Review Article Open Access
Pain Assessment and Treatment Guidelines across Addiction Status
Lacy A. Fabian1*, Meridith H Thanner1 and Katherine Kim21Health and Analytics, Battelle, Baltimore, MD 21209, USA
2School of Pharmacy, Notre Dame of Maryland University, Baltimore, USA
- Corresponding Author:
- Lacy A. Fabian
Health and Analytics, Battelle, Baltimore, MD 21209, USA
Tel: +1-410-372-2731
Fax: +1-614-485-0661
E-mail: FabianL@Battelle.org
Received date: March 14, 2014; Accepted date: April 23, 2014; Published date: April 30, 2014
Citation: Fabian LA, Thanner MH, Kim K (2014) Pain Assessment and Treatment Guidelines across Addiction Status. J Addict Res Ther 5:182. doi:10.4172/2155-6105.1000182Copyright: © 2014 Fabian LA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Addiction Research & Therapy
Abstract
Chronic pain is one of the most widespread and costly complaints facing healthcare today. In developing appropriate care plans for managing chronic pain it is critical to adequately assess risk factors that may alter the course of care. Notably, addiction operates along many of the same pathways as pain and can serve as a moderating factor in caring for those in pain (i.e., considering medications to prescribe for the management of pain in those with an active addiction) or a mediating factor whereby a new addiction develops in the course of managing the pain. This review examines addiction risk assessment tools specifically in the realm of alcohol, opioid use, and tobacco, which can be used in developing chronic pain care plans. Though the importance of assessing risk factors associated with addiction is well recognized, given the implications for the management of chronic pain, the means for adequately utilizing the assessments are limited by a lack of standardization and even barriers in the healthcare setting, such as time with which to perform the assessments. Efforts to develop psychometrically strong risk assessments tailored for use in various healthcare settings that allow for stratification into levels of risk and also employ strategies beyond mere subjective self-report are needed. As pain is such a prevalent experience and is considered a vital sign of health status, appropriate pain management is critical to managing healthcare costs and preventing undue stress on individuals, particularly those with various addictions.
Keywords
Pain assessment; Addiction; Treatment guidelines; Substance use disorder; Tobacco; Alcohol; Opioids
Introduction
Chronic pain is one of the most widespread complaints in healthcare settings [1]. It is both a persistent and costly concern, especially among those with a substance use disorder (SUD). Among this group, however, the coexistence of these disorders often remains inadequately assessed and treated, often because medical providers are ill equipped to evaluate substance use and abuse in pain patients [2]. Prevalence of chronic pain in this group is estimated to be higher than in the general population [3,4] with more than a third of the population reporting comorbid pain and substance abuse. Potter and colleagues [5] found, for example, in a multi-site treatment outcome study that one forth to one third of people seeking treatment for SUDs experienced moderate to severe chronic pain in the previous year. Their findings show the presence of a sizable proportion of patients with both SUD and pain who would benefit from the use of a valid risk assessment in developing their pain management care plan.
Chronic Pain Experience in Addiction
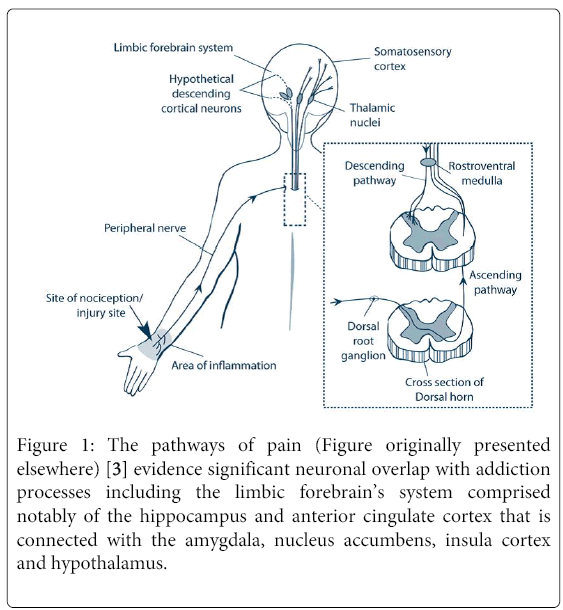
Substance use and pain are interrelated, with each experience influencing the persistence and treatment of the other [4]. Dependence on alcohol, drugs, and tobacco, for example, and chronic pain share common neural circuits, with the same pathways and brain regions, such as the anterior cingulate cortex [6], that regulate addiction also mediating chronic pain (Figure 1). It is anticipated that pain could affect substance use patterns while dependence conversely could influence pain sensitivity [7]. Thus, effective management of either condition requires that issues associated with both be simultaneously addressed [3,8].
Figure 1: The pathways of pain (Figure originally presented elsewhere) [3] evidence significant neuronal overlap with addiction processes including the limbic forebrain’s system comprised notably of the hippocampus and anterior cingulate cortex that is connected with the amygdala, nucleus accumbens, insula cortex and hypothalamus.
Given the interplay between the pathways that regulate addiction and pain, dissecting “true pain from drug-seeking behaviour” to properly manage pain in patients with a comorbid SUD can be challenging [9]. There is great overlap in physiologic states between addiction and pain in patients with SUD, and this can affect how pain is processed and tolerated, as having both conditions coexist can lead to a reorganization of the brain’s baseline perceptual pathways. SUD patients, for instance, have been found to have levels of pain tolerance different from, greater than or less than, those not with an addiction [4]. This line of research related to understanding the neuro-biologic interactions between chronic pain and SUD is still developing and a review of current practice and guidelines in support of producing more effective care plans for managing chronic pain is warranted.
Pain Assessment Guidelines
In addition to better identifying how to most effectively and efficiently assess and treat the SUD population also presenting with chronic pain, a gap in understanding exists in how such assessments and care plans should be modified to improve outcomes given individual patient characteristics such as age, gender, race/ethnicity, etc. Despite a recognition by researchers and practitioners that pain and responses to pain vary by such factors as gender, age, and other health/bio-psychosocial factors and thus, that the consideration of individual characteristics matters [3,10-14] – when looking at pain and SUD independently as well as together – research, practice, and implementation has been mixed.
Pain Assessment Guidelines
In addition to better identifying how to most effectively and efficiently assess and treat the SUD population also presenting with chronic pain, a gap in understanding exists in how such assessments and care plans should be modified to improve outcomes given individual patient characteristics such as age, gender, race/ethnicity, etc. Despite a recognition by researchers and practitioners that pain and responses to pain vary by such factors as gender, age, and other health/bio-psychosocial factors and thus, that the consideration of individual characteristics matters [3,10-14] – when looking at pain and SUD independently as well as together – research, practice, and implementation has been mixed.
A better understanding of these mechanisms overall, as well as by individual differences, is needed to inform assessments and interventions designed to alleviate both pain as well as treat coexisting addiction. In particular, an understanding of the various pathways between pain and addiction (addiction anteceding or following the onset of chronic pain) and how these may differ based on individual characteristics, has implications for how best to assess and then treat these comorbid conditions within different groups of patients. The literature to date is mixed [4,15-17] and the present review examines the current state of the field, including a review of prevalence rates, assessment tools, and treatment guidelines for alcohol, opioid and tobacco addiction’s implications for chronic pain within a bio-psychosocial framework (Table 1).
| Screening Tools | Assessment | Scoring | Pros | Cons | |
|---|---|---|---|---|---|
| Opioids | |||||
| Screening Tool for Addiction Risk (STAR) [2] | Self- administered survey tool to help identify risk for addiction in chronic pain patient receiving opioids treatment | 14 true-or-false questions | Self- administered questionnaires | Does not detect active substance abuse in patient with chronic pain | |
| Derived from validated tests such as MAST and CAGE | Not generalizable; sampling bias | ||||
| Tobacco abuse as a possible predictor for active substance abuse | No prospective follow up of patients | ||||
| Pain Medication Questionnaire (PMQ) [40] | An ongoing tool to identify patients on a range of potential risk factors through self-reports of addiction related behaviors | 26 questions represented on a 5-point Likert scale format | Good reliability and predictive validity | It is not a diagnostic tool | |
| Cut off point at 22 PMQ, suitable to separate between patients at risk | Prospective follow up in 2 week and retested: results show very strong correlation | Risk for false positives (56%) at cut-off score of 22. | |||
| Patient classification: High-PMQ or Low-PMQ groups | Validity of PMQ was assessed by correlation to Portenoy’s Criteria (PC) | Not generalizable: Translation and cultural difference between the Danish and American population may require different cut-off points for pain. | |||
| A strong tool to screen at risk patient, follow up, and intervene to manage therapy | Samples in the study was a mixture of two pain populations: cancer and non-cancer pain | ||||
| Screener and Opioid Assessment for Patient with Pain (SOAPP) | A self-reported tool to predict aberrant medication related behaviors among chronic pain patient | 14-item self-reported questionnaire | N/A | Scoring difficulties due to only 14-items | |
| Patient’s easily categorized into high-risk category | |||||
| Screener and Opioid Assessment for Patient with Pain –Revised (SOAPP-R) [46] | A risk assessment tool to helps predicts possible opioid abuse in patient with chronic pain | 24-item self- reported questionnaire | Addressed limitation in original SOAPP | Tool is not applicable to every population: must be considering long-term therapy with opioids | |
| Greater sensitivity and specificity to prediction score | |||||
| Possible to detect potential risk early and intervene accordingly | |||||
| Distinguish risk groups between high and low risk patients | |||||
| Opioid Risk Tool (ORT) [47] | A self-administered risk assessment tool to help predict aberrant behaviors while on opioids therapy to manage chronic pain | Potential risk factors were awarded a specific point value based responder’s sex. | Risk factors comparable to what is found in scientific literature for substance abuse | Small sample size relative to the number of risk factors | |
| The total score used to stratified patient into three risk groups: Low (0-3), Moderate (4-7), and High (> 8) | Prospective follow up of cohort for 12 months after initial visit. | Tested in only one clinic site, thus may not be universally applicable | |||
| Greatly distinguish risk between high and low risk patients, and between males and females | Clinicians who recorded patient’s aberrant behavior were not blinded to patient’s ORT score. | ||||
| Diagnosis, Intractability, Risk, Efficacy (DIRE) Score [34] | A risk assessment tool to help identify chronic pain patients at risk for addiction while receiving opioids treatment, and help assess whether patient will remain compliant to long-term opioids therapy | Scoring criteria consist of four main factors that are broken four subcategorizes | Strong correlation to compliance | Selective for primary care setting only to be used by trained clinician | |
| Scoring: Not a suitable candidate (7-13), Good candidate (14-21) | Allows for a rapid assessment of aberrant behavior | Moderate correlation to efficacy reported in study | |||
| Distinguishable risk groups between high and low risk patients | |||||
| Prospective follow up of cohort for over 12 months | |||||
| May be concurrently use or complement other documentation tools such as Pain Assessment and Documentation Tool (PADT) | |||||
| Alcohol | |||||
| CAGE Assessment [48] | A screening tool for alcohol use disorders (AUD) | 4-item questionnaire: Cut down, Annoyed, Guilty, Eye-Opener | May be self-administered | Does not differentiate between past and active alcohol abuse | |
| Scoring: Probable alcoholism (2+) | Completion Time: 30 seconds | Widely varying sensitivity based on the cut-off point used | |||
| Simple and easy to use | Sensitivity low in elderly and psychiatric population | ||||
| Identify life-time prevalence | |||||
| Adapted version to include risk for drug abuse: CAGE-AID | |||||
| Michigan Assessment Screening Test (MAST) [48] | A screening tool for alcohol abuse/dependence and hazardous drinking | 22-item questionnaire awarded specific point value based on yes or no response | Identify life-time prevalence | Varying specificity when cut off score lower than 5 | |
| Scoring: Indicate alcohol abuse/dependence (6+) | High sensitivity | Longer completion time: 5 minutes | |||
| High specificity with standard cut-off at 5 | |||||
| Different versions of the tool to assess elderly population: MAST-G, SMAST-G | |||||
| Alcohol Use Disorder Identification Test (AUDIT) [48] | A screening tool for alcohol abuse/dependence and hazardous drinking | 10-item questionnaire awarded specific point value | May be self-administered | AUDIT is less effective in elderly population | |
| Scoring: Indicate alcohol abuse/ dependence (8+) | Completion Time: 2-3 minutes | ||||
| Identify active and current alcohol abuse | |||||
| New version AUDIT-5: shown to outperform AUDIT and CAGE in elderly and psychiatric patients |
Table 1: Summary of initial screening tools used for risk assessment to establish addiction status in individuals presenting with chronic pain
Alcohol addiction
Prevalence: Based on findings from the National Epidemiologic Survey on Alcohol and Related Conditions Prevalence, prevalence of lifetime and 12-month alcohol abuse is 17.8% and 4.7% respectively and prevalence of lifetime and 12-month alcohol dependence is 12.5% and 3.8%. For respondents who have lifetime alcohol dependence, only 24.1% seek treatment for alcohol dependence. Additionally, respondents with lifetime alcohol dependence were likely to have fewer comorbid disabilities (social, emotional, or mental) compared to those who have never received treatment [18]. In another study, Sheu and colleagues [16] found that patients in an alcoholic outpatient treatment program were more likely to cite physical pain compared to other factors as the impetus for alcohol abuse during the prior three months. Of this group, only 13% were receiving any form of care for their chronic pain, though 72% expressed an interest in “treatment”. Given the association between chronic pain and alcohol abuse, and the belief that the majority of chronic pain problems will not resolve while there is ongoing alcohol (or other drug) abuse [3,19], the ability for physicians to identify and simultaneously manage co-occurring pain and alcohol addiction is critical. With so few patients receiving adequate pain management, findings further support the need to better assess and address pain problems in this substance abusing population and to engage the healthcare team.
Treatment guidelines: It is the consensus of researchers that until alcohol abuse is brought under control, treatment of chronic pain will be ineffective. Once a patient presenting with chronic pain is assessed as having a coexisting SUD, it is recommended that s/he first be referred to an addiction treatment provider for treatment of their alcohol dependence while continuing under the care of their primary care physician for pain management [3]. However, most primary care physicians are ill equipped to adequately assess this, or any other, substance use disorder due to lack of available time to conduct screenings, knowing whether a screening is warranted for the particular patient, knowing the best screener to use, and administering the screener in way that is cost effective [20]. The culmination of these barriers can result in missed detection of an alcohol dependence problem altogether, thus further complicating treatment of chronic pain [21,22]. Moreover, individual characteristics with respect to the presentation, assessment, and treatment of both pain and alcohol addiction must be considered.
Assessment tools: When working with chronic pain patients, it is important to determine as early as possible in the development of a pain management care plan whether coexisting alcohol dependence exists. At present, the authors were unable to identify a single assessment tool that accomplished this end specifically for chronic pain patients. As such, practitioners must rely on a combination of assessment tools to help fully characterize an understanding of their patient’s condition [3]. Furthermore, as part of conducting the overall pain assessment, including SUD assessment, often based on patient self-reports, collected information is most beneficial when corroborated with other sources of information such as medical records, family interviews, lab work, etc. [23]. Without a valid assessment tool, it is difficult for a primary care physician to identify a patient with a coexisting SUD. For identifying the ‘unobvious’ alcohol dependent patient presenting with chronic pain, the CAGE (Cut-down, Annoyed, Guilty, Eye-Opener), consisting of only four questions, has been shown to be a valid and reliable screening tool for detection of problem alcohol use in such populations as general practice medical patients (inpatient and ambulatory), surgical inpatients, and psychiatric inpatients [24].
Evidence and recommendations: Broadly, research has found that patients who present with chronic and severe pain are significantly more likely to also present with a comorbid SUD. Sheu et al. [16], for example, further found that patients with coexisting pain and SUD also report that their physical pain led to their alcohol (and other substance) use. A few studies have examined comorbid chronic pain and alcohol abuse with a specific focus on examining individual characteristics. In an older investigation of patients with chronic lower back pain, compared on age, sex, civil status, and income to matched control groups, investigators found that alcohol abuse was significantly more frequent among the male patients [25]. In a more recent examination, Parks et al. [26], in an on-going study of phenotypes of alcohol dependence among Alaska Natives by gender, found that male and female subjects reported similar experiences with alcohol-related health problems, though women reported more chronic pain related complaints and complications. These reports of more pain symptoms by women, in addition to increased use of and increased negative general health consequences compared with men in this alcohol dependent sample, suggests additional considerations for treatment planning and intervention [26].
Opioid addiction
Prevalence: Misuse of opioids in the United States and abroad has reached epidemic proportions that is attributed to their availability and misconceptions on proper use [1,27,28] with estimates ranging from 14% to 19% [29]. Given the prevalence of chronic pain, it poses a challenge to clinicians of just how to provide pain relief and accurately account for existing addiction or addictive tendencies.
Treatment guidelines: Common guidelines with the most evidentiary support show a need for caution at doses greater than 90-200 mg of morphine equivalents per day, fentanyl patch risks, 25-50% dose reduction when switching opioids, as well as risk assessment tools, treatment agreements and urine drug assessments [30]. Treatment for co-occurring chronic pain and opioid use disorders only receives general guidance on what constitutes a valid risk assessment tool [27] with standard International Classification of Disease (ICD-10) criteria being the “gold standard” [29] focusing on tolerance, physical dependence, and withdrawal [1]; the criteria, however, may also need to include preoccupation with obtaining opioids, loss of control over use, and adverse consequences of use [1].
Assessment tools: Screening tools do exist that focus on the occurrence of prior substance abuse; however, this can fall short when the risk of developing a new opioid addiction during management of chronic pain is estimated between 3-12% [27]. A recent study comparing Portenoy’s criteria with the ICD-10 [29,31] found factors such as younger age, alcohol use, and higher prescription doses of opioids to be significant risk factors for opioid addiction when using either criteria. Other studies have also found tobacco use to be risk factor for substance abuse generally, and non-medical opioid abuse specifically [2,32]; depression and anxiety, in addition to tobacco use, have also been found predictive of opioid addiction [33]. Others [31] have combined existing assessment tools focusing on past drug and alcohol use, family and personal history of use, abuse and psychosis; aberrant behavior; and other pain treatment methods in order to develop a rapid assessment tool in ambulatory clinics, with the consistent finding that of primary importance was a space for clinicians to summarize overall risk level [32,33] and also tools to assess likely compliance such as the Diagnosis, Intractability, Risk and Efficacy (DIRE) score [34]. Additionally, the type of pain itself may also be relevant to risk of addiction with more severe illnesses, inability to easily access medications, and persistence in pursuing accurate treatment and diagnosis being associated with a lower prevalence of an SUD [15].
Evidence and recommendations: Long-term consequences of chronic opioid use to manage pain are still not well understood despite the widespread use with current guidelines focused on developing clear risk stratification and close monitoring [35]. Fear of addiction is commonly cited, particularly among the elderly, as a reason to not want opioid medication, which can provide relief to chronic pain. Continued efforts, such as those tailored for use with chronic pain populations [29] and pharmacokinetic-pharmacodynamic modelling [30,36], to validate assessment tools beyond just self-report, possibly with urinary drug assessments [37,38] to identify risk factors and create tailored treatment plans will come to provide assurance for those suffering chronic pain. In individuals recovering from addiction or at high-risk for addiction, as evidenced by risk factors warrant structure and monitoring, as well as a balanced treatment approach that focuses on overall risk and quality of life as opposed to just comfort [1,38].
Tobacco addiction
Prevalence: Although the overall smoking prevalence has declined from 2005 (20.9%), there are still an estimated 42.1 million (18.1%) adults who smoke in the United States. Smoking related deaths accounts for approximately 480,000 a year, or one in every five, while smoking related complications such as lung cancer and COPD is prevalent in about 16 million people in the U.S. [39] Given the current prevalence of smoking and the growing popularity of other tobacco products, it becomes more urgent to understand how tobacco dependence may impact the management of chronic pain or increase risk for other substance, namely opioid, abuse.
Treatment guidelines: Treatment guidelines for chronic pain across various organizations such as the American Chronic Pain Association (ACPA) and Institute of Medicine (IOM) focus more on smoking cessation as a means for better pain control and management. However, these guidelines do not recognize tobacco dependence as a possible predictor or risk factor to help identify or potentiate opioid abuse in chronic pain patients with or without addictive tendencies, as recent research may suggest is warranted. Several studies suggest that heavy smoking in chronic pain patients were indicative of future SUDs or addiction to opioids [17,40,41], while other studies suggest tobacco abuse as a predictor for substance abuse [42].
Assessment tools: Although there are many screening tools available to help assess future risk for addiction, many of these tools focus on personal and family history of abuse to alcohol and prescription and illicit drugs. Seemingly often these tools do not assess smoking history or status of the individual. Thus, screening tools for addiction risk (STAR) was derived from questions utilized in the Michigan Alcohol Screening Test (MAST) and CAGE to assess smoking history of an individual and prior treatment to substance abuse. Results show that smoking related questions listed in STAR help identify pain patients with substance abuse and addictive tendencies from pain patients without history of substance abuse. Furthermore, it is evident that prior treatment for SUD was as a potential risk factor in patients receiving opioids for chronic pain [42].
Evidence and recommendation: Current evidence suggests that nicotine exerts its effects on various neurobiological systems, which may alter pain perception and endogenous pain regulatory mechanisms. Chronic smokers may have a diminished physiological response to various stressors, often due to the absence of certain stress-induced analgesia and relatively low release of cortisol and ACTH, during high times of stress when compared to non-smokers. Due to this, chronic smokers may present with higher sensitivity to pain, which may often manifest as higher pain ratings [43]. These “blunted” responses to pain may pose a risk of over or under utilization of opioids or other medications to manage chronic pain. The lack of current screening tools that assess tobacco addiction in chronic pain populations, and the growing body of evidence that reveals smoking as a predictor or risk factor to substance abuse or opioid addiction, it is therefore recommended that inquiries of tobacco use be taken into account when developing risk assessment tools for chronic pain populations.
Conclusion
Alcohol, opioid, and tobacco use addictions have implications in the management of chronic pain, and their interrelation is an area in need of more standardized risk assessment protocols that move beyond mere self-report. Alcohol use can moderate and mediate the pain experience [16], yet, a risk assessment tool validated in the chronic pain population is not readily available. Likewise, not only are the long-term effects of opioid use not well understood [32,33,44], there is also concern of noncompliance and under-reported use [45]. Furthermore, tobacco use appears a critical addition to a risk assessment tool, given the implications for the pain experience and addiction potential. Healthcare facilities at all points of a patient treatment cycle such as ambulatory clinics, primary care, and pharmacies need access to tailored risk assessment tools that allow for summative assessment and stratification into risk levels. Ideally, such risk assessment tools will provide clinicians with rapid assessment options, with validated approaches, that rely on more than self-report and questions that recognize the overlap in underlying neural physiology and pathways.
References
- Miotto K, Kaufman A, Kong A, Jun G, Schwartz J (2012) Managing co-occurring substance use and pain disorders. PsychiatrClin North Am 35: 393-409.
- Friedman R, Li V, Mehrotra D (2003) Treating pain patients at risk: evaluation of a screening tool in opioid-treated pain patients with and without addiction. Pain Med 4: 182-185.
- Managing Chronic Pain in Adults With or in Recovery From Substance Use Disorders. Rockville, MD: Substance Abuse and Mental Health Services Administration, 2011 HHS Publication No. (SMA) 12-4671.
- Prince V (2001) Pain Management in Patients with Substance-Use Disorders. Lenexa KS (edn) Chronic Illnesses. American College of Clinical Pharmacotherapy.
- Potter JS, Prather K, Weiss RD (2008) Physical pain and associated clinical characteristics in treatment-seeking patients in four substances use disorder treatment modalities. Am J Addict. 17: 121-125.
- Weston CS (2012) Another major function of the anterior cingulate cortex: the representation of requirements. NeurosciBiobehav Rev 36: 90-110.
- Apkarian AV, Neugebauer V, Koob G, Edwards S, Levine JD, et al. (2013) Neural mechanisms of pain and alcohol dependence. PharmacolBiochemBehav 112: 34-41.
- Trafton JA, Oliva EM, Horst DA, Minkel JD, Humphreys K (2004) Treatment needs associated with pain in substance use disorder patients: implications for concurrent treatment. Drug Alcohol Depend 73: 23-31.
- Mitchell AM, Dewey CM (2008) Chronic pain in patients with substance abuse disorder: general guidelines and an approach to treatment. Postgrad Med 120: 75-79.
- Dixon KE, Thorn BE, Ward LC (2004) An evaluation of sex differences in psychological and physiological responses to experimentally-induced pain: a path analytic description. Pain 112: 188-196.
- Edwards RR, Fillingim RB (2001) Age-associated differences in responses to noxious stimuli. J Gerontol A BiolSci Med Sci 56: M180-185.
- Fabian LA1, McGuire L, Goodin BR, Edwards RR (2011) Ethnicity, catastrophizing, and qualities of the pain experience. Pain Med 12: 314-321.
- Fillingim RB, Edwards RR, Powell T (2000) Sex-dependent effects of reported familial pain history on recent pain complaints and experimental pain responses. Pain 86: 87-94.
- Geisser ME, Roth RS (1998) Knowledge of and Agreement with Chronic Pain Diagnosis: Relation to Affective Distress, Pain Beliefs and Coping, Pain Intensity, and Disability. Journal of Occupational Rehabilitation 8: 73-88.
- Proctor SL, Estroff TW, Empting LD, Shearer-Williams S, Hoffmann NG (2013) Prevalence of substance use and psychiatric disorders in a highly select chronic pain population. J Addict Med 7: 17-24.
- Sheu R, Lussier D, Rosenblum A, Fong C, Portenoy J, et al. (2008) Prevalence and characteristics of chronic pain in patients admitted to an outpatient drug and alcohol treatment program. Pain Med 9: 911-917.
- Ekholm O, Grønbaek M, Peuckmann V, Sjøgren P (2009) Alcohol and smoking behavior in chronic pain patients: the role of opioids. Eur J Pain 13: 606-612.
- Hasin DS, Stinson FS, Ogburn E, Grant BF (2007) Prevalence, correlates, disability, and comorbidity of DSM-IV alcohol abuse and dependence in the United States: results from the National Epidemiologic Survey on Alcohol and Related Conditions. Arch Gen Psychiatry 64: 830-842.
- Weaver MF, Schnoll SH (2002) Opioid treatment of chronic pain in patients with addiction. J Pain Palliat Care Pharmacother 16: 5-26.
- Screening for Alcohol Use and Alcohol Related Problems (2005) National Institute on Alcohol Abuse and Alcoholism (NIAAA), Alcoholism NIoAAa, Rockville, MD.
- Aertgeerts B, Buntinx F, Ansoms S, Fevery J (2002) Questionnaires are better than laboratory tests to screen for current alcohol abuse or dependence in a male inpatient population. ActaClinBelg 57: 241-249.
- Aertgeerts B, Buntinx F, Kester A (2004) The value of the CAGE in screening for alcohol abuse and alcohol dependence in general clinical populations: a diagnostic meta-analysis. J ClinEpidemiol 57: 30-39.
- Katz N, Fanciullo GJ (2002) Role of urine toxicology testing in the management of chronic opioid therapy. Clin J Pain 18: S76-82.
- Dhalla S, Kopec JA (2007) The CAGE questionnaire for alcohol misuse: a review of reliability and validity studies. Clin Invest Med 30: 33-41.
- Sandström J, Andersson GB, Wallerstedt S (1984) The role of alcohol abuse in working disability in patients with low back pain. Scand J Rehabil Med 16: 147-149.
- Parks CA, Hesselbrock MN, Hesselbrock VM, Segal B (2001) Gender and reported health problems in treated alcohol dependent Alaska natives. J Stud Alcohol 62: 286-293.
- Ling W, Mooney L, Hillhouse M (2011) Prescription opioid abuse, pain and addiction: clinical issues and implications. Drug Alcohol Rev 30: 300-305.
- Jamison RN, Edwards RR (2013) Risk factor assessment for problematic use of opioids for chronic pain. ClinNeuropsychol 27: 60-80.
- Højsted J, Nielsen PR, Guldstrand SK, Frich L, Sjøgren P (2010) Classification and identification of opioid addiction in chronic pain patients. Eur J Pain 14: 1014-1020.
- Nuckols TK, Anderson L, Popescu I, Diamant AL, Doyle B, et al. (2014) Opioid prescribing: a systematic review and critical appraisal of guidelines for chronic pain. Ann Intern Med 160: 38-47.
- Pink LR, Smith AJ, Peng PW, Galonski MJ, Tumber PS, et al. (2012) Intake assessment of problematic use of medications in a chronic noncancer pain clinic. Pain Res Manag 17: 276-280.
- Holliday S, Hayes C, Dunlop A (2013) Opioid use in chronic non-cancer pain--part 2: prescribing issues and alternatives. AustFam Physician 42: 104-111.
- Holliday S, Hayes C, Dunlop A (2013) Opioid use in chronic non-cancer pain--part 1: known knowns and known unknowns. AustFam Physician 42: 98-102.
- Belgrade MJ, Schamber CD, Lindgren BR (2006) The DIRE score: predicting outcomes of opioid prescribing for chronic pain. J Pain 7: 671-681.
- Cheatle M, Comer D, Wunsch M, Skoufalos A, Reddy Y (2014) Treating pain in addicted patients: recommendations from an expert panel. Popul Health Manag 17: 79-89.
- Yang H, Feng Y, Xu XS (2014) Pharmacokinetic and pharmacodynamicmodeling for acute and chronic pain drug assessment. Expert Opin Drug MetabToxicol 10: 229-248.
- Dupouy J, Mémier V, Catala H, Lavit M, Oustric S, et al. (2014) Does urine drug abuse screening help for managing patients? A systematic review. Drug Alcohol Depend 136: 11-20.
- Ballantyne JC (2013) Treating pain in patients with drug-dependence problems. BMJ 347: f3213.
- Current Cigarette Smoking Among Adults-USA, 2005–2012.
- Højsted J, Nielsen PR, Kendall S, Frich L, Sjøgren P (2011) Validation and usefulness of the Danish version of the Pain Medication Questionnaire in opioid-treated chronic pain patients. ActaAnaesthesiolScand 55: 1231-1238.
- Michna E, Ross EL, Hynes WL, Nedeljkovic SS, Soumekh S, et al. (2004) Predicting aberrant drug behavior in patients treated for chronic pain: importance of abuse history. J Pain Symptom Manage 28: 250-258.
- Jiang HJ, Friedman B, Jiang S (2013) Hospital cost and quality performance in relation to market forces: an examination of U.S. community hospitals in the "post-managed care era". Int J Health Care Finance Econ 13: 53-71.
- al'Absi M, Nakajima M, Grabowski J (2013) Stress response dysregulation and stress-induced analgesia in nicotine dependent men and women. Biological psychology 93:1-8.
- Marhe R, Luijten M , Franken IH (2014) The Clinical Relevance of Neurocognitive Measures in Addiction. Front Psychiatry 4: 185.
- Kipping K, Maier C, Bussemas HH, Schwarzer A (2014) Medication compliance in patients with chronic pain. Pain Physician 17: 81-94.
- Fulton AT, Rhodes-Kropf J, Corcoran AM, Chau D, Castillo EH (2011) Palliative care for patients with dementia in long-term care. ClinGeriatr Med 27: 153-170.
- Webster LR, Webster RM (2005) Predicting aberrant behaviors in opioid-treated patients: preliminary validation of the Opioid Risk Tool. Pain Med 6: 432-442.
- O'Connell H, Chin AV, Hamilton F, Cunningham C, Walsh JB, et al. (2004) A systematic review of the utility of self-report alcohol screening instruments in the elderly. Int J Geriatr Psychiatry 19: 1074-1086.
Relevant Topics
- Addiction Recovery
- Alcohol Addiction Treatment
- Alcohol Rehabilitation
- Amphetamine Addiction
- Amphetamine-Related Disorders
- Cocaine Addiction
- Cocaine-Related Disorders
- Computer Addiction Research
- Drug Addiction Treatment
- Drug Rehabilitation
- Facts About Alcoholism
- Food Addiction Research
- Heroin Addiction Treatment
- Holistic Addiction Treatment
- Hospital-Addiction Syndrome
- Morphine Addiction
- Munchausen Syndrome
- Neonatal Abstinence Syndrome
- Nutritional Suitability
- Opioid-Related Disorders
- Relapse prevention
- Substance-Related Disorders
Recommended Journals
Article Tools
Article Usage
- Total views: 15891
- [From(publication date):
April-2014 - Aug 30, 2025] - Breakdown by view type
- HTML page views : 11201
- PDF downloads : 4690