Case Report Open Access
Novel Use of Budesonide MMX in the Treatment of Lymphocytic Colitis
Nicklesh Singh1 and Anish Patel2*
1Department of Family Practice, Carl R. Darnall Army Medical Center, Ft. Hood, TX, USA
2Department of Gastroenterology/Hepatology, Carl R. Darnall Army Medical Center, Ft. Hood, TX, USA
- *Corresponding Author:
- Anish Patel, Physician
Carl R Darnall Army Medical Center
Gastroenterology, 36000 darnall loop
ft. hood, tx 76544, USA
Tel: 6093528222
E-mail: anishpa81@gmail.com
Received date: March 30, 2016; Accepted date: April 28, 2016; Published date: April 30, 2016
Citation: Singh N, Patel A (2016) Novel Use of Budesonide MMX in the Treatment of Lymphocytic Colitis. J Gastrointest Dig Syst 6:423. doi:10.4172/2161-069X.1000423
Copyright: © 2016 Singh N, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Gastrointestinal & Digestive System
Abstract
Background: Microscopic colitis, in particular, lymphocytic colitis is a relatively common cause of chronic diarrhea. The incidence of lymphocytic colitis is reported up to 63.7 per 100,000 person-years. Unfortunately, limited pharmacologic interventions are available with durable remission. We present a case of lymphocytic colitis refractory to conventional therapy with efficacy with budesonide MMX therapy.
Case Presentation: A 60-year-old Caucasian retired Army service member presented to a civilian Gastroenterology clinic with complaint of chronic diarrhea. He reported having up to 6-8 liquid bowel movements per day with Bristol 5-6 consistency for over 4 months with an associated 10 pound weight loss. He was initially discontinued on esomeprazole therapy and started on loperamide with no improvement in symptoms after 4 weeks. He was started on bismuth subsalicylate for another 4 weeks with minimal to no response. He attempted a short course of oral prednisone therapy but was intolerant due to significant side effects including headaches and migraines. He underwent an EGD and repeat colonoscopy for second opinion evaluation. Random duodenal biopsies were negative for enteritis (to include celiac) and repeat colonic biopsies with persistent lymphocytic colitis but negative for infectious agents and/or inflammatory bowel disease. Patient was started on budesonide MMX therapy daily for 2 months. He noted complete resolution of symptoms with normalization of bowels within 1 week of therapy. He remained asymptomatic at follow-up 3 months after therapy completion.
Discussion: Lymphocytic colitis is a common colonic disorder associated with chronic watery diarrhea. When medication side effects and food-related diarrhea is eliminated, pharmacological options are limited with short rates of durable remission. Budesonide MMX is a novel therapy option due to its formulation and colonic delivery design allowing high rates of colonic penetration with minimal systemic absorption.
Keywords
Lymphocytic colitis; Budesonide; Microscopic colitis
Introduction
Lymphocytic colitis is a relatively uncommon cause of chronic diarrhea. The incidence of lymphocytic colitis is reported up to 4.0 cases per 100,000 person-years [1]. Unfortunately, limited pharmacologic interventions are available with durable remission. We present a case of lymphocytic colitis refractory to conventional therapy with efficacy with budesonide multi-matrix formulation (MMX) therapy.
Case Report
A 60 year-old Caucasian retired Army service member presented to a civilian Gastroenterology clinic with complaint of chronic diarrhea. He had an associated history of GERD and hemochromatosis and no history of alcohol abuse and/or pancreatic insufficiency. He reported having up to 6-8 liquid bowel movements per day with Bristol 5-6 consistency for over 4 months. He reported having associated mild weight loss of 10 pounds over the past 2 months but no nocturnal stooling. He reported no exacerbation of symptoms with lactose products and/or gluten-containing products. He reported no exposure to sick contacts and/or well water use. His prescription medications included only esomeprazole. Serological evaluation was normal, to include negative celiac testing. He underwent a colonoscopy with reports demonstrating a normal mucosal appearance but with random biopsies consistent with lymphocytic colitis.
He was initially started on loperamide 2 mg up to four times a day with no improvement in symptoms after 4 weeks. He was started on bismuth subsalicylate 786 mg three times a day for another 4 weeks with minimal to no response. He attempted a course of budesonide EC 9 mg but with limited effectiveness.
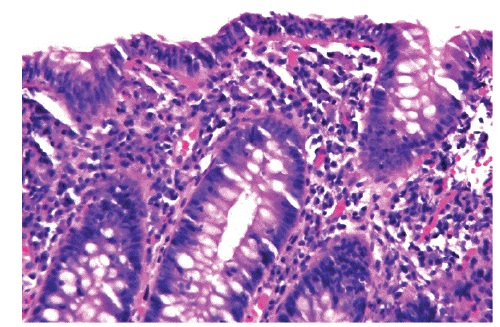
He underwent an EGD and repeat colonoscopy for second opinion evaluation. Random duodenal biopsies were negative for enteritis (to include celiac) and repeat colonic biopsies with persistent lymphocytic colitis but negative for infectious agents and/or inflammatory bowel disease (Figure 1). All specimens were reviewed by a gastropathologist.
Patient was started on budesonide MMX 9 mg daily for 2 months. He noted complete resolution of symptoms with normalization of bowels within 1 week of therapy. He remained asymptomatic at followup 3 months after therapy completion.
Discussion
Lymphocytic colitis (LC) is characterized by chronic watery diarrhea with a relation to collagenous colitis, and together are more broadly characterized as microscopic colitis. Pooled analysis estimate the incidence of LC to be approximately 4.14 per 100,000 person-years and a prevalence of 63.05 cases per 100,000 person-years [2]. Clinically, the predominant symptom for LC is chronic diarrhea affecting a mean age of 63 years and further histological evaluation demonstrating an increased amount of intraepithelial lymphocytes (IEL) in conjunction with an inflammatory infiltrate in the lamina propria [3,4]. The preponderance of data for medication causes are including but not limited to nonsteroidal anti-inflammatory drugs (NSAID) and proton pump inhibitors (PPI). Medical therapy particularly for LC has been limited.
One of the first reported cases of the use of budesonide extendedrelease (budesonide EC) in LC was first described in 1998 in patient refractory to 5-aminosalicylate therapy (5-ASA) [5]. To date, only one randomized double-blinded, placebo-controlled trial has been performed for the treatment of LC with budesonide EC [6]. Of the 41 patients with LC, 86% achieved a clinical response compared to 48% in the placebo group with an NNT of 3. Up to 46% of patients had a relapse after discontinuation of budesonide EC with a mean follow-up of 14 months. The use of mesalamine was shown to be effective in about 85% of patients, but no increased benefit with the addition of cholestyramine [7]. Limited by a small sample size, bismuth subsalicylate was found to have improvement in clinical symptoms versus placebo for LC [8].
As budesonide EC has become the mainstay of therapy for refractory LC, the ideal mechanism of therapeutic benefit is not well understood. Budesonide, in general, has a high first past hepatic metabolism, and the extended-release formulation was formulated to delay the dissolution of budesonide, targeting release primarily into the ileum and ascending colon [9]. Using pharmacoscintigraphy, the delivery and site of absorption for budesonide EC was primarily in the ileum and ascending colon. Up to 71% absorption was demonstrated in the ileocolonic region in Crohn’s disease patients and healthy volunteers [10]. It is hard to ascertain the pharmacological benefit of budesonide EC as lymphocytic colitis tends to have a more distal distribution within the colon [11].
To the best of our knowledge, this is the first report of budesonide MMX being used in the treatment of lymphocytic colitis. Budesonide MMX formulation includes a budesonide-containing lipophilic matrix which allows for the release of budesonide throughout the colon, as opposed to isolated release in the ileocolonic region [12]. With pharmacoscintigraphy, up to 96% of its absorption occurred in the region between the ascending and descending/sigmoid colon, illustrating its suitability for colonic delivery for LC [13].
We suspect the MMX formulation of budesonide may be more efficacious in the treatment of lymphocytic colitis than the extendedrelease. Based on the pharmacokinetics of budesonide MMX, it appears to have a more targeted area of therapy for LC. Further randomized controlled-studies are needed in order generalize the benefit of budesonide MMX for the treatment of lymphocytic colitis.
References
- Agnarsdottir M, Gunniaugsson O, Orvar KB, Cariglia N, Birgisson S, et al. (2002) Collagenous and lymphocytic colitis in Iceland. Dig Dis Sci. 47: 1122-1128.
- Tong J, Zheng Q, Zhang C, Lo R, Shen J, et al. (2015) Incidence, prevalence, and temporal trends of microscopic colitis: a systematic review and meta-analysis. Am J Gastroenterol 110: 265-276.
- Rasmussen MA, Munck LK (2012) Systematic review: are lymphocytic colitis and collagenous colitis two subtypes of the same disease - microscopic colitis? Aliment Pharmacol Ther 36: 79-90.
- Temmerman F, Baert F (2009) Collagenous and lymphocytic colitis: systematic review and update of the literature. Dig Dis 27: 137-145.
- Van Gossum A, Schhmit A, Peny MO (1998) Oral budesonide for lymphocytic colitis. Am J Gastroenterol 93: 270.
- Miehlke S, Madisch A, Karimi D, Wonschik S, Kuhlisch E, et al. (2009) Budesonide is effective in treating lymphocytic colitis: a randomized double-blind placebo-controlled study. Gastroenterology 136: 2092-2100.
- Calabrese C, Fabbri A, Areni A, Zahlane D, Scialpi C, et al. (2007) Mesalazine with or without cholestyramine in the treatment of microscopic colitis: randomized controlled trial. J Gastroenterol Hepatol 22: 809-814
- Fine K, Ogunji F, Lee E, Lafon G, Tanzi M (1999) Ranndomized, double-blind, placebo-controlled trial of bismuth subsalicylate for microscopic colitis. Gastroenterology 116: G3825.
- Edsbacker S, Andersson T (2004) Pharmacokinetics of Budesonide (Entocort EC) Capsules for Crohn's Disease. Clin Phamacokinet 43: 803-821.
- Edsbacker S, Bengtsson B, Larsson P, Lundin P, Nilsson A, et al. (2003) A pharmacoscintigraphic evaluation of oral budesonide given as controlled-release (Entocort) capsules. Aliment Pharmacol Ther 17:525-536.
- Thijs WJ, van Baarlen J, Kleibeuker JH, Kolkman JJ (2005) Microscopic colitis: prevalence and distribution throughout the colon in patients with chronic diarrhoea. Neth J Med 63:137-140.
- (2014) Inc. S. Uceris(budesonide) extended release tablets, for oral use: prescribing information.
- Brunner M, Ziegler S, Di Stefano AF, Dehghanyar P, Kletter K, et al. (2006) Gastrointestinal transit, release and plasma pharmacokinetics of a new oral budesonide formulation. Br J Clin Pharmacol 61:31-38.
Relevant Topics
- Constipation
- Digestive Enzymes
- Endoscopy
- Epigastric Pain
- Gall Bladder
- Gastric Cancer
- Gastrointestinal Bleeding
- Gastrointestinal Hormones
- Gastrointestinal Infections
- Gastrointestinal Inflammation
- Gastrointestinal Pathology
- Gastrointestinal Pharmacology
- Gastrointestinal Radiology
- Gastrointestinal Surgery
- Gastrointestinal Tuberculosis
- GIST Sarcoma
- Intestinal Blockage
- Pancreas
- Salivary Glands
- Stomach Bloating
- Stomach Cramps
- Stomach Disorders
- Stomach Ulcer
Recommended Journals
Article Tools
Article Usage
- Total views: 12190
- [From(publication date):
April-2016 - Aug 17, 2025] - Breakdown by view type
- HTML page views : 11205
- PDF downloads : 985