regele Mihai I al Romanei” Timisoara, Romania.
- *Corresponding Author:
- Butnariu M
Banat’s University of Agricultural Sciences and
Veterinary Medicine King Michael I of Romania from Timisoara
300645, Calea Aradului 119, Timis, Romania
Tel: +40-0-256-277
E-mail: monicabutnariu@yahoo.com
Received Date: May 04, 2016; Accepted Date: June 15, 2016; Published Date: June 21, 2016
Citation: Butnariu M (2016) Methods of Analysis (Extraction, Separation, Identification and Quantification) of Carotenoids from Natural Products. J Ecosys Ecograph 6: 193. doi:10.4172/2157-7625.1000193
Copyright: © 2016 Butnariu M. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Ecosystem & Ecography
Abstract
Developing techniques for isolation and identification of biocompounds, from natural products, resulted in a rapid enrichment of carotenoid pigments number. For their isolation from biometabolites mixtures, the physical and chemical properties are taken into account. Carotenoids are sensitive to light, heat, oxygen, acids and alkaline bases. The exposure to light (direct sunlight/ UltraViolet), causes cis–trans photoisomerization, which may lead to their photodestruction. Biological materials containing carotenoids and their solutions must be protected from the action of light. Many carotenoids are thermolabile (xanthophylls); their heating being indicated only when it is absolutely necessary. The separation of carotenoids is done at room temperature or up to –20°C, in the dark. In the case of hot saponification they should be protected by a low–boiling solvent (30–60°C). Carotenoids may be oxidized in the presence of oxygen or peroxides, because of their sensitivity to oxygen in the adsorbed state (in thin layer or column chromatograms). It is necessary to operate in inert conditions (under nitrogen or vacuum). The oxidation during the extraction and saponification can be minimized if it is carried out in a nitrogen atmosphere. The exposure of carotenoids to acids, leads to changes such as: the oxidative decomposition, cis–trans isomerization and isomerization of 5,6–epoxides and 5,8–epoxides. The inconveniences are minimized by neutralization (calcium carbonate, pyridine, dimetilalanine). It works with purified solvents, freshly distilled, chlorinated derivatives (dichloromethane or solvents containing hydrochloric acid). The storage of carotenoids should be done in the dark, under an atmosphere of nitrogen or in vacuum, at a temperature of –20°C. The best preservation method is in the crystalline state. The current research techniques are using spectral methods, which provide accurate information on the structure and properties of organic biosubstances. Compared to chemical methods of identification, spectral methods have the advantage that it provides data faster, are accurate, require small amounts of material and enable continuous analysis at different stages of processing of the compound extracted without changing the composition of the biosubstance investigated, which enables its recovery. This chapter presents the main methods of extraction, separation and identification of organic compounds with direct applications on carotenoids.
Keywords
Carotenoids; Isolation; Extraction; Separation; Identification; Quantification; Natural products; Biosubstance
Introduction
From chemical point of view carotenoids (fat-soluble plant pigments that provide much of the color in nature) are polyisoprenic compounds, consisting of isoprene units (eight units, forty carbon atoms).
There are about 600 compounds in the carotenoid group that can be classified as carotenes or carotenoids (presenting in the structure only carbon and hydrogen atoms) and xanthophylls, which are oxygenated hydrocarbon compounds having at least one oxygen atom, hydroxy, keto, epoxy, methoxy or carboxyl groups.
The last category forms apocarotenoids, of smaller dimensions, typically formed by cleavage of carotenoids. In green plants, carotenoids are found in chloroplasts, as part of biosynthesis system, but they are more numerous, more visible, and more coloured in roots, fruits and flowers [1].
Carotenoids are yellow, red or orange pigments which are spread in both plant and animal world. They came with green chlorophyll in leaves and herbs and are present in many flowers, fruits, seeds or roots.
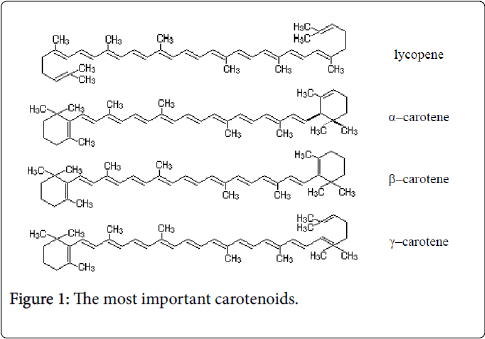
They are synthesized only in plants. Carotenoid pigments are plant carotenoids. They are the most widespread of all vegetal pigments. In the animal kingdom, these pigments originate from foods of plant origin. They can be found in free state or in combination with holoproteides and carbohydrates (carotenoproteides, carotenoid glycosides). Carotenoid pigments are dispersed in all plant organs with or without chlorophyll (leaves, fruits, stems, bulbs, seeds, etc.). Due to the hydrocarbon structure, the carotenoid pigments are hydrophobic substances, soluble only in organic solvents, oils and fats. From chemical point of view, carotenoid pigments are characterized by a structure presenting conjugated double bonds, determining the unsaturated nature and therefore can cause the oxidation and autoxidation reactions (in open air), the absorption capacity of the illuminating radiation, etc. The biochemical role of carotenoids is determined by their chemical structure and properties listed above. Carotenoids, as well as terpenes, sterols, phytol, vitamin K, vitamin E, etc., present as repetitive structural unit an activated isoprene which can form hydrocarbon chains of carotenoid, steroid type [2]. Most of the 80 carotenoids that can be extracted from plants contain 40 carbon atoms. Only a few are smaller molecules, probably being results of the oxidative degradation of carotenoids. The high number of conjugated double bonds gives them a specific colour ranging from yellow to orange and red to purple. The xanthophylls comprises (numerous other biocompounds) lutein, zeaxanthin, neoxanthin, violaxanthin, and α− and β−cryptoxanthin. Their structural characteristic is double conjugated bonds that impact chemical, biochemical, and physical features. Carotenoid pigments play a role in photosynthesis and in protection of self-photo-destruction of chlorophyll molecules and other active substances (cytochromes, peroxidases, catalases, flavonoid pigments, vitamin B12, E, K). Carotenoids prevent damage caused by excessive irradiation activating directly triplet chlorophyll (³Chl) or singlet oxygen (¹O2), compounds formed during photodynamic reactions, through a direct contact mechanism of carotenoids with photosensitive materials (chlorophyll). This photo-protection process can be achieved by carotenoid compounds (primary carotenoids are integrated in photosynthesis systems) that are associated with chlorophylls, since they can fix oxygen forming unstable oxygenated compounds, via oxidoreduction processes' intervention. Carotenoids can also form intermediary metabolites that stimulate or inhibit plant development, important for their activity in photo-tropes and photoaxes. Lutein is one of valuable antioxidants: its antioxidant activity is superior to that of tocopherol, ascorbic acid, β–carotene, lycopene. Lutein (xanthophylls) has same chromophore and same spectrum as its parent carotenoid, α−carotene. Lutein (principal natural stereoisomer is (3R, 3′R, 6′R) −β, ε−carotene−3, 3′−diol) differs of zeaxanthin only in location of one of terminal conjugated double bonds; however, chromatographic separation of these compounds, while difficult, it protects oxidation photosynthesis apparatus. Presence of two hydroxy groups can be confirmed by acetylation; allylic position of one of hydroxyls is verified by its positive response to methylation with acidic methyl alcohol, producing a compound that behaves like a monohydroxy carotenoid on chromatography. Other mechanisms include involvement of also synthesised lutein (by high stability, innocuity, high security) molecules that achieve their photo-protection (reducing photon excess), with strong impacts on curing eye diseases, averting degeneration of eye muscles effectively, improving eyesight, availing to cure heart disease, resisting arteriosclerosis, averting blood vessel blocking up, cutting down occurring of cancer and resisting aging. Excellency of antioxidant impact of lutein in prevention of lipid oxidation is free of nature of pro–oxidant factor. In organic solutions, lutein is a trap for singlet oxygen, an inhibitor of peroxyl radicals that result by lipid oxidation. Lutein has been legitimated as a dietary agent that can delay onset of age-related. Carotenoids containing oxygen in their molecule (xanthophylls) are yellow [3]. The presence of double bonds in the molecules of carotenoids leads to cis–trans isomerism, fact that determines the presence of many geometric isomers. Most carotenoids have a trans configuration. The carbon chain is made up of eight isoprene units giving the carotenoids the solubility in fats (also called lipochrome substances). All carotenoids absorb light in the blue and ultraviolet region of the spectrum. Carotenoids are only slightly soluble hydrophobic molecules, or no soluble in water. They are restricted by hydrophobic areas of the cell, and inside the membranes, except the cases when they are present in combination with proteins in an aqueous medium. Polar functional groups alter the interaction with other molecules. Carotenoids isoprene chain ends at the terminations with two ionone cycles, one of which may have an α–ionone structure, and the other is usually a β–ionone ring. β–ionone ring is required for the carotens acting as provitamin A. α–ionone in diluted solution has a characteristic odor of violets. Carotenoids were identified in a free form in natural products. Carotenoids are divided into: carotenoid hydrocarbons and oxygenated derivatives of hydrocarbon carotenoids. Hydrocarbon carotenoids are carotenoids with 40 carbon atoms, formula C40H56. The most important of these are: lycopene (red carotenoid pigment), α–carotene (alpha-carotene is a precursor to retinoic acid, or a provitamin A compound), β–carotene, γ–carotene (Figure 1).
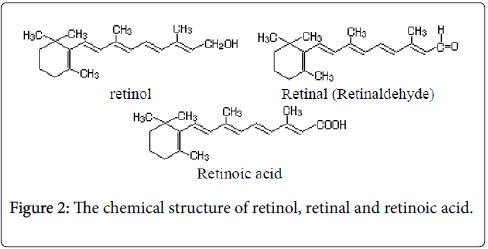
Lycopene (acyclic carotenoid formed from the sequential desaturation) is a crystalline material, red–purple, insoluble in water, soluble in organic solvents [4]. It gives the color of fruits and tomatoes. α–carotene (form of carotene with a β-ionone ring at one end and an α-ionone ring at the opposite end) is a copper–colored crystalline substance soluble in organic solvents. Alpha-carotenes are antioxidant substances that give color and flavor, orange- and red-colored fruits and vegetables. By heating, α–carotene may convert to β–carotene (is composed of two retinyl groups). It is found in plants in smaller quantities than β–carotene. β–carotene (is a red/orange-colored fatsoluble terpenoid with antioxidant properties; it may be enzymatically cleaved to generate vitamin A and retinoic acid (Figure 2)) is in the form of violet crystals, soluble in organic solvents.
Retinaldehyde is the direct precursor to retinoic acid (the form of Vitamin A used directly by the organism). It is widespread throughout the plant kingdom permanently accompanying chlorophyll. Through oxidative enzymatic hydrolysis, β–carotene is converted to two molecules of vitamin A1. β–carotene is the main provitamin A. γ– carotene (contain a β-end group) takes the form of red crystals with blue reflexes, soluble in organic solvents. γ–carotene is less widespread in the plant kingdom (being most commonly found in carrots). Oxygenated derivatives of carotenoids are alcohols, ketones, acids, etc., derived from hydrocarbon carotenoids. Apocarotenoids are carotenoid derivatives formed by the removal of fragments of the carbon backbone from either or both ends of a C40 precursor (as lycopene or beta, beta-carotene). These modifications originate in the oxidative degradation at the level of the terminal rings; they can be the result of nonspecific mechanisms (lipoxygenase, photo-oxidation) as well as of specific mechanisms (dioxygenases). They have significant roles as developmental and environmental response signals. Apocarotenoids make important contributions to flavor and nutritional quality of foods (fruits, tea and wine) and tobacco. Bixin and crocetin are apocarotenoids, which have economic importance as pigments and aroma in foods.
Naturally occurring carotenoids
Hydrocarbons: Hexahydrolycopene, Lycopersene, Phytofluene, Torulene, α–zeacarotene
Alcohols: Alloxanthin, Crustaxanthin, Cryptomonaxanthin, Cynthiaxanthin, Gazaniaxanthin, Loroxanthin, Lutein, Lycoxanthin, OH–chlorobactene, Pectenoxanthin, Rhodopin, Rhodopinol or Warmingol, Saproxanthin and Zeaxanthin
Glycosides: Oscillaxanthin, and Phleixanthophyll
Ethers: Rhodovibrin, and Spheroidene
Epoxides: Citroxanthin, Diadinoxanthin, Foliachrome, Luteoxanthin, Mutatoxanthin, Neochrome, Trollichrome, Vaucheriaxanthin, and Zeaxanthin
Aldehydes: Rhodopinal, Torularhodinaldehyde, and Warmingone
Acids and acid esters: Torularhodin, and Torularhodin methyl ester
Ketones: 2,2'–diketospirilloxanthin, 3'–hydroxyechinenone, 3–OH– canthaxanthin or adonirubin or Phoenicoxanthin, Astacene, Astaxanthin, Canthaxanthin or Aphanicin, Chlorellaxanthin, Capsanthin, Capsorubin, Cryptocapsin, Echinenone, Flexixanthin, Hydroxyspheriodenone, Okenone, Pectenolone, Phoeniconone or Dehydroadonirubin, Phoenicopterone, Rubixanthone, and Siphonaxanthin
Esters of alcohols: Astacein, Fucoxanthin, Isofucoxanthin, Physalien, Siphonein, and Zeaxanthin
Apocarotenoids: Apo–2–lycopenal, Apo–6'–lycopenal 6'–apo–y– caroten–6'–al, Azafrinaldehyde, Bixin, Citranaxanthin, Crocetin, Crocetinsemialdehyde, Crocin, Hopkinsiaxanthin, Methyl apo–6'– lycopenoate, Paracentrone, Sintaxanthin, and β–apo–2'–carotenal
Nor– and seco–carotenoids: β–carotenone, Actinioerythrin, Peridinin, Triphasiaxanthin, Pyrrhoxanthininol, Semi–α–carotenone, and Semi–β–carotenone
Retro–carotenoids and retro–apo–carotenoids: Eschscholtzxanthin, Eschscholtzxanthone, Rhodoxanthin, and Tangeraxanthin
Higher carotenoids: Bacterioruberin, C.p. 450, C.p. 473, Decaprenoxanthin and nonaprenoxanthin [5]
There are known many representatives of these derivatives such as: xanthophylls, carotenoids ketones, carotenoid acids.
Xanthophylls
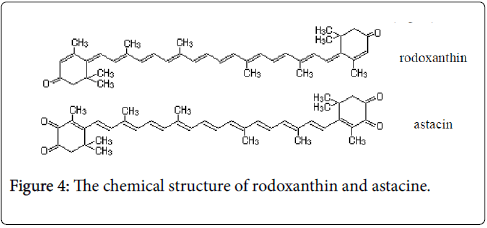
Xanthophylls are hydroxyl derivatives of carotenoid hydrocarbons, which comprise a diverse group of oxygenated carotenoids with varied structures and complexes functions. There are known many xanthophylls, of which the most important are lutein and zeaxanthin (Figure 3).
Lutein is a yellow crystalline substance with purple gloss, accompanying β–carotene and chlorophyll in all green plants. It gives the yellow color of the flowers. It is also found in egg yolk, animal tissues, and particularly in the ovary. Zeaxanthin takes the form of orange crystals. From structural point of view it differs from lutein by the position of a double bond (2 cycles β–ionic) [6]. Lutein is isomeric with zeaxanthin, varying just in the position of one twofold bond. The essential natural stereoisomer characteristic of lutein is (3R, 3′R, 6′R)- beta, epsilon-carotene-3,3′-diol. Lutein is a lipophilic compound and is for the most part insoluble in water. The vicinity of the long chromophore of conjugated twofold bonds (polyene chain) gives the particular light-retaining properties.
The polyene chain is vulnerable to oxidative debasement because is chemically unstable in acids and it depends on environmental conditions (by light or heat). Lutein is available in plants as fatty-acid esters, with one or two fatty acids bound to the two hydroxyl-groups. Hence, saponification (de-esterfication) of lutein esters to yield free lutein may yield lutein in any proportion from 1:1 to 1:2 molar proportion with the saponifying fatty acid. Fucoxanthin with formula C42H58O6 is a carotenoid (xanthophyll), found in brown seaweed that is being investigated for its fat burning abilities. It is found as a biocompounds (accessory pigment) in the chloroplasts of brown algae and most other heterokonts, giving them a brown or else olive-green color. This biocompounds absorbs light primarily in the blue-green to yellow-green part of the visible spectrum, peaking at around 510-525 nm and absorbing significantly in the range of 450 to 540 nm.
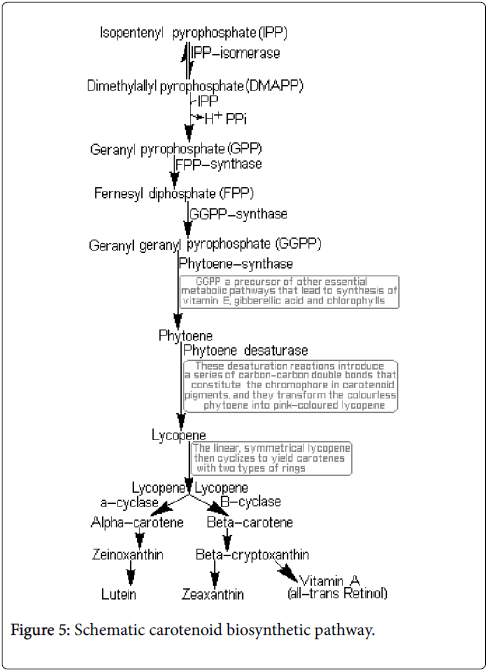
Carotenoid ketones
Among carotenoid ketones are to be mentioned the rodoxanthin and astacin (Figure 4).
Rodoxanthin is a substance colored in red–blue, widespread in aquatic plants and conifers. It is a carotenoid diketone. Astacin is violet colored substance. From the chemical point of view is a carotenoid tetracetone [7].
Carotenoid acids
Among the carotenoid acids, with less than 40 carbon atoms, the most important are Bixin and Crocetin, yellow pigments. Given their ubiquity in the plant kingdom, total annual carotenoid biosynthesis was estimated at 100 million tons. Their structural feature is the system of conjugated double bonds, which influence their chemical, biochemical and physical properties [8].
Carotenoids biosynthetic pathway
It is important to know the way is taking place biosynthesis (also called biogenesis or anabolism) the various types of carotenoids, with different carotenoid backgrounds co-transformed with plasmids containing carotenogenic genes from plants, in order to more easily identify the modalities of extraction, separation, identification and quantification of carotenoids from natural products. Carotenoid pigments are involved in photosynthesis, with a role both in absorbing light energy and in protecting autophotodestruction of chlorophyll molecules and other active biosubstances (cytochromes, peroxidases, catalase, flavonoid pigments, vitamin B12, vitamin E, vitamin K). Carotenoids may prevent excessive radiation damage by activating directly the chlorophyll triplet (³Chl) or singlet oxygen (¹O2), compounds formed in photodynamic reactions.
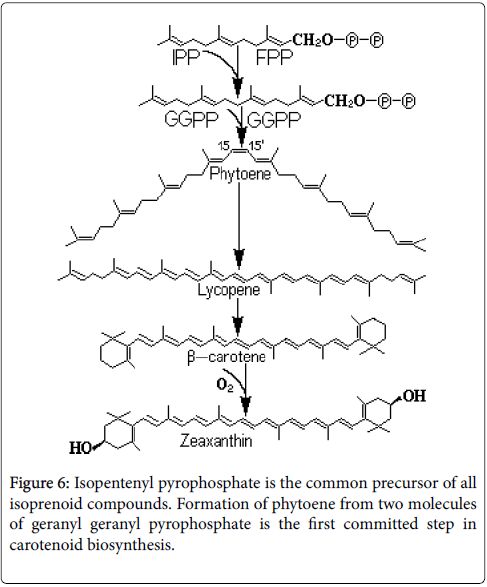
This mechanism requires direct contact of carotenoids with photosensitive biosubstances (chlorophyll). This kind of protection can be achieved by carotenoid compounds (primary carotenoids are embedded in the photosynthetic systems), which are associated with chlorophylls. As they can fix oxygen forming oxygenated compounds less stable, carotenoid pigments are involved in redox processes. They can form intermediate metabolites that stimulate or inhibit the plant growth [9,10]. They are important for the action that they may have on phototropism (use light for ATP synthesis) and phototaxis (positive or negative movement along a light gradient or vector) [11]. Combination of carotenoid pigments from chloroplast (carotene in reduced state and xanthophyll in oxidized state) and chromoplasts (α–carotene, β– carotene, γ–carotene δ–carotene, lycopene, cryptoxanthin, xanthophyll, lutein, capsanthin, canthaxanthin, violaxanthin and anteraxanthin) gives the colors of the petals of flowers, fruit, leaves and roots. Carotenoids are biosynthesized in the chloroplasts (Figure 5) from the intermediate products originating from the degradation of sugars, namely acetyl coenzyme A.
The enzymes that catalyze the biosynthesis are encoded by genes, transported in chloroplasts and chromoplasts after transcription and translation. Most chloroplast carotenoid pigments accumulate in complex with lipoproteins. These complexes contain galactolipids, phospholipids and one protein called fibrillin. The genes that modify the plastidial enzymes: geranyl–geranyl pyrophosphate synthase (GGPP), phytoene synthase (Psy), phytoene desaturase (Pds), and capsanthin-capsorubin synthase (Ccs), determine the coloring during maturation. The expression of these genes can be triggered by mere damage of tissue. The expression of the genes encoding the farnesyl pyrophosphate (FPP) synthase synthesis in the cytoplasm is triggered only by elicitors. It results that izoprenoids biosynthesis in plastids and cytoplasm is partly controlled by signals from the environment [10].
In higher plants, there are two separate pathways, biochemical different for the biosynthesis of isopentenyl–pyrophosphate (IPP). Isopentyl–pyrophosphate is synthesized in the cytosol by acetate / mevalonate pathway and in the chloroplasts by 1–deoxy–D–xilulosa– 5–phosphate (Figure 6). Carotenoid synthesis main constituent is isopentenyl–pyrophosphate and its allyl isomer dimethylallyl– pyrophosphate. The isomerization reaction is catalyzed by the enzyme isopentenyl pyrophosphate isomerase [12]. Genes for the enzymes involved in the biosynthesis of carotenoids in the plastids are located in the nucleus and the products of these genes are transported to the chloroplast. The next step in the biosynthetic chain of carotenoid is the desaturation of phytoene. Two enzymes, functionally and structurally similar, phytoene desaturase and zeta ( ζ) -carotene desaturase convert the phytoene in lycopene by four desaturation reactions.
The first two stages of desaturation are catalyzed by phytoen– desaturase and are completed with ζ–carotene formation; the other two desaturation reactions are catalyzed by ζ–carotene desaturase in the result of which is formed lycopene. Lycopene cyclization occurs towards α–carotene and β–carotene. In the formation of β–carotene is involved the enzyme lycopene–β–cyclase. Zeaxanthin specific biosynthesis steps are catalyzed by β–carotene and carotene– hydroxylase ketolase. By the β–carotene–ketolase are introduced two cetogroups in the positions 4 and 4' of the β–carotene to form cantaxanthine. Two hydroxyl groups are introduced at the 3 and 3' of canthaxanthin to form lutein and zeaxanthin. Zeaxanthin is one of the most valuable antioxidants, its antioxidant activity being higher than that of tocopherol, ascorbic acid, β–carotene, lycopene. Zeaxanthin is localized in lipid globules in the cytoplasm, absorbs light in the blue region and protects the photosynthetic system by oxidation. Nonpolar carotenoids in plasma (β–carotene, α–carotene, lycopene) are transported by low density and very low density lipoproteins and polar carotenoids by high density lipoproteins. The mobility of zeaxanthin ensures its ability of penetration in various structures of the body and mediates the generalized positive effect on many structures and functions [13]. A significant cycle is the violaxanthin-anteraxanthinzeaxanthin cycle adjusts the interconversion of epoxidated xanthophyll (violaxanthin) to de-epoxidated forms anteraxanthin and zeaxanthin. The mechanism of antioxidant activity of carotenoids is based on the absorption of reactive oxygen species during oxidative chain. Carotenoids inhibit peroxyl radicals in microsome systems and in unilamellar vesicles. They inactivate singlet oxygen and inhibit the photooxidation of unilamellar liposomes. The antioxidant property is superior to other antioxidants used to prevent the oxidation of polyene fatty acid with peroxides formation and degradation of cell membranes. The superiority of the antioxidant activity in prevention of lipid oxidation occurs irrespective of the nature of the prooxidant factor. In organic solutions the cartenoids are a trap for singlet oxygen and peroxyl radical inhibitor, arising from lipid peroxidation [14].
Extraction Methods
Lately carotenoids have represented another possibility for the food and pharmaceutical industry. The practice of post-harvest residues is a good option for obtaining biosubstances (analytes) of interest. The implementation of enzymatic pretreatment over plant matrices has resulted in an increase in the quantity of extraction and lowered operating costs. At the moment, the use of organic solvents in this process is very often, regardless of how, new assay with novel environmentally friendly solvents (non-toxic) assurance new cleaner technologies. A simple, reliable, and gentle saponification method for the quantitative determination of carotenoids in plant material was developed. The method involves an extraction procedure with acetone and the selective removal of the chlorophylls and esterified fatty acids from the organic phase using a strongly basic resin. Extracts from common plant material were analyzed by HPLC for their content of major carotenoids before and after action of basic resin. Regardless of the extraction method, for the extraction of carotenoids from the samples and for their solubilization are used organic polar solvents: acetone, methylic alcohol, ethylic alcohol, dimethylsulphoxide, dichloromethane, and mixtures of solvents (ethylic alcohol / diethyl ether, hexane / acetone / ethylic alcohol, acetone / methylic alcohol, dichloromethane / methylic alcohol). Based on the toxicity of most solvents, studies were conducted to remove the organic solvent used for extraction of antioxidants from the vegetal material [15]. These include saponification, selective solvent extraction, transesterification followed by molecular distillation and further purification by adsorption using synthetic resins, silica gel and reverse phase C18 silica, adsorption chromatography and membrane methods. Even though there are different methods, but there is one same feature which is the use of solvent. Solvent plays an important role in most of the methods. It can be used either as a pre-extraction solvent, main solvent or co-solvent. The problem of most solvents which are used nowadays is that they possess potential fire health and environmental hazards.
General extraction methods
Before making a carotenoids extract you first need to consider the chemical and physical properties of the substance you want to extract from the vegetal material. Most extractions are based on solubility of the chemicals of interest. The distribution to a variety of materials and fluids of the biologically active compounds made it impossible to adopt a generally–available extraction method that can be applied universally as a standard technique. This is due to the various structures of biologically active compounds. Even those belonging to the same class differ by the chain length, by the specific functional groups or configuration, by different solubility in solvents etc.
Carotenoids are soluble in apolar solvents, including edible fats and oils. Basically carotenoids are lip soluble; they are usually extracted from the plant sources with organic solvents (chloroform, hexane, acetone, petroleum ether, etc.). The plant material can contain the large quantity of water; water-miscible organic solvents such as ethylic alcohol are also used. One of the inconveniences is the elimination of the residual solvents to obtain a safe extract; this can be avoided by using food grade solvents such as ethylic alcohol, found that the yield of each carotene from plant material was perceptibly higher with extraction performed with ethylic alcohol than with that using ethyl acetate. The method of extraction is selected according to the nature of the plant material, the ease of extraction solvent, the amount and properties of extracted compounds (solubility termolability), etc.
Taking into consideration the nature of the biosubstance to be extracted, liquid or solid, a special type of extraction is applied. This may be solid–liquid extraction or liquid–liquid extraction, i.e., extraction of the active principles which are liquid or embedded a liquid, with another immiscible liquid [16].
Fresh or dried plant product is sprayed or fine crushed to achieve the suitable structure needed for extraction. The choice of solvent is determined by the nature of the active ingredients, the physico– chemical properties of the solvent, the selectivity of the solvent. Various methods have been developed in order to recover carotenoids from being destroyed by the process of separate.
These include saponification, selective solvent extraction, transesterification followed by molecular distillation and further purification by adsorption using synthetic resins, silica gel and reverse phase C18 silica, adsorption chromatography and membrane method. Although the though there are diverse methods, all they have in common the use of solvents. Solvent has a significant role in most of the methods. It can be used either as a pre-extraction solvent, main solvent or co-solvent. The impediment to of most solvents which are used is that they have potential fire health and environmental hazards [17]. Thermolabile biosubstances are cold extracted by leaching or agitation. For the hot extraction are used several methods, i.e., the reflux extraction, solid–liquid or liquid–liquid continue extraction. The best results are obtained by applying the countercurrent extraction. An effective extraction method is the ultrasonication of the material extracted with solvents.
Methods of extraction of carotenoids
Extraction of carotenoids from the vegetable product is made non– polar organic solvent, typically diethyl ether or chloroform, by maceration, mechanical stirring or refluxing in boiling water bath.
One study a specialty literature was done and has been presented to the methods currently used for the extraction of carotenoids: lowpressure solvent extraction by different techniques and supercritical fluid extraction. The process parameters and their influence on carotenoids yield was done considered. It requires a high degree of grinding as nonpolar solvent does not penetrate the cell membrane to dissolve carotenoids [18]. Extracted plant material can be fresh or dried. If the extracted material is fresh, this implies the existence of water in the tissues and it is necessary to remove it by drying prior to the extraction with water–miscible solvents (acetone, methylic alcohol, and ethylic alcohol).
The plant material must be sufficiently dehydrated to allow further extractions with non–aqueous solvents. In the case of dry material, the extraction is carried out with water–immiscible solvents [19].
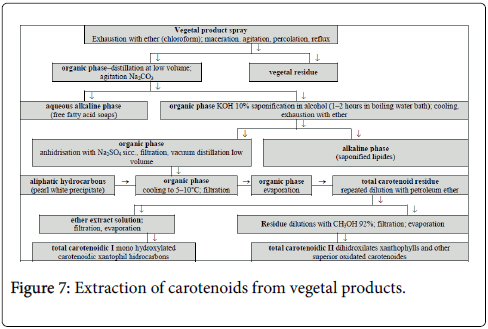
Figure 7 presents a general method for the extraction of carotenoids. The organic extract solution contains the carotenoid compounds, along with a number of ballast substances such as chlorophyll.
Extraction from the tissues of higher plants
The general scheme for the extraction of carotenoids from vegetal products is shown in Figure 7. For the higher plants tissues are known a variety of extraction methods and are described only the ones which are simplified versions of the general method of extraction [18].
Method I
The plant material is finely ground and stirred with acetone for 1–2 minutes in a nitrogen atmosphere. The homogenate is filtered, and the residue is extracted further with acetone until it becomes colorless. Acetone extracts are combined and concentrated to a small volume by rotary evaporation. Over the acetone extract is added an equal volume of freshly distilled diethyl ether, homogenizing the mixture and then washing it with a solution of sodium chloride (for diethyl ether to remove easily and to avoid emulsification of the mixture) to complete the removal of the acetone.
This method is proper when using green tissue of plants, flowers and fruits for extraction.
Method II
This method is useful when working with small amounts of substance. The tissue is ground and triturated with anhydrous sodium sulphate, to give a fine powder which is then extracted with acetone followed by diethyl ether. The extract is centrifuged and the precipitate obtained will be further extracted until it becomes a colorless solution. The supernatants are mixed and concentrated, and then are transferred in an equal volume of diethyl ether as in the case of Method I.
For both methods the saponification is necessary, which allows removal of ballast substances, especially chlorophyll, and xanthophyll esters hydrolysis [19,20]. The saponification is carried out by addition of potassium hydroxide 15% over the extract (1:10, v/v). This saponification can be done in two ways:
Alkaline mixture is heated on a water bath for 10–15 minutes, in a nitrogen atmosphere, in dark; Keeping at room temperature or 5°C, in dark for 10–15 hours.
It is preferred the method of saponification by cooling due to the fact that some carotenoid pigments (such as xanthophylls) are thermally labile, degradation being possible [21]. Types of mixtures solvent tested in order to standardize: acetone, acetone: water (9:1 v/v); acetone: water (8:2, v/v); dimethylformamide (DMF); dimethylsulphoxide (DMSO); ethyl ether; hexane; methyl alcohol and toluene, they were used for identification purposes for the most efficient solvent in the extraction procedure.
Extraction from algae
In the case of extraction of carotenoids from algae it does not apply the general method, due to the fact that, for efficient extraction it is required a mechanical disruption or pre-treatment. Extraction of carotenoids from algae is carried out with more polar solvents than acetone (ethylic alcohol or acetone: ethylic alcohol), because they have high polarity and allow the saponification on the initial extract.
The pre–treatment may consist of a suspension of algae in distilled water and cooling for 5–8 hours before the actual extraction, and the extraction with methyl alcohol. Another pre–treatment consists in the preliminary bleaching treatment with water vapor or hot water for 2–3 minutes and rapidly cooling with ice. This latter method enables extraction of carotenoids, but it has a drawback, namely the possibility of destruction of enzymes that act on carotenoids and chlorophylls. For efficient isolation of carotenoids from algae, they can be subjected to drying, although, in this process, some of the compounds may suffer modifications, and it may be deceased the yield of the extraction [22].
A process that involves drying the algae prior to the extraction of carotenoids was used for Fucus serratus . The algae were dried, grounded, powdered and then mixed with water (800 mL water to 500 g of powder) the mixture was eluted through a column by leaching with a mixture of benzene: methyl alcohol (1:10, v/v). The green eluates were discarded and the crude material was eluted with solvent, concentrated, and calcium carbonate column chromatographed.
Methods of Separation
Methods of separation are selected according to the physical and chemical properties of the biosubstances to be separated. Among the commonly used methods of separation in the separation of mixtures of biosubstances, may be mentioned:
Distillation (is a process of separating the component substances from a liquid mixture by selective evaporation and condensation);
Crystallization (is the process of formation of solid crystals precipitating from a solution, melt or more rarely deposited directly from a gas);
Dialysis (is the process of separating molecules in solution by the difference in their rates of diffusion through a semipermeable membrane, such as dialysis tubing);
Electrophoresis (is a method for separation and analysis of macromolecules and their fragments, based on their size and charge/ separates organic molecules based on their different interaction with a gel under an electric potential i.e., different travel);
Column chromatography and thin layer (is a physical method of separation that distributes components to separate between two phases, one stationary (stationary phase), the other (the mobile phase) moving in a definite direction; subtle differences in a compound's partition coefficient result in differential retention on the stationary phase and thus changing the separation), etc.
Overview on general methods of separation
Distillation
Distillation is the separation operation by boiling a mixture on the basis of the difference of volatility of the biocomponents. Distillation has not much to do with solubility but rather vaporization points.
One other method of extracting carotenoids from plants is by distillation. It allows a volatile substance to be separated from a nonvolatile substance. Distillation is probably most widely used for producing alcoholic beverages after fermentation of plant sugars by yeasts. Ethylic alcohol boils at 78°C while water boils at 100°C. Because of its lower boiling point, ethylic alcohol will begin to boil and evaporate (turn to gas) when heated before the water does. Using a proper apparatus to collect and condense the steam, ethylic alcohol will be separated from the water. This process is of course not complete because much of the water will evaporate at the same time. Plant carotenoids can be separated by distillation in the same manner. Water and plants are put together into a flask or pot and heated until the temperature reaches a point where steam is formed. The steam as well as anything with a boiling point near that of water will evaporate (turn into gas) to enter the column part of the still and then the condenser to return to a liquid form. Plant carotenoids that enter the distillate portion include a complex mixture of essential oil, water, and organic plant acids. If enough volume is distilled, eventually the distillate will separate out into oil soluble and water soluble components with the essential oil floating on top of the water soluble components. The watery portion of the distillate is considered waste. Because of this difference in volatility it is provided a different composition of the liquid phase against the vapor in equilibrium with this, thereby allowing separation of bioelements.
Simple distillation allows incomplete separation, between the volatile components and the less volatile, by a sequence of two steps: evaporation and condensation. If this sequence of steps is repeated many, on the same device can be achieved a high number of liquid– vapor equilibrium, the operation is called correction. There are many variations of distillation:
• Distillation at atmospheric pressure
• Steam distillation
• Reduced pressure (applies when atmospheric distillation lead to degradation biosubstances)
• Distillation at high vacuum (used to distilling high–boiling compounds that are very sensitive to heating) [23].
When using the purification by column chromatography, carotenoid extract must be concentrated to be added to the column once. In this case, for the concentration of the mixture of carotenoids at a smaller volume is used the distillation.
Dialysis
It is the process of separation and purification based on the difference in diffusion of the biocomponents through the semipermeable membrane. The membranes act as "molecular sieves", having pores through which may pass certain molecular species while others, with larger molecules are retained. "The theory of phase distribution" states, however, that those bioelements who pass through the membrane "dissolve" in its material, and cross by diffusion and are eluted on the other side. Irrespective of the mechanism of the process, the driving force of dialysis is the difference in concentration between the two sides of the membrane. According to Fick law, the amount of substance diffused, m, is proportional to the time τ, the active surface of the membrane, A, and the concentration gradient between the two sides of the membrane, (C1–C2) eq. (1):

Where k is a constant dependent on the nature of the membrane and of the solution in question. Carotenoids from plants oil were recovered through a process involving neutralization and transesterification of plants oil followed by molecular distillation of the esters. Molecular distillation process a carotenoids represents a special type of vaporization at low pressures and low temperatures, finding great usefulness in the separation and purification of materials with molecules of high molecular weight, as well as for those thermally sensitive (as the carotenoids), minimizing losses by thermal decomposition. These particularities show the high potential of this process in the separation, purification and/or concentration of carotenoids, usually constituted by complex and thermally sensitive molecules [20].
This separation process cannot be applied to carotenoid mixture, due to the fact that these pigments have low molecular weight and values relatively close to each other.
Electrophoresis
It is the method for separating compounds dissolved or suspended in the liquid, on the basis of different migration rates in an electric field. Some macromolecular compounds (polysaccharides, proteins) are able to fix ions to the surface of their molecules. The number of positive or negative ions depends on the nature of the macromolecule, pH and ionic strength of the environment. Sometimes, the number of ions of one kind detained on the macromolecules differs from the number of ions of opposite sign. So the macromolecule has a net, positive or negative charge. The principle of separation by capillary electrophoresis (CE) is based on differences in the mobilities of the analytes under the electrical field which is created by the application of high voltage between the electrodes replaced in a buffer solution. Carotenoids are highly hydrophobic, neutral, and huge molecules, and their effective separation is difficult because of their similar properties. Therefore, capillary zone electrophoresis is not the most convenient CE method for this type of analytes. Recently, neutral analytes were separated by interacting with ionic surfactant monomers added to the buffer in the presence of high concentration of organic solvents. This special CE method is currently called as capillary electrokinetic chromatography. Nonaqueous systems are especially preferred for the analysis of highly hydrophobic compounds. Organic solvents create highly interesting alternatives for the separation mechanisms (some interactions which cannot take place or are too weak to be measured in aqueous media, such as solvophobic, electrostatic, donor–acceptor, and analyte–additive interactions etc.) [21].
There were studies that investigated CE for the separation of the carotenoids (chlorophyll derivatives as well as photosynthetic pigments) together. Numerous CE assays, such as micellar electrokinetic chromatography, capillary electrokinetic chromatography, and nonaqueous capillary electrophoresis were welltried, with coated and uncoated capillary columns to evaluate optimal separation conditions using diode array detection. The effect of distinct type and composition of organic solvents and/ or surfactants on the separation that analysed; some carotenoids (beta-carotene and lycopene) were analysed by CE with a methacrylate ester-based monolithic column. The effects of the porogenic solvent ratio, and the hydrophobicity of bulk monomer employed were examined on carotenoids separations [22]. Fixing of the positive or negative ions was observed for some larger particles such as cells of bacteria, viruses, etc. The momentary electric charge of a particle is an integer, but may be varied in both directions; because, on the one hand, fixing ions is a reversible process, and on the other, is intervening fixing or separation of ions with opposite sign. Such a micro particle, placed in a saline medium, disturbs the normal distribution of the anions and cations around them favorable for the formation of an "ion cloud" in which are predominant ions of opposite sign [24]. If in a medium containing such particles are inserted two electrodes between which an electric field is created, the micro particles will be attracted to one pole (the one with opposite sign their overall charge) and "ionic clouds" towards the pole of opposite sign. Depending on the difference between the two attraction forces, the micro particles will move to the corresponding terminal at a rate dependent on the charge of the particle size and viscosity [25]. Carotenoids cannot be separated by electrophoresis (these pigments have no charge and do not migrate in an electric field).
Chromatography
It is a physical separation method based on the distribution of the biocomponents in a mixture, it has two stages: a fixed one, called stationary phase, and the mobile phase, which passes through the stationary phase. The stationary phase can be a solid adsorbent (adsorption chromatography), a liquid deposited on the surface of a solid base (partition chromatography), ion exchanger (ion exchange chromatography) or gel (steric exclusion chromatography) [26]. Chromatography (techniques designed for the separation or analysis of complex mixtures) is applied for the purpose of isolation of biocomponents by their separation at the elution of the column (column chromatography), by scraping the spots of the chromatoplates (thin layer chromatography), or by cutting the spots on the paper (paper chromatography). Chromatography term which means "color writing" (chrom = color in Greek) was given by TVET (1906), when separating of dyes from green leaves (chlorophyll, carotenoids), by liquid–solid adsorption.
The principle of TVET consist of a balance in the process of adsorption–desorption kinetics between fixed solid phase and mobile liquid phase. The stationary phase is formed by the adsorbent material, granular, placed in a column and the mobile phase is a solution of the mixture to be separated in a suitable solvent. The solution is transported thorough the column by gravity or by a pressure difference. For the spacing of the areas in which are adsorbed various biocomponents of the mixture, a solvent is added at the top of the column. The bioelements migrate at different rates because they are retained differently by the stationary phase. The process is called the chromatogram development.
Complete separation of the biocomponents in the column, named elution of the chromatogram is carried out by adding different solvents and collecting the in different vessels. The bioelements come out of the column in the reverse order of their adsorbability in stationary phase. Among the chromatographic methods, gas chromatography and gas– liquid chromatography are widespread because they can be applied to any mixture of liquid bioelements with low temperature boiling [27].
For low volatility organic compounds, it is used thin layer chromatography and paper chromatography. In the paper chromatography, the stationary phase consists of a sheet of filter paper, on which is applied to one end a drop of the mixture to be analyzed. The paper is then introduced drop wise into an eluent in a covered container. The eluent rises by capillarity and gets out the components in order of increasing Rf value, which is the ratio of the velocity of component migration and migration velocity of the eluent (in cm) eq. (2):
Rf= Vc/Ve (2)
Basically it is measured the migration distance of the solvent and of the component. Purity determination of carotenoids extract was performed according to the eq. (3):
% Purity= Standard peak area ⋅ 100/ Total area (3)
ß-carotene extract concentration was corrected according to the eq. (4):
C (μg/g) = C (μg/g) ⋅ % Purity/ 100 (4)
Identification of the colorless bioelements is performed using a specific reagent that gives colored biocompounds. When it is desired to separate a mixture of bioelements, two–dimensional chromatography is achieved when, after first developing, the paper is dried, rotated to 90° and introduced in a new eluent. In thin layer chromatography, the stationary phase is a layer of adsorbent (silica gel or alumina) with a thickness of part of a millimeter deposited on a glass plate [28].
The principle of separation is the same as paper chromatography, but has the advantage of a faster and more precise separation.
Separation methods for carotenoids
Separation of carotenoids from a mix is dependent of their nature, especially of the polarity of the molecule and the functional groups of the molecule, which determine their hydrophilic character.
Because of these properties there are two separation methods for carotenoids:
• Repartition between 2 liquid phases immiscible;
• Separation by adsorbtion of adsorbant [29].
• The general scheme of separation implies the next stages:
• The organic phase, obtained after the saponification of the extract (according to the paragraph Extraction methods for carotenoids). The extract is anhydrified with sodium sulphate sicc., is repartitioned between petroleum ether–methylic alcohol 92% for the separation of the carotenoid hydrocarbons and the monohydroxilated xanthophylls which go into epiphase (ether), and the dihydroxilated xanthophylls and other superior oxidized carotenoids go into hypophase (alcohol);
• Each phase is subject to column chromatography with aluminium oxide (aluminium oxide standardized–Brockmann II–III) or other adsorbant, using as eluants the ether, ethylic alcohol: ether (1:9, v/v), absolute ethylic alcohol, acetone. There are retained fractions of 50–100 mL, for which the purity is determined by thin layer chromatography, using as stationary phase the silicagel, and mobile phase a solvent system.
Separation by liquid–liquid repartition
A system of repartition is comprised of petroleum ether and methyl alcohol. By this method can be separated the hydrocarbon carotenoids by xanthophylls. The separation is used as a preliminary phase for column chromatography or TLC (TLC is a much faster procedure than column chromatography ).
The separation by repartition between 2 liquids can be an alternative for saponification, for the purpose of removing the chlorophyll from the extracts that cannot be saponified by the addition of bases (some carotenoids are sensitive to these substances) [30]. This process implies the passing of xanthophyll’s in the methyl alcoholic hypophase, the chlorophylls passing in the epiphase. Another variant is the extraction of a methyl alcoholic solution of a pigment with petroleum ether, until the chlorophylls are removed. In both cases, the xanthophylls from the hypophase are passed in the ethylic ether and then washed with water. This process is not so much used because of the sensitivity of the chromatographic methods, by repartition the separation yield being quite low. The carotenoids, because of their different solubility, are able to exist in both repartition fractions. In the case of a carotenoid that is soluble only in one of the solvents (glycoside or acid), this process can be applied, although, the chromatographic methods are preferred.
Separation by the counter–current distribution method
The process comprises of repeated repartition between two solvents, reaching the method called “counter–current distribution”. After a high enough number of repartitions, the original mix is separated in fractions, each having certain solubility: carotenoid hydrocarbons, mono– and dihydroxilated xanthophylls, etc. This process is followed by a column chromatography fractionation, by different adsorbtion due to the specific groups of each carotenoid group. This process is useful in the case of separation of polar xanthophylls which are strongly adsorbed [31]. For the separation of carotenoids are used the chromatographic methods, both on column and TLC.
Thin layer chromatography
TLC is a technique used in the separation and purification of carotenoids. The degree of purification by this method is higher, compared to column chromatography. Depending on the nature of the carotenoid the stationary phase is chosen, also the mobile phase and the plate elution system. In TLC for carotenoids there are some precautions that must be taken:
• Fast application of the sample
• Application of the sample in low light
• Immediate developing of the chromatogram
• Developing the chromatogram in the dark
• Using a nitrogen atmosphere
The biological materials that contain carotenoids or their solutions must be protected from the action of light; because by exposing carotenoids to light, especially direct solar light or UV light, the cis– trans photoisomerization is done, that can lead to their photodestruction. Many carotenoids are thermolabile, especially xanthophylls, their heating up being done only in the situation when it is absolutely necessary. Separation of carotenoids must be done at room temperature, up to –20°C and in dark [32]. In the case of warm saponification, the solutions must be protected by using solvents with boiling points not very high (30–60°C). In the presence of oxygen (air) or peroxides, many carotenoids can be oxidized, being sensitive to oxygen in adsorbed state (in chromatograms, on column and thin layer) [33].
Choosing the Stationary Phase
There can be used as stationary phase a lot of adsorbants, as can be seen from the Table 1.
| S No. | Adsorbant |
|---|---|
| 1 | Al2O3 |
| 2 | Cellulose |
| 3 | CaCO3:MgO:Ca(OH)2 (30:6:5, m/m/m/) |
| 4 | Ca(OH)210. MgO:silicagel (1:1, m/m) |
| 5 | Ca(OH)2:silicagel (1:1, m/m) |
| 6 | Kieselguhr |
| 7 | Manitol |
| 8 | MgO |
| 9 | MgO:celite (1:2, m/m) |
| 10 | MgO:silicagel (1:1, m/m) |
| 11 | Silicagel |
| 12 | Saccharose |
| 13 | ZnCO3 |
Table 1: Adsorbants used for carotenoid TLC.
The most used systems in TLC consist of separating carotenoids on layer with silicagel and developing with petroleum ether (5% ethylic ether) or on magnesium oxide and developing with benzene: petroleum ether (9:1, v/v). The cromatoplates that have as stationary system zinc carbonate , developed with hexane–alcohol, give very good resolutions, for xanthophylls, while calcium carbonate, magnesium oxide and calcium hydroxide are useful both in separating carotenoid hydrocarbons and xanthophylls.
Choosing the solvent
The solvents used in the separation of carotenoids by TLC are weak eluents, like ethylic ether, benzene, as well as a mix of solvents, acetone–ethylic ether, ethylic alcohol–ethylic ether, ethyl acetate– benzene, etc. Solvents that give a high resolution, because of their properties, are tertiary alcohols. Chloroform is generally omitted because of traces of chlorhidric acid contained and that may lead to carotenoid transformations. Acetic acid is used only in the case of carotenoids strongly adsorbed by the adsorbent.
Detection of carotenoids
Due to the fact that these pigments are coloured, there can be done also the visual identification (it can be visually detected until 0.05 μg ß–caroten). Carotenoids are rapidly getting discoloured on the chromatographic plate, for this reason being necessary to spray the plate with a solution of paraffin in petroleum ether. Some carbonylic carotenoids or products of oxidative decomposition can be identified also with a solution of rodamine 1% in acetone or by spraying with a solution of SbCl3 in chloroform. The most sensitive method is the identification with iodine vapours, when brown spots are formed, by this method being able to identify the eventual impurities [34]. The colourless biocompounds can be detected by visualizing the green fluorescence characteristic to absorption in UV.
Elution of carotenoids from chromatoplates
The elution is done depending on the adsorbing layer and the polarity of carotenoids. For silicagel G chromatography, the elution is done with ethylic ether or ethylic ether + 5–10% ethylic alcohol (polar carotenoids), and for the magnesium oxide chromatography, the elution is done with acetone (carotenoid hydrocarbons and xanthophylls). The elution is done after the removal of the coloured area from the support, followed by filtration [35].
Column chromatography
The most important method for carotenoid separation, column chromatography, was recently replaced with TLC by its adsorption characteristics. This method is used by its improvement along with the discovery of high performance liquid chromatography (HPLC), method that presents the advantages of using low quantities of substance and high separation speeds, but limited to a certain scale, not being able to be used in preparative purposes. According to the nature of the carotenoid, the adsorbent will be chosen and the adequate developing solvent. Of the adsorbents used in column chromatography for the separation of carotenoids, one can add cellulose, calcium hydroxide (separation of carotenoid hydrocarbons), saccharose, starch, silicagel, aluminium oxide (separation of monohydroxicarotenoids), calcium carbonate and magnesium oxide (separation of intermediate polarity carotenoids, such as xanthophylls). On the same column there can be used different adsorbents placed according to the adsorption power, the most powerful being placed in the superior side of the column. The solvent used in column chromatography for carotenoids are weak eluents.
Usually it is worked with a mix of solvents, such as acetone–ethylic ether, ethylic alcohol–ethylic ether, ethyl acetate–benzene, etc. The adsorption column must be loaded with adsorbent in proportion of 3, a part being left for the solvent and the extract. The adsorbent must be very well compacted in the column, for this reason the column fill being done with small portions of adsorbent that must be pressed after [36].
An example of high-performance liquid chromatography analyses of concentrated methanolic extracts could be conducted using an HPLC (with an 18 column). Carotenoids could be detected at 470 nm by using an online photodiode array detector with a 600 nm reference wavelength.
Peak areas generated by an HPLC auto-integrator could be used for carotenoid quantification as all detected carotenoids shared similar absorption maxima and their absorption coefficients were unknown. The carotenoid extract must be very concentrated for being able to be added to the column once. After the pigment mix is introduced in the column, the developing is started with different solvents, starting with the most nonpolar. In column chromatography there can be distinguished three base processes for carotenoid separation, as follows:
• zone chromatography
• stage elution chromatography
• gradient elution chromatography
Zone chromatography: The carotenoid mix that is in a nonpolar solvent is applied on the upper side of the column. For the separation of zones on the column there is used an adequate solvent for developing. The zones are pulled from the column, each zone being then eluted individually with a polar solvent.
Stage elution chromatography: In this method it is applied the general principle of column chromatography, the introduction of carotenoid mix in the superior side of the column, the applying the solvents, according to the polarity. The pigments are eluted in the order of growth of their asdorbtion affinity and collected. Each eluted zone is then evaporated, redissolvedi a small volume of solvent, in view of spectrophotometrication [37].
Identification Methods
Identification of any biosubstance can be done by physical methods (spectral) and/or chemical.
Below it will be presented general methods of identification of carotenoids.
Physical methods
By development of theoretical and experimental physics, with its different branches, like optics, atomic physics and nuclear, magnetism etc., were optimized methods for the precise determination of some characteristic physical properties of carotenoids (optical spectra, NMR spectra, electric spin resonance spectra etc.).
The physical methods present undeniable advantages, comparative with chemical methods, like:
• Wide applicability
• Sample integrity conservation
• Fast determinations
• Perfect result reproducibility
• High sensitivity
Another important aspect is the possibility of obtaining a big variety of data and information for the carotenoid structure. In Table 2 are presented the physical methods used in establishing the chemical structure of carotenoids.
| Researched state | Methods | Used agent | Obtained structural information |
|---|---|---|---|
| Excitation Spectrometry of: UV, VIS, IR, Raman and microunde | Ultraviolet (UV) Visible | Electromagnetic radiations | Presence of different structural particularities; presence, nature and intensity of atomic interaction in organic molecules. |
| Infrared (IR) | |||
| Combined diffusion (Raman) | |||
| Microunde | |||
| Energetic | Calorimetry | Heat | thermodynamic data; link energies; Conjugation energies and effects |
| Mass spectrometry (SM) | Beam of e– and electromagnetic field | ||
| Geometric | Roentgenography | Beam of X rays | Electronic densities; interatomic distances; Valence angles; Elementary crystalline cell; Spatial disposition of atoms. |
| Electronography | Beam of electrons | ||
| Neutronography | Beam of neutrons | ||
| MagneticSpectrometry of: NMR, SER, etc. | Nuclear magnetic resonance; (NMR) | Magnetic field | Nature, number and disposition of hydrogen atoms in molecule |
| Spin electric resonance (SER). | |||
| Electric | Molecular polarization | Electric field | Electric moment of molecule; Atomic effects and interactions. |
| Dipole–moment | |||
| Optic | Light Diffusion | Monocromatic light Polarized light |
Presence of some structural particularities; Nature and intensity of some atomic interactions; Optic activity. |
| Light Refraction | |||
| Rotating optic dispersion | |||
| Circular dicroism (CD) | |||
| Rotating power |
Table 2: Physical methods used in establishing the chemical structure of carotenoids.
Some subtle details of configuration, conformation, rapid changes of structure etc. would not be discovered by use of chemical methods. It must be underlined the value of each method in a certain field (for exp. the spin electric resonance in the research of free radicals, UV and visible spectrometry to analyse the electronic structure of the researched substances, IR spectrometry for the determination of functional groups, NMR for the analysis of number, position and bonds of different nuclei etc.). There were even developed mixed methods, for example double electronic–nuclear resonance, mixed technique gas chromatography–mass spectrometry [38]. With the help of physical methods it is studied and, in most cases it is automatically recorded, the different physical states of the organic molecules in relation to their behavior towards the action of different physical agents used in the study, such as electromagnetic radiations, elementary particle beams, magnetic fields, polarized light etc. Spectral methods are based on the interaction of electromagnetic radiations with the carotenoid molecules that can absorb radiations at different wavelengths.
The energy, being directly proportional with the frequency and inversely proportional with the wavelength, has different values in different spectral regions (Table 3).
| Change | Spin | Orientation | Configuration | Electronic distribution | ||
|---|---|---|---|---|---|---|
| RMN | RES | Microwaves | IR | UV–Vis | x rays | |
| Wave number | 1 cm–1 | 102 cm–1 | 104 cm–1 | 106cm–1 | ||
| Wavelength | 100 m | 1m | 1 cm | 100μm | 1 μm | 100 A |
| Frequency (Hz) | 3 x 105 | 3 x 108 | 3 x 1010 | 3 x 1012 | 3 x 1014 | 3 x 1016 |
| Energy(kcal/mol) | 0,0003 | 0,03 | 3 | 3 x 102 | 3 x 104 | 3 x 106 |
Table 3: Energy values according to the spectral region.
This leads to the change of the molecules energy which is proportional with frequency eq. (5):
ΔE=Ef–Ei hν=hc/ λ (5)
Where ΔE is the variation of the energy of system, Ef and Ei energy of the system in final and initial stage, h – Planck constant, ν –radiation frequency, c –light speed, λ –radiation wavelength. If Ef > Ei , energy absorption occurs, case which corresponds to absorption spectrometry, and when Ef < Ei , energy emission occurs, case of emission spectrometry.
UV–VIS spectra
The absorption of electromagnetic waves in the visible field (400– 800 nm) and ultraviolet (140–400 nm) is produced with changes of energy of valence electrons. The spectra obtained at the absorption of radiation with wavelength between 120 and 1000 nm in the UV, visible and near IR field are named electronic spectra, because are due to electronic transitions in orbitals σ, π or n, from a low energy state to an excited state richer in energy. The electronic transitions happen from a link orbital σ or π, occupied with electrons in fundamental state or with electrons that do not participate in an anti–link orbital σ* or unoccupied in fundamental state, but occupiable in excited state.
• σ→σ* transitions are manifested at the carotenoid units with σ links, require high energies, for which the corresponding bands are at low wavelengths in vacuum UV
• n→σ* and n→π* transitions are characteristic to carotenoid units that contain atoms with unparticipating electrons, require lower energies, the corresponding absorption bands being at wavelengths higher than those corresponding to the σ→σ* transitions
• π→π* transitions are specific to carotenoid units with double bonds and determine the absorptions at wavelengths who’s values depend on the nature of the substituents
Absorption of radiations in visible and UV by the molecules of carotenoids takes place after the Lambert–Beer law eq. (6):
A = lg (I0 / I) = ε⋅l⋅c (6)
Where A is the absorbance (optic density), I0 and I are the incident light intensity, and transmitted respectively, c–solution concentration (in mol/L), l–thickness of the solution layer (in cm), and ε–molar absorptivity (named also extinction molar coefficient). Weak absorptions, with ε = 10 – 100 indicate n → π* transitions. A strong O absorption at 200–260 nm, with ε = 100 – 4000 indicate a n → σ* transition, and a very strong absorption with ε = 10 000 – 20 000 indicate a π → π* transition, attributed to 200 nm to a conjugated system of double bonds [39]. UV-visible light (UV-Vis) absorption spectra of crude methanolic extracts were determined using a spectrophotometer. Wavelengths from 400 nm to 800 nm were recorded at 0.5 nm intervals. For carotenoids for which the extinction molar coefficients for the wavelength corresponding the absorption maximum are known, quantitative determinations can be done [40].
IR spectra
Absorption of IR radiations is accompanied by transitions between vibration levels. The necessary energy being of few kcal/mol, the vibration transitions are accompanied by rotation transitions.
By mixing these two types of transitions it results the characteristic bands of IR spectra. Carotenoids contain the π-electron-conjugated carbon-chain molecules and are similar to polyenes with regard to their structure and optical properties. Structurally, these molecules have a linear, chain-like conjugated carbon backbone that consists of alternating carbon single and double bonds with varying numbers of conjugated double bonds and number of functionalities in various positions such as keto, aldehyde, ester, hydroxy, methoxy and glycoside groups. IR spectroscopy allows resolving some structure problems for carotenoids; because it can be attributed a certain maximum of absorption to some vibration transition characteristic to some groups of atoms or links [41]. By comparing the spectra of a number of carotenoids that contain in the molecule the same structural unit, it is attributed the band with frequency almost constant to the common structural unit. The group characteristic vibrations are quasi– independent of the rest of the molecule. The IR spectrum is a characteristic property of a pure compound, offering the possibility of a more precise identification than the melting point, refractive index or density. Some carotenoids could be quantified by means of FT-IR spectroscopy.
The IR bands of the characteristic functional groups (CH3, CH2, C=C, C=O, OH, etc.) could be assigned when possible. Some special functional groups – without H-atoms – such as C=C, ‘cross epoxides', etc., which cannot be easily identified by 1H-NMR methods, were also detected in the FT-IR spectra [40]. Also, it offers the possibility of qualitative and quantitative analysis of some mixtures (absorption of a carotenoid being proportional with its concentration in the mixture). This method is useful in the fast analysis of the biologically important carotenoids especially if there are small samples available.
Raman spectra
At the passing of a strong monochromatic light beam through a carotenoid, it is obtained a diffused light line spectrum. The molecules take the energy of the light beam quanta which, losing energy, have a lower frequency and is scattered in all directions. A Raman spectrophotometer allows the analysis of the diffused light, in direction perpendicular on the propagation direction of the incident light.
The determined frequencies have the same level with the values that are obtained in IR. Some frequencies of the Raman spectrum can be missing in the IR spectrum and in reverse, especially in the case of symmetric molecules. Resonance Raman spectra of naturally occurring carotenoids could be obtained under 514.5 nm. The spectra acquired from the carotenoids are simple, and are exclusively dominated by bands at 1510–1520 cm−1 assigned to ν (C=C) in-phase stretching, 1150–1160cm−1 assigned to ν (C−C) stretching, and 1010 cm−1 assigned to a combination of δ (C=CH) methyl in-plane rocking and δ (C−H) out-of-plane bending modes. Of engrossing, an intense band at 1450 cm−1 is the dominant mode and is assigned to δ (CH2) scissoring mode of methylene. The spectra contain several bands between 1390 and 1000cm−1 which are assigned to ν (C−C) stretching modes from the main hydrocarbon chain, and several bands between 1000 and 800cm−1 that are assigned to a combination of δ (C=CH) methyl in-plane rocking and δ (C−H) out-of-plane bending modes [41].
Like IR spectra, Raman spectra give information on the molecular structure of carotenoids and apply for the qualitative and quantitative analysis of carotenoids, but on a low scale.
Microwave spectra
With the help of microwave spectra there were calculated the inertia moments of small molecules, after which could be determined the lengths of bonds and valence angles of carotenoids. Microwave spectra give information on repartition of electronic density in the molecule of carotenoids, allowing the evaluation of the hybridization degree. Applying this method is though limited only to molecules with permanent electric moment that give microwave spectra.
Nuclear magnetic resonance spectra
NMR constitutes a physical method efficient for the identification of carotenoids. The phenomenon of nuclear magnetic resonance is based on the property of nuclei of some atoms of having a rotation movement around their own axis. Base information regarding the molecular structure given by the NMR spectra is supplied by:
• Signal number, which states how many types of protons are in the molecule of carotenoids
• Signal position in the spectrum, states the nature of the respective protons (alkane, alkene, aromatic, benzilic, hydroxilic, aldehidic etc.)
• Signal intensity, which sets the number of protons that generate each signal
• Signal breaking in doublets, triplets, multiplets, which give information regarding the number of protons situated on the carbon atom neighbour with the carbon atom carrying the broken signal proton
With the help of this technique the elucidation became possible for some structural details, like the ones related to the biopolymer structure that are the base of the vital phenomena in plants. The fact that the energies involved in the onset of the NMR phenomenon are very low, give the guarantee that the studied carotenoids are not altered by the impact of the energy with the carienoidic compounds subjected to investigation [42]. Conformational analysis that deals with the study of stability and reactivity of conformational isomers would be unconceivable without the value added by the dynamic NMR, method capable of surprising structural aspects characteristic to carotenoid molecules with rigid structures by cooling to low temperatures. Studies in the fields: hydrogen bonds, tautomery, proton exchange phenomena, stereochemistry, reaction mechanisms, quantitative organic chemistry are due to the nuclear magnetic resonance, protonic resonance or the one in which 13C, 19F or 31P atoms are involved.
Mass spectrometry
Mass spectrometry (MS) is based on the transformation of the researched carotenoids in ions, by electron bombarding, after which the ions are accelerated, separated and recorded. Taking into account the relative abundance of all ionic species formed, important conclusions can be drawn regarding the carotenoid structure, can be set the molecular mass or the isotopic composition. As a follow–up of the impact between an electron and a molecule of carotenoid (Car), two ionization processes can occur:
Car + e– → Car–;
Car + e–→ Car + + 2e–.
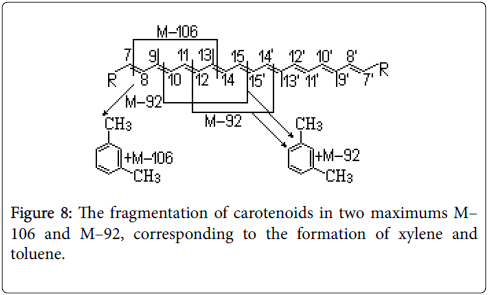
In the first case a negative ion–radical was formed, and in the second case, by removing an electron from the carotenoid particle, a positive ion–radical resulted. If the stream contains electrons with energy equal to the ionization energy of the molecule, the formed cation–radical is called molecular ion or pariate ion and has the same mass as the molecule from which it came. By determining the mass of the molecular ion it can be set the exact value of the molecular mass. Usually, the electron beam has an energy of about 70 eV, which makes the molecular ion to be very unstable, decomposing in a way specific to each carotenoid, by identifying the resulted fragments being able to obtain information regarding the structure of the researched carotenoid. On the basis of the information obtained from the specialty literature and experiments, have been featured properties characteristic carotenoid ions that can be associated to functional groups in the structures of these biocompounds.
Furthermore, biocompounds with a very similar structure have been differentiated through comparison of the intensities of their fragments. The data provide a basis for the structural elucidation of carotenoids by MS [43].
Chemical Methods
Carotenoids can be identified by reactions characteristic to different functional groups, with specific reagents. By an assembly of operations and chemical reactions the following functional groups can be identified: hydroxyl (–OH), ether (–O–), carbonyl (C=O), carboxyl (– COOH), ester (–COOR), anhydride (–CO)2O, amide (–CONH2), nitrile (–C≡N), nitro (–NO2), nitrozo (–N=O), amine (–NH2), azo (– N=N–) etc. Investigation by chemical way of the carotenoid structure, even though successfully used for long time, is linked to some advantages, like long time (hours, days), necessity of some big quantities of substances, its irreversible consuming, limited structural information etc. (these are disadvantages of the method). Most of these inconvenients were resolved by introducing physical methods for the determination of carotenoids chemical structure.
Carotenoid identification methods
The most important characteristic of carotenoids is the absorption spectrum that is a function of the chromophore of carotenoids. The most important identification process of carotenoids is the determination of its absorption spectrum in visible, in different solvents. The absorption characteristics are researched by column chromatography or TLC. Identification of carotenoids can be done also by chemical tests, which detect the different functional groups in the molecule of the pigment, by color reactions. The main physical and chemical methods of identification of carotenoid pigments.
Identification of carotenoids by spectroscopic methods
Spectroscopic methods used frequently in carotenoid identification are spectroscopy of absorption in visible, IR, nuclear magnetic resonance and mass.
Identification by spectroscopy in visible
This method was used for the first time for identification of carotenoids and their metabolites.
Characterizing with precision of carotenoid chromophore is possible due to the correlation between the electronic structure and absorption, position of absorption maximums being a function of the number of the conjugated double bonds in the molecule. In the case in which the chromophore contains triple bond C≡C instead of the double bond C=C, λ max will decrease. Carotenoids present three fundamental maximums in the visible field, their spectrum having a smooth structure. For the determination of the chemical structure it is important to know the level of influence on the spectrum by the existence of some existent groups in the molecule. Thus, the double bonds C=C and C=O influence very much the electronic spectra, in this way being able to set a link between the electronic spectra and the chromophore systems. In the case of existence of conjugated double bonds, the maximum wavelength will raise, this increase not being constant [44]. By comparison λ max of an unknown carotenoid with the λ max values of a known carotenoid, control, the number of double bonds in the chromophore can be estimated. Absorption maximums depend also on the used solvent, in Table 4 being presented some absorption maximums of some carotenoids in different solvents.
|
Carotenoid |
Solvent |
Absorption maximum (nm) |
||
|---|---|---|---|---|
|
α–Carotene |
acetone |
424 |
448 |
476 |
|
α–Carotene |
chloroform |
433 |
457 |
484 |
|
α–Carotene |
petroleum ether |
422 |
444 |
473 |
|
β–Carotene |
carbon sulfide |
|
472 |
502 |
|
β–Carotene |
hexane |
(425) |
450 |
477 |
|
β–Carotene |
petroleum ether |
(425) |
448 |
475 |
|
β–Carotene |
ethylic alcohol |
(427) |
449 |
475 |
|
γ–Carotene |
benzene |
447 |
477 |
510 |
|
γ–Carotene |
acetone |
(439) |
461 |
491 |
|
Lycopene |
benzene |
455 |
487 |
522 |
|
Luteine |
carbon sulfide |
445 |
475 |
508 |
|
Violaxantine |
hexane |
|
443 |
472 |
|
Zeaxantine |
ethylic alcohol |
(428) |
450 |
478 |
Table 4: Absorption maximums of some carotenoids in different solvents.
Carotenoid spectrum in visible and the absorption maximums depend on the nature of the carotenoid, as follows:
• According to the nature of the polyenic extremity
• According to the presence of the C=O conjugated groups
• According to the cis–trans configuration
Moreover, an array of methods is used in the analysis and quantification of these carotenoids, among which UV /visible light spectrophotometry and, more are recently, HPLC and or high performance liquid chromatography coupled with tandem mass spectrometry (HPLC-MS). The carotenoids have been quantified by HPLC using external calibration curves constructed for carotenoids. The curves have been constructed using standard solutions of known concentrations and analysed by HPLC under the same conditions as the samples. These concentrations were correlated with the respective peak areas (obtained using the chromatograms) by simple linear regression. The mass spectra were acquired at a m/z interval acquainted.
Carotenoids have been separated in a column using a mobile phase.
The carotenoids and have been identified according to the retention times in the column, the UV/visible spectra (λmax, fine structure, cis peak intensity) and mass spectra, based on comparison with informations and confirmed by comparison with the retention times of standards.
Influence of the terminal cyclic group
Depending on the position of the absorption maximums the nature of the polyenic system that is present in the molecule of the carotenoid can be distinguished. In the case of the removal of a double bond from the conjugation with the polyenic system, the maximums are hypsochromically moved.
For the aliphatic carotenoids, the electronic spectrum is characterized by three net marked maximums, while the passing to the monocyclic and bicyclic carotenoids affects the spectra, which are progressively attenuated.
Influence of the C=O group
Influence of the ketone group on the absorption spectrum depends both on its position compared to the polyenic system in the carotenoid molecule, and on the solvent in which the determination is done.
The ketone group conjugated with the polyenic system in the molecule of carotenoids determines a bathochromic move of the absorption maximums.
At the same time with the movement of the maximums, occurs a change in the electronic structure of the spectrum, which has as effect the disappearing of the three characteristic maximums and the appearing of a single one. In the case in which the ketone group is not conjugated with the polyenic system of carotenoids, there will not be an effect on the position of the absobtion maximums.
Influence of the cis–trans configuration
Carotenoids that contain groups in the trans position are transformed easily in cis, this being done under the influence of light, temperature, acids etc. In this case, the smooth structure of the spectrum is diminished, and the absorption maximums are moved towards lower wavelengths.
Characteristic to the carotenoids that have double bonds with the cis configuration is the appearing of a maximum in UV.
Identification by IR spectroscopy
Due to the fact that the aIR absorption spectra present a high number of bands, thus, present a richer informative material, they are more advantageous in determining the structure.
With the help of IR spectra there can be set both the presence and the position of different functional groups in the molecule of carotenoids, and the nature of the polyenic system [40].
In order not to have interferences, carotenoids are tableted in solid phase in KBr or solubilized in different solvents (CCl4, CHCl3, CS2 etc.).
Frequency of Substituents
Nature and position of hydroxyl group
Hydroxy–carotenoids can contain a primary, secondary or tertiary hydroxyl.
In Table 5 are presented frequencies characteristic to the oxygenated groups in the carotenoid molecule.
| Group | ν (C–O), cm–1 | |
|---|---|---|
| OH–primary | 1005–1025 | |
| OH–secondary | allelic (cyclic) | 1020–1030 |
| allelic (acyclic) | 1045 | |
| Non–allelic | 1040–1050 | |
| OH–tertiary | 1140–1150 | |
| 905 | ||
| OH–enolic | 1050–1060 | |
| 1200–1240 | ||
| metoxyl tertiary | 1070–1080 | |
| furanoid (5,8–epoxid) (C–O–C) | 1070 | |
| OH–acyl | 1730–1745 | |
Table 5: Frequencies characteristic to the oxygenated groups in the carotenoid molecule.
For the elucidation of the nature and position of the –OH group it is studied, with the help of IR spectroscopy, the 1000–1200 cm–1 region, in which occurs the C–O bond, this being closer to the skeleton of the carotenoid molecule and more sensitive to changes in the carotenoid chain than the vibration of the –O–H bond.
Nature and position of the carbonyl group
In the IR spectrum, the carbonyl group is envisaged by a band that appears between 1650 and 1690 cm–1. The position of this band depends on the nature and position of the carbonyl group. Aldo– carotenoids present a band at 1685 cm–1, and keto–carotenoids between 1650–1670 cm–1 and even at lower values [45]. Carotenoids that contain the ketone group and, to a lesser extent, aldehyde, are C40 carotenoids.
Frequencies due to the molecular skeleton
Due to the vibration of deformation outside the plane of C–H bond from the rest of –CH=C–, the molecule skeleton produces a band at 870 cm–1. In the region of high frequencies (3100–2800 cm–1), all carotenoids present absorption bands.
Identification by NMR spectroscopy
With the help of NMR method it could be sett the structure of carotenoids.
The most used solvents are deutero–chloroform, chloroform and carbon tetrachloride, using as internal control the tetramethylsilane. By NMR it is emphasized the presence of protons by signals that appear in the spectrum, their size depending on the carotenoid structure, the arrangement of neighbour atoms and the nature and number of protons that are in a certain group. In case of carotenoids, the proton source is constituted by the groups –CH3,–CH2–, –OH, –OCH3, CHO etc. there will be obtained spectra cu different sized signals, which allows to emphasize them. The highest intensity will be for the signals given by the –CH3 groups, because they are the rischest in protons, leading to the possibility of determining both the number and types of the –CH3 groups, and the protons that come from other groups [46].
Identification by mass spectrometry
This method lies in the very precise fragmentation and characteristic to the different types of carotenoids in molecular ions. All carotenoids give a maximum of intensity which is situated at the value of their molecular mass, caused by the molecular ion M+ or carotenoid+. Due to the fact that these pigments are instable and have low volatility, for their volatilization and achievement of molecular ions, heating up until 30°C is required. Maximums and their intensity are determined by the fragmentation mode of the carotenoid molecule, as well as by the stability of the formed ions.
All carotenoids give two maximums M–106 and M–92, corresponding to the formation of xylene and toluene which come from the break of the polyenic chain and from cyclization (Figure 8). The molecular ions that give rise to the M–92 and M–106 maximums are the most abundant and significant in the fragmentation of carotenoids, reason why these two ions represent important indicators in the structural and molecular mass determinations.
M–92 and M–106 maximums do not appear in the case of carotenoids with less the 9 conjugated double bonds. Epoxy– carotenoids are characteristically breaking in the ion at M–70.
Chemical methods for elucidating the structure of carotenoids
Carotenoids can be identified by reactions characteristic to different functional groups or by color reactions that these pigments give with specific reagents. Carotenoids give color reactions with:
• Stibium trichloride (Carr–Price reaction), solution saturated in chloroform, which determines a blue coloration that passes quickly in red–purple
• Concentrated sulfuric acid (Winterstein reaction) which leads to a blue–green of green–blue coloration
• Concentrated hydrochloric acid, mono–epoxids and furanic oxides giving a green–blue coloration
Identification of carotenoid functional groups
Identification of hydroxyl–carotenoids
Identification of hydroxyl–carotenoids is done by acetylation with acetic anhydride in pyridine, at cold. The reaction supplies information regarding the nature of the –OH group (primary or secondary).
By this reaction one can separate the carotenoid acetate by other pigments in the mix, due to the diminishing of molecule polarity. The NMR spectrum will supply further information regarding the position of the hydroxyl. Hydroxy–carotenoids with tertiary hydroxyl can be identified by the formation of trimethylsillilated ether and by dehydration with phosphorus oxychloride. Hydroxy–carotenoids that have a secondary allelic HO– have a high chemical reactivity, being able to be oxidized to keto–carotenoids with silver-oxide or can eliminate the –OH group by treatment with chloroformic hydrochloric acid, passing to retrodehydro–carotenoids, these transformations being emphasized by reading λ max [47].
Identification of keto–carotenoids and aldehyde carotenoids
Keto–carotenoids can be identified by their treatment with sodium borohydride (NaBH4) which reduces them to hydroxyl–carotenoids, which are then identified by spectroscopy methods.
By condensation with orthophenylenediamine there are emphasized the α –diketone groups, forming phenazinic derivatives, colored specifically (blue). Monoketones do not give this reaction, which proves the specificity of the reagent. The aldehyde carotenoids can be identified by reaction with 2–phenyl–1,3–indandione–1hidrazone, when stable complexes are formed, this reaction occurring with keto– carotenoids also. Both keto–carotenoids and aldehyde carotenoids by treatment with hydroxylamine chlorhydrate in pyridine form oxymes, this thing being emphasized by increasing polarity and hypsochromic movement of λ max.
Identification of carotenoids with epoxy groups
A reaction specific to all epoxy–carotenoids is the treatment with hydrochloric acid, in the case of 5,8–epoxy–carotenoids being obtained a blue coloration. Reaction of epoxy–carotenoids with mercuric chloride leads to the formation of a colored complex, which can be identified by the absorption spectra. The formation of the colored complexes with the mercuric chloride and their absorption maximums represent characteristics of 5,6– and 5,8–epoxy–carotenoids. At the treatment of 5,8–epoxy–carotenoids in benzenic solution with titanium tetrachloride (TiCl4), results a dark–blue complex, which presents a λ max with a strong bathochromic absorption [48]. These complexes can be broken by acetone–lysis to retrodehydro– carotenoids.
The quantitative determination methods for carotenoids
The spectrophotometric method represents the general method used in the quantitative determination of carotenoids, the essential condition being that the pigments to be of analytical purity.
The most used solvents for the dosing of hydrocarbon carotenoids are petroleum ether and hexane, and for xanthophylls the polar solvents. The quantity of carotenoid is calculated taking into account the volume V of the solution (mL), the extinction ε read at the given wavelength and the substance quantity G taken into work (g) eq. (7) and (8):


The coefficient of specific extinction, noted 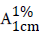 , represents the extinction at a certain wavelength and in a certain solvent of concentration 1%, in a cuvette with a diameter of 1 cm. In the reference cuvette is the respective solvent. For determinations in the UV area are used quartz cuvettes.The specific extinction is determined by the maximum wavelength (λ max), in the case of solutions that cannot spectrophotometrate due to the very high concentration of the solution, this is diluted, and the dilution value is introduced in the counter of the relation, by amplification [49]. From the value of the coefficient of specific molar extinction ε, which is a characteristic of the carotenoid chromophore, it can be done the calculation
, represents the extinction at a certain wavelength and in a certain solvent of concentration 1%, in a cuvette with a diameter of 1 cm. In the reference cuvette is the respective solvent. For determinations in the UV area are used quartz cuvettes.The specific extinction is determined by the maximum wavelength (λ max), in the case of solutions that cannot spectrophotometrate due to the very high concentration of the solution, this is diluted, and the dilution value is introduced in the counter of the relation, by amplification [49]. From the value of the coefficient of specific molar extinction ε, which is a characteristic of the carotenoid chromophore, it can be done the calculation 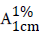 the relation between them being eq. (9):
the relation between them being eq. (9):
ε= 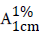 (molecular mass /10) (9)
(molecular mass /10) (9)
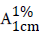 (A) of a carotenoid A can be calculated from
(A) of a carotenoid A can be calculated from 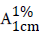 (B) of carotenoid B with an identical chromophore, with relation eq. (10):
(B) of carotenoid B with an identical chromophore, with relation eq. (10):
ε= 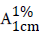 (B) [molecular mass (B)/ molecular mass (A)] (10)
(B) [molecular mass (B)/ molecular mass (A)] (10)
If the value 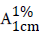 is not known, are taken into consideration the following values presented in Table 6.
is not known, are taken into consideration the following values presented in Table 6.
| Carotenoids | 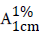 |
λ (nm) | Solvent |
|---|---|---|---|
| α–Carotene | 2800 | 444 | petroleum ether |
| α–Carotene | 2180 | 477 | carbon sulphur |
| β–Carotene | 2008 | 484 | carbon sulphur |
| β–Carotene | 2592 | 453 | petroleum ether |
| β–Carotene | 2592 | 453 | hexane |
| β–Carotene | 2620 | 453 | ethylic alcohol |
| β–Carotene | 2337 | 465 | benzene |
| β–Carotene | 2396 | 465 | chloroform |
| Phytoene (15–cis) | 757 | 286 | hexane |
| Phytoene (trans–total) | 951 | 286 | hexane |
| Phytofluene | 1577 | 347 | hexane |
| Fucoxanthin | 2036 | 478 | carbon sulphur |
| Lycopene | 3450 | 472 | petroleum ether |
| Lycopene | 3240 | 508 | acetone |
| Luteine | 2550 | 445 | ethylic alcohol |
| Luteine | 2236 | 458 | benzene |
| Retrodehidro–β–carotene | 3205 | 472 | petroleum ether |
| Retrodehidro–β–carotene | 3130 | 473 | cyclohexane |
| Violaxantine | 2550 | 443 | ethylic alcohol |
| Zeaxantine | 2340 | 452 | acetone |
Table 6: Values of the coefficient of specific extinction.
Sometimes it is necessary to determine the concentrations of the two carotenoids with the help of absorption spectra, the chromatographic method not allowing this. The total content of carotenoids from 100 g tissue (dry matter-DM%) can be calculated after the relation eq. (11):
mg carotenoid/100g tissue=( DM⋅V⋅103)/[tissue weight (g)] (11)
Percentage of carotenoids (reported that the dry matter -DM%) can be calculated with the cu relation eq. (12):
% pigment= [100 V DM λ max (for each fraction)] / [V DM λ max (for each fraction)] (12)
In some methods, expressing the quantity of pigment is done through the relation eq. (13):
(μg ß–carotene) 100 mL= Ax 400 (13)
In which Ax represents the absorption of the carotenoid to be researched at 450 nm [50]. Phytofluene is a carotenoid (the second product of carotenoid biosynthesis) with an orange color found naturally in tomatoes and other vegetables. It is formed from phytoene in a desaturation reaction leading to the formation of five conjugated double bonds. In the following step, addition of carbon-carbon conjugated double bonds leads to the formation of z-carotene and appearance of visible color.
This biocompound has absorption spectra in the UVA range, with maximal absorption at 348 nm and with ε1% of 1557. Quantitative determination of individual carotenoids can be done also by automated chromatography of a mix of pigments, and that is by high performance liquid chromatography.
The degradation of carotenoids
The carotenoids are degraded (for example by the oxidation mechanisms), including pathways induced by light, heat, acid, oxygen, metals, or interactions with radical species. What's more, a few carotenoid delivery systems are evaluated for their potential to decrease carotenoid degradation in food (or functional food products). When exposed to oxygen, all carotenoids (except beta-cryptoxanthin), degraded at lower rates. The degradation of all the carotenoids followed zero-order reaction kinetics.
The carotenoids are constantly synthesized and degraded. This maintains carotenoids at physiological levels, in green tissues, where a constant ratio of chlorophylls to carotenoids has to be maintained to ensure the integrity of the photosynthesis system. Carotenoid degradation by enzymatic oxidative cleavage produces an array of terpenoid products collectively known as apocarotenoids (i.e., abscisic acid and strigolactones, and other volatile and non-volatile compounds), which are known in industries for their use as aromas, flavors and fragrances (while β-ionone, are known to role in plantinsect interactions). During processing, carotenoids may isomerize to cis-isoforms with the existence of heat and, or oil, or for the time of dehydration. Additionally, for the time of storage and, or processing carotenoids undergoes further geometric (cis/trans) isomerism, mainly cis- to trans retro-isomerisation. With long heating times or temperatures above 50°C, degradation proceeds faster than isomerization, the stability of carotenoids isomers decreases in the order: 5-cis > all-trans > 9-cis > 13-cis > 15-cis > 7-cis > 11-cis. Beginning from the all-trans-carotenoids, isomerization is characterized first by the formation and then by the disappearance of the unstable 13-cis-isomer. The isomerization and degradation are competitive and contemporaneous processes. It is generally accepted that oxidation of carotenoids begins with epoxidation and cleavage to apocarotenals prior a transformation into other derivatives [51]. Several epoxycarotenoids and apocarotenals were observed in experimental oxidation models but also some were identified in processed foods.
Concluding Remarks
Now, that a big accent is put on the superior valorization of culture plants and those from the spontaneous flora, of natural sources, according to their chemical composition, knowing the biologically active substances is imposed as a request of first. Only by knowing the chemical composition of plants, the biochemical mechanisms that are the base of the biological phenomena, then it can be obtained new plant hybrids, with high content of biologically active substances that increase the biological value of the plants in which they are found. To set the molecular formula, molecular structures and the physical– chemical properties of a bioactive compound, it is necessary to first isolate it from the mix of natural biocompounds.
Extraction methods of biologically active biocompounds are chosen depending on the properties of the biocompounds subjected to extraction. Extraction of carotenoids from the plant products is done with nonpolar organic solvents (ethylic ether, chloroform), by maceration, mechanic agitation or by refluxing on the bath of boiling water, involving grinding, due to the fact that the nonpolar solvent does not reach through the cell membrane in order to dissolve the carotenoids.
Separation methods are chosen according to the physical and chemical properties of the substances that are to be separated. Of the frequently used methods for separation of carotenoids, a big importance is shown for the chromatographic methods, both on the column and in thin layer, as well as the separations by liquid–liquid repartition and by the counter–current distribution method.
For the identification of carotenoids there can be used bot chemical methods as well as spectral methods, which present the advantages of wide applicability, high sensitivity, perfect result reproducibility, sample integrity conservation and determination rapidity. Of the spectral methods used in detecting carotenoids there can be enumerated visible spectroscopy, IR NMR, mass spectrometry.
For the quantitative determination carotenoids, the generally used method is the spectrophotometric method, the essential condition being that the pigments are of analytical purity. The storage of carotenoids should be done in the dark, under an atmosphere of nitrogen or in vacuum, at a temperature of –20°C. The best preservation method is in the crystalline state. In solution, the storage should be done under petroleum ether, hexane or benzene. Determination of physical constants such as melting temperature, boiling point, density, refraction index, absorption intensity at certain wavelengths etc. provides data that allow individualization and characterization of an organic compound. Carotenoids have numerous uses in pharmaceutical industry, cosmetics, food industry (as antioxidants and natural colorants), zootechnics (animal feed) etc. Flower colour is correlated with carotenoids content in flowers and weakly correlated with the carotenoids content in fruits, so flower colour cannot be used as credible indicator of carotenoids content in fruit.
References
- Zhao S, Tuan PA, Kim JK, Park WT, Kim YB, et al. (2014) Molecular characterization of carotenoid biosynthetic genes and carotenoid accumulation in Lyciumchinense. Molecules 19: 11250-11262.
- Butnariu M, Coradini CZ (2012) Evaluation of biologically active compounds from Calendula officinalis flowers using spectrophotometry. Chemistry Central Journal 6: 35.
- Montero O, Sánchez-Guijo A, Lubián LM, Martínez-Rodríguez G (2012) Changes in membrane lipids and carotenoids during light acclimation in a marine cyanobacteriumSynechococcus sp. Journal of Biosciences 37: 635-645.
- Butnariu MV, Giuchici CV (2011) The use of some nanoemulsions based on aqueous propolis and lycopene extract in the skin's protective mechanisms against UVA radiation. Journal of Nanobiotechnology 9: 3.
- Armstrong GA, Hearst JE (1996) Carotenoids 2: Genetics and molecular biology of carotenoid pigment biosynthesis. FASEB J 10: 228-237.
- Armstrong GA, Hearst JE (1996) Carotenoids 2: Genetics and molecular biology of carotenoid pigment biosynthesis. FASEB J 10: 228-237.
- Wichuk K, Brynjólfsson S, Fu W (2014) Biotechnological production of value-added carotenoids from microalgae: Emerging technology and prospects. Bioengineered 5: 204-208.
- Choi S, Koo S (2005) Efficient Syntheses of the Keto-carotenoids Canthaxanthin, Astaxanthin, and Astacene. J Org Chem 70: 3328-3331.
- Filho Had S, Pacheco LC, Andrade Eda S, Correa MJ, Araújo RN (2014) Xanthones from the roots of MoutabeaguianensisAubl. Molecules 20:127-134.
- Choi S, Koo S (2005) Efficient Syntheses of the Keto-carotenoids Canthaxanthin, Astaxanthin, and Astacene. J Org Chem 70: 3328-3331.
- Zorin B, Grundman O, Khozin-Goldberg I, Leu S, Shapira M, et al. (2014) Development of a Nuclear Transformation System for Oleaginous Green Alga Lobosphaera (Parietochloris) incisa and Genetic Complementation of a Mutant Strain, Deficient in Arachidonic Acid Biosynthesis. PLoS One 9: e105223.
- Shete V, Quadro L (2013) Mammalian metabolism of β-carotene: gaps in knowledge. Nutrients 5: 4849-4868.
- Gáspár J (2009) Evolution of phototaxis. Philosophical transactions of the Royal Society of London. Series B, Biological sciences Royal Society 364: 2795-2808.
- Othman R, MohdZaifuddin FA, Hassan NM (2014) Carotenoid biosynthesis regulatory mechanisms in plants. Journal of oleo science 63: 753-760.
- Prabhu S, Rekha PD, Arun AB (2014) Zeaxanthin biosynthesis by members of the genus Muricauda. Polish journal of microbiology 63: 115119.
- Corniani N, Velini ED, Silva FM, Nanayakkara NP, Witschel M (2014) Novel bioassay for the Discovery of Inhibitors of the 2-C-Methyl-D-erythritol 4-Phosphate (MEP) and Terpenoid Pathways Leading to Carotenoid Biosynthesis. PLoS One 9: e103704.
- Altemimi A, Lightfoot DA, Kinsel M, Watson DG (2015) Employing Response Surface Methodology for the Optimization of Ultrasound Assisted Extraction of Lutein and β-Carotene from Spinach. Molecules 20: 6611-6625.
- Hochberg U, Degu A, Toubiana D, Gendler T, Nikoloski Z, et al. (2013) Metabolite profiling and network analysis reveal coordinated changes in grapevine water stress response. BMC Plant Biology 13: 184.
- Radzali SA, Baharin BS, Othman R, Markom M, Rahman RA (2014) Co-solvent Selection for Supercritical Fluid Extraction of Astaxanthin and Other Carotenoids from Penaeusmonodon Waste. Journal of oleo science 63: 769-777.
- Smita S, Rajwanshi R, Lenka SK, Katiyar A, Chinnusamy V, et al. (2013) Expression profile of genes coding for carotenoid biosynthetic pathway during ripening and their association with accumulation of lycopene in tomato fruits. Journal of genetics 92: 363-368.
- Zheljazkov VD, Astatkie T, Schlegel V (2014) Hydrodistillation extraction time effect on essential oil yield, composition, and bioactivity of coriander oil. Journal of oleo science 63: 857-865.
- Medina-Ceja L, Pardo-Peña K, Morales-Villagrán A, Ortega-Ibarra J, López-Pérez S (2015) Increase in the extracellular glutamate level during seizures and electrical stimulation determined using a high temporal resolution technique. BMC Neuroscience 16: 11.
- Naziri D, Hamidi M, Hassanzadeh S, Tarhriz V, MalekiZanjani B, et al.(2014) Analysis of Carotenoid Production by Halorubrum sp. TBZ126; an Extremely HalophilicArcheon from Urmia Lake. Advanced Pharmaceutical Bulletin 4: 61-67.
- Zou TB, Jia Q, Li HW, Wang CX, Wu HF (2013) Response surface methodology for ultrasound-assisted extraction of astaxanthin from Haematococcuspluvialis. Marine Drugs 11: 1644-1655.
- Belin GK, Gülaçar FO (2007) Separation of chlorins and carotenoids in capillary electrophoresis. Journal of Chromatographic Science 45: 593-599.
- Hong E, Kim GH (2013) GC-MS analysis of the extracts from ko rean cabbage (Brassica campestris L. ssp. pekinensis) and its seed. PrevNutr Food Sci 18: 218-221.
- Brown HH, Borchelt DR (2014) Analysis of mutant SOD1 electrophoretic mobility by blue native gel electrophoresis; Evidence for soluble multimeric assemblies. PLoS One 9: e104583.
- Mustafa A, Trevino LM, Turner C(2012) Pressurized hotethanol extraction of carotenoids from carrot by-products. Molecules 17: 1809-1818.
- Karthikeyan R, Devadasu C, Babu SP (2015) Isolation, Characterization, and RP-HPLC Estimation of P-Coumaric Acid from Methanolic Extract of Durva Grass (Cynodondactylon Linn) (Pers). International Journal of Analytical Chemistry 2015: 201386.
- Hong E, Kim GH (2013) GC-MS analysis of the extracts from ko rean cabbage (Brassica campestris L. ssp. pekinensis) and its seed. PrevNutr Food Sci 18: 218-221.
- Ligor M, Kováčová J, Kopciuch G, Studzi�?ska S, Bocian S, et al.(2014) Study of RP HPLC retention behaviours in analysis of carotenoids. Chromatographia77:1047-1057.
- Hubicka U, Witek ZB, Piotrowska J, Krzek J (2015) Determination of neomycin in the form of neomycin derivative with dabsyl chloride by thin layer chromatography and densitometry. ActaPoloniaePharmaceutica 72: 31-37.
- Karthikeyan R, Devadasu C, Babu SP (2015) Isolation, Characterization, and RP-HPLC Estimation of P-Coumaric Acid from Methanolic Extract of Durva Grass (Cynodondactylon Linn) (Pers). International Journal of Analytical Chemistry 2015: 201386.
- Durante M, Lenucci MS, Mita G (2014) Supercritical carbon dioxide extraction of carotenoids from pumpkin (Cucurbitaspp): a review. International Journal of Molecular Sciences 15: 6725-6740.
- Stirbet A, Riznichenko GY, Rubin AB, Govindjee (2014) Modeling chlorophyll a fluorescence transient: relation to photosynthesis. Biochemistry 79: 291-323.
- Cai H, Cao G, Li L, Liu X, Ma X, et al. Profiling and characterization of volatile components from non-fumigated and sulfur-fumigated FlosLoniceraeJaponicae using comprehensive two-dimensional gas chromatography time-of-flight mass spectrometry coupled with chemical group separation. Molecules 18: 1368-1382.
- Karthika K, Paulsamy S (2015) TLC and HPTLC fingerprints of various secondary metabolites in the stem of the traditional medicinal climber, solenaamplexicaulis. Indian Journal of Pharmaceutical Sciences 77: 111-116.
- Hitaka Y, Nakano A, Tsukigawa K, Manabe H, Nakamura H (2013) Characterization of carotenoid fatty acid esters from the peels of the persimmon Diospyros kaki. Chemical and Pharmaceutical Bulletin 61: 666-669.
- Yang Y, Gu L, Xiao Y, Liu Q, Hu H, et al. (2015) Rapid identification of α-glucosidase inhibitors from Phlomistuberosa by Sepbox chromatography and thin-layer chromatography bioautography. PLoS One 10: e0116922.
- Nolan JM, Meagher K, Kashani S, Beatty S (2013) What is meso-zeaxanthin, and where does it come from? Eye 27: 899-905.
- Butnariu M, Caunii A, Putnoky S (2012) Reverse phase chromatographic behaviour of major components in Capsicum Annuum extract. Chemistry Central Journal 6: 146.
- Fleshman MK, Riedl KM, Novotny JA, Schwartz SJ, Harrison EH (2012) An LC/MS method for d8-β-carotene and d4-retinyl esters: β-carotene absorption and its conversion to vitamin A in humans. Journal of Lipid Research 53: 820-827.
- Shinopoulos KE, Yu J, Nixon PJ, Brudvig GW (2014) Using site-directed mutagenesis to probe the role of the D2 carotenoid in the secondary electron-transfer pathway of photosystem II. Photosynthesis Research 120: 141-152.
- Krasnovsky AA, Kovalev YV (2014) Spectral and kinetic parameters of phosphorescence of triplet chlorophyll a in the photosynthetic apparatus of plants. Biochemistry 79: 349-361.
- Faheem S, Kim SH, Nguyen J, Neravetla S, Ball M (2012) Wax-tear and meibum protein, wax-β-carotene interactions in vitro using infrared spectroscopy. Experimental Eye Research 100: 32-39.
- Shi Q, Wang H, DU C, Zhang W, Qian H (2013) Tentative identification of torulenecis/trans geometrical isomers isolated from Sporidioboluspararoseus by high-performance liquid chromatography-diode array detection-mass spectrometry and preparation by column chromatography. Analytical Sciences 29: 997-1002.
- Kopec RE, Schweiggert RM, Riedl KM, Carle R, Schwartz SJ (2013) Comparison of high-performance liquid chromatography/tandem mass spectrometry and high-performance liquid chromatography/photo-diode array detection for the quantitation of carotenoids, retinyl esters, α-tocopherol and phylloquinone in chylomicron-rich fractions of human plasma. Rapid Communications in Mass Spectrometry 27: 1393-1402.
- Dong L, Shion H, Davis RG, Penak DB, Perez DJ, et al. (2010) Collision cross-section determination and tandem mass spectrometric analysis of isomeric carotenoids using electrospray ion mobility time-of-flight mass spectrometry. Analytical Chemistry 82: 9014-9021.
- Souza ADS, Camara CA, Silva MSDE, Silva TM (2013) Composition and Antioxidant Activity of Geopropolis Collected by Meliponasubnitida (Jandaíra) Bees. Evidence-Based Complementary and Alternative Medicine 2013:801383.
- Barrero AF, Herrador MM, Artega P, González JA, Arteaga JF (2012) A minor dihydropyranapocarotenoid from mated cultures of Blakesleatrispora. Molecules 17: 12553-12559.
- Filipowicz A, Wo�?owiecS (2012) Bioconjugates of PAMAM dendrimers with trans-retinal, pyridoxal, and pyridoxal phosphate. International Journal of Nanomedicine 7: 4819-4828.
Relevant Topics
- Aquatic Ecosystems
- Biodiversity
- Conservation Biology
- Coral Reef Ecology
- Distribution Aggregation
- Ecology and Migration of Animal
- Ecosystem Service
- Ecosystem-Level Measuring
- Endangered Species
- Environmental Tourism
- Forest Biome
- Lake Circulation
- Leaf Morphology
- Marine Conservation
- Marine Ecosystems
- Phytoplankton Abundance
- Population Dyanamics
- Semiarid Ecosystem Soil Properties
- Spatial Distribution
- Species Composition
- Species Rarity
- Sustainability Dynamics
- Sustainable Forest Management
- Tropical Aquaculture
- Tropical Ecosystems
Recommended Journals
Article Tools
Article Usage
- Total views: 130931
- [From(publication date):
June-2016 - Aug 16, 2025] - Breakdown by view type
- HTML page views : 120384
- PDF downloads : 10547