Research Article Open Access
Microorganisms Metabolism during Bioremediation of Oil Contaminated Soils
| Xenia ME1 and Refugio RV2* | |
| 1Universidad Autónoma Metropolitana-Iztapalapa, Departamento de Ingeniería de Procesos e Hidráulica, San Rafael Atlixco No. 186, Colonia Vicentina, Delegación Iztapalapa, C.P. 09340, México City, Mexico | |
| 2Departamento de Biotecnología y Bioingeniería, Centro de Investigación y de Estudios Avanzados del Instituto Politécnico Nacional. Av. IPN No. 2508 Col. San Pedro Zacatenco, Cd. de México, CP. 07360, Mexico | |
| *Corresponding Author : | Refugio RV Departamento de Biotecnología y Bioingeniería Centro de Investigación y de Estudios Avanzados del Instituto Politécnico Nacional. Av. IPN No. 2508 Col. San Pedro Zacatenco Cd. de México, CP. 07360, Mexico Tel: +52(55)57473316 Fax: +52(55)50613313 E-mail: rrodrig@cinvestav.mx |
| Received: January 27, 2016 Accepted: March 04, 2016 Published: March 11, 2016 | |
| Citation: Xenia ME, Refugio RV (2016) Microorganisms Metabolism during Bioremediation of Oil Contaminated Soils. J Bioremed Biodeg 7:340. doi: 10.4172/2155-6199.1000340 | |
| Copyright: © 2016 Xenia ME, et al. This is an open-a ccess article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Visit for more related articles at Journal of Bioremediation & Biodegradation
Abstract
The development of some industrial processes, such as extraction, refining and oil transportation has been the cause of environmental degradation caused by oil spills and their extraction processes involved. In order to reduce these contaminants, among others, physicochemical technologies have been applied, which haven't solved the contamination problem because in many of these processes, new toxic compounds are generated. Technologies based in the use of microorganisms, plants and other biological species are called bioremediation. This is an alternative to the physical and chemical processes of treatment that, although slower in response, are more sustainable. Therefore, in this review, bioremediation technologies to treat oil contaminated soils are analyzed as a convenient alternative for the restoration of impacted soils, against progressive deterioration of environmental quality. Additionally, the microorganisms as well as the required conditions for which bioremediation is considered a sustainable process to undertake will also be presented.
| Keywords |
| Bioremediation; Microorganisms; Hydrocarbons |
| Introduction |
| Soil is a natural nonrenewable resource; it is the most easily affected space of the oil industry (exploration, drilling and exploitation) because of the residue the industry generates. In countries where oil activity is fundamental, the ground and subsurface is highly polluted [1]. This contamination can be carried to the aquifers; this is why soil rehabilitation is a priority. |
| There are different physicochemical techniques to control and mitigate hydrocarbon contamination. Among the methods used to restore contaminated ground there is incineration, thermal desorption, and ground confinement to locations destined for its dispose [2]. Although considered effective, incineration and desorption strategies prompt harsh damage to the soil; the treatment causes losses in its nutritional value. These methods also generate carbon dioxide, carbon monoxide, sulfur dioxide and nitrogen oxides, which are gasses with greenhouse effect [2]. The confinement method consists in removing residue and/or contaminated soil and releasing it to a localized area or in covering it with an impermeable coating, that allows the permanence of hydrocarbon in the subsoil, which lixiviates to the shallow groundwater reserves or spread on the surface. |
| These methods are expensive, cost time and are not a real solution to the problem on the long run [3]. In the past years, the use of biological technologies has demonstrated to be efficient and adequate, and cause less impact to the location where the problem is found. These techniques are denominated bioremediation, which is the technology that uses microorganisms’ natural metabolic potential to transform contaminants into simple compounds, that are slightly or nonpollutant. Bioremediation uses environmentally sustainable methods [4]. Bioremediation does neither alter nor modify the food chain in the treated ecosystems: it respects the flora and fauna of the contaminated site. This is a biotechnological practice that has gained great importance worldwide, because it is friendly with the environment and very low in cost [5]. The following example demonstrates the value in dollars for each one of the treatments applied in one ton of contaminated soil (value in dollars): deposit or confinement: $140-200, incineration: $140-150, stabilization: between $100 y 200 and bioremediation: between $15 y 170 [6]. The application of bioremediation techniques reduce or eliminate the costs for soil transportation because the ground can be treated directly in the contaminated area. This is a feasible technique that can be applied in areas that are difficult to access, where, once the site has been cleansed, it may be once more used as landfill for industrial areas, if completely restored, it may be used for housing developments and, in some cases, even for agricultural purposes. |
| Toxicity in contaminated ground by hydrocarbon has been proven by toxicity bioassays, which can provide information about the characterization process and the ecological risks in the contaminated site. Toxicity in petroleum hydrocarbons in strongly correlated with a low boiling point and especially with those that are within the range of C10-C19. Some metabolic compounds such as aldehydes, aromatic ketones, carboxylic acids, fatty acids and esters, also contribute to the contamination of the soil. There is proof that allows the evaluation of the toxicity in soil, such as Microtox, the use of earthworms and seed germination trials. The time of exposition of the ground to it may favor the decrease of toxicity in the ground by means of the humic acid activity in the soil. However, the change in the toxicity under different conditions and time exposition is still unknown because of the intervening variables [7]. |
| Hydrocarbon biodegradation |
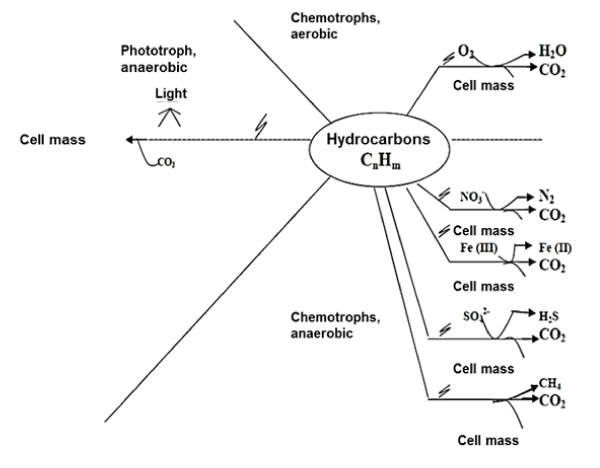
| The process of digestion, assimilation and hydrocarbon metabolism carried out by bacteria, fungi, protozoa and other organisms (Figure 1). The process of biodegradation includes the following reactions: oxidation-reduction, adsorption processes and ion exchange, as well as chelation reactions that result in metal buildup. Microbial biodegradability allows the transformation of hydrocarbons of complex structure into a more simple chemical structure [8]. When the transformation is simple, it is called "primary", when complete, "mineralization", in the latter, hydrocarbon is decomposed into inorganic compounds and/or cellular constituents [9,10]. The acceleration in hydrocarbon biodegradation may be carried out by native microorganisms or bio-augmentation, with appropriate nutritional and environmental conditions [11]. |
| Hydrocarbon biodegradative microorganisms |
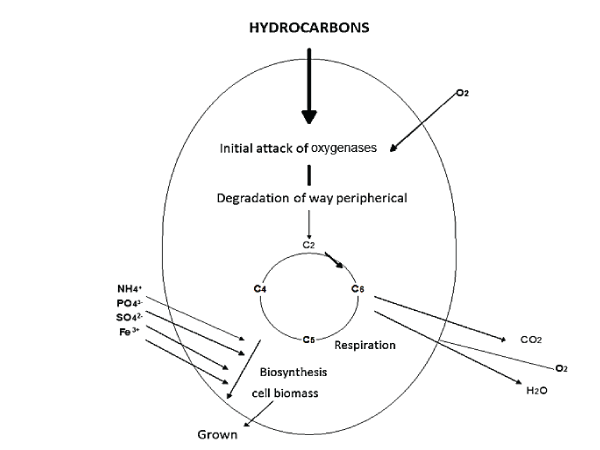
| Bacteria: Among the most studied bacteria in this process are Pseudomonas, Alcaligenes, Sphingomonas, Rhodococcus and Mycobacterium, these degrade aliphatic and aromatic hydrocarbons, using them as the only source of carbon and energy. Aerobic degradation is carried out by dioxygenases enzymes that use oxygen during the degradation process (Figure 2). Microorganisms first oxidize the carbon molecule through a complex multi-enzymatic (oxygenase type enzymes) that incorporate an oxygen molecule. This is how a hydrocarbon with an alcohol group molecule (a more reactive one) is obtained. Other enzymes oxidize the alcohol group to an aldehyde group and finally to a carboxylic acid. Because of this, a molecule, similar to a fatty acid, is obtained and it may be degraded to an acetyl-CoA by beta oxidation. This process of oxidation can occur in non-terminal carbons, giving place to two fatty acids that are processed by beta oxidation [12]. |
| Biodegradation can also be done in the absence of oxygen, as it happens in the deepest hydrocarbon sediments or reservoirs from which anaerobic bacteria has been isolated [13,14]. Bacteria use nitrates, sulfates and iron as electron acceptors for their metabolism [15]. Example of it is the Archeas bacteria that oxidize methane in association with sulfate reducing bacteria [16]. Other anaerobic bacteria that have been reported with hydrocarbon biodegradable capacity are Dechloromonas aromatica and some species from the genus Azoarcus, Thauerasp [17,18]. |
| Fungi: These microorganisms are known for their diversity and for their capacity to degrade natural, persistent and complex material [19]. The mycelium contains insoluble substances. With them, it begins the process of biodegradation, allowing for subsequent entrance or succession of other microorganisms and facilitating compound decomposition [20]. Fungi can extend by means of their hyphae and grow in stress conditions that bacteria cannot resist due to low disposition to nutrients, low pH and low humidity [1]. In hydrocarbon bioremediation, the fungi of white rot (lignin fungi) have been mostly studied [21,22]. Among these species there is Tyromyces palustris, Gloeophyllum trabeum, Trametes versicolor, which oxidize hydrocarbons by the action of the laccase enzyme [23]; Phanerochaete chrysosporium, oxidizes the anthracene and the pyrene through lignin peroxidase enzymes (LiP) and manganese peroxidase (MnP) [24,25], Stropharia coronilla oxidizes benzopyrene through MnP [26] and Pleurotus ostreatus which decomposes the benzanthracene, chrysene, benzofluoranthene, benzopyrene, dibenzanthracene and benzoperylene through the laccase and the MnP [27]. |
| Filamentous fungi, native to the ground [21] can also be decomposed; however, in many cases they must first be adapted by developing them on solid supports (sugar cane bagasse, sorghum or other agricultural residue) [28,29], with small contaminant quantities in order to place them later on the contaminated site. This process of bioaugmentation may also be applied to the lignin fungi [30]. |
| Yeast: Yeasts may also be used in bioremediation of hydrocarbons, among which there are Debayomyces, Saccharomyces, Yarrowia, Pichia and some species of Candida [31,32]. Yeast Candida viswanathii has demonstrated a high percentage of removal of phenanthrene (77.21-89.76 %), as this it is a low- molecular weight hydrocarbon, it also demonstrated high efficiency of removal (55.53-60.77 %) of benzopyrene, which is a high-molecular weight hydrocarbon [33]. The isolated strain Pichia anomala 2.2540, from a contaminated ground, demonstrated high removal at 96 hours, in cultures with naphtalene, dibenzothiophene, phenanthrene and chrysene, both in individual as in combined forms [34]. Yeasts have the capacity of adaptability, such is the example of the Candida digboiensis, strain, isolated from the contaminated ground by hydrocarbons and acidic pH [35]. |
| Algae: Walker et al. in 1975 [36] were the first to report the isolation of algae Prototheca zopfi, which decomposes petroleum and other hydrocarbons. Marine algae (cyanobacteria, green algae and diatoms) are capable of metabolizing naphtalene into a series of metabolites [37]. The metabolic route of decomposition of hydrocarbons of the genus Oscillatoria and Agmenellum spp is similar to that of the fungi. In addition, algae possess an important potential in the consortia with bacteria, for example: Pseudomonas migulae and Sphingomonas yanoikuyae that decompose phenanthrene. Microalgae S. costatum and Nitzschia sp. can accumulate and decompose two polycyclic hydrocarbons simultaneously [38]. |
| Protozoa: The contribution of protozoa in bioremediation of hydrocarbons is considered controversial since it does not have the capacity to use them directly as an energy source: protozoa are considered to have a negative effect, due to the fact that they feed off the bacterium that contributes to decomposing hydrocarbons. However, other investigations consider that protozoa have a positive effect in the bioremediation systems, because they contribute to keep porosity in the contaminated ground by regulating bacterial count [39]. |
| Microbial consortia: In nature, microorganisms are generally found in consortia that interact synergistically, allowing a greater removal of pollutants in the ground [40,41]. When exposed to contaminated areas during long periods of time, the microbial community present in the ground adapts to the pollutants [42]. This benefits the process of bioremediation. Microbial consortia have proven to render satisfactory results in reducing contaminants in ground that was previously isolated [43]. Nevertheless, there are consortia that have been tested in extreme conditions because of its adaptability. Such is the case of the consortia formed by Acinetobacter sp., Pseudomonas sp., Ralstonia sp. and Microbacterium sp, which decompose aromatic hydrocarbons of high-molecular weight at low temperatures [44]. The enzyme that is activated for the biodegradation of the hydrocarbon depends on the microorganism and the type of hydrocarbon (Table 1) [45]. |
| Microbial metabolism for the decomposition of hydrocarbons: In the processes of bioremediation there are interactions between the enzymes that degrade hydrocarbons, which modify their structure as well as their toxicity, in aerobic or anaerobic conditions. This depends on the systems conditions and the microorganisms that develop in it [61]. |
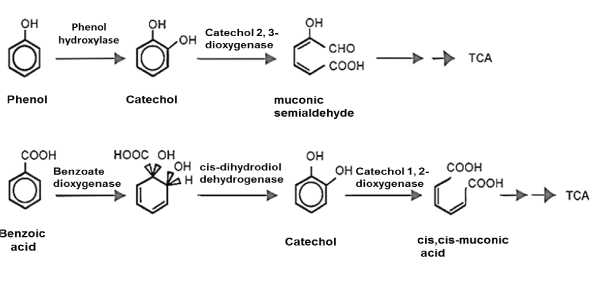
| Aerobic route: The aerobic degradation is carried out within the cytoplasmic membrane or on the cellular surface. The product of oxidation is transported inside the cell [62], where the microorganisms oxidize the carbon molecule through a multi-enzyme complex (oxygenases enzymes) that incorporates an oxygen molecule [59]. This results in a hydroxylated hydrocarbon (alcohol), a more reactive molecule. Subsequently, the alcohol group is oxidized to an aldehyde and finally to a carboxylic acid, resulting in a molecule similar to a fatty acid, which can be decomposed into an acetyl-CoA by ß-oxidation. This process of oxidation can occur in non-terminal carbons, giving place to two fatty acids [15]. Oxidase enzymes intervene in the metabolic routes of both alkanes such as aromatic hydrocarbons, by bacteria of the genus Mycobacterium, Rhodococcus and Pseudomonas, as well as by fungi of the genus Aspergillus and Fusarium [46]. In the case of polyaromatic hydrocarbons, the reaction begins with an oxidation, where two oxygenases enzyme groups participate: dioxygenases for bacteria and monoxygenases for fungi (Figure 3). In bacteria, oxygenases catalyze molecular oxygen (O2) to the substrate, forming the intermediary cis-dihydrodiol that subsequently oxides, causing the rupture of the aromatic ring into muconic acids [62]. Oxidation by fungi is done by means of the P-450 Cytochrome, catalyzed by monoxygenases enzymes, to form a furan oxide (epoxide), which experiments a rearrangement to hydrolyzed phenol or a trans dihydrodriol. |
| Phenol is transformed into sulfates or glycosides, these compounds can originate in the detoxification of other systems that are carried out by fungi of white rot, in which the peroxidase lignin enzyme of Phanerochaete chrysosporium, oxidizes the dihydroxide to form quinones, which are highly reactive compounds [46]. |
| Anaerobic route: When the contamination is not superficial, the degradation is carried out mainly in anaerobic manner, using a variety of electron acceptors (NO3-, Fe2+, SO4-, CH2) [63]. Among the microorganisms capable of degrading hydrocarbons anaerobically, the following are found: Rhodopseudomonas palustris CGA009 strain, Magnetospirillum magneticum AMB-1 strain, Azoarcus sp. EbN1 strain, Geobacter metallireducens GS-15 y Syntrophus aciditrophicus. Anaerobic degradation is a slower process than that of the aerobic. Additionally, it requires less energy, that is to say, the growth (and assimilation) is slower [64]. |
| Monochromatic compound degradation: One of the best characterized routes is by toluene, where the participation of sulfatereducing and denitrifying bacteria takes place. This also implies the addition of fumarate to toluene, followed by the activation with acetyl CoA. A benzoyl-CoA molecule is formed previously to a series of additional steps. Such molecule is susceptible to loss of aromaticity through processes of reduction, eliminating two of the three double bonds of the molecular [64]. |
| Alkane degradation has been observed in two bacteria, a sulfatereducing and a denitrifying (nitrate-reducing); the activation mechanism of the alkane implies the addition of a fumarate molecule, forming a diacid that is likely to be metabolized by means of oxidation of the fatty acids [17]. |
| It has been determined that petroleum and its products degradation is quicker in the presence of oxygen than without it, this has been demonstrated in comparative studies of cultures with presence and absence of oxygen (Table 2). Although the degraded hydrocarbon spectrum in the presence of oxygen is greater, it has been observed that benzene, toluene, xylene and ethylbenzene are degraded in anaerobic conditions, and these are more soluble in water than the aromatic hydrocarbons, which make them more accessible to degradation by anaerobic microorganisms. |
| Conclusions |
| Bioremediation is considerably beneficial to the environment if it is adequately performed. To achieve this, it is crucial to develop technologies proper to the location to be decontaminated, considering previous physicochemical and microbial characterization of the site: determining with it, the concentration, the type and toxicity of the contaminant, the impacted ground area and volume, and the nutritional and environmental conditions of the site. Subsequently, it is highly recommended to perform treatability tests in the laboratory, pilot level, to define the most appropriate treatment conditions, decide the type of process to be applied and reduce time without increasing costs for its application on a large scale. |
| When applying a bioremediation system, it is important to consider using the organisms (fungi, bacteria, yeast and plants) that have survived the ground contamination; because of their adaptability in contaminated ground, they are crucial candidates as bioremediators. Using their tolerance, defense and biodegradation mechanisms is a fast and efficient process that does not generate pollutants, is low in cost, and allows to restore hydrocarbon contaminated soils. |
| Acknowledgement |
| The authors acknowledge the financial support of provided by CONACyT to Xenia Mena Espino (Cátedra: 1665). |
References
|
Tables and Figures at a glance
| Table 1 | Table 2 |
Figures at a glance
 |
 |
 |
| Figure 1 | Figure 2 | Figure 3 |
Relevant Topics
- Anaerobic Biodegradation
- Biodegradable Balloons
- Biodegradable Confetti
- Biodegradable Diapers
- Biodegradable Plastics
- Biodegradable Sunscreen
- Biodegradation
- Bioremediation Bacteria
- Bioremediation Oil Spills
- Bioremediation Plants
- Bioremediation Products
- Ex Situ Bioremediation
- Heavy Metal Bioremediation
- In Situ Bioremediation
- Mycoremediation
- Non Biodegradable
- Phytoremediation
- Sewage Water Treatment
- Soil Bioremediation
- Types of Upwelling
- Waste Degredation
- Xenobiotics
Recommended Journals
Article Tools
Article Usage
- Total views: 18833
- [From(publication date):
March-2016 - Aug 17, 2025] - Breakdown by view type
- HTML page views : 17102
- PDF downloads : 1731
