Make the best use of Scientific Research and information from our 700+ peer reviewed, Open Access Journals that operates with the help of 50,000+ Editorial Board Members and esteemed reviewers and 1000+ Scientific associations in Medical, Clinical, Pharmaceutical, Engineering, Technology and Management Fields.
Meet Inspiring Speakers and Experts at our 3000+ Global Conferenceseries Events with over 600+ Conferences, 1200+ Symposiums and 1200+ Workshops on Medical, Pharma, Engineering, Science, Technology and Business
Mini Review Open Access
Mini-Review on the Use of Liquid Membranes in the Extraction of Platinum Group Metals from Mining and Metal Refinery Wastewaters/Side-Streams
| Francis Moyo and Roman Tandlich* | |
| Division of Pharmaceutical Chemistry, Faculty of Pharmacy, Rhodes University, P.O. Box 94, Grahamstown, 6140, South Africa | |
| Corresponding Author : | Roman Tandlich Division of Pharmaceutical Chemistry Faculty of Pharmacy, Rhodes University P.O. Box 94, Grahamstown, 6140 South Africa Tel: +27-46-603-8825 Fax: +27-46-636-1205 E-Mail: roman.tandlich@gmail.com |
| Received May 09, 2014; Accepted May 28, 2014; Published Jun 01, 2014 | |
| Citation: Moyo F, Tandlich R (2014) Mini-Review on the Use of Liquid Membranes in the Extraction of Platinum Group Metals from Mining and Metal Refinery Wastewaters/Side-Streams. J Bioremed Biodeg 5:228. doi:10.4172/2155-6199.1000228 | |
| Copyright: © 2014 Tandlich R, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Related article at Pubmed Pubmed  Scholar Google Scholar Google |
|
Visit for more related articles at Journal of Bioremediation & Biodegradation
Abstract
The current mini-review focuses on the use of liquid membranes in the platinum group metal (PGM/PGMs) extraction from various types of wastewaters to prevent environmental pollution; and for the metal recovery to address the scarcity of the PGMs in the industrial cycles. The bulk liquid membranes have been used to the extracted PGMs from the (acidic) aqueous media with recoveries of up to 96.3 ± 2.5% of the original PGM amount. The extraction time generally ranges from 2 to 24 hours. The bulk membrane liquid in the PGM extraction will depend on the covalent structure of the extractant, the feed phase PGM concentration and the complex of the PGM in question that is actually extracted from the aqueous environment. The advantages of this type of liquid membrane include its operational simplicity, but the disadvantages include limited possibility to improve the extraction performance of the system. Literature data are encouraging as they indicate that extraction of PGMs from mining and metal-refinery side-streams does not suffer from interference from metal contaminants that are commonly found in the mining and metal refinery side-streams, e.g. iron. Thus further research should focus on the application of ELM to extraction of PGMs from said wastewaters and major research drive should focus on the use of the Taylorvortex column and the non-Newtonian ELMs. With the supported liquid membranes, 78-82% of the original PGM content could be recovered from model side-streams. The selectivity of the extraction for individual PGMs can be controlled by the extractant used.
Abstract
The current mini-review focuses on the use of liquid membranes in the platinum group metal (PGM/PGMs) extraction from various types of wastewaters to prevent environmental pollution; and for the metal recovery to address the scarcity of the PGMs in the industrial cycles. The bulk liquid membranes have been used to the extracted PGMs from the (acidic) aqueous media with recoveries of up to 96.3 ± 2.5% of the original PGM amount. The extraction time generally ranges from 2 to 24 hours. The bulk membrane liquid in the PGM extraction will depend on the covalent structure of the extractant, the feed phase PGM concentration and the complex of the PGM in question that is actually extracted from the aqueous environment. The advantages of this type of liquid membrane include its operational simplicity, but the disadvantages include limited possibility to improve the extraction performance of the system. Literature data are encouraging as they indicate that extraction of PGMs from mining and metal-refinery side-streams does not suffer from interference from metal contaminants that are commonly found in the mining and metal refinery side-streams, e.g. iron. Thus further research should focus on the application of ELM to extraction of PGMs from said wastewaters and major research drive should focus on the use of the Taylor-vortex column and the non-Newtonian ELMs. With the supported liquid membranes, 78-82% of the original PGM content could be recovered from model side-streams. The selectivity of the extraction for individual PGMs can be controlled by the extractant used.
Keywords
PGMs; Demulsification; Non-Newtonian liquids; BLM; ELM; SLM; Diluent
Introduction
A membrane is a thin, film-like structure separating two adjacent phases [1]. If the rates of mass transfer through the membrane are different among the various components of a mixture, then such a membrane can be deemed semi-permeable [1]. It allows some chemical molecules to pass through but remains impermeable to others [2]. Membranes are used to separate mixtures of various components in two different phases based on variations in the mass transfer of mixture components through the membrane [3]. Advantages of using membranes in industrial chemical processes include their conceptual simplicity, cost-effective operation and their low energy consumption [1]. If the membrane-based systems are used in liquid-liquid extraction and the semipermeable membrane is liquid in nature or can be classified as an emulsion, then this gives rise to the concept of liquid membranes (LM/LMs) [1]. The LMs can be divided into the bulk liquid membranes (BLM/BLMs) [4], the emulsion liquid membranes (ELM/ELMs) [5] and the supported liquid membranes (SLM/SLMs) [6]. Different LM configurations allow for different LM application in processes such as the platinum-group metal (PGM/PGMs) recovery from ore leachates and aqueous waste side-streams [7].
The LM systems for wastewater treatment have three main components. The first one is the source/treated wastewater (so-called the feed phase or external phase). The second one is the LM itself which is made up of an organic diluent, in the case of metal extraction contains a ligand, i.e. extractant or carrier, a surfactant and sometimes a modifier [3]. Finally, the LM systems contain the receiving (stripping) phase in which the extracted metal is recovered [1,3]. The particular LM should be immiscible with the feed phase and the extractant should have low aqueous solubility [3]. The LM technology has been used in the recovery of heavy metals [1] and PGMs [4]. Thus extensive literature exists on the subject of solvents metal extraction using the LM technology. However, to date the extraction of PGMs has not been systematically reviewed and critically evaluated.
Palladium is the most studied PGM with respect to extraction from wastewater using the LM technology. Extractants that have been used in this regard include di-2-ethylhexyl-thiophosphoric acid [8], 8-quinolone derivatives [9] and Cyanex 471 [10]. Various arrangements of the LM have been used, e.g. the ELM with LIX 984N-C as the extractant [11]. The SLMs have been used to extract various PGMs from wastewater using the following extractants: Aliquat 336 for Pt (IV) [12], Pd (II) [13] and Rh (III) [14]; and MSP-8 for Pd(II) [15]. Quantitative extraction is generally achieved within 2 to 24 hours [16]. Stripping phases generally contain HCl or HNO3 and sometimes this is supplemented by thiourea [17]. The current mini-review focuses on the use of LMs in the PGM extraction from various types of wastewaters. The article will cover the metal removal from the mining and metal-refinery side-streams, wastewaters from battery production and other potential sources of environmental pollution; and metal recovery to address the scarcity of the PGMs in the industrial cycles.
The LM systems for wastewater treatment have three main components. The first one is the source/treated wastewater (so-called the feed phase or external phase). The second one is the LM itself which is made up of an organic diluent, in the case of metal extraction contains a ligand, i.e. extractant or carrier, a surfactant and sometimes a modifier [3]. Finally, the LM systems contain the receiving (stripping) phase in which the extracted metal is recovered [1,3]. The particular LM should be immiscible with the feed phase and the extractant should have low aqueous solubility [3]. The LM technology has been used in the recovery of heavy metals [1] and PGMs [4]. Thus extensive literature exists on the subject of solvents metal extraction using the LM technology. However, to date the extraction of PGMs has not been systematically reviewed and critically evaluated.
Palladium is the most studied PGM with respect to extraction from wastewater using the LM technology. Extractants that have been used in this regard include di-2-ethylhexyl-thiophosphoric acid [8], 8-quinolone derivatives [9] and Cyanex 471 [10]. Various arrangements of the LM have been used, e.g. the ELM with LIX 984N-C as the extractant [11]. The SLMs have been used to extract various PGMs from wastewater using the following extractants: Aliquat 336 for Pt (IV) [12], Pd (II) [13] and Rh (III) [14]; and MSP-8 for Pd(II) [15]. Quantitative extraction is generally achieved within 2 to 24 hours [16]. Stripping phases generally contain HCl or HNO3 and sometimes this is supplemented by thiourea [17]. The current mini-review focuses on the use of LMs in the PGM extraction from various types of wastewaters. The article will cover the metal removal from the mining and metal-refinery side-streams, wastewaters from battery production and other potential sources of environmental pollution; and metal recovery to address the scarcity of the PGMs in the industrial cycles.
BLM in the extraction of PGMs
The LM technology has been reported as a tool in the preparation of the PGM particles [18]. The role of surfactants in LM was also investigated in the PGM extraction from aqueous matrices [19]. Recoveries of up to 97% of Pd (II) were reported from model solutions with the initial metal concentration around 100 mg/L [20]. Carriers such as N2-substituted N1-phenylbenzamidines and N'-di-substituted N-benzoyl thiourea derivatives with thiocyanates and thiourea in the stripping agents have been applied in 1994 [6]. The thiocyanate anion has been shown to stabilise the extracted complex composition of Pd (II) in aqueous environments [21]. The extent of the effect is, however, dependent on the extractant which is used as demonstrated through the experimental results with triphenylphosphine sulphide and triisobutylphosphine sulphide [21]. The BLMs are the simplest type of LMs [1,3]. Generally, a diluent with extractant is placed between the feed phase and the stripping phase and the mass transfer through the BLM takes place by diffusion and/or ion-pairing [1,3]. After extraction, the loaded metal is stripped from the stripping phase in a separate step. The BLM applications in the extraction of PGMs from aqueous solutions are outlined below.
The BLM schematic representation can be found in Figure 1. The treated wastewater, i.e. the feed phase is layered above or below the BLM as function of the density ratio. The BLM is a solution of the extractant in the diluent. The extracted metal diffuses through the feed phase to the feed phase/BLM interface where it reacts with the extractant and a complex is formed. This complex then diffuses through the BLM to the BLM/stripping interface where the metal is released from the complex with the extractant and sequestered in the stripping. Sequestration leads to the maintenance of the metal activity in the stripping phase near zero, thus maintaining the metal gradient between the feed phase and the stripping phase. This in turn drives the extraction to completion.
Palladium followed first-order extraction kinetics during extraction using a BLM with bis(2-ethylhexyl)monothiophosphoric acid dissolved in EXXSOL D-80 at 303 K [22]. The extractant formed a dimer in the diluent and Pd (II) was extracted as a 1:2 complex with the extractant [22]. Chloroform was used as the diluent in a BLM containing thioridazine hydrochloride and oleic acid as extractants [23]. The extracted metal was Pd (II) which formed a stable complex with the thioridazine hydrochloride and oleic acid; and the BLM extracted the metal into the stripping phase (a NaNO2 aqueous solution) [23]. After a 120 minute extraction 96.3 ± 2.5% of the initial Pd (II) amount was recovered in the stripping phase and this value was unaffected by the presence of the following cations in the source phase extractants [23]: K+, Mg2+, Ca2+, Sr2+, Ba2+, Co2+, Ni2+, Cu2+, Zn2+, Pb2+, Cd2+, Fe3+, Cr3+, and Al3+.
Some authors have proposed and tested the use of surfactants as extractants and emulsifiers at the same time [24]. These bi-functional surfactants have been reported to lower the required concentration of surfactant in the liquid membrane [24], thus increasing the rates of mass transfer of the metal due to decrease in the LM viscosity [3]. The metal transport through the BLM is faster if a more hydrophobic surfactant is used [24]. The BLMs in the extraction of PGMs have seen reports on the use of extractants such as crown ethers [25] and the pyrimidium surfactants have strong affinity for the metal-halide complexes of PGMs [26,27]. Trioctyl amine has been used in the extraction of molybdenum [28] and other extractants such as LiX84-I for palladium [17] from wastewaters.
Alizadeh et al. [29] reported on the use of a BLM containing hexadecyl pyrimidium bromide (HDPB) in the extraction of Pd (II) from a 0.1 M HCl solution. The BLM consisted of a 0.0002 M solution of HDPB and the stripping phase was 10 ml of 4 M HBr with 0.0002 M addition of thiourea [29]. Extraction of Pd (II) was performed from the feed phase, i.e. 0.0005 M Pd (II) solution, with varying addition of NaBr [29]. The transported species of Pd was the [Pd(Br)4]2- complex and the maximum rate of extraction was obtained 0.05 M NaBr concentration in the feed phase [29]. The Pd recovery in the stripping phase was a function of the HDPB concentration in the BLM with the optimum value equal to 0.0002 M of HDPB [29]. The maximum recovery ranged from 93-95% of the initial Pd(II) amount in the feed phase, if the bromide concentration in feed phase was 0.05 M or higher [29].
If the BLM concentration of HDPB increased above 0.0002 M, then aggregation of the extractant in the chloroform BLM took place. This resulted in the decrease of the number of HDPB molecules available to reaction with [Pd(Br)4]2- and which in turn led to a drop in the recovery of Pd (II) in the stripping phase [29]. The stripping phase had to contain 4 M HBr to maintain stability of the [Pd(Br)4]2- complex and the thiourea concentration had to be kept at 0.0004 M [29]. The maximum recovery of Pd (II) was achieved after continuous extraction for 110 minutes. During these experiments, the maximum recovery on average was equal to 97.9 ± 1.1% [29]. The rate-limiting step of the extraction was the release of the Pd from the carrier complex and the diffusion of said complex through the BLM; or the complexation of Pd (II) with thiourea in the stripping phase [29].
Low hydration of Pd (II) in acidic and chloride-rich media has been listed in the literature as an important factor in the extraction of the metal [30]. Khayatian and Shamsipur [31] studied the use of a crown ether, namely using NH4+-dibenzyldiaza-18-crown-6 as carrier, for the extraction of [Pd(Cl)4]2- from 0.000088 M solution of palladium with 0.2 M NH4Cl [31]. The stripping phase was 1M KSCN in water and the BLM was made up of 20 ml of CHCl3 containing 0.01 M of the crown ether [31]. Presence of NH4+ cation is required to facilitate the fit of [Pd(Cl)4]2- into the cavity of the crown ether, i.e. it increases the stability constant of the extracted palladium species [31]. The best extraction efficiency was reached for Pd if the concentration of NH4+ was equal to 0.2 M [31]. The SCN-anion was the best stripping agent for Pd and the extraction efficiency was detrimentally affected by exchange of the isothiocyanate for EDTA and other stripping agents [31].
The extraction did not take place in the absence of the NH4+-dibenzyldiaza-18-crown-6 from the BLM [31]. The presence of dibenzyl substituents in the structure of the crown ether increased the lipophilicity of the compound and facilitated higher Pd extraction efficiencies. The extraction reached a maximum recovery of Pd in the stripping phase after 100 minutes and the average recovery of 92.2 ± 1.4% of the initial Pd amount in the feed phase [31]. Only significant interference of the [Pd(Cl)4]2- ion was observed with Hg2+ which forms similar chloro complexes as Pd under the conditions of the feed phase [31]. This will have significant influence on the extraction of PGMs from waste materials containing trace amounts of mercury, either through material origin or follow-up/environmental contamination.
Reddy et al. [16] stated that application of surfactants as extractants can be used to perform the metal extraction independently on the pH adjustment of the feed phase. A mixed-micelle BLM was prepared from trioctyl methyl ammonium chloride (TOAC) TX-100 in trichloroethylene (TCE) as diluent to perform Pt and Pd extraction from spent catalytic converters [16]. The concentrations of the components of BLM were as follows: 0.05% (w/v) TOAC and 1% (w/v) of TX-100 in TCE [16]. Solutions of Pt and Pd were prepared by dissolution of the pure metals in 10% HCl and aqua regia [16]. Extraction of metals was performed using the BLM out of the Cu/Ni matte and a spent catalytic converter with 200 mg of the solids mixed with 4 ml of aqua regia [16].
The suspensions were micro-waved for two cycles and the resulting solution was diluted to 50 ml with deionised water [16]. Five millilitres of the resulting solution was then spiked with KI (final weight fraction 0.4% w/w), 5.0% HCl and 2.5% of HNO3 (both concentrations in w/v) and finally 0.25% of TX-100 [16]. Chemical additives were dosed into the aqueous phase to stabilise the dominant metal complexes and thus speed up extraction. The final solution was then extracted with 20 ml of 0.05% solution of TOAC in TCE [16]. The extraction efficiency was comparable to the other BLM type studies on PGMs and palladium recovery. The practical problem with the application of this system would the dependence of stability of the aqueous Pd (II) solutions depending on the pH and chemical composition of the feed phase [7].
One of the practical problems in the solvent extraction of PGMs is its slow kinetics due to limited by the interfacial area, but the BLM technology has found use in testing the potential and extraction specificity of newly synthesized extractants [4,32]. Covalent structure of the calix [4] arene group of ligands facilitates the structural arrangement that allow for selectivity of the ligand to form complexes with high stability constants with a wide variety of metals [33,34]. Akin et al. [4] examined the efficiency of three calix [4] arenes as extractants for Pd (II) from model wastewaters. During the treatment of the model treated wastewater, 10 mL of 0.001 M solution of Pd (II) with 1 M KCl [4]. On the other hand, the stripping phase consisted of 10 mL of 1 M HCl and the BLM was a solution of calix [4] arene with a concentration of 0.001 M in CH2Cl2 [4]. Optimum feed phase pH for extraction is 2.0, or inside the range 1.0-4.0 [4]. The optimum concentration of calixarenes inside the BLM is 0.001 M [4]. Extraction percentage increased with extraction time and reached a maximum of 82% after 24 hours [4]. Mechanism of extraction is based on the ion pair formation and complexation [4].
The facts mentioned in section 2 demonstrate the use potential of the BLM technology in the extraction of the PGMs. The efficiency will depend on the covalent structure of the extractant, the feed phase PGM concentration and the complex of the PGM in question that is actually extracted from the aqueous environment. Advantages of the BLM in PGM extraction include the operational simplicity, in line with the general properties of the LM systems. The limited nature for making changes to the design is, however, the major problem with the BLM in the PGM metal/solvent in extraction. This is addressed by the use of the ELMs and the SLMs which are described in the next two sections of this mini-review.
The BLM schematic representation can be found in Figure 1. The treated wastewater, i.e. the feed phase is layered above or below the BLM as function of the density ratio. The BLM is a solution of the extractant in the diluent. The extracted metal diffuses through the feed phase to the feed phase/BLM interface where it reacts with the extractant and a complex is formed. This complex then diffuses through the BLM to the BLM/stripping interface where the metal is released from the complex with the extractant and sequestered in the stripping. Sequestration leads to the maintenance of the metal activity in the stripping phase near zero, thus maintaining the metal gradient between the feed phase and the stripping phase. This in turn drives the extraction to completion.
Palladium followed first-order extraction kinetics during extraction using a BLM with bis(2-ethylhexyl)monothiophosphoric acid dissolved in EXXSOL D-80 at 303 K [22]. The extractant formed a dimer in the diluent and Pd (II) was extracted as a 1:2 complex with the extractant [22]. Chloroform was used as the diluent in a BLM containing thioridazine hydrochloride and oleic acid as extractants [23]. The extracted metal was Pd (II) which formed a stable complex with the thioridazine hydrochloride and oleic acid; and the BLM extracted the metal into the stripping phase (a NaNO2 aqueous solution) [23]. After a 120 minute extraction 96.3 ± 2.5% of the initial Pd (II) amount was recovered in the stripping phase and this value was unaffected by the presence of the following cations in the source phase extractants [23]: K+, Mg2+, Ca2+, Sr2+, Ba2+, Co2+, Ni2+, Cu2+, Zn2+, Pb2+, Cd2+, Fe3+, Cr3+, and Al3+.
Some authors have proposed and tested the use of surfactants as extractants and emulsifiers at the same time [24]. These bi-functional surfactants have been reported to lower the required concentration of surfactant in the liquid membrane [24], thus increasing the rates of mass transfer of the metal due to decrease in the LM viscosity [3]. The metal transport through the BLM is faster if a more hydrophobic surfactant is used [24]. The BLMs in the extraction of PGMs have seen reports on the use of extractants such as crown ethers [25] and the pyrimidium surfactants have strong affinity for the metal-halide complexes of PGMs [26,27]. Trioctyl amine has been used in the extraction of molybdenum [28] and other extractants such as LiX84-I for palladium [17] from wastewaters.
Alizadeh et al. [29] reported on the use of a BLM containing hexadecyl pyrimidium bromide (HDPB) in the extraction of Pd (II) from a 0.1 M HCl solution. The BLM consisted of a 0.0002 M solution of HDPB and the stripping phase was 10 ml of 4 M HBr with 0.0002 M addition of thiourea [29]. Extraction of Pd (II) was performed from the feed phase, i.e. 0.0005 M Pd (II) solution, with varying addition of NaBr [29]. The transported species of Pd was the [Pd(Br)4]2- complex and the maximum rate of extraction was obtained 0.05 M NaBr concentration in the feed phase [29]. The Pd recovery in the stripping phase was a function of the HDPB concentration in the BLM with the optimum value equal to 0.0002 M of HDPB [29]. The maximum recovery ranged from 93-95% of the initial Pd(II) amount in the feed phase, if the bromide concentration in feed phase was 0.05 M or higher [29].
If the BLM concentration of HDPB increased above 0.0002 M, then aggregation of the extractant in the chloroform BLM took place. This resulted in the decrease of the number of HDPB molecules available to reaction with [Pd(Br)4]2- and which in turn led to a drop in the recovery of Pd (II) in the stripping phase [29]. The stripping phase had to contain 4 M HBr to maintain stability of the [Pd(Br)4]2- complex and the thiourea concentration had to be kept at 0.0004 M [29]. The maximum recovery of Pd (II) was achieved after continuous extraction for 110 minutes. During these experiments, the maximum recovery on average was equal to 97.9 ± 1.1% [29]. The rate-limiting step of the extraction was the release of the Pd from the carrier complex and the diffusion of said complex through the BLM; or the complexation of Pd (II) with thiourea in the stripping phase [29].
Low hydration of Pd (II) in acidic and chloride-rich media has been listed in the literature as an important factor in the extraction of the metal [30]. Khayatian and Shamsipur [31] studied the use of a crown ether, namely using NH4+-dibenzyldiaza-18-crown-6 as carrier, for the extraction of [Pd(Cl)4]2- from 0.000088 M solution of palladium with 0.2 M NH4Cl [31]. The stripping phase was 1M KSCN in water and the BLM was made up of 20 ml of CHCl3 containing 0.01 M of the crown ether [31]. Presence of NH4+ cation is required to facilitate the fit of [Pd(Cl)4]2- into the cavity of the crown ether, i.e. it increases the stability constant of the extracted palladium species [31]. The best extraction efficiency was reached for Pd if the concentration of NH4+ was equal to 0.2 M [31]. The SCN-anion was the best stripping agent for Pd and the extraction efficiency was detrimentally affected by exchange of the isothiocyanate for EDTA and other stripping agents [31].
The extraction did not take place in the absence of the NH4+-dibenzyldiaza-18-crown-6 from the BLM [31]. The presence of dibenzyl substituents in the structure of the crown ether increased the lipophilicity of the compound and facilitated higher Pd extraction efficiencies. The extraction reached a maximum recovery of Pd in the stripping phase after 100 minutes and the average recovery of 92.2 ± 1.4% of the initial Pd amount in the feed phase [31]. Only significant interference of the [Pd(Cl)4]2- ion was observed with Hg2+ which forms similar chloro complexes as Pd under the conditions of the feed phase [31]. This will have significant influence on the extraction of PGMs from waste materials containing trace amounts of mercury, either through material origin or follow-up/environmental contamination.
Reddy et al. [16] stated that application of surfactants as extractants can be used to perform the metal extraction independently on the pH adjustment of the feed phase. A mixed-micelle BLM was prepared from trioctyl methyl ammonium chloride (TOAC) TX-100 in trichloroethylene (TCE) as diluent to perform Pt and Pd extraction from spent catalytic converters [16]. The concentrations of the components of BLM were as follows: 0.05% (w/v) TOAC and 1% (w/v) of TX-100 in TCE [16]. Solutions of Pt and Pd were prepared by dissolution of the pure metals in 10% HCl and aqua regia [16]. Extraction of metals was performed using the BLM out of the Cu/Ni matte and a spent catalytic converter with 200 mg of the solids mixed with 4 ml of aqua regia [16].
The suspensions were micro-waved for two cycles and the resulting solution was diluted to 50 ml with deionised water [16]. Five millilitres of the resulting solution was then spiked with KI (final weight fraction 0.4% w/w), 5.0% HCl and 2.5% of HNO3 (both concentrations in w/v) and finally 0.25% of TX-100 [16]. Chemical additives were dosed into the aqueous phase to stabilise the dominant metal complexes and thus speed up extraction. The final solution was then extracted with 20 ml of 0.05% solution of TOAC in TCE [16]. The extraction efficiency was comparable to the other BLM type studies on PGMs and palladium recovery. The practical problem with the application of this system would the dependence of stability of the aqueous Pd (II) solutions depending on the pH and chemical composition of the feed phase [7].
One of the practical problems in the solvent extraction of PGMs is its slow kinetics due to limited by the interfacial area, but the BLM technology has found use in testing the potential and extraction specificity of newly synthesized extractants [4,32]. Covalent structure of the calix [4] arene group of ligands facilitates the structural arrangement that allow for selectivity of the ligand to form complexes with high stability constants with a wide variety of metals [33,34]. Akin et al. [4] examined the efficiency of three calix [4] arenes as extractants for Pd (II) from model wastewaters. During the treatment of the model treated wastewater, 10 mL of 0.001 M solution of Pd (II) with 1 M KCl [4]. On the other hand, the stripping phase consisted of 10 mL of 1 M HCl and the BLM was a solution of calix [4] arene with a concentration of 0.001 M in CH2Cl2 [4]. Optimum feed phase pH for extraction is 2.0, or inside the range 1.0-4.0 [4]. The optimum concentration of calixarenes inside the BLM is 0.001 M [4]. Extraction percentage increased with extraction time and reached a maximum of 82% after 24 hours [4]. Mechanism of extraction is based on the ion pair formation and complexation [4].
The facts mentioned in section 2 demonstrate the use potential of the BLM technology in the extraction of the PGMs. The efficiency will depend on the covalent structure of the extractant, the feed phase PGM concentration and the complex of the PGM in question that is actually extracted from the aqueous environment. Advantages of the BLM in PGM extraction include the operational simplicity, in line with the general properties of the LM systems. The limited nature for making changes to the design is, however, the major problem with the BLM in the PGM metal/solvent in extraction. This is addressed by the use of the ELMs and the SLMs which are described in the next two sections of this mini-review.
ELMs in the extraction of PGMs
An emulsion is a dispersion consisting of at least two immiscible phases, one of which is finely subdivided and uniformly dispersed as globules (dispersed phase) throughout the other continuous liquid phase [35]. This is achieved using mechanical agitation of the two immiscible liquids. Usually one liquid in an emulsion is polar/aqueous, while the other is relatively non-polar (e.g. an organic solvent phase otherwise known as diluent). The liquid usually used is water to which water-immiscible organic solvents are added. Emulsions are thermodynamically unstable and thus a third component, the emulsifying agent (surfactant), is required to stabilize the emulsion [35,36]. In the oil-in-water emulsion (o/w), the oil (organic) phase/diluent is dispersed as globules throughout an aqueous continuous phase, while in the water in oil (w/o) emulsions; the oil/diluent serves as the continuous phase with water being the disperse phase. An ELM results from combining the concepts of the LMs with that of emulsions.
The ELMs separation technique was invented by Li [37-39]. An ELM is a three-phase dispersion system [40], with the general composition of a LM [38]. The schematic representation of the ELM globule immersed in the respective feed phase and can be modeled by the shrinking core model depicted in figure 2 [41,42]. The extracted metal moves through the feed phase towards the interface with the ELM, where it becomes complexed with the extractant on the diluent side of the feed/ELM interface. The required number of H+ is released from the ELM extractant into the feed phase to maintain electroneutrality of the system [1].
The metal/extractant complex then diffuses through the diluent towards the stripping phase microdroplets (see space between the circumference of the ELM globule and the dashed line, seconds from the outside of the globule of the ELM in Figure 2) [41,42]. At the diluent/stripping phase interface, the metal ion is released into the inner droplet, and the required number of H+ protons is bound to the extractant molecule to maintain electroneutrality of the diluent pat of the ELM. The closer the particular stripping phase microdroplet is towards the diluent/feed phase interface, the more likely and the faster the extracted PGM will get sequestered in it. There is always a reactive front (see the dark globules adjacent to the dashed line in Figure 2). Once the layer closest to the feed phase/diluent interface becomes depleted of protons, no more metal ions can be extracted from the complex. The complex molecule diffuses further inside the ELM globules as depicted by the open globules adjacent to the dashed line in Figure 2.
In comparison with BLM and SLM, the ELM membrane phase contains the extractant and the surfactant to stabilize the emulsion and stripping phase droplets dispersed in it [40]. During the extraction process, an ELM is then dispersed in the external/feed phase by (mechanical) agitation. ELMs have been applied in the fields of environmental and pharmaceutical engineering, hydrometallurgy and the food industry [39]. The ELM technique has been successfully applied to extraction of heavy metals, organic acids, amines and phenols from industrial, mining and metal refinery wastewaters [37,38,40]. Matrices and metals that have been treated and recovered using the ELMs technology include radioactive wastes and the rare earth metals [43]. On the other hand, up to 98% of lanthanides was recovered from model wastewater after 5 minutes of extraction, if the volume ratio of ELM to wastewater was equal to 1 to 32 [44].
For palladium extraction, an ELM with the N7301 extractant (a mixture of tertiary amines) with the alkyl chains ranging from 8 to 12 carbon atoms with the average molecular weight 400 g/mol was prepared [45]. SPAN 80, i.e. sorbitan monooleate, was used as the surfactant and kerosene as the diluent, while an acidic aqueous solution of EDTA was used as the stripping phase [45]. The system provided a 96% recovery of palladium in the stripping phase, with interference from the presence of Zn2+, Cu2+, Cd2+, Pb2+, Ni2+ and Fe2+ [45]. This observation could be explained by the complex chemistry of palladium in aqueous environments, i.e. presence in the form of negatively charged chloro-complexes [7]. Thus the extraction of Pd using the ELM technology is likely to be affected by the similar chloro-complex-forming metals, i.e. the other members of the PGMs group [7]. However, the results are encouraging for practical applications of the ELM extraction on the large scale as the efficiency of the process is unaffected by common contaminant in the metal refinery side-streams such as iron. Thus practical applications of the ELM will be dependent on the selectivity of the actual extraction protocol used (see below).
The ELM literature for the extraction of PGMs is limited to date. In comparison with BLMs and SLMs, this type of LM can be applied to existing contactors already installed and routinely used in the solvent extraction of metals in the PGM refineries. Thus in the next part of this section, the general properties of the ELMs are reviewed in detail and possibilities for extension of their practical application in PGM extraction and processing are outlined. At first, the general properties and types of ELMs are reviewed and this is followed by the individual steps in the extraction processes. Some of the outstanding research questions and less commonly reviewed types of extraction systems are presented, together with the existing and possible future applications in the PGM extraction and recovery from waste side-streams in the metal-refinery and mining operations.
The ELMs separation technique was invented by Li [37-39]. An ELM is a three-phase dispersion system [40], with the general composition of a LM [38]. The schematic representation of the ELM globule immersed in the respective feed phase and can be modeled by the shrinking core model depicted in figure 2 [41,42]. The extracted metal moves through the feed phase towards the interface with the ELM, where it becomes complexed with the extractant on the diluent side of the feed/ELM interface. The required number of H+ is released from the ELM extractant into the feed phase to maintain electroneutrality of the system [1].
The metal/extractant complex then diffuses through the diluent towards the stripping phase microdroplets (see space between the circumference of the ELM globule and the dashed line, seconds from the outside of the globule of the ELM in Figure 2) [41,42]. At the diluent/stripping phase interface, the metal ion is released into the inner droplet, and the required number of H+ protons is bound to the extractant molecule to maintain electroneutrality of the diluent pat of the ELM. The closer the particular stripping phase microdroplet is towards the diluent/feed phase interface, the more likely and the faster the extracted PGM will get sequestered in it. There is always a reactive front (see the dark globules adjacent to the dashed line in Figure 2). Once the layer closest to the feed phase/diluent interface becomes depleted of protons, no more metal ions can be extracted from the complex. The complex molecule diffuses further inside the ELM globules as depicted by the open globules adjacent to the dashed line in Figure 2.
In comparison with BLM and SLM, the ELM membrane phase contains the extractant and the surfactant to stabilize the emulsion and stripping phase droplets dispersed in it [40]. During the extraction process, an ELM is then dispersed in the external/feed phase by (mechanical) agitation. ELMs have been applied in the fields of environmental and pharmaceutical engineering, hydrometallurgy and the food industry [39]. The ELM technique has been successfully applied to extraction of heavy metals, organic acids, amines and phenols from industrial, mining and metal refinery wastewaters [37,38,40]. Matrices and metals that have been treated and recovered using the ELMs technology include radioactive wastes and the rare earth metals [43]. On the other hand, up to 98% of lanthanides was recovered from model wastewater after 5 minutes of extraction, if the volume ratio of ELM to wastewater was equal to 1 to 32 [44].
For palladium extraction, an ELM with the N7301 extractant (a mixture of tertiary amines) with the alkyl chains ranging from 8 to 12 carbon atoms with the average molecular weight 400 g/mol was prepared [45]. SPAN 80, i.e. sorbitan monooleate, was used as the surfactant and kerosene as the diluent, while an acidic aqueous solution of EDTA was used as the stripping phase [45]. The system provided a 96% recovery of palladium in the stripping phase, with interference from the presence of Zn2+, Cu2+, Cd2+, Pb2+, Ni2+ and Fe2+ [45]. This observation could be explained by the complex chemistry of palladium in aqueous environments, i.e. presence in the form of negatively charged chloro-complexes [7]. Thus the extraction of Pd using the ELM technology is likely to be affected by the similar chloro-complex-forming metals, i.e. the other members of the PGMs group [7]. However, the results are encouraging for practical applications of the ELM extraction on the large scale as the efficiency of the process is unaffected by common contaminant in the metal refinery side-streams such as iron. Thus practical applications of the ELM will be dependent on the selectivity of the actual extraction protocol used (see below).
The ELM literature for the extraction of PGMs is limited to date. In comparison with BLMs and SLMs, this type of LM can be applied to existing contactors already installed and routinely used in the solvent extraction of metals in the PGM refineries. Thus in the next part of this section, the general properties of the ELMs are reviewed in detail and possibilities for extension of their practical application in PGM extraction and processing are outlined. At first, the general properties and types of ELMs are reviewed and this is followed by the individual steps in the extraction processes. Some of the outstanding research questions and less commonly reviewed types of extraction systems are presented, together with the existing and possible future applications in the PGM extraction and recovery from waste side-streams in the metal-refinery and mining operations.
Types of ELM systems
Depending of the composition of the ELM and the mass transfer mechanism during extraction, ELMs can be classified into two main groups, namely the type 1 systems and the type 2 systems [46]. Thermodynamic driving force of the metal mass transfer from the treated wastewater into the ELMs is the difference between the metal’s chemical potential in the feed phase and the stripping phase of the ELM [47]. In the ELM system 1, the extracted metal is soluble in all three phases and thus no extractant addition is required. The concentration of the extracted metal and its chemical potential are higher in the feed phase than in the stripping phase [3]. Diffusion of the metal along the concentration gradient through the diluent into the stripping phase is easy during the extraction process [3]. Once in the stripping phase, the metal ions are sequestered by complexation or pH change with respect to the feed phase [3]. This maintains the chemical potential of the extracted metal near zero inside the stripping phase, which in turn maintains a high gradient of the metal’s chemical potential between the feed phase and the stripping phase [3], facilitating continuation of the metal extraction [46,48,49].
If an ELM is a type 2 extraction system, the metal which is being extracted from the feed phase, namely the solute being studied, is insoluble in the diluent phase of the ELM [50]. This results in the need for a carrier/extractant to be dissolved in the diluent phase of the ELM. The extractant dissolution then facilitates the metal movement across the membrane into the stripping phase [50]. The first reaction occurs at the feed/diluent interphase, where the solute reacts with the extractant undergoes the formation of a coordination covalent bond with the extracted metal, i.e. the solute/extractant complex is formed [3]. This complex diffuses through the diluent phase and reaches the diluent/stripping interface where the extracted metal dissociates from the carrier complex [3]. Subsequently, the stripped metal enters the stripping phase and becomes sequestered. Thus the extractant is regenerated to be reused to form a coordination covalent bond with another metal ion [50]. More details can be found by the reader in the PhD thesis of Park [50], the book chapter by Tandlich [3] and the book edited by Kislik [1].
The most common ELM carriers can be divided into three classes. The first class contains the proton acceptors such as the secondary and tertiary amines; and their derivatives [51]. The second class contains organic acids and their derivatives [40]. The third class is extractants with solvating and electron donor or acceptor properties [52]. Up to date, all ELM application with PGMs were the type 2 systems Prapruike et al. [11] used an ELM with Exxol™ D80 as diluent, SPAN 80 as surfactant and LIX 984N-C as the extractant [11]. The ultra-fine PGM particles were prepared a type 2 ELM [18]. Use of a mixture of tertiary amines in kerosene with SPAN 80 as the surfactant was also applied to the palladium extraction by Wang et al. [45]. As most of the PGM are extracted from the aqueous solutions with highly acidic pH, the extracted species of the metal is generally some sort of a chloro or halide-anion complex [7]. Some PGMs have also been reported to have limited hydration in aqueous environments. This can lead to the requirement for the extractant presence in the ELM, where the halide-anion complexes form coordination covalent bonds or ion pairs with the extractant.
The carrier properties influence the ELM performance as such these compounds should be highly selective to the extracted metal, i.e. the stability constant of the extractant/carrier-metal complex should be high in value [53]. This enables the carrier to extract the solute from the feed into the liquid membrane phase at the feed-LM interface [53]. Breaking of the coordination covalent bond of the extracted metal from the carrier-solute complex at the ELM-stripping phase interface and the metal sequestration into the stripping phase should facilitate the regeneration of the carrier and enable the continuity of the metal extraction. At the same time, the diffusion kinetics of the metal extractant complex through the diluent should be rapid for successful extraction. There should not be formation of any covalent bonds between the carrier and the diluent or any other components of the ELM; and the carrier should have suitable viscosity, density and surface tension. It should not react with other components of the LM [53].
If an ELM is a type 2 extraction system, the metal which is being extracted from the feed phase, namely the solute being studied, is insoluble in the diluent phase of the ELM [50]. This results in the need for a carrier/extractant to be dissolved in the diluent phase of the ELM. The extractant dissolution then facilitates the metal movement across the membrane into the stripping phase [50]. The first reaction occurs at the feed/diluent interphase, where the solute reacts with the extractant undergoes the formation of a coordination covalent bond with the extracted metal, i.e. the solute/extractant complex is formed [3]. This complex diffuses through the diluent phase and reaches the diluent/stripping interface where the extracted metal dissociates from the carrier complex [3]. Subsequently, the stripped metal enters the stripping phase and becomes sequestered. Thus the extractant is regenerated to be reused to form a coordination covalent bond with another metal ion [50]. More details can be found by the reader in the PhD thesis of Park [50], the book chapter by Tandlich [3] and the book edited by Kislik [1].
The most common ELM carriers can be divided into three classes. The first class contains the proton acceptors such as the secondary and tertiary amines; and their derivatives [51]. The second class contains organic acids and their derivatives [40]. The third class is extractants with solvating and electron donor or acceptor properties [52]. Up to date, all ELM application with PGMs were the type 2 systems Prapruike et al. [11] used an ELM with Exxol™ D80 as diluent, SPAN 80 as surfactant and LIX 984N-C as the extractant [11]. The ultra-fine PGM particles were prepared a type 2 ELM [18]. Use of a mixture of tertiary amines in kerosene with SPAN 80 as the surfactant was also applied to the palladium extraction by Wang et al. [45]. As most of the PGM are extracted from the aqueous solutions with highly acidic pH, the extracted species of the metal is generally some sort of a chloro or halide-anion complex [7]. Some PGMs have also been reported to have limited hydration in aqueous environments. This can lead to the requirement for the extractant presence in the ELM, where the halide-anion complexes form coordination covalent bonds or ion pairs with the extractant.
The carrier properties influence the ELM performance as such these compounds should be highly selective to the extracted metal, i.e. the stability constant of the extractant/carrier-metal complex should be high in value [53]. This enables the carrier to extract the solute from the feed into the liquid membrane phase at the feed-LM interface [53]. Breaking of the coordination covalent bond of the extracted metal from the carrier-solute complex at the ELM-stripping phase interface and the metal sequestration into the stripping phase should facilitate the regeneration of the carrier and enable the continuity of the metal extraction. At the same time, the diffusion kinetics of the metal extractant complex through the diluent should be rapid for successful extraction. There should not be formation of any covalent bonds between the carrier and the diluent or any other components of the ELM; and the carrier should have suitable viscosity, density and surface tension. It should not react with other components of the LM [53].
The ELM extraction process
There are four main steps involved in the ELM extraction process: emulsion preparation, the metal extraction, emulsion separation and demulsification. The properties of the extractant, diluent, surfactant and the extracted metal will determine the ELM extraction efficiency. Diluent should have [47]: (a) low solubility in the feed and stripping phases to avoid losses during emulsion preparation and solute extraction; (b) compatibility with surfactant and the extractant dissolved; (c) moderate viscosity balancing the membrane stability and permeability; (d) enough density difference from the aqueous phase for the fast process of emulsion separation; (e) low cost of production and (f) low toxicity because of environmental concerns. The extractant should [50]: (a) thermodynamically favour the formation of the extractant/solute complex from the feed phase; (b) react fast enough to form this complex for fast solute extraction and (c) should easily dissociate at the stripping phase.
Emulsification of two immiscible phases is the actual preparation of the ELM. It can be achieved through the use of high pressure homogenisation, rotor stator devices, mechanical agitation, ultrasonic and static mixer techniques [54]. During emulsification, the stripping phase is introduced to the membrane phase prepared. The stripping phase contains a stripping agent which is thermodynamically favourable for the stripping process and exhibits a fast reaction with a solute-extractant complex for successful stripping [50]. The microdroplet diameter is also important for high extraction efficiency and high stability of the membrane [55]. Large microdroplet diameters produce poor extraction efficiency, resulting in poor membrane stability [56] due to microdroplet rupture and coalescence [37]. For good stability and rapid extraction, the ELM microdroplets should be in the range of 0.3-10 µm, but preferably between 0.8 and 3 µm [46,56]. Smaller droplets have higher extraction efficiency as well as a better breaking resistance compared to larger droplets [54]. Praipruke et l. [11] used a drip-wise addition of HCl as the stripping phase to the solution of LIX 984N-C in Exxol™ D80 with addition of SPAN 80; and the emulsification was conducted in a high-speed homogeniser at 12000 rpm for 30 minutes.
The metal extraction process commences by contacting the prepared ELM with the feed phase. These emulsions are mixed or agitated in the mixing reactor, leading to the dispersion of the emulsion form globules of 0.1-3 mm in diameter [46,57]. This is sufficient to form the required size globules for efficient extraction but should be low enough to reduce shear rate and shear stress, the main causes of emulsion instability leading to emulsion rapture and reducing the extraction efficiency. Therefore optimum speed of agitation is required in this stage. Agitation is stopped after solute extraction and the system is left to achieve phase separation. The emulsion separation/setting from the treated feed phase is governed by the density [50]. Times of 40 to 90 minutes have been reported to for extraction equilibrium, i.e. maximum recovery of PGMs [11,18]. After this process demulsification of the metal loaded stripping phase begins.
Demulsification means that the stripping phase is separated from the membrane phase through coagulation and coalescence of microdroplets, or destabilization of the emulsion [58-60]. This unit operation in the ELM process can be based physical and/or chemical treatment. The efficiency of demulsification dictates metal recovery and it can be affected by the physical characteristic of the stripping phase, namely viscosity of the ELM, microdroplet size and concentration of the surfactant [58-62]. In chemical demulsification, studies have shown that non-ionic surfactants such as Tweens and surfactants characterized by a high number of hydrophilic groups have also shown to be good demulsifiers [61,62]. The hydrophilic groups lead to the formation of hydrogen bonds with water in the stripping phase hence leading to demulsification [63]. The addition of the demulsifier prevents the reuse of the ELM in another extraction [50].
Various studies have shown that successful metal recovery with chemical demulsification [63-65]. Sometimes combinations of chemical and physical approaches are necessary for successful demulsification to take place [59]. The physical approach involves heat, centrifugation, porous glass, ultrasonication, electrostatic enhancement and microwave radiation [50,66]. Studies have shown that W/O emulsions are broken using these methods [58,67-69]. Praipruke et al. [11] used simple phase separation to recover the ELM extracted Pd (II). More research is required on the recovery of PGMs from the loaded ELMs as the interfacial area increase by using ELMs in comparison to the classical solvent extraction could provide faster extraction kinetics and more economical process [70].
The main problems in the stability of ELMs are swelling and leakage of the stripping phase into the feed phase or the diluent [37,55]. Stability of the membrane is defined as its ability to resist leakage or rupture in the process of solute extraction in the contactor [71]. Leakage is normally due to the high shear stress generated in the process of solute extraction [71]. Together with ELM swelling, it is the most serious problem with the use of the ELM in metal extraction from the treated side-stream or wastewater [72-74]. Swelling occurs when the interface between feed phase and the ELM diluent is compromised. Resulting drawbacks are [39,71]: (a) the reduction of membrane thickness; (b) viscosity of the ELM is compromised, impeding the extraction process from the feed phase and (c) it leads to the dilution of the final concentration of the solute. The actual swelling can be caused by one or more of the following factors [39,55]: pH, temperature, ionic strength of the feed and stripping phases, diluent and water volumetric ratio in the ELM, properties of diluents, surfactants and extractants, microdroplet size, entrainment and osmosis.
In entrainment swelling, the feed phase is drawn into the stripping phase as a result of coalescence of emulsion globules during the extraction process [39]. Osmotic swelling may be caused by the surfactant movement in the ELM [75]. Hydration of the surfactant takes place at the feed phase/diluent interface, where the hydrophilic head of the surfactant forms a complex with a water molecule [47]. The other process by which osmotic swelling occurs in the liquid membranes is the formation of the reversed micelles [50]. In the reversed micelle formation the hydrophilic heads of the surfactants are attracted by the aqueous core and they face each other or aggregate creating a hydrophilic core, while the hydrophobic tails of the surfactant are attracted to the diluent part of the ELM membrane [76,77].
Water is easily solubilised in the hydrophilic core and hence the reversed micelles can increase the size of the stripping phase microdroplets [47,78]. Swelling, coalescence and leakage lead to the reduced extraction efficiency for metals [79]. The ELM microdroplet stability also depends on the agitation shear, concentrations of the surfactant and extractant; and occlusion [37,80]. Swelling can be reduced by addition of hydrocarbons and cyclohexanone to the ELM [81]. The information on swelling of the ELM during PGM recovery is limited and needs to be investigated further in literature. Due to this fact, it can be similar to other metals as outlined by previous authors [1,3,50]. The article continues with coverage of the experimental measures which can be taken to prevent microdroplet swelling and coalescence.
Emulsification of two immiscible phases is the actual preparation of the ELM. It can be achieved through the use of high pressure homogenisation, rotor stator devices, mechanical agitation, ultrasonic and static mixer techniques [54]. During emulsification, the stripping phase is introduced to the membrane phase prepared. The stripping phase contains a stripping agent which is thermodynamically favourable for the stripping process and exhibits a fast reaction with a solute-extractant complex for successful stripping [50]. The microdroplet diameter is also important for high extraction efficiency and high stability of the membrane [55]. Large microdroplet diameters produce poor extraction efficiency, resulting in poor membrane stability [56] due to microdroplet rupture and coalescence [37]. For good stability and rapid extraction, the ELM microdroplets should be in the range of 0.3-10 µm, but preferably between 0.8 and 3 µm [46,56]. Smaller droplets have higher extraction efficiency as well as a better breaking resistance compared to larger droplets [54]. Praipruke et l. [11] used a drip-wise addition of HCl as the stripping phase to the solution of LIX 984N-C in Exxol™ D80 with addition of SPAN 80; and the emulsification was conducted in a high-speed homogeniser at 12000 rpm for 30 minutes.
The metal extraction process commences by contacting the prepared ELM with the feed phase. These emulsions are mixed or agitated in the mixing reactor, leading to the dispersion of the emulsion form globules of 0.1-3 mm in diameter [46,57]. This is sufficient to form the required size globules for efficient extraction but should be low enough to reduce shear rate and shear stress, the main causes of emulsion instability leading to emulsion rapture and reducing the extraction efficiency. Therefore optimum speed of agitation is required in this stage. Agitation is stopped after solute extraction and the system is left to achieve phase separation. The emulsion separation/setting from the treated feed phase is governed by the density [50]. Times of 40 to 90 minutes have been reported to for extraction equilibrium, i.e. maximum recovery of PGMs [11,18]. After this process demulsification of the metal loaded stripping phase begins.
Demulsification means that the stripping phase is separated from the membrane phase through coagulation and coalescence of microdroplets, or destabilization of the emulsion [58-60]. This unit operation in the ELM process can be based physical and/or chemical treatment. The efficiency of demulsification dictates metal recovery and it can be affected by the physical characteristic of the stripping phase, namely viscosity of the ELM, microdroplet size and concentration of the surfactant [58-62]. In chemical demulsification, studies have shown that non-ionic surfactants such as Tweens and surfactants characterized by a high number of hydrophilic groups have also shown to be good demulsifiers [61,62]. The hydrophilic groups lead to the formation of hydrogen bonds with water in the stripping phase hence leading to demulsification [63]. The addition of the demulsifier prevents the reuse of the ELM in another extraction [50].
Various studies have shown that successful metal recovery with chemical demulsification [63-65]. Sometimes combinations of chemical and physical approaches are necessary for successful demulsification to take place [59]. The physical approach involves heat, centrifugation, porous glass, ultrasonication, electrostatic enhancement and microwave radiation [50,66]. Studies have shown that W/O emulsions are broken using these methods [58,67-69]. Praipruke et al. [11] used simple phase separation to recover the ELM extracted Pd (II). More research is required on the recovery of PGMs from the loaded ELMs as the interfacial area increase by using ELMs in comparison to the classical solvent extraction could provide faster extraction kinetics and more economical process [70].
The main problems in the stability of ELMs are swelling and leakage of the stripping phase into the feed phase or the diluent [37,55]. Stability of the membrane is defined as its ability to resist leakage or rupture in the process of solute extraction in the contactor [71]. Leakage is normally due to the high shear stress generated in the process of solute extraction [71]. Together with ELM swelling, it is the most serious problem with the use of the ELM in metal extraction from the treated side-stream or wastewater [72-74]. Swelling occurs when the interface between feed phase and the ELM diluent is compromised. Resulting drawbacks are [39,71]: (a) the reduction of membrane thickness; (b) viscosity of the ELM is compromised, impeding the extraction process from the feed phase and (c) it leads to the dilution of the final concentration of the solute. The actual swelling can be caused by one or more of the following factors [39,55]: pH, temperature, ionic strength of the feed and stripping phases, diluent and water volumetric ratio in the ELM, properties of diluents, surfactants and extractants, microdroplet size, entrainment and osmosis.
In entrainment swelling, the feed phase is drawn into the stripping phase as a result of coalescence of emulsion globules during the extraction process [39]. Osmotic swelling may be caused by the surfactant movement in the ELM [75]. Hydration of the surfactant takes place at the feed phase/diluent interface, where the hydrophilic head of the surfactant forms a complex with a water molecule [47]. The other process by which osmotic swelling occurs in the liquid membranes is the formation of the reversed micelles [50]. In the reversed micelle formation the hydrophilic heads of the surfactants are attracted by the aqueous core and they face each other or aggregate creating a hydrophilic core, while the hydrophobic tails of the surfactant are attracted to the diluent part of the ELM membrane [76,77].
Water is easily solubilised in the hydrophilic core and hence the reversed micelles can increase the size of the stripping phase microdroplets [47,78]. Swelling, coalescence and leakage lead to the reduced extraction efficiency for metals [79]. The ELM microdroplet stability also depends on the agitation shear, concentrations of the surfactant and extractant; and occlusion [37,80]. Swelling can be reduced by addition of hydrocarbons and cyclohexanone to the ELM [81]. The information on swelling of the ELM during PGM recovery is limited and needs to be investigated further in literature. Due to this fact, it can be similar to other metals as outlined by previous authors [1,3,50]. The article continues with coverage of the experimental measures which can be taken to prevent microdroplet swelling and coalescence.
The proposed solutions of membrane instability
Stability of ELMs may be improved by increasing the concentration of surfactants [82], increasing the concentration of the extractant which results in the increase in membrane viscosity [83], the use of membrane diluent with high viscosity [84] and the use of non-Newtonian modifiers for the membrane phase. The last point has not received much attention in the research literature and previous review articles and will thus be a major part of this section of the current mini-review. The non-Newtonian liquids can be achieved by the addition of the high molecular weight polymers such as polyisobutylene into the diluent of the ELM [50]. Polymers are dissolved in the membrane phase, causing the membrane to behave like a non-Newtonian fluid i.e. a pseudo-plastic fluid [85]. Optimizations of the operational conditions such as stirring speed, composition of emulsion and temperature have been investigated in order to try and improve ELM stability [42,86,87].
Importance of surfactant and the effects of its concentration
Surfactant selection during the ELM preparation is important because the surfactant has a strong influence on potential swelling and breakage of the ELM microdroplets [39]. Poor selection may lead to an ineffective ELM process [39,81]. Surfactants are amphiphilic compounds [36,55], which makes them soluble in both water and organic solvents [88,89]. In ELMs, they have the ability to reduce the interfacial tension between the diluent and the feed/stripping phase and influence the ELM stability, as well as mass transfer characteristics [39,55]. If one liquid is dispersed in another liquid and the two are mutually immiscible, then the two liquids are first dispersed as small droplets in one another; and then mostly separate rapidly into two clearly defined layers. This is because the strength of the cohesive forces surpasses that of adhesive forces. Cohesive forces in individual phases are manifested as an interfacial energy or tension at the interface between the liquids [90]. Interfacial energy is decreased by adsorption of a surfactant at that interface [91]. This is due to the formation of the interfacial film around dispersed the ELM microdroplets, which in turn prevents the microdroplet coagulation and flocculation and stabilises the ELM [92,93].
Addition of the surfactant also modifies the mass transfecr resistance, water solubility and osmosis of the resulting ELM [39,93,94]. Interfacial tension is inversely proportional to the concentration of surfactant and curvature of the microdroplets [91]. It decreases as the concentration of surfactant increases which leads to the formation of smaller droplets and hence a more stable ELM. This will reduce the internal leakage of the membrane [95]. But increased concentration of the surfactant leads to the increase in swelling [39,96,97]. Surfactant concentration also affects the viscosity of the ELM [53]. Viscosity increases with increased surfactant concentration. Increases in viscosity of the membrane phase lead to a mechanically more stable ELM [98-101]. However it has its drawbacks. It leads to a decrease in the diffusion and mass transfer coefficients, resulting in a decrease in the extraction efficiency of the ELM [84,102]. This can be explained using the Eyring-Stokes-Einstein equation [84]. It can be noted that the increase in viscosity of the membrane phase leads to the decrease in the metal’s diffusion coefficient inside the ELM leading to reduced extraction rates of the ELM [71,84,103].
Therefore the concentration of the surfactant is vital to the ELM process, with the optimum weight fraction ranging from 2 to 5% (w/w%) [55]. For a successful ELM process, the surfactant must be insoluble in the feed and the stripping phases and must be soluble in the membrane phase so as to prevent the loss of the surfactant during the whole process. It should not react with the extractant and should be chemically stable in the presence of the stripping phase. Its resistance to mass transfer should be low and, finally, the demulsification process must not be hindered by the surfactant [50]. The hydrophile–lipophile balance (HLB) value is also important when choosing a surfactant, as it determines the type of emulsion formed. The HLB values from 1-10 make the surfactant more soluble in diluents compared to water, while the opposite is the case if the HLB ranges from 10 to 20. Table 1 describes the type of emulsion formed as function of the HLB values [53].
Hydration capacity and the molecular weight of the particular surfactant are important in the ELM context. High molecular weight lower the diffusivity of the surfactant [39], which is likely to result in higher degree of ELM droplet dispersion during the extraction process. This is likely going to result in the increased interfacial area for metal extraction and faster completion of the metal recovery. Low hydration capacity is preferred for a stable surfactant [39], i.e. the ester-type surfactants such as SPAN 80 are less likely to undergo hydrolysis and thus the stability of the ELM structure will not be compromised.
Addition of the surfactant also modifies the mass transfecr resistance, water solubility and osmosis of the resulting ELM [39,93,94]. Interfacial tension is inversely proportional to the concentration of surfactant and curvature of the microdroplets [91]. It decreases as the concentration of surfactant increases which leads to the formation of smaller droplets and hence a more stable ELM. This will reduce the internal leakage of the membrane [95]. But increased concentration of the surfactant leads to the increase in swelling [39,96,97]. Surfactant concentration also affects the viscosity of the ELM [53]. Viscosity increases with increased surfactant concentration. Increases in viscosity of the membrane phase lead to a mechanically more stable ELM [98-101]. However it has its drawbacks. It leads to a decrease in the diffusion and mass transfer coefficients, resulting in a decrease in the extraction efficiency of the ELM [84,102]. This can be explained using the Eyring-Stokes-Einstein equation [84]. It can be noted that the increase in viscosity of the membrane phase leads to the decrease in the metal’s diffusion coefficient inside the ELM leading to reduced extraction rates of the ELM [71,84,103].
Therefore the concentration of the surfactant is vital to the ELM process, with the optimum weight fraction ranging from 2 to 5% (w/w%) [55]. For a successful ELM process, the surfactant must be insoluble in the feed and the stripping phases and must be soluble in the membrane phase so as to prevent the loss of the surfactant during the whole process. It should not react with the extractant and should be chemically stable in the presence of the stripping phase. Its resistance to mass transfer should be low and, finally, the demulsification process must not be hindered by the surfactant [50]. The hydrophile–lipophile balance (HLB) value is also important when choosing a surfactant, as it determines the type of emulsion formed. The HLB values from 1-10 make the surfactant more soluble in diluents compared to water, while the opposite is the case if the HLB ranges from 10 to 20. Table 1 describes the type of emulsion formed as function of the HLB values [53].
Hydration capacity and the molecular weight of the particular surfactant are important in the ELM context. High molecular weight lower the diffusivity of the surfactant [39], which is likely to result in higher degree of ELM droplet dispersion during the extraction process. This is likely going to result in the increased interfacial area for metal extraction and faster completion of the metal recovery. Low hydration capacity is preferred for a stable surfactant [39], i.e. the ester-type surfactants such as SPAN 80 are less likely to undergo hydrolysis and thus the stability of the ELM structure will not be compromised.
Newtonian and non-Newtonian fluids
Non-Newtonian liquids have been theoretically understood for several decades. However, their properties have been used in the ELM metal extraction only in the last twenty years or so. This section will outline the use of non-Newtonian ELMs in metal extraction and the main research findings to date will be reviewed. At the end of this section, the proposals will be made to the application of the non-Newtonian ELMs in the PGM extraction from mining and metal-refinery side-streams. The description starts with outlining the differences between Newtonian and non-Newtonian liquids. In Newtonian fluids, shear stress and shear rate are directly proportional, as shown in equation 1 [35,36].
σ=ηγ (1)
In Equation 1, σ is shear stress (unit of Pa) and γ is shear rate (unit of s-1). The proportionality constant in equation 1 η is the (dynamic) viscosity (unit of Pa.s). If the relationship between shear stress and shear rate does not follow equation 1, and viscosity varies as a function of shear rate, then the fluid/ELM becomes non-Newtonian [3,50].
In non-Newtonian fluids, the constant coefficient of viscosity cannot be defined and so the diffusion coefficient of the extracted metal is independent of the ELM viscosity [104]. The Newtonian diluent is modified to a non-Newtonian one by dissolution of a high-molecular-weight polymer such as polyisobutylene in the diluent [105]. A critical concentration of the polymer in the diluent exists. Below the critical polymer concentration and at low shears, the viscosity of the ELM is directly proportional to the shears [50]. If the addition of the polymer increases the diluent viscosity in a non-Newtonian ELM and this in turn reduces coalescence and movement of the ELM microdroplets [85]. As a result, the interfacial area of the ELM microdroplets increases and the mass transfer rate of the extracted metal do not change. Reduced coalescence of the microdroplets leads to a more stable and more efficient ELM [85]. At the same time, this reduces the concentration of the surfactant used, hence leading to the lower affinity for water of the ELM, reducing the rates of swelling [3]. Combination of all these factors increases the stability of the ELM and improves the efficiency of the solvent metal extraction.
The critical concentration of the polymer (Ccritical; mg/L or mol/L) can be calculated as derived by Skelland and Meng [106] as shown in equation 2.
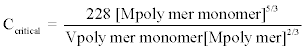 (2)
(2)
In equation 2, Mpolymer monomer is the molecular weight of the monomeric unit of the polymer that is added to the diluent and used to prepare a non-Newtonian ELM (g/mol). At the same time, Vpolymer monomer stands for the liquid molar volume of this monomeric subunit of the polymer at the normal pressure and boiling point (cm3/mol). Finally, Mpolymer represents for the mean molecular weight of the polymer modifier, e.g. polyisobutylene (g/mol) [106]. The non-Newtonian liquid’s effectiveness was first investigated by Skelland and Meng [85] in their study of extraction of benzoic acid, phenol and ammonia from simulated wastewater. Recently it was successfully applied to the extraction of metals such as Zn and Cu [50]. Reduced ELM leakage and improved stability were demonstrated, especially if the metal extraction was performed in the Taylor vortex column.
The Taylor vortex column consists of two cylinders, namely the outer static cylinder and the inner rotating cylinder. Schematic representation of the Taylor-vortex column can be found in Figure 3. The Taylor vortex column is advantageous over the continuously stirred tanks which have been used in most ELM studies. The disadvantages of continuously stirred tanks are drag and large shear around the impeller surface [107]. This leads to the breakage of emulsion droplets due to the turbulences created around the impeller [107]. These turbulences are a result of the unequal dissipation of volumetric energy in the reactor. Most volumetric energy dissipation occurs near the impeller tips [50]. Forney et al. [108] concluded that shear increases with the increase in the size of the contactor and that more electrical energy is required in scale-up devices used in industry [108]. The Taylor vortex column nullifies these disadvantages by distributing the power evenly per unit volume throughout the volume of the contactor and the rotor and tank stirrers are roughly equal in diameter [50].
The Taylor vortex column produces the Taylor-Couette flow pattern, reducing the maximum shear stress by one to two orders of magnitude compared to a stirred tank [50]. This reduction leads to an increase in the area inside the contactor exposed to constant maximum shear based on the friction drag on the large cylindrical surfaces near the boundaries of the Taylor-Couette flow [108,109]. The use of Taylor vortex columns instead of conventional mixing reactors has also improved the stability of the ELMs [71]. A major advantage of Taylor-column devices is the simplicity of scale-up, i.e. constant value of the dimensionless Taylor number [108]. This criterion is defined in equation 3.
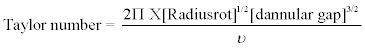 (3)
(3)
In equation 3, Radiusrot stands for the radius of the Taylor vortex column rotor (m), while X is the rotational speed of the inner cylinder (s-1). On the other hand, dannular gap is the annular gap width (m) which is the width of the space between the two cylinders of the Taylor vortex column. Finally, v stands for the kinematic viscosity of the liquid inside the Taylor-vortex column (m2s-1). More information can be found in reference [3] on pages 371-373 or the PhD thesis by Park [50]. Taylor-vortex columns as contactors and non-Newtonian fluid have been used in the extraction of benzoic acid [71], phenol and substituted phenols [49,50], of Zn, Pb, Ni and Cd [50,90] from model industrial wastewater. Park [50] used the shrinking core mathematical model of Liu and Liu [41] for quantitative description of the mass transfer kinetics of the process (see Figure 2 for details) [41,50]. These publications indicate that quantitative extraction can be achieved after time periods comparable with the classical solvent metal extraction [41, 50].
Limited studies to date have been applied to the extraction of PGMs from model mining and metal-refinery wastewaters to date [11]. They show potential of the ELM technique in the recovery of the PGMs from mining waste water. Major advantages of this approach include simultaneous purification and concentration of the solute because the processes of extraction and stripping are combined in one stage [55]. Use of ELM can be conducted with classical solvent contactors and could be up to 40% cheaper than those of solvent extraction [55]. Other advantages over liquid-liquid extraction are improved kinetics, selection of species to be removed and decreases in the volume ratio of organic phase to aqueous feed solution necessary [110]. Encouraging are so far the data that indicate that the ELM extraction of PGMs does not suffer from interference from metal contaminants that are commonly found in the mining and metal refinery side-streams, e.g. iron [23]. Thus further research should focus on the application of ELM to extraction of PGMs from said wastewaters and major research drive should focus on the use of the Taylor-vortex column and the non-Newtonian ELMs.
σ=ηγ (1)
In Equation 1, σ is shear stress (unit of Pa) and γ is shear rate (unit of s-1). The proportionality constant in equation 1 η is the (dynamic) viscosity (unit of Pa.s). If the relationship between shear stress and shear rate does not follow equation 1, and viscosity varies as a function of shear rate, then the fluid/ELM becomes non-Newtonian [3,50].
In non-Newtonian fluids, the constant coefficient of viscosity cannot be defined and so the diffusion coefficient of the extracted metal is independent of the ELM viscosity [104]. The Newtonian diluent is modified to a non-Newtonian one by dissolution of a high-molecular-weight polymer such as polyisobutylene in the diluent [105]. A critical concentration of the polymer in the diluent exists. Below the critical polymer concentration and at low shears, the viscosity of the ELM is directly proportional to the shears [50]. If the addition of the polymer increases the diluent viscosity in a non-Newtonian ELM and this in turn reduces coalescence and movement of the ELM microdroplets [85]. As a result, the interfacial area of the ELM microdroplets increases and the mass transfer rate of the extracted metal do not change. Reduced coalescence of the microdroplets leads to a more stable and more efficient ELM [85]. At the same time, this reduces the concentration of the surfactant used, hence leading to the lower affinity for water of the ELM, reducing the rates of swelling [3]. Combination of all these factors increases the stability of the ELM and improves the efficiency of the solvent metal extraction.
The critical concentration of the polymer (Ccritical; mg/L or mol/L) can be calculated as derived by Skelland and Meng [106] as shown in equation 2.
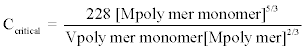 (2)
(2)In equation 2, Mpolymer monomer is the molecular weight of the monomeric unit of the polymer that is added to the diluent and used to prepare a non-Newtonian ELM (g/mol). At the same time, Vpolymer monomer stands for the liquid molar volume of this monomeric subunit of the polymer at the normal pressure and boiling point (cm3/mol). Finally, Mpolymer represents for the mean molecular weight of the polymer modifier, e.g. polyisobutylene (g/mol) [106]. The non-Newtonian liquid’s effectiveness was first investigated by Skelland and Meng [85] in their study of extraction of benzoic acid, phenol and ammonia from simulated wastewater. Recently it was successfully applied to the extraction of metals such as Zn and Cu [50]. Reduced ELM leakage and improved stability were demonstrated, especially if the metal extraction was performed in the Taylor vortex column.
The Taylor vortex column consists of two cylinders, namely the outer static cylinder and the inner rotating cylinder. Schematic representation of the Taylor-vortex column can be found in Figure 3. The Taylor vortex column is advantageous over the continuously stirred tanks which have been used in most ELM studies. The disadvantages of continuously stirred tanks are drag and large shear around the impeller surface [107]. This leads to the breakage of emulsion droplets due to the turbulences created around the impeller [107]. These turbulences are a result of the unequal dissipation of volumetric energy in the reactor. Most volumetric energy dissipation occurs near the impeller tips [50]. Forney et al. [108] concluded that shear increases with the increase in the size of the contactor and that more electrical energy is required in scale-up devices used in industry [108]. The Taylor vortex column nullifies these disadvantages by distributing the power evenly per unit volume throughout the volume of the contactor and the rotor and tank stirrers are roughly equal in diameter [50].
The Taylor vortex column produces the Taylor-Couette flow pattern, reducing the maximum shear stress by one to two orders of magnitude compared to a stirred tank [50]. This reduction leads to an increase in the area inside the contactor exposed to constant maximum shear based on the friction drag on the large cylindrical surfaces near the boundaries of the Taylor-Couette flow [108,109]. The use of Taylor vortex columns instead of conventional mixing reactors has also improved the stability of the ELMs [71]. A major advantage of Taylor-column devices is the simplicity of scale-up, i.e. constant value of the dimensionless Taylor number [108]. This criterion is defined in equation 3.
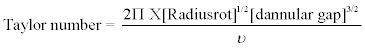 (3)
(3)In equation 3, Radiusrot stands for the radius of the Taylor vortex column rotor (m), while X is the rotational speed of the inner cylinder (s-1). On the other hand, dannular gap is the annular gap width (m) which is the width of the space between the two cylinders of the Taylor vortex column. Finally, v stands for the kinematic viscosity of the liquid inside the Taylor-vortex column (m2s-1). More information can be found in reference [3] on pages 371-373 or the PhD thesis by Park [50]. Taylor-vortex columns as contactors and non-Newtonian fluid have been used in the extraction of benzoic acid [71], phenol and substituted phenols [49,50], of Zn, Pb, Ni and Cd [50,90] from model industrial wastewater. Park [50] used the shrinking core mathematical model of Liu and Liu [41] for quantitative description of the mass transfer kinetics of the process (see Figure 2 for details) [41,50]. These publications indicate that quantitative extraction can be achieved after time periods comparable with the classical solvent metal extraction [41, 50].
Limited studies to date have been applied to the extraction of PGMs from model mining and metal-refinery wastewaters to date [11]. They show potential of the ELM technique in the recovery of the PGMs from mining waste water. Major advantages of this approach include simultaneous purification and concentration of the solute because the processes of extraction and stripping are combined in one stage [55]. Use of ELM can be conducted with classical solvent contactors and could be up to 40% cheaper than those of solvent extraction [55]. Other advantages over liquid-liquid extraction are improved kinetics, selection of species to be removed and decreases in the volume ratio of organic phase to aqueous feed solution necessary [110]. Encouraging are so far the data that indicate that the ELM extraction of PGMs does not suffer from interference from metal contaminants that are commonly found in the mining and metal refinery side-streams, e.g. iron [23]. Thus further research should focus on the application of ELM to extraction of PGMs from said wastewaters and major research drive should focus on the use of the Taylor-vortex column and the non-Newtonian ELMs.
SLMs in PGMs extraction
In the SLM, the solution of extractant in the diluent is impregnated onto a solid support. The transport takes place through the pore of the support using an analogical principle as with BLM and ELMs [1]. Schematic representation of the possible SLM arrangement can be seen in Figure 4. The SLM have been applied in various studies to the extraction of PGMs from mining and metal-refinery side-streams. Chaturabul et al. [111] reported on 78-82% recovery and extraction efficiency for Pd by an SLM from a feed phase of pH=2.0 and the stripping into a 0.03 M NaNO2 solution. The SLM contained 0.005 M thioridazine and 0.05 M oleic acid as extractants and the palladium extraction process followed the first-order kinetics [111].
At the flow rate of 100 mL/min, the extraction rate constant of the same PGM was equal to 0.0140/s, while the stripping rate constant was equal to 0.0248/s [111]. Zaghbani et al. [112] used thiacalix [4] arene as a carrier in an SLM to separate Pd and Au. The diluent was 1-(2-nitrophenoxy) octane and the stripping phase was 0.5 M solution of NaSCN with pH adjusted to 2.0 [112]. Selective extraction of PGMs from acidic aqueous solutions can be achieved by using selective extractants as demonstrated for Pd over Pt and Rh in an SLM, if benzyl (2-methoxy-3-diphenylphosphino) propyl ether is used as the extractant and decalin as the diluent [113] and by N-benzoyl-N',N'-diethylthiourea [114].
Fontas et al. [115] examined the influence of the SLM diluent on the membrane stability. Previous studies have indicated that an ideal diluent should have the following properties [116]: low aqueous solubility, high boiling point and interfacial tension at the diluent/water interface. For aromatic diluents, the literature data seems to indicate that SLMs suffer from lower stability in these solvents in comparison to their aliphatic counterparts [117,118]. The extractant used [119] and the preparation procedures of the LM in question [120] have also been shown to influence its stability. Stability of the SLMs is also a function of the pore size of the solid support with the stability of the SLM increasing with decreasing pore diameter [115]. Fontas et al. [115] studied the influence of the diluent solvent on the stability of SLMs. Cumene and 3-chlorotoluene as diluents led to higher permeability of Pd (II) through a Cyanex 471 impregnated SLM [115]. On the other hand, decaline improved the membrane stability at 24 hours of extraction/exposure [115].
Wu et al. [121] studied the transport of Pd2+ in the dispersion SLM which contained N, N-di (1-methylheptyl) acetamide as extractant and kerosene as diluent. These membrane components were impregnated into the polyvinylidene fluoride membrane [121]. A solution of potassium thiocyanate was used as the stripping phase [121]. The Pd2+ transport and extraction characteristics were studied as function of the feed-phase HCl molarity, the volumetric ratio of the diluent phase to the stripping phase and finally the stripping phase salt molarity [121]. The optimised conditions for Pd2+ were achieved under the following conditions: 0.10 mol/L of HCl in the feed phase, the diluent phase-to-the stripping phase ratio is 1:1 and the stripping phase is a 1.60 M potassium thiocyanate solution [121]. At the initial feed phase concentration of Pd2+ of 0.00005 M, 79.8% of the initial amount of palladium was recovered in the stripping phase after 130 minutes of extraction [121].
Ruhela et al. [122] reported a study on the extraction efficiency for palladium with a SLM that consisted as 0.05 M N,N,N',N'-tetra-(2-ethylhexyl) thiodiglycolamide as the extractant and n-dodecane as the diluent. The metal was dissolved in 3.0 M HNO3 solution and the stripping phase was a solution of 0.01 M thiourea in 2.0 M nitric acid [122]. The palladium mass transfer through the SLM was examined as function of the pH of the feed phase, the SLM concentration of the extractant and the pore size distribution of the membrane support/complete SLM [122]. The diffusion coefficient of Pd2+ through the SLM was equal to 3.56×10-5 cm2/s [122]. The extractant was 1000 more selective for palladium than for the interfering impurities and the loss of extractant into the feed phase limited [122]. Similar observations were reported by Panja et al. [123]. More studies are needed on the separation of the individual PGMs from their mixtures as described for the extraction of Ph, Pd and Pt from automotive catalytic converters was examined [124].
At the flow rate of 100 mL/min, the extraction rate constant of the same PGM was equal to 0.0140/s, while the stripping rate constant was equal to 0.0248/s [111]. Zaghbani et al. [112] used thiacalix [4] arene as a carrier in an SLM to separate Pd and Au. The diluent was 1-(2-nitrophenoxy) octane and the stripping phase was 0.5 M solution of NaSCN with pH adjusted to 2.0 [112]. Selective extraction of PGMs from acidic aqueous solutions can be achieved by using selective extractants as demonstrated for Pd over Pt and Rh in an SLM, if benzyl (2-methoxy-3-diphenylphosphino) propyl ether is used as the extractant and decalin as the diluent [113] and by N-benzoyl-N',N'-diethylthiourea [114].
Fontas et al. [115] examined the influence of the SLM diluent on the membrane stability. Previous studies have indicated that an ideal diluent should have the following properties [116]: low aqueous solubility, high boiling point and interfacial tension at the diluent/water interface. For aromatic diluents, the literature data seems to indicate that SLMs suffer from lower stability in these solvents in comparison to their aliphatic counterparts [117,118]. The extractant used [119] and the preparation procedures of the LM in question [120] have also been shown to influence its stability. Stability of the SLMs is also a function of the pore size of the solid support with the stability of the SLM increasing with decreasing pore diameter [115]. Fontas et al. [115] studied the influence of the diluent solvent on the stability of SLMs. Cumene and 3-chlorotoluene as diluents led to higher permeability of Pd (II) through a Cyanex 471 impregnated SLM [115]. On the other hand, decaline improved the membrane stability at 24 hours of extraction/exposure [115].
Wu et al. [121] studied the transport of Pd2+ in the dispersion SLM which contained N, N-di (1-methylheptyl) acetamide as extractant and kerosene as diluent. These membrane components were impregnated into the polyvinylidene fluoride membrane [121]. A solution of potassium thiocyanate was used as the stripping phase [121]. The Pd2+ transport and extraction characteristics were studied as function of the feed-phase HCl molarity, the volumetric ratio of the diluent phase to the stripping phase and finally the stripping phase salt molarity [121]. The optimised conditions for Pd2+ were achieved under the following conditions: 0.10 mol/L of HCl in the feed phase, the diluent phase-to-the stripping phase ratio is 1:1 and the stripping phase is a 1.60 M potassium thiocyanate solution [121]. At the initial feed phase concentration of Pd2+ of 0.00005 M, 79.8% of the initial amount of palladium was recovered in the stripping phase after 130 minutes of extraction [121].
Ruhela et al. [122] reported a study on the extraction efficiency for palladium with a SLM that consisted as 0.05 M N,N,N',N'-tetra-(2-ethylhexyl) thiodiglycolamide as the extractant and n-dodecane as the diluent. The metal was dissolved in 3.0 M HNO3 solution and the stripping phase was a solution of 0.01 M thiourea in 2.0 M nitric acid [122]. The palladium mass transfer through the SLM was examined as function of the pH of the feed phase, the SLM concentration of the extractant and the pore size distribution of the membrane support/complete SLM [122]. The diffusion coefficient of Pd2+ through the SLM was equal to 3.56×10-5 cm2/s [122]. The extractant was 1000 more selective for palladium than for the interfering impurities and the loss of extractant into the feed phase limited [122]. Similar observations were reported by Panja et al. [123]. More studies are needed on the separation of the individual PGMs from their mixtures as described for the extraction of Ph, Pd and Pt from automotive catalytic converters was examined [124].
Conclusions
Advantages of the BLM in the PGM extraction originate from the operational simplicity, but they suffer from the limited nature for adaptation of the given system design in the particular PGM metal/solvent extraction. In contrast to BLMs, the ELM extraction can be conducted in the “classical” solvent contactors with cost savings of up to 40%, compared with the simple metal-solvent extraction. The up-to-date data of the ELM application in the PGM does not suffer from interference caused by the common metal contaminants found in the mining and metal refinery side-streams, e.g. iron. Thus further research should focus on the application of ELM to extraction of PGMs from said wastewaters using selective extractants such as crown ethers, which provide selectivity for a given PGM and eliminate the majority of potential interferences. At the same time, more research should be focused on the use of the Taylor-vortex column and the non-Newtonian ELMs that improve the ELM stability and extraction kinetics. For aromatic diluents, the literature data seems to indicate that SLMs suffer from lower stability in these solvents in comparison to their aliphatic counterparts. The SLM application in the PGM extraction from mining wastewater and side-streams should be further investigated. However, based on the currently available data ELMs seem to be the most practical liquid membranes for the practical application in the PGM extraction from mining-related wastewaters. The main focus should be on the optimisation of the process and the competition studies with all six PGMs.
Acknowledgements
The authors are grateful to the Rhodes Research Council Fund, Institute of Water Research, and the Water Commission of South Africa (Grant number K5/2011/3) for financial support and the Faculty of Pharmacy at Rhodes University and the laboratory colleagues from the Environmental Health and Biotechnology Research Group in the Faculty of Pharmacy of Rhodes University, South Africa.
References
- Kislik VS 2010 Chapter 1 - Introduction, General Description, Definitions, and Classification: Liquid Membranes Amsterdam,(Vladimir S. Kislikedn), Elsevier, 1-15.
- Davis RH 1997 Microfiltration and ultrafiltration: Principles and applications: Leos J. Zeman and Andrew L. Zydney, Marcel Dekker, New York, 1996. J MembrSci 134: 273-274.
- Tandlich R (2010) Application of Liquid Membranes in Wastewater Treatment: Liquid Membranes Amsterdam. (Vladimir S. Kislikedn), Elsevier, 357-400.
- Akin I, Erdemir S, Yilmaz M, Mustafa Ersoz M (2012) Calix[4]arene derivative bearing imidazole groups as carrier for the transport of palladium by using bulk liquid membrane. J Hazard Mater 223-224.
- Hirai T, Komasawa I (2001) Preparation and microscopic structure control of fine particle materials by using emulsion liquid membrane system as a microreactor. Kagaku KogakuRonbun 27: 311-313.
- Anticó E, Masana A, Hidalgo M, Salvadó V, Valiente M (1994) New sulphur-containing reagents as carriers for the separation of palladium by solid supported liquid membranes. Hydrometallurgy 35: 343-352.
- Dobson RS, Burgess JE (2007) Biological treatment of precious metal refinery wastewater. MinerEng 20: 519-532.
- Rovira M, Sastre AM (1998) Modelling of mass transfer in facilitated supported liquid-membrane transport of palladium(II) using di-(2-ethylhexyl) thiophosphoric acid. J MembrSci 149: 241-250.
- Fu J, Nakamura S, Akiba K (1995) Carrier-mediated transport of platinum(IV) through a supported liquid membrane impregnating an 8-quinolinol derivative. J MembrSci 105: 281-286.
- Hidalgo M, Masana A, Salvadò V, Freiser H, Al-Bazi S, et al. (1991) Accelerated mass transfer of palladium(II) through a selective solid-supported liquid membrane containing Cyanex 471. Anal ChimActa 251: 233-239.
- Praipruke S, Kriausakul K, Tantayanon S (2012) Extraction of Palladium from Acidic Chloride Media into Emulsion Liquid Membranes using LIX 984N-C. Int J NonferrMetall 1: 13-22.
- Fontàs C, Salvadó V, Hidalgo M (1999) Solvent extraction of Pt(IV) by Aliquat 336 and its application to a solid supported liquid membrane system. Solvent Extr Ion Exc 17: 149-162.
- Kolev SD, Sakai Y, Cattrall RW, Paimin R, Potter ID (2000) Theoretical and experimental study of palladium(II) extraction from hydrochloric acid solutions into Aliquat 336/PVC membranes. AnalChimActa 413: 241-246.
- Fontàs C, Anticó E, Salvadó V, Valiente M, Hidalgo M (1997) Chemical pumping of rhodium by a supported liquid membrane containing Aliquat 336 as carrier. AnalChimActa 346: 199-206.
- Kakoi T, Horinouchi N, Goto M, Nakashio F (1996) Selective recovery of palladium from a simulated industrial waste water by liquid surfactant membrane process. J MembrSci 118: 63-71.
- Reddy TR, Merravali NN, Reddy AVR (2013) Novel reverse micelle mediated transport of Pt and Pd through a bulk liquid membrane from real samples. Sep PurifTechnol 103: 71-77.
- Wongkaew K, Pancharoen U, Phatanasri S, Leepipatpiboon N, Lothongkum AWEffect of diluent polarity on membrane stability in the separation of trace Pd(II) from wastewater by HFSLM using LIX84-I. J IndEng Chem.
- Majima H, Hirato T, Awakura Y, Hibi T (1991) Preparation of monosized ultrafine particles of precious metals utilizing an emulsion-type liquid membrane technique. Metall Trans B 22: 397-404.
- Kakoi T, Kondo K, Coto M, Nakashio F (1993) Extraction mechanism of palladium with didodecylmonothiophosphoric acid in heptane diluent. Solvent Extr 11: 627-643.
- Zhang R, Quan S (1993) Extraction of palladium (II) by emulsion liquid membrane. Water Treat 8: 431-438.
- Anticó E, Hidalgo M, Masana A, Salvadó V, Valiente M (1996) SCN- effect on the palladium(II) transfer in two and three phases systems using triphenylphosphine sulfide as a carrier. React FunctPolym 28: 103-109.
- Alam MS, Inoue K, Yoshizuka K (1996) Kinetics of palladium extraction with bis(2-ethylhexyl)monothiophosphoric acid in a hollow fiber membrane extractor. IndEngChem Res 35: 3899-3906.
- Farhadi K, Shamsipur M (2000) Separation study of palladium through a bulk liquid membrane containing thioridazine. HCl and oleic acid.SeparSciTechnol 35: 859-868.
- Kakoi T, Goto M, Nakashio F (1996) Separation of platinum and palladium by liquid surfactant membranes utilising a novel bi-functional surfactant. J MembrSci 120: 77-80.
- Kakoi T, Goto M, Nakashio F (1997) Separation of palladium and silver from a nitric acid solution by liquid surfactant membranes. SeparSciTechnol 32: 1415-1432.
- Vytras K, Kalous J,Simickova B, Cerna J, Silena I (1988) Potentiometric titration of palladium(II) halide complex anions based on ion-pair formation. Anal ChimActa 209: 357-361.
- Wisniewski M, Szymanowski J (1998) Palladium(II) Extraction and Extractant Adsorption at a Liquid/Liquid Interface. Anal Sci 14: 241-245.
- Ashraf Chaudry M, Malik MT, Ali A (1990) Transport of Mo(VI) ions through tri-n-octylamine-xylene based supported liquid membranes. SeparSciTechnol 25: 263-291.
- Alizadeh N, Salimi S, Jabbari A (2002) Transport study of palladium through a bulk liquid membrane using hexadecylpyrimidium as carrier. Sep PurifTechnol 28: 173-180.
- Lokhande TN, Anuse MA, Chavan MB (1998) Liquid-liquid extraction of palladium(II) with N-n-octylaniline from hydrochloric acid media. Talanta 46: 163-169.
- Khayatian G, Shamsipur M (1999) Highly selective and efficient membrane transport of palladium as PdCl42- ion using NH4+-dibenzyldiaza-18-crown-6 as carrier. Sep PurifTechnol 16: 235-241.
- Abbaspour A, Tavakol F (1999) Highly selective transport of palladium through a liquid membrane. Anal Sci 15: 1005-1008.
- Kakoi T, Toh T, Kubota F, Goto M, Shinkai S, et al. (1998) Liquid-liquid extraction of metal ions with a cyclic ligand calixarene carboxyl derivative. Anal Sci 14: 501-506.
- Memon S, Memon N, Memon S, Latif Y (2011) An efficient calix[4]arene based silica sorbent for the removal of endosulfan from water. J Hazard Mater 186: 1696-1703.
- Remington JP (2005) The science and practice of pharmacy.(21stedn), Lippincott Williams & Wilkins, New York, USA, 331-338.
- Allen LV, Popovich NG (2005)Ansel's pharmaceutical dosage forms and drug delivery systems. (9thedn), Lippincott Williams & Wilkins, New York, USA, 383-405.
- Patnaik PR (1995) Liquid emulsion membranes: Principles, problems and applications in fermentation processes. BiotechnolAdv 13: 175-208.
- Kargari A, Kaghazchi T, Sohrabi M, Soleimani M (2004) Batch extraction of gold(III) ions from aqueous solutions using emulsion liquid membrane via facilitated carrier transport. J MembrSci 233: 1-10.
- Wan Y, Zhang X (2002) Swelling determination of W/O/W emulsion liquid membranes. J MembrSci 196: 185-201.
- Fouad EA, Bart H (2008) Emulsion liquid membrane extraction of zinc by a hollow-fiber contactor. J MembrSci 307: 156-168.
- Liu X, Liu D (2000) Mass transfer resistance analysis of L-tryptophan extraction in an emulsion liquid membrane system. SeparSciTechnol 35: 2707-2724.
- Chakraborty M, Bhattacharya C, Datta S (2004) Study of the Stability of W/O/W-Type Emulsion During the Extraction of Nickel via Emulsion Liquid Membrane. SeparSciTechnol 39: 2609-2625.
- Ho WSW, Li NN (1996) Recent Advances in emulsion liquid membranes. AcsSymSer 642: 206-221.
- Teramoto M, Sakuramoto T, Koyama T, Matsuyama H, Miyake Y (1986) Extraction of lanthanoids by liquid surfactant membranes. SeparSciTechnol 21: 229-250.
- Wang J, Feng Y, Yin X, Yue M (1998) Studies on transport and separation of Pd(II) by the emulsion liquid membrane method. Chin JInorgChem 14: 157-161.
- Chanukya BS, Rastogi NK (2013) Extraction of alcohol from wine and color extracts using liquid emulsion membrane. Sep PurifTechnol 105: 41-47.
- Gu Z, Ho WSW, Li N (1992) Emulsion liquid membranes: Design considerations. Membrane Handbook.VanNostrand Reinhold, Chapman & Hall, New York, NY, USA.
- Chanukya B, Rastogi NK (2010) Extraction of alcohol using emulsion liquid membrane consisting of paraffin oil as an organic phase and lecithin as a surfactant. J ChemTechnolBiotechnol 85: 243-247.
- Park Y, Skelland AHP, Forney LJ, Kim J (2006) Removal of phenol and substituted phenols by newly developed emulsion liquid membrane process. Water Res 40: 1763-1772.
- Park Y (2006) Development and Optimization of Novel Emulsion Liquid Membranes Stabilized by Non-Newtonian Conversion in Taylor-Couette Flow for Extraction of Selected Organic and Metallic Contaminants. PhD thesis, Georgia Institute of Technology, Georgia, USA.
- Kumbasar R, Sahin I (2008) Separation and concentration of cobalt from ammoniacal solutions containing cobalt and nickel by emulsion liquid membranes using 5,7-dibromo-8-hydroxyquinoline (DBHQ). J MembrSci 325: 712-718.
- Hasan MA, Selim YT, Mohamed KM (2009) Removal of chromium from aqueous waste solution using liquid emulsion membrane. J Hazard Mater 168: 1537-1541.
- KislikVS (2009) Liquid membranes: principles and applications in chemical separations and wastewater treatment.Elsevier Science, Amsterdam, Netherlands.
- Mohan S, Narsimhan G (1997) Coalescence of protein-stabilized emulsions in a high-pressure homogenizer. J Colloid Interface Sci 192: 1-15.
- Ahmad AL, Kusumastuti A, Derek CJC, Ooi BS (2011) Emulsion liquid membrane for heavy metal removal: An overview on emulsion stabilization and destabilization. ChemEng J 171: 870-882.
- Li N, Cahn R, Naden D, Lai R (1983) Liquid membrane processes for copper extraction. Hydrometallurgy 9: 277-305.
- Chowta S, Mohapatra PK, Tripathi SC, Tomar BS, Manchanda VK (2010) Recovery and pre-concentration of americium (III) from dilute acid solutions using an emulsion liquid membrane containing di-2-ethylhexyl phosphoric acid (D2EHPA) as extractant. J RadioanalNucChem285: 309-314.
- Sun D, Duan X, Li W, Zhou D (1998) Demulsification of water-in-oil emulsion by using porous glass membrane. JMembrSci 146: 65-72.
- Al-Sabagh AM, Nasser NM, Abd El-Hamid TM (2013) Investigation of Kinetic and Rheological Properties for the Demulsification Process. Egyptian Journal of Petroleum 22: 117-127.
- Kim YH, Wasan DT (1996) Effect of demulsifier partitioning on the destabilization of water-in-oil emulsions. IndEngChem Res 35: 1141-1149.
- Shetty C, Nikolov A, Wasan D, Bhattacharyya B (1992) Demulsification of water in oil emulsions using water soluble demulsifiers. J Dispersion SciTechnol13: 121-133.
- Bhardwaj A, Hartland S (1998) Studies on buildup of interfacial film at the crude oil/water interface. J Dispersion SciTechnol 19: 465-473.
- Roodbari NH, Badiei A, Soleimani E, Khaniani Y (2012) Tweens demulsification effects on heavy crude oil/water emulsion. Arabian Journal of Chemistry.
- Wang J, Hu F, Li C, Li J, Yang Y (2010) Synthesis of dendritic polyether surfactants for demulsification. Sep PurifTechnol 73: 349-354
- Tong K, Zhang Y, Chu PK (2013) Evaluation of calcium chloride for synergistic demulsification of super heavy oil wastewater. Colloids Surf PhysicochemEng Aspects 419: 46-52.
- Eow JS, Ghadiri M (2002) Electrostatic enhancement of coalescence of water droplets in oil: a review of the technology. ChemEng J 85: 357-368.
- Hosseini M, Shahavi MH (2012) Electrostatic Enhancement of Coalescence of Oil Droplets [in Nanometer Scale] in Water Emulsion. Chin J ChemEng 20: 654-658.
- Mousavichoubeh M, Shariaty-Niassar M, Ghadiri M (2011) The effect of interfacial tension on secondary drop formation in electro-coalescence of water droplets in oil. ChemEngSci 66: 5330-5337.
- Mousavichoubeh M, Ghadiri M, Shariaty-Niassar M (2011) Electro-coalescence of an aqueous droplet at an oil–water interface. Chemical Engineering and Processing: Process Intensification 50: 338-344.
- Peng W, Jiao H, Shi H, Xu C (2012) The application of emulsion liquid membrane process and heat-induced demulsification for removal of pyridine from aqueous solutions. Desalination 286: 372-378.
- Park Y, Forney LJ, Kim JH, Skelland AHP (2004) Optimum emulsion liquid membranes stabilized by non-Newtonian conversion in Taylor–Couette flow. ChemEngSci 59: 5725-5734.
- Borwankar R, Chan C, Wasan D, Kurzeja R, Gu Z, et al. (1988) Analysis of the effect of internal phase leakage on liquid membrane separations. AIChE J 34: 753-762.
- Bhowal A, Datta S (1997) Facilitated transport through liquid surfactant membrane: Analysis of breakage model. JMembrSci 135: 245-250.
- Bandyopadhyaya R, Bhowal A, Datta S, Sanyal SK (1998) A new model of batch-extraction in emulsion liquid membrane: Simulation of globule-globule interaction and leakage. ChemEngSci 53: 2799-2807.
- Colinart P, Delepine S, Trouve G, Renon H (1984) Water transfer in emulsified liquid membrane processes. J MembrSci 20: 167-187.
- Ghosh S (2011) Comparative studies on brij reverse micelles prepared in benzene/surfactant/ethylammonium nitrate systems: Effect of head group size and polarity of the hydrocarbon chain. J Colloid Interface Sci 360: 672-680.
- Mohd-Setapar SH, Mohamad-Aziz SN, Harun NH, Mohd-Azizi CY (2012) Review on the Extraction of Biomolecules by Biosurfactant Reverse Micelles. APCBEE Procedia 3: 78-83.
- Malik MA, Wani MY, Hashim MA (2012)Microemulsion method: a novel route to synthesize organic and inorganic nanomaterials: 1st nano update. Arabian Journal of Chemistry 5: 397-417.
- Gu Z, Ho WSW, Li N (1992) Emulsion liquid membranes: Design considerations. IN: Membrane Handbook.Van Nostrand Reinhold, Chapman & Hall, New York, NY, USA, 656-700.
- Stroeve P, Varanasi PP (1984) An experimental study on double emulsion drop breakup in uniform shear flow. J Colloid Interface Sci 99: 360-373.
- Gadekar PT, Mukkolath AV, Tiwari KK (1992) Recovery of nitrophenols from aqueous solutions by a liquid emulsion membrane system. SeparSciTechnol 27: 427-445.
- Li NN (1971) Separation of hydrocarbons by liquid membrane permeation. Industrial & Engineering Chemistry Process Design and Development10: 215-221.
- Manzak A, Sonmezoglu M (2010) Extraction of acetic acid from aqueous solutions by emulsion type liquid membranes using Alamine 300 as a carrier. Ind J ChemTechnol 17: 441-445.
- Terry RE, Li NN, Ho WS (1982) Extraction of phenolic compounds and organic acids by liquid membranes. J MembrSci 10: 305-323.
- Skelland AHP, Meng X (1999) Non-Newtonian conversion solves problems of stability, permeability, and swelling in emulsion liquid membranes. J MembrSci 158: 1-15.
- Correia PFMM, de Carvalho JMR (2003) Recovery of phenol from phenolic resin plant effluents by emulsion liquid membranes. JMembrSci 225: 41-49.
- Pfeiffer R, Bunge A, Cheung H, Shere A (1992) Corrected analysis of the effect of preparation parameters on leakage in liquid surfactant membrane systems. SeparSciTechnol 27: 753-763.
- Chakrabarty K, Saha P, Ghoshal AK. Simultaneous separation of mercury and lignosulfonate from aqueous solution using supported liquid membrane. JMembrSci 346: 37-44.
- Chakrabarty K, Saha P, Ghoshal AK (2010) Separation of lignosulfonate from its aqueous solution using emulsion liquid membrane. J MembrSci 360: 34-39.
- Aulton ME, Wells T (2002) Pharmaceutics: The science of dosage form design, (2nd edn), Churchill Livingstone, New York, USA, 41-86.
- Chakraborty M, Bhattacharya C, Datta S (2010) Chapter 4 - Emulsion Liquid Membranes: Definitions and Classification, Theories, Module Design, Applications, New Directions and Perspectives. (Vladimir S. Kislik editor), Liquid Membranes Amsterdam: Elsevier141-199.
- Basualto C, Poblete M, Marchese J, Ochoa A, Acosta A, et al. (2006) Extraction of cadmium from aqueous solutions by emulsion liquid membranes using a stirred transfer cell contactor. J BrazChemSoc 17: 1347-1354.
- Kakoi T, Goto M, Nakashio F (1996) Separation of platinum and palladium by liquid surfactant membranes utilizing a novel bi-functional surfactant. J MembrSci 120: 77-88.
- Nakashio F, Goto M, Matsumoto M, Irie J, Kondo K (1988) Role of surfactants in the behavior of emulsion liquid membranes--development of new surfactants. J MembrSci 38: 249-260.
- Gasser M, El-Hefny N, Daoud J (2008) Extraction of Co(II) from aqueous solution using emulsion liquid membrane. J Hazard Mater 151: 610-615.
- Sengupta B, Sengupta R, Subrahmanyam N (2006) Process intensification of copper extraction using emulsion liquid membranes: Experimental search for optimal conditions. Hydrometallurgy 84: 43-53.
- Yan J, Pal R (2004) Effects of aqueous-phase acidity and salinity on isotonic swelling of W/O/W emulsion liquid membranes under agitation conditions. J MembrSci 244: 193-203.
- Lin CC, Long RL (1997) Removal of nitric acid by emulsion liquid membrane: Experimental results and model prediction. J MembrSci 134: 33-45.
- Kumbasar RA (2009) Cobalt–nickel separation from acidic thiocyanate leach solutions by emulsion liquid membranes (ELMs) using TOPO as carrier. Sep PurifTechnol 68: 208-215.
- Li Q, Liu Q, Li K, Tong S (1997) Separation study of cadmium through an emulsion liquid membrane. Talanta 44: 657-662.
- Chakravarti A, Chowdhury S, Mukherjee D (2000) Liquid membrane multiple emulsion process of separation of copper (II) from waste waters. Colloids Surf PhysicochemEng Aspects 166: 7-25.
- Lacks DJ, Rear DB, Van Orman JA (2007) Molecular dynamics investigation of viscosity, chemical diffusivities and partial molar volumes of liquids along the MgO-SiO2 join as functions of pressure. GeochimCosmochimActa 71: 1312-1323.
- Wang C, Bunge A (1990)Multisolute extraction of organic acids by emulsion liquid membranes. I. Batch experiments and models. J MembrSci 53: 71-103.
- Skelland AHP (1967) Non-Newtonian flow and heat transfer. Wiley, New York, USA.
- Bianchi U, Peterlin A (1969) Time Dependence of Viscosity of Dilute Polyisobutylene Solutions. Colloid PolymSci 232: 749-752.
- Skelland A, Meng XM (1996) A new solution to emulsion liquid membrane problems by non-Newtonian conversion. AIChE J 42: 547-561.
- Groeneweg F, van Dieren F, Agterof WGM (1994) Droplet break-up in a stirred water-in-oil emulsion in the presence of emulsifiers. Colloids Surf PhysicochemEng Aspects 91: 207-214.
- Forney L, Skelland A, Morris J, Holl R (2002) Taylor vortex column: large shear for liquid–liquid extraction. SeparSciTechnol 37: 2967-2986.
- Richter O, Hoffmann H, Kraushaar-Czarnetzki B (2008) Effect of the rotor shape on the mixing characteristics of a continuous flow Taylor-vortex reactor. ChemEngSci 63: 3504-3513.
- El-Reefy SA, Selim YT, Hassan MA, Aly HF (2003) Assessment of liquid emulsion membrane for cleanup of aqueous waste effluents from hazardous elements. WM’03 conference, Arizona; 2003, WM’03 Conference, Tucson, AZ.
- Chaturabul S, Wongkaew K, Pancharoen U (2013) Selective Transport of Palladium through a Hollow Fiber Supported Liquid Membrane and Prediction Model Based on Reaction Flux. SeparSciTechnol (Philadelphia) 48: 93-104.
- Zaghbani A, Tayeb R, Dhahbi M, Hidalgo M, Vocanson F, et al. (2007) Selective thiacalix[4]arene bearing three amide groups as ionophore of binary Pd(II) and Au(III) extraction by a supported liquid membrane system. SepPurifTechnol 57: 374-379.
- Fontàs C, Compañl L, Polo A, Salvadó V, Hidalgo M (2001) Benzyl (2-Methoxy-3-Diphenylphosphino) propyl ether as a carrier for the selective transport of Pd(II) through a solid supported liquid membrane. Solvent Extr Ion Exc 19: 329-344.
- Domínguez M, Anticó E, Beyer L, Aguirre A, García-Granda S, et al. (2002) Liquid-liquid extraction of palladium(II) and gold(III) with N-benzoyl-N',N'-diethylthiourea and the synthesis of a palladium benzoylthiourea complex. Polyhedron 21: 1429-1437.
- Fontàs C, Salvadó V, Hidalgo M (2003) Selective enrichment of palladium from spent automotive catalysts by using a liquid membrane system. JMembrSci 223: 39-48.
- Deblay P, Delepine S, Minier M, Renon H (1991) Selection of organic phases for optimal stability and efficiency of flat-sheet supported liquid membranes. SeparSciTechnol 26: 97-115.
- Takeuchi H, Takahashi K, Goto W (1987) Some observations on the stability of supported liquid membranes. J MembrSci 34: 19-31.
- Barnes DE, Marshall GD, Van Staden JF (1995) Rapid optimization of chemical parameters affecting supported liquid membranes. SeparSciTechnol 30: 751-776.
- Chiarizia R (1991) Stability of supported liquid membranes containing long-chain aliphatic amines as carriers. J MembrSci 55: 65-77.
- Yang XJ, Fane T (1997) Effect of membrane preparation on the lifetime of supported liquid membranes. J MembrSci 133: 269-273.
- Wu X, Yao B, Feng J (2012) Study on transportation of Pd(II) by dispersion supported liquid membrane system of N503-kerosene-KSCN. Rare Metal Mat Eng 41: 78-81.
- Ruhela R, Panja S, Sharma JN, Tomar BS, Tripathi SC, et al. Facilitated transport of Pd(II) through a supported liquid membrane (SLM) containing N,N,N',N'-tetra-(2-ethylhexyl) thiodiglycolamide T(2EH)TDGA: A novel carrier. J Hazard Mater 229-230: 66-71.
- Panja S, Ruhela R, Das A, Tripathi SC, Singh AK, et al. (2014) Carrier mediated transport of Pd(II) from nitric acid medium using Dithiodiglycolamide(DTDGA) across a supported liquid membrane (SLM). J Membrane Sci 449: 67-73.
- Fontàs C, Salvadó V, Hidalgo M (2002) Separation and concentration of Pd, Pt, and Rh from automotive catalytic converters by combining two hollow-fiber liquid membrane systems. IndEngChem Res 41: 1616-1620.
Tables and Figures at a glance
| Table 1 |
Figures at a glance
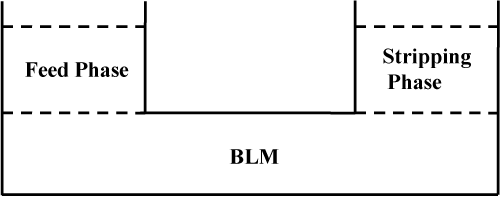 |
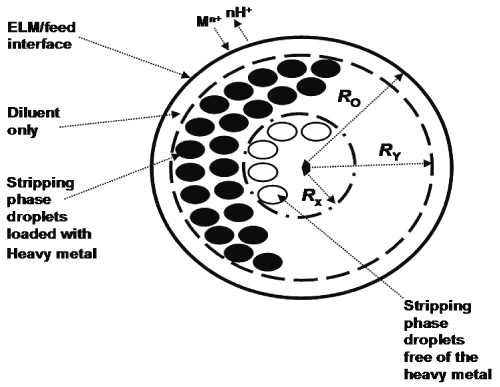 |
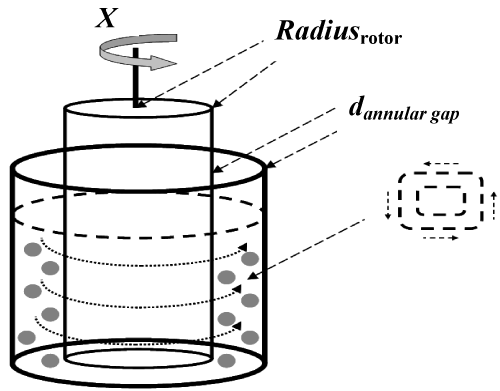 |
 |
|
| Figure 1 | Figure 2 | Figure 3 | Figure 4 |
Post your comment
Relevant Topics
- Anaerobic Biodegradation
- Biodegradable Balloons
- Biodegradable Confetti
- Biodegradable Diapers
- Biodegradable Plastics
- Biodegradable Sunscreen
- Biodegradation
- Bioremediation Bacteria
- Bioremediation Oil Spills
- Bioremediation Plants
- Bioremediation Products
- Ex Situ Bioremediation
- Heavy Metal Bioremediation
- In Situ Bioremediation
- Mycoremediation
- Non Biodegradable
- Phytoremediation
- Sewage Water Treatment
- Soil Bioremediation
- Types of Upwelling
- Waste Degredation
- Xenobiotics
Recommended Journals
Article Tools
Article Usage
- Total views: 16531
- [From(publication date):
July-2014 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 11274
- PDF downloads : 5257
