Review Article Open Access
Oral Lichen Planus: A Review of Etiopathogenesis, Clinical, Histological and Treatment Aspects
Ravi Teja Chitturi1*, A Santha Devy2, R Madhavan Nirmal3 and PM Sunil4
1Department of Oral Pathology, SIBAR Institute of Dental Sciences, Guntur, Andhra Pradesh, India
2Department of Oral Pathology, Indira Gandhi Institute of Dental Sciences, Puducherry, India
3Department of Oral Pathology, Rajah Muthiah Dental College and Hospital, Annamalai Nagar, Tamil Nadu, India
4Department of Oral Pathology, Sree Anjaneya Institute of Dental Sciences, Calicut, Kerala, India
- Corresponding Author:
- Dr. Ravi Teja Chitturi
Senior Lecturer
Department of Oral Pathology
SIBAR Institute of Dental Sciences
Takkellapadu, Guntur – 522 509, India
Tel: +91-9676767387
E-mail: dr.raviteja@gmail.com
Received date: July 4, 2014; Accepted date:August 4, 2014; Published date: August 11, 2014
Citation:Chitturi RT, Devy AS, Nirmal RM, Sunil PM (2014) Oral Lichen Planus: A Review of Etiopathogenesis, Clinical, Histological and Treatment Aspects. J Interdiscipl Med Dent Sci 2:142. doi:10.4172/2376-032X.1000142
Copyright: © 2014 Chitturi, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at JBR Journal of Interdisciplinary Medicine and Dental Science
Abstract
It is a well known fact that oral lichen planus (OLP) is a non infectious disease affecting the oral mucous membrane. It is considered to be an autoimmune disorder mediated mainly by the T-lymphocytes. It affects 1-2% of the general population with maximum prevelance seen among women above the age of 40. WHO considers it to be a potentially malignant disorder and the rate of malignant transformation has been put between 0.5-2%. This article mainly reviews the various pathogenetic mechanisms by which this unique disorder occurs along with the clinical, histopathological and treatment aspects.
Keywords
Oral lichen planus; Etiopathogenesis; Autoimmune disorder
Introduction
Lichen planus (LP) is a non infectious disease that mainly affects the skin and mucous membrane of the oral cavity, oesophagus & genitals. It was first described and named by a famous British physician named Erasmus Wilson in 1869 [1]. The word ‘lichens’ means primitive plants composed of symbiotic algae and fungi and the term ‘planus’ is from Latin which means flat. Apart from the aforementioned sites this disease may also affect other parts of the body such as hair follicles, nails, larynx and eyes [2,3]. LP of the skin typically present as small (2 mm) pruritic, white to violaceous flat-topped papules, which can increase in size to as much as 3 cm. The following clinical presentations of LP of skin have been observed: annular, linear, atrophic, bullous, hypertrophic, ulcerated and pigmented. It is considered to have an acute progression and also a spontaneous remission. The skin lesions tend to affect the wrists, ankles and also the nails which can show pitting or permanent loss. The disease has a predilection for the flexor surfaces of the forearms and legs. Koebner’s phenomenon is commonly seen feature of LP [4,7].
The disease when it affects the oral cavity is termed oral lichen planus (OLP). Unlike the skin lesions, mucosal lesions follow a more chronic course. It follows a cyclic pattern of exacerbation or flares followed by quiescence. It has been observed that the oral lesions may persist upto 25 years. Flare-ups may be triggered by spicy and acidic foods and by stress. In the oral cavity, the buccal mucosa, tongue and gingiva are the most favoured sites whereas lesions of the palate are very rare. The lesions very occasionally seem to be unilateral or affect any one part of the oral cavity. It is usually bilateral or affects more than one part of the oral cavity and also may involve other mucous membranes [6].
Epidemiology
Various studies have been done across the world to record the incidence of OLP in various populations. In general it considered to affect about 1-2% of the general adult population, although young adults and children may also be affected [8-10]. The estimates of prevelance vary among different populations (0.5% in Japaneese, 1.9% in Swedish, 2.6% in Indian and 0.38% in Malay), but it does not appear to exhibit a racial predliction [11-12]. OLP has been described as a disease of the middle aged, predominantly in adults over the age of 40, and more common in women than men in ratio 1.4:1 [13,14]. It is considered to be the most common non infectious oral mucosal disease that is referred to dental clinics [15].
Clinical Features
OLP may present anywhere in the oral cavity. The buccal mucosa, tongue and gingival are the most common sites whereas palatal lesions are uncommon. They are usually symmetrical and bilateral lesions or multiple lesions in the mouth [15]. Andreasen in 1968 clinically divided OLP into six types: Papular, reticular, plaque type, atrophic, erosive and bullous. The reticular, papular and plaque-like forms are usually painless and appear clinically as white keratotic lesions. The erosive, atrophic and bullous forms are often associated with a burning sensation and in many cases can cause severe pain. These lesions occur singly or in combined forms [16].
The reticular type is the most common form of OLP. Characteristically, it presents as a series of fine, radiant, white striae known as ‘Wickham striae’, which may be surrounded by a discrete erythematous border. The buccal mucosa is the site most commonly involved. The striae are typically bilateral in a symmetrical form on the buccal mucosa. They may also be seen on the lateral border of the tongue and less often on the gingiva and the lips. Reticular lichen planus is likely to resolve in 41% of cases [17].
The papular form presents as small white pinpoint papules about 0.5 mm in size. It is rarely seen and because the lesions are small it is possible to overlook them during a routine oral examination [6].
The plaque type is seen as homogenous white patches. It may range from a slightly elevated, smooth patch to a slightly irregular form and may also be multifocal. The primary sites for this type are over the dorsum of the tongue and the buccal mucosa. They resolve by themselves in only 7% of cases. This form is significantly more common among tobacco smokers [16].
The atrophic type of OLP is diffuse, red and there are usually white striae around the lesion. Such striae that radiate peripherally are usually evident at the margins of the atrophic zones of the lesion. The attached gingiva is often involved and the condition is commonly referred to as ‘chronic desquamative gingivitis’. The atrophic form can display a symmetrical patchy distribution over all four quadrants. The lingual gingiva is usually less severely involved. This condition can cause a burning sensation particularly when in contact with certain foods. About 12% of atrophic lesions will resolve spontaneously [6].
Bullous OLP appears as small bullae or vesicles that tend to rupture easily. The bullae or vesicles range from a few millimeters to several centimeters in diameter. When they rupture they leave an ulcerated, painful surface. This form is rarer than the other forms of OLP. The bullous form is commonly seen on the buccal mucosa, particularly in the posteroinferior areas adjacent to the second and third molar teeth. The next most common site is the lateral margins of the tongue. The lesions are rarely seen on the gingiva or inner aspect of the lips [17].
Erosive OLP is the second most common type. The lesions are usually irregular in shape and covered with a fibrinous plaque or pseudomembrane where there is an erosion. The periphery of the lesion is usually surrounded by reticular or finely radiating keratotic striae. It is painful when the pseudomembrane or fibrinous plaque is disturbed. It is thought that erosive oral lichen planus has a greater potential to undergo malignant change along with atrophic type [18].
Association with Systemic Disorders
Various systemic disorders have been associated with OLP such as diabetes, hepatitis–C, and hypertension. Sometimes the appearance of OLP in diabetic patients has been attributed to the fact that drugs taken by these patients might induce lichenoid like reactions. Hypertensive drugs have also known to induce such lesions. This triad of OLP, diabetes mellitus and hypertension has been termed ‘‘Grinspan’s syndrome” in which the white lesions are due to the drugs used for the aforementioned systemic diseases.
Both cutaneous LP and OLP are associated with chronic liver disease secondary to HCV infection. Antibodies to the basal cells of all keratin producing epithelia have been expressed in chronic hepatitis B. Lichen planus may also associate with other immune mediated diseases including alopecia areata, dermatomyositis, lichen sclerosis atrophicus, morphea, myasthenia gravis, primary biliary cirrhosis, ulcerative colitis and vitiligo [5].
Etiology and Pathogenesis
OLP over the years has been enigma to pathologists as far as the etiology is concerned. Various factors have been proposed for the etiology including a genetic background, dental materials, drugs, infectious agents such as bacteria & viral, immunodeficiency, food allergies and stress. But recently authors have suggeted that it is T-cell-mediated chronic inflammatory oral mucosal disease [9].
The pathogenesis of OLP after its suggestion being a T-cell mediated autoimmune disorder has been described in detail and reviewed by various authors. The 4 chief mechanisms decribed are as follows:
•Cell-mediated immune response
•Non-specific mechanisms
•Autoimmune response
•Humoral immunity
Cell-mediated immune response
Though OLP is considered an autoimmune disease the precise antigen responsible for autoimmunity is yet to be identified. This hypothesis refers to the antigen as a self-peptide. This antigen may considered to be expressed in the kerationocytes due to variety of reasons such as a trauma or due to the presence of contact allergens such as a tooth paste or even a dental restorative material. It has also been hypothesized that these antigens can be activated by an infectious agent such as a virus or bacteria [15].
It has also been found that heat shock proteins (HSP) are upregulated in OLP and some authors feel that these might be the probable antigens for the immune response. But there are controversial studies which have proven that there is an over expression of these proteins due to variety of other exogenous agents such as drugs or allergens and as a result of final mechanism pathway for these agents OLP may develop. A dysregulation in the gene responsible for expressing these proteins in the epithelial cells or the inability by it to suppress an immune response following self-HSP recognition as a foreign substance is considered to provoke an immune response. MHC class I and II molecules activate the CD4+ T helper cells and CD8+ cytotoxic cells [15,19].
As the antigens presented MHC class II molecules are processed by endosomal cellular pathway and the antigens presented via the MHC class I molecules are processed through cytosolic cellular pathway, there must be more than one antigen involved in the pathogenesis of this disease. It can quite confidently be stated that cell mediated immunity appears to play a major role in the pathogenesis of OLP as either way T lymphocytes are involved in the course of the lesion which is also evident by immunohistochemical studies.
When it has been said that OLP is T cell mediated immune disease the sequence of events that occurs in such disorders can be described as follows,
•Migration of T lymphocytes into the epithelium;
•Activation of the T-lymphocytes;
•Killing of keratinocytes.
Migration of the T-lymphocytes
There are currently two hypothesis on how the T lymphocytes enter the oral epithelium. One theory states that T lymphocytes enter the epithelium as a routine surveillance measure. This theory has been named ‘chance encounter hypothesis’ as the antigens are recognised as a mere chance rather than anything else. The second theory postulates that the affected keratinocytes secretes certain cytokines that are responsible for drawing the attention of the T cells [13].
Activation of the T lymphocytes
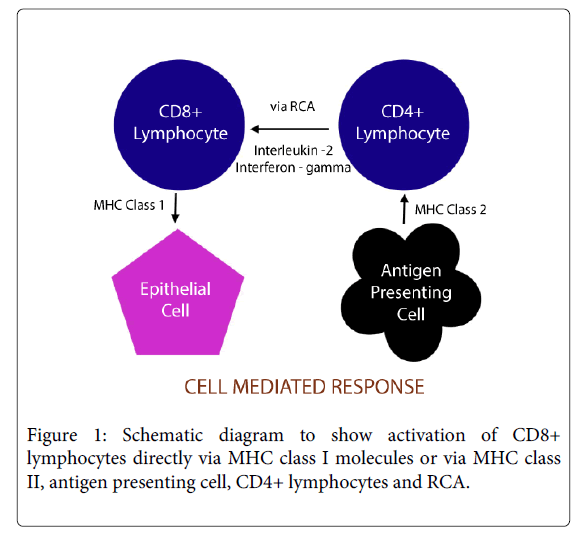
The ultimate result in OLP is the destruction of the basal keratinocytes by CD8+ cytotoxic cells. This may occur either by direct activation of CD8+ cytotoxic by MHC class I molecules expressed by the epithelial cells or via the CD4+ helper cells. The langerhans cells (LC) play a major role in the latter where they present the responsible antigen to the helper cells via the MHC class II molecules which in turn activate CD8+ cells via RCA (request cytotoxic activity) receptor (figure 1) and by the action of Interleukin-2 (IL2) and Interferon gamma (Ifn γ) [9].
Killing of the keratinocytes
The death of the basal keratinocytes is brought about by the cytotoxic T cells. It has been thought to induce apoptosis in the epithelial cells by activating the caspase cascade via various mechanisms such as by secreting TNF α and granzyme B or via the Fas ligand [15].
Non specific mechanisms in OLP
By immunocytochemical means it has been found that some of the T cells in the OLP lymphocytic infiltrate are not specific. They may be attracted to and retained within oral lichen planus lesions by various mechanisms associated with pre-existing inflammation which may be by the epithelial basement membrane, matrix metalloproteinases, chemokines or the mast cells.
It is a well known fact that the keratinocytes produce the basement membrane, and one of the functions of it is that it contains cell survival signals and inhibits apoptosis. When the cytotoxic T cells destroy the basal keratinocytes the basement membrane loses this ability to inhibit apoptosis and also attracts non specific T cells to the site [13].
The matrix metallo proteinases (MMP) especially MMP 9 is overexpressed in the lesional OLP tissues compared the control counterparts. The function of MMP 9 is to cleave type IV collagen. Thus its overexpression in OLP can be considered to have a non specific role in pathogenesis.
Chemokines are nothing but pro inflammatory cytokines, where RANTES (regulated on activation, normal T cell expressed and secreted) is one of the member of β-chemokine family which is produced by various cells. Activated lymphocytes and keratinocytes also produce cytokine like RANTES which recruits both acute and chronic inflammatory cells. It also stimulates mast cell degranulation in OLP which might in turn upregulate the RANTES secretion by the T cells. This cyclic mechanism might underlie the disease chronicity [9].
There is an increased mast cell density in OLP and about 60% of the cells undergo degranulation. Mast cell degranulation might result in release of a range of inflammatory mediators like TNF-α, Chymase and Tryptase. The mediators upregulate the endothelial adhesion molecule and also stimulates secretion of RANTES, which might further attract inflammatory cells which inturn leads to disease chronicity. Chymase a mast cell protease activates the MMP-9 which results in basement membrane disruption in the lesional area [19].
Autoimmunity
Various clinical parameters of OLP support the hypothesis that it is an autoimmune disorder, like disease’s chronicity, adult onset, female predilection, associated with other autoimmune disease, occasional tissue type association, depressed immune suppressor activity & the presence of auto-cytotoxic T cell clones in lichen planus lesion.
TGF β1 has been found to have an immunosupressive effect against self antigens. Also its deficiency in various studies has proved that it predisposes the individual to autoimmune diseases. Its underexpression in OLP tissues has been observed thus hypothesizing that OLP can be an autoimmune disorder [13].
Keratinocytes in oral lichen planus show increased expression of heat shock proteins. The upregulation may be triggered by drugs, infections, bacterial products and trauma. T cells proliferate in response to these proteins. Heat shock proteins are suspected to be autoantigenic for oral lichen planus, thus supporting the autoimmune hypothesis.
The LC mediated autoimmune hypothesis is a widely accepted theory where the apoptotic bodies of the epithelial cells are phagocytosed by the LC’s which activate the T helper cells and which inturn activate the CD8+ cytoxic cells, thus producing more cell death [15].
Humoral immunity
Circulating antibodies have been identified including autoantibodies against desmogleins 1 and 3 in OLP which indicates a role of humoral immunity in OLP [20]. But further studies are needed to know the exact role of humoral immunity in the pathogenesis of OLP.
Histopathologic Features of OLP
The features are similar to those of cutaneous lichen planus, and show focal parakeratosis, acanthosis, thickening of the granular cell layer, basal cell liquefaction degeneration, and blunted rete ridges. In skin lesions the rete ridges have a ‘saw tooth’ appearance. The characteristic histopathological feature of OLP is the presence of a band of dense sub-epithelial lympho-histiocytic infiltrate, penetration of lymphocytes intraepithelially and degeneration of basal keratinocytes. Degenerating basal keratinocytes form inclusion bodies which appear as homogenous eosinophilic masses and are termed a variety of names such as colloid, civatte, hyaline, or cytoid bodies. Another feature of the OLP is the presence of degenerated basement membrane. Degeneration of basal keratinocytes and disruption of the epithelial basement membrane and basal keratinocytes anchoring elements produce weakness at the epithelial-connective tissue interface which may result in histological cleft formation (Max-Joseph space) and clinical blistering of the oral mucosa (bullous lichen planus). B cells and plasma cells are infrequent in OLP. Direct immunofluorescence studies show presence of immunoglobulins IgA, IgG, and IgM. These features suggest a cell-mediated immune response [6,21].
Diagnosis of OLP
Various diagnostic features have been described over the years. It must be noted that diagnosis of OLP cannot be made on strictly clinical or histological grounds alone. A combined clinical and histopathological investigation is required for making a diagnosis. The widely used definition for the diagnosis of OLP was the criteria introduced by World Health Organization (WHO) in 1978.
World Health Organization diagnostic criteria (1978) of oral lichen planus (OLP) [22]
Clinical criteria
•Presence of white papule, reticular, annular, plaque-type lesions, gray-white lines radiating from the papules.
•Presence of a lace-like network of slightly raised gray-white lines (reticular pattern).
•Presence of atrophic lesions with or without erosion, and also in the form of bullae.
Histopathologic criteria
•Presence of thickened ortho or parakeratinized layer in sites with normally keratinized mucosa, and if site normally is nonkeratinized this layer may be very thin.
•Presence of civatte bodies in basal layer, epithelium and superficial part of the connective tissue.
•Presence of a well-defined band like zone of cellular infiltration that is confined to the superficial part of the connective tissue, consisting mainly of lymphocytes.
•Signs of ‘liquefaction degeneration’ in the basal cell layer.
Management
In all OLP patients, it is important to remove local exacerbating factors. The teeth should be scaled to remove plaque and calculus, and the patient should be instructed in thorough oral hygiene. Teeth associated with oral lesions should be examined and sharp cusps or edges reduced. Dental restorations associated with oral lesions should be mirror polished or replaced if contact sensitivity is suspected [10].
Many drugs have been tried, including corticosteroids, retinoids and cyclosporine. Corticosteroids have been found to be the most useful agent in the treatment of oral lichen planus. They can be applied topically on the lesion and can also be administered systemically. The topical application of a steroid seems to be safer when applied to mucous membranes. Though its use must be carefully observed for patients may develop candidiasis if used for a prolonged time and also may develop a potential for adrenal suppression. Retinoids were first used for the treatment of asymptomatic, white, reticulated oral lichen planus. In a study 71% of atrophic and erosive lesions had improved, whereas the other lesions were unchanged or worse. The systemic use of vitamin A is limited because of its toxicity and side-effects including skin dryness and hair loss [23,24].
Damage to the basement membrane in oral lichen planus is the result of the production of lymphokines such as interferon gamma by activated T lymphocytes. Cyclosporin is an immunosuppressant and reduces the production of lymphokines. It inhibits the proliferation and function of T lymphocytes. Its main adverse reaction is renal dysfunction as a result of prolonged use, so patients taking cyclosporin need to be monitored closely [21].
Cryosurgery and carbon dioxide laser ablation have been suggested for the surgical treatment of OLP. However, excision should not be a primary method of treatment as it is an inflammatory condition that can recur. In addition, surgical management is not suitable for the erosive and atrophic types because the surface epithelium is eroded. Surgical treatment is more applicable to the plaque like lesions, because the affected surface epithelium can be removed easily [6].
Malignant Transformation
OLP has moved from being regarded as an innocuous benign condition to being defined as a potentially malignant disorder by the world health organization (2005). WHO defines OLP as the risk of malignancy being present in a lesion or condition either at time of initial diagnosis or at a future date and included the oral lesions of lichen planus in its classification as a potentially malignant disorder [25]. Numerous reports have been published assessing the malignant potential of OLP. In general a 0.5 – 2 % chance of OLP turning into oral squamous cell carcinoma has been described [26-31].
Summary
OLP is a non infectious disease that affects the oral mucous membrane. It is seen mainly in women and people in the age group above 40. Though many etiological factors have been described over the years, it is primarily considered to be a T cell mediated autoimmune disorder in the the basal keratinocytes are attacked by the lymphocytes. OLP is considered a potentially malignant disorder. The risk of mailgnant transformation is considered to be between 0.5 – 2 % and therefore regular and periodic checkups are advised for patients and addressing the etiology and treating according is very essential.
References
- Wilson E (1869) On lichen planus. J Cutan Med Dis Skin 3:117-132.
- Bajaj DR, Khoso NA, Devrajani BR, Matlani BL, Lohana P (2010) Oral lichen planus: a clinical study. J Coll Physicians Surg Pak 20: 154-157.
- Farhi D, Dupin N (2010) Pathophysiology, etiologic factors, and clinical management of oral lichen planus, part I: facts and controversies. ClinDermatol 28: 100-108.
- Freedber IM (2033) Fitzpatrick's dermatology in general medicine. (6th edn),McGraw-Hill, New York.
- Lozada-Nur F, Miranda C (1997) Oral lichen planus: epidemiology, clinical characteristics, and associated diseases. SeminCutan Med Surg 16: 273-277.
- Zakrzewska JM (2001) Re: Mollaoglu N. Oral lichen planus: a review. Br J Oral MaxillofacSurg 2000; 38: 370-377. Br J Oral MaxillofacSurg 39: 407.
- Berger, James WD, Elston DM, Timothy G. Andrews' Diseases of the skin : clinical dermatology. (11th edn), Saunders/ Elsevier, London.
- Sousa FA, Rosa LE (2008) Oral lichen planus: clinical and histopathological considerations. Braz J Otorhinolaryngol 74: 284-292.
- Roopashree MR, Gondhalekar RV, Shashikanth MC, George J, Thippeswamy SH, et al. (2010) Pathogenesis of oral lichen planus--a review. J Oral Pathol Med 39: 729-734.
- Sugerman PB, Savage NW, Zhou X, Walsh LJ, Bigby M (2000) Oral lichen planus. ClinDermatol 18: 533-539.
- Carrozzo M, Thorpe R (2009) Oral lichen planus: a review. Minerva Stomatol 58: 519-537.
- Ismail SB, Kumar SK, Zain RB (2007) Oral lichen planus and lichenoid reactions: etiopathogenesis, diagnosis, management and malignant transformation. J Oral Sci 49: 89-106.
- Lodi G, Scully C, Carrozzo M, Griffiths M, Sugerman PB, et al. (2005) Current controversies in oral lichen planus: Report of an international consensus meeting. Part I. Viral infections and etiopathogenesis. Oral Surg Oral Med Oral Pathol Oral RadiolEndod 100: 40-51.
- Sugerman PB, Savage NW (2002) Oral lichen planus: causes, diagnosis and management. Aust Dent J 47: 290-297.
- Sugerman PB, Savage NW, Walsh LJ, Zhao ZZ, Zhou XJ, et al. (2002) The pathogenesis of oral lichen planus. Crit Rev Oral Biol Med 13: 350-365.
- Andreasen JO (1968) Oral lichen planus. 1. A clinical evaluation of 115 cases. Oral Surg Oral Med Oral Pathol 25: 31-42.
- Canto AM, Müller H, Freitas RR, Santos PS (2010) Oral lichen planus (OLP): clinical and complementary diagnosis. An Bras Dermatol 85: 669-675.
- Gorsky M, Raviv M, Moskona D, Laufer M, Bodner L (1996) Clinical characteristics and treatment of patients with oral lichen planus in Israel. Oral Surg Oral Med Oral Pathol Oral RadiolEndod 82: 644-649.
- Zhou XJ, Sugerman PB, Savage NW, Walsh LJ, Seymour GJ (2002) Intra-epithelial CD8+ T cells and basement membrane disruption in oral lichen planus. J Oral Pathol Med 31: 23-27.
- Lukac J, Brozovic S, Vucicevic-Boras V, Mravak-Stipetic M, Malenica B, et al. (2006) Serum autoantibodies to desmogleins 1 and 3 in patients with oral lichen planus. Croat Med J 47: 53-58.
- Eisen D (2002) The clinical features, malignant potential, and systemic associations of oral lichen planus: a study of 723 patients. J Am AcadDermatol 46: 207-214.
- Rad M, Hashemipoor MA, Mojtahedi A, Zarei MR, Chamani G, et al. (2009) Correlation between clinical and histopathologic diagnoses of oral lichen planus based on modified WHO diagnostic criteria. Oral Surg Oral Med Oral Pathol Oral RadiolEndod 107: 796-800.
- Lodi G, Scully C, Carrozzo M, Griffiths M, Sugerman PB, et al. (2005) Current controversies in oral lichen planus: Report of an international consensus meeting. Part II.Clinical management and malignant transformation. Oral Surg Oral Med Oral Pathol Oral RadiolEndod 100:164-178.
- Vincent SD, Fotos PG, Baker KA, Williams TP (1990) Oral lichen planus: the clinical, historical, and therapeutic features of 100 cases. Oral Surg Oral Med Oral Pathol 70: 165-171.
- George A, Sreenivasan BS, Sunil S, Varghese SS, Thomas J (2011) Potentially malignant disorders of oral cavity. Oral & Maxillofacial Pathology Journal 2: 95-100.
- Carrozzo M, Carbone M, Gandolfo S, Valente G, Colombatto P, et al. (1997) An atypical verrucous carcinoma of the tongue arising in a patient with oral lichen planus associated with hepatitis C virus infection. Oral Oncol 33: 220-225.
- Silverman S Jr, Gorsky M, Lozada-Nur F (1985) A prospective follow-up study of 570 patients with oral lichen planus: persistence, remission, and malignant association. Oral Surg Oral Med Oral Pathol 60: 30-34.
- Lo Muzio L, Mignogna MD, Favia G, Procaccini M, Testa NF, et al. (1998) The possible association between oral lichen planus and oral squamous cell carcinoma: a clinical evaluation on 14 cases and a review of the literature. Oral Oncol 34: 239-246.
- Gandolfo S, Richiardic L, Carrozzo M, Broccolettia R, Carbonea M, et al. (2004) Risk of oral squamous cell carcinoma in 402 patients with oral lichen planus: a follow-up study in an Italian population. Oral Oncology 40:77–83.
- van der Meij EH, Mast H, van der Waal I (2007) The possible premalignant character of oral lichen planus and oral lichenoid lesions: a prospective five-year follow-up study of 192 patients. Oral Oncol 43: 742-748.
- Bermejo-Fenoll A, Sanchez-Siles M, López-Jornet P, Camacho-Alonso F, Salazar-Sanchez F (2009) Premalignant nature of oral lichen planus. A retrospective study of 550 oral lichen planus patients from south-eastern Spain. Oral Oncology 45: 54–56.
Relevant Topics
- Cementogenesis
- Coronal Fractures
- Dental Debonding
- Dental Fear
- Dental Implant
- Dental Malocclusion
- Dental Pulp Capping
- Dental Radiography
- Dental Science
- Dental Surgery
- Dental Trauma
- Dentistry
- Emergency Dental Care
- Forensic Dentistry
- Laser Dentistry
- Leukoplakia
- Occlusion
- Oral Cancer
- Oral Precancer
- Osseointegration
- Pulpotomy
- Tooth Replantation
Recommended Journals
Article Tools
Article Usage
- Total views: 16474
- [From(publication date):
October-2014 - Aug 30, 2025] - Breakdown by view type
- HTML page views : 11560
- PDF downloads : 4914