Review Article Open Access
Relationship between Years of Marijuana Use and the Four Main Diagnostic Criteria for Metabolic Syndrome among United States Adults
Barbara A Yankey1*, Richard Rothenberg2, Sheryl Strasser3, Kim Ramsey-White4 and Ike S Okosun11Georgia State University, School of Public Health, Division of Epidemiology and Biostatistics, 140 Decatur Street NE, Suite 848, Atlanta, GA 30303, USA
2Georgia State University, School of Public Health, Division of Research and Faculty Development, USA
3Tobacco Center of Regulatory Science, 140 Decatur Street NE., Suite 857 Urban Life Bldg, Atlanta, GA 30303, USA
4Georgia State University, School of Public Health, Division of Health Promotion and Behavior, 140 Decatur Street NE, Suite 848, Atlanta, GA 30303, USA
- Corresponding Author:
- Barbara A
Yankey, Division of Epidemiology and Biostatistics
Georgia State University School of Public Health, USA
Tel: 404-519-0105
Email: byankey1@student.gsu.edu
Received date: January 09, 2017; Accepted date: February 12, 2017; Published date: February 19, 2017
Citation: Yankey BA, Rothenberg R, Strasser S, White KR, Okosun IS (2017) Relationship between Years of Marijuana Use and the Four Main Diagnostic Criteria for Metabolic Syndrome among United States Adults. J Addict Res Ther S11:017. doi:10.4172/2155-6105.1000S11-017
Copyright: © 2017 Yankey BA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Addiction Research & Therapy
Abstract
Objective: Research on marijuana use suggests a protective effect on metabolic syndrome. National Cholesterol Education Program, Adult Treatment Panel III, World Health Organization, European Group for the study of Insulin Resistance and International Diabetes Federation has different criteria for metabolic syndrome. Definitions of both marijuana use and criteria for metabolic syndrome may influence the observed effects. We examine the relationship of years of marijuana use with the four common definitions of metabolic syndrome.
Method: This is a cross-sectional study of 3051 adults aged ≥ 20 years who participated in the National Health and Nutrition Examination Survey 2011-2012. Only participants who responded to the question, "Have you ever even once used marijuana or hashish?" were enrolled. Using multivariate logistic regression, we estimated odds ratios for metabolic syndrome with each year of marijuana use.
Results: Adjusted odds ratios (AOR) for having metabolic syndrome with each increase in year of marijuana use was 1.05 (95% CI: 1.02, 1.08) using National Cholesterol Education Program Adult Treatment Panel III (ATP III) criteria. Respective AOR using International Diabetes Federation (IDF) was 1.08 (95% CI: 1.04, 1.13) and 1.05 (95% CI: 1.04, 1.13) using World Health Organization (WHO) or European Group for the study of Insulin Resistance (EGIR) criteria. Using ATP III or IDF criteria, the adjusted odds ratio of having hypertension (AOR Hyp) for each year of marijuana use was 1.07 (95% CI: 1.03, 1.12). Using WHO criteria, AOR Hyp was 1.05 (95% CI: 1.01, 1.09) and 1.08 (95% CI: 1.03, 1.12) using EGIR. All the applicable criteria show increased odds for abdominal obesity: AOR 1.06 (95% CI: 1.00, 1.11) (ATP III), 1.09 (95% CI: 1.05, 1.14) (EGIR) or 1.07 (95% CI: 1.01, 1.13) (IDF). Adjusted odds ratio for having high oral glucose tolerance test levels was 1.12 (95% CI: 1.07, 1.18) using WHO and EGIR criteria.
Conclusion: Irrespective of the criteria for metabolic syndrome, each year of marijuana use showed increased odds of having metabolic syndrome, hypertension or high oral glucose tolerance test levels. This increased odd is in contrast to most findings in literature. The small, yet consistent increase in odds for hypertension was slightly higher than that observed with cigarette smoking. Recreational marijuana use may be detrimental to cardiovascular health. A standardized definition of marijuana use will be relevant for further investigation.
Keywords
Cannabis; Cardiovascular disease; Cigarette; Marijuana; Metabolic syndrome; Tobacco
Introduction
Marijuana is a psychoactive substance that induces relaxation and euphoria. Marijuana is classified as a schedule 1 drug by the drug enforcement administration (DEA) and is an illicit compound under federal law. However, by the end of election 2016, 28 states had legalized medical marijuana. Eight states and Washington DC also permit adult recreational marijuana use. Support for legalization of marijuana is on ascendancy [1]. Like cigarette, the main route of administration of marijuana is smoking and whereas the detrimental effect of tobacco/cigarette on cardiovascular health is established, that of marijuana is unknown.
Metabolic syndrome (MetS) is a constellation of cardiovascular risk factors and is a condition associated with detrimental cardiovascular prognosis. Because cardiovascular disease (CVD) is a leading cause of mortality worldwide [2], the prevalence of metabolic syndrome may be an important determinant of the health status of a nation. The prevalence of metabolic syndrome generally increases with age. During the period 2003 to 2012, metabolic syndrome prevalence in the United States (US) was about 18.0% among adults aged 20-39 years, 35.0% among adults aged 40-59 years and 46.7% among adults aged 60 years and above [3]. In 2012, an estimated 31.0% of all global deaths were due to CVDs [4]. Studies on tobacco and marijuana are inconclusive on their associations with metabolic syndrome and its components [2,5]. Metabolic syndrome has varying criteria. National Cholesterol Education Program, Adult Treatment Panel III (ATP), World Health Organization (WHO), European Group for the study of Insulin Resistance (EGIR) and International Diabetes Federation (IDF) have different criteria for metabolic syndrome. Definitions of both marijuana use and criteria for metabolic syndrome may influence the observed effects. We examine the relationship of years of marijuana use with the four common definitions of metabolic syndrome. Our hypothesis is that the definition used for metabolic syndrome may change the estimates of the associations between marijuana use and metabolic syndrome.
Literature Review
Metabolic syndrome is a co-occurrence of hypertension, hyperlipidemia, hyperglycemia and visceral obesity. Metabolic syndrome is associated with cardiometabolic pathology [6]. There is no unified definition [7,8] for MetS, however, the definition by the National Cholesterol Education Program, Adult Treatment Panel III (ATP III) is widely adopted because of its clinical applicability [8]. In accordance with ATP III, MetS is a co-occurrence of any three of the following: Hypertension, hyperglycemia, abdominal obesity, reduced high density lipoprotein cholesterol (HDL-C) or hypertriglyceridemia. By WHO standard, MetS is a diagnosis of diabetes or increased two hour oral glucose tolerance test (OGTT) or fasting insulin levels plus any two or more of the following: hypertension, obesity, high plasma triglycerides, low plasma HDL-C or albumin creatinine ratio ≥ 30. By EGIR criteria, MetS consists of fasting insulin level above 75th percentile of cohorts, and two or more of the following: hypertension, abdominal obesity, hypertriglyceridemia or low HDL-C. The IDF criteria require increases in ethnicity-specific waist circumference and any two or more of the following: hypertension, hypertriglyceridemia, low plasma HDL-C and high fasting plasma glucose or diagnosis of diabetes [8].
Results from research on marijuana use and MetS suggest a protective effect of marijuana use for MetS and some of its components [5,9]. Although some therapeutic effects of extracts of cannabis (marijuana plant) can be anticipated [10], these benefits may not apply to recreational use of marijuana. In the US, tobacco and marijuana are the most common substances of abuse after alcohol [11]. Statistics from the 2014 National Survey of Drug Use and Health, under the Substance Abuse and Mental Health Services Administration, show that among US adults aged 18-25 years, the lifetime prevalence of alcohol, cigarette and marijuana use were 83.4%, 56.1% and 52.6%, respectively whilst among US adults aged 26 years and above, lifetime prevalence were 88.3%, 67.5% and 46.1%, respectively [12]. With a likely increase in marijuana use arising from legalization of marijuana, it is important to assess the relationship with determinants of cardiovascular disease.
Methods
Data and variables
This is a cross-sectional study of adults aged 20 years and above who participated in the National Health and Nutrition Examination Survey (NHANES) 2011-2012. Only participants who responded to the question, “Have you ever even once used marijuana or hashish?” were enrolled.
Dependent variable: Our main dependent variable was MetS. We used the four most widely accepted definitions of MetS. In accordance with 2005 modification of ATP III criteria for Mets, we classified participants as having MetS if they had a co-occurrence of three or more of the following: Hypertension-an average blood pressure >130/85 mm Hg or use of medication for hypertension; Hyperglycemia-defined as fasting plasma glucose (FBG) ≥ 100 mg/dl or use of medication for diabetes; Abdominal obesity or high waist circumference-defined as females with waist circumference >88.0 cm and males with waist circumference >102.0 cm; Low HDL cholesterolemia-defined as plasma HDL-C levels <50 mg/dl for females and <40 mg/dl for males or use of medications for hypercholesterolemia; and Hypertriglyceridemia-defined as plasma triglycerides ≥ 150 mg/dl or use of medication for hypercholesterolemia. Details of laboratory and clinical procedures are described in the NHANES manual.
By WHO criteria, participants who said they had been diagnosed with diabetes by a doctor or were using medications for diabetes, or had a two hour oral glucose tolerance test (OGTT) result 140 mg/dl, or fasting insulin levels >25.2 μIU/ml and had any two or more of the following: average blood pressure 140/90 mmHg; body mass index >30 kg/m2; plasma triglycerides 150 mg/dl; plasma HDL-C levels <39 mg/dl (for females) or <35 mg/dl (for males); and albumin creatinine ratio 30.
By EGIR criteria, participants whose fasting insulin level fell above 75th percentile of this study group and had two or more of the following: average blood pressure ≥ 140/90 mm Hg or use of medications for hypertension; waist circumference ≥ 94 cm if male or ≥ 80 cm if female; plasma triglyceride ≥ 150 mg/dl; HDL-C ≥ 39 mg/dl.
By IDF criteria, ethnicity-specific waist circumference being ≥ 94 cm (for black males) or ≥ 80 cm (for black females); ≥ 102 cm (for white males) or ≥ 88 cm (for white female), and ≥ 94 cm or ≥ 80 cm for males and females respectively who were Asians/Mexican American/ Multiracial and had any two or more of the following: average blood pressure>130/85 mmHg or on medication for hypertension; plasma triglyceride ≥ 150 mg/dl or on anti-cholesterol medications; plasma HDL-C ≤ 50 mg/dl (for females) or ≤ 40 mg/dl (for males); and fasting plasma glucose ≥ 100 mg/dl diagnosis of diabetes by a doctor.
Main independent variable: According to the questions in NHANES, participants who had never used marijuana/hashish were categorized as never marijuana users. Those who said they had used marijuana/hashish but not up to once a month for more than a year were classified as non-regular marijuana users and those who had used marijuana or hashish at least once a month for more than a year were classified as regular marijuana users.
We estimated years of marijuana use by subtracting each participant’s age at regular marijuana use from their current age. For participants who were non-regular users of marijuana, we assigned zero years of marijuana use. Our multivariate logistic analysis included only marijuana users (regular users or non-regular-users) to enable us assess the effect among those who had ever used marijuana and avoid placing non-regular marijuana users and never marijuana users on the same level.
Other independent variables: We controlled for cigarette smoking. We classified participants who reported they have smoked at least 100 cigarettes their entire life and still smoke every day or some days as current cigarette smokers. Those who have smoked at least 100 cigarettes but do not currently smoke at all were past smokers. Those who had never smoked cigarettes were non-smokers. Non-smokers or past smokers were assigned zero years of smoking cigarettes. Years of smoking for current smokers was estimated by subtracting reported initial age at regular smoking from their current age.
In the multivariate model, we controlled for age of participant, gender, race, education, marital status, poverty to income ratio (PIR), participation in at least moderate physical activity, days of alcohol use in a week, other recreational substance use (methamphetamine, heroin or cocaine) and participation in rehabilitation. Details of the measurement of these control variables are described in NHANES manual.
Statistical analysis
We used Stata/IC 14.0 software package for analysis. We estimated the proportions of demographic and clinical variables by race to have an appreciation of the differences. Disparities in socioeconomic factors as well as race/ethnicity have been described as important factors for metabolic abnormalities as well as recreational substance use [13,14]. Using logistic regression analysis, we estimated unadjusted and adjusted odds ratios for MetS among regular and non-regular marijuana users. In all analyses we applied the appropriate weights for the NHANES multi-stage survey design and used a two-tailed significance level of α=0.05 (http://www.cdc.gov/nchs/data/nhanes/nhanes3/cdrom/NCHS/MANUALS/WGT_EXEC.PDF).
Results
Demographic and metabolic syndrome characteristics of study participants
Characteristics for the basic demographics and MetS with its components are shown in Tables 1a and 1b, respectively. Overall, 26.2% of participants were regular marijuana users and 24.0% were current cigarette smokers (Table 1a). Among the different racial/ethnic groups, people of other race/Multi-racials had the highest prevalence for marijuana use (40.7%) with Asians having the least (7.6%). Among all participants, the prevalence of other illicit drug use (cocaine, heroin or methamphetamine) was 17.7% whilst multiracial had the highest prevalence (28.7%). A higher proportion of Non-Hispanic Blacks (7.8%) have had rehabilitation compared to Multiracial (7.4%).
| Variable | Sample Size (%) | Race | |||||
|---|---|---|---|---|---|---|---|
| NHW | NHB | MA | OHISP | ASIANS | ORACE | ||
| Marijuana Use*** | |||||||
| Never | 1427 (46.80) | 33.78 | 42.86 | 61.09 | 58.72 | 73.3 | 33.56 |
| Non-regular | 824 (27.03) | 33.51 | 25.66 | 23.1 | 21.71 | 18.74 | 28.7 |
| Regular | 798 (26.17) | 32.7 | 31.48 | 15.81 | 19.57 | 7.6 | 40.74 |
| Cigarette smoking*** | |||||||
| Never | 1807 (59.25) | 48.43 | 63.26 | 64.13 | 62.63 | 74.94 | 55.56 |
| Past | 510 (16.72) | 20.49 | 11.74 | 19.15 | 19.22 | 13.11 | 14.81 |
| Current | 733 (24.03) | 31.09 | 25 | 16.72 | 18.15 | 11.94 | 29.63 |
| Gender* | |||||||
| Male | 1552 (50.87) | 52.02 | 46.66 | 54.71 | 48.04 | 53.4 | 55.56 |
| Female | 1499 (49.13) | 47.98 | 53.34 | 45.29 | 51.96 | 46.6 | 44.44 |
| Marital Status*** | |||||||
| Married | 1371 (44.94) | 49.15 | 31.65 | 52.28 | 41.28 | 56.91 | 38.89 |
| Other | 1680 (55.06) | 50.85 | 68.35 | 47.72 | 58.72 | 43.09 | 61.11 |
| Country of Birth*** | |||||||
| USA | 2154 (70.65) | 95.06 | 89.91 | 41.77 | 26.79 | 19.67 | 80.56 |
| Other Countries | 895 (29.35) | 4.94 | 10.09 | 58.23 | 73.21 | 80.33 | 19.44 |
| Education*** | |||||||
| ≤ High School Graduate | 1172 (38.41) | 33.42 | 38.97 | 69.6 | 52.31 | 19.44 | 29.63 |
| ≥ Some College | 1879 (61.59) | 68.58 | 61.03 | 30.6 | 47.69 | 80.56 | 70.37 |
| Age groups (Years)** | |||||||
| 20-25 | 543 (17.80) | 15.09 | 20.43 | 15.5 | 17.08 | 19.44 | 28.7 |
| Above 25 | 2508 (82.20) | 84.91 | 79.57 | 84.5 | 82.92 | 80.56 | 71.3 |
| PIR*** | |||||||
| <1.00 | 753 (26.39) | 24.91 | 31.12 | 30.95 | 30.15 | 15.56 | 27.18 |
| 1.00 to 2.99 | 1048 (36.73) | 36.02 | 36.93 | 42.52 | 43.13 | 29.16 | 38.83 |
| 3.00 to 4.99 | 555 (19.45) | 19.26 | 19.09 | 18.03 | 17.94 | 24.04 | 14.56 |
| >5.00 | 497 (17.42) | 19.81 | 12.86 | 8.5 | 8.78 | 31.2 | 19.42 |
| Other Drug Use*** | |||||||
| No | 2504 (82.23) | 74.41 | 87.01 | 80.49 | 87.14 | 94.6 | 71.3 |
| Yes | 541 (17.77) | 25.59 | 12.99 | 19.51 | 12.86 | 5.4 | 28.7 |
| Ever had rehabilitation*** | |||||||
| No | 2877 (94.30) | 92.54 | 92.18 | 97.57 | 96.44 | 99.3 | 92.59 |
| Yes | 174 (5.70) | 7.46 | 7.82 | 2.43 | 3.56 | 0.7 | 7.41 |
Table 1a: Proportions of recreational substance use and demographic characteristics of participants stratified by race, Percentages are column percentages. Chi square tests (*P<0.05, **P<0.01, ***P<0.001) show significant differences among the various racial ethnic groups. MA: Mexican Americans; NHW: Non-Hispanic Whites; NHB: Non-Hispanic Blacks; OHISP: Other Hispanics; ORACE: Other Race or Multiracial; PIR: Family Income to Poverty Ratio.
| Variable | Diagnostic Criteria | |||
|---|---|---|---|---|
| WHO | EGIR | ATP III | IDF | |
| Metabolic Syndrome | ||||
| Overall (Yes) | 9.21 | 8.55 | 23.17 | 23.37 |
| By substance use (Yes) | ||||
| Regular marijuana | 8.4 | 8.4 | 20.93 | 21.3 |
| Current cigarette smoker | 9.41 | 9.14 | 24.83 | 24.69 |
| By Race (Yes) | ||||
| NHW | 9.43 | 9.97 | 24.71 | 22.01 |
| NHB | 9.21 | 8.07 | 25.73 | 25.98 |
| MA | 14.89 | 10.33 | 24.62 | 28.27 |
| OHISP | 7.83 | 5.69 | 21.71 | 25.27 |
| ASIANS | 5.39 | 6.79 | 15.46 | 18.03 |
| ORACE | 8.33 | 6.48 | 18.52 | 19.44 |
| Hypertension | ||||
| Overall (Yes) | 10.56 | 23.89 | 31.69 | 31.69 |
| Hyperglycemia | ||||
| Overall (Yes) | 19.04 | 8.19 | 21.86 | 23.43 |
| Hyperinsulinemia | ||||
| Overall (Yes) | 3.61 | 61.42 | - | - |
| High OGTT Level | ||||
| Overall (Yes) | 5.97 | 5.97 | - | - |
| Hypertriglyceridemia | ||||
| Overall (Yes) | 11.67 | 11.67 | 18.49 | 18.49 |
| Low HDL-C | ||||
| Overall (Yes) | 10.49 | 15.34 | 34.81 | 34.81 |
| High WC | ||||
| Overall (Yes) | - | 69.59 | 49.8 | 64.15 |
| NHW | - | - | - | 50.07 |
| NHB | - | - | - | 69.59 |
| ASIANS/MA/OHISP/OR | - | - | - | 74.57 |
| BMI >30 kg/m2 | ||||
| Overall (Yes) | 35.59 | - | - | - |
| High Albumin/Creatinine Ratio | ||||
| Overall (Yes) | 13.27 | - | - | - |
Table 1b: Prevalence in percentages of metabolic syndrome diagnosis and its components of participants by the different criteria, Chi square tests (P<0.01 was obtained for racial differences of metabolic syndrome prevalence for WHO, ATP III, IDF) show significant differences among the various racial ethnic groups. NHW: Non-Hispanic Whites; NHB: Non-Hispanic Blacks; MA: Mexican Americans; OHISP: Other Hispanics; ORACE: Other Race or Multiracial; OGTT: Oral Glucose Tolerance Test; HDL-C: High Density Lipoprotein Cholesterol; WC: Waist Circumference; BMI: Body Mass Index.
Of the four criteria, ATP III and IDF classify more people as having MetS (23.2% and 23.4%, respectively) and WHO criteria classify the least (9.2%) and EGIR (8.6%) (Table 1b). This pattern is also seen for MetS prevalence among marijuana users and cigarette smokers: the proportion of MetS among marijuana users was 21.3% (IDF) and 20.9% (ATP III) whilst the proportion of MetS among cigarette smokers was 26.0% (IDF) and 25.7% (ATP III). By race/ethnicity, ATP III classifies the 25.7% of non-Hispanic Blacks as having MetS. All other criteria predominantly classify Mexican Americans (MA) as having MetS (28.3%-IDF, 14.9%-WHO and 10.3%-EGIR).
Hypertension, hypertriglyceridemia, and Low HDL-C are more prevalent (31.7%, 18.5%, 34.8%, respectively) using ATP III and IDF criteria. The prevalence of hyperglycemia is 21.9% using ATP III and 23.4% using IDF. Disparities in prevalence for other components are shown in Table 1b.
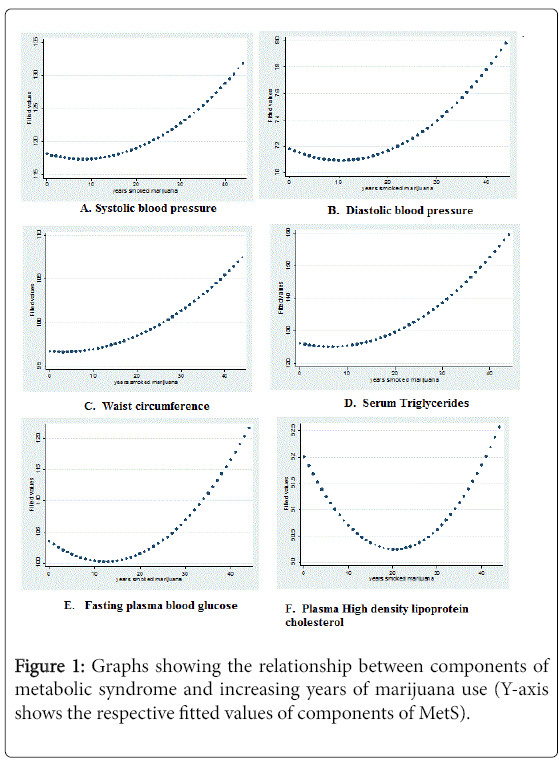
Components of metabolic syndrome with years of marijuana use
The relationship between components of MetS and years of marijuana use are shown in Figure 1. Curvilinear relationships between years of marijuana use and components of MetS are apparent. The relationship of systolic blood pressure (SBP), diastolic blood pressure (DBP), waist circumference (WC), plasma triglycerides (TG) and fasting blood glucose (FBG) with years of marijuana used tend to be J-Shaped. This shows an initial decrease in values but eventual increase. The relationship between plasma high density lipoprotein cholesterol HDL-C and years of marijuana used was U-shaped. This shows an initial decrease and eventual increase.
Bivariate analysis of metabolic syndrome and its components with years of marijuana use and cigarette smoking
By all the criteria, unadjusted analysis showed a universal increase in odds of having MetS with every year of marijuana use. With every year increase in marijuana use, the odds ratios (OR) for having MetS are 1.06 (95% CI: 1.03, 1.08) for ATP III, 1.06 (95% CI: 1.04, 1.08) for WHO, 1.05 (95% CI: 1.02, 1.08) for EGIR and 1.05 (95% CI: 1.03, 1.07) for IDF (Table 2a). By all applicable criteria, for each year of marijuana use, a significant increase in odds is observed for hypertension, hyperglycemia, high oral glucose tolerance test levels, hypertriglyceridemia, abdominal obesity and obesity.
| Variable | Diagnostic Criteria | |||
|---|---|---|---|---|
| ATP III | WHO | EGIR | IDF | |
| Metabolic Syndrome | 1.06 (1.03-1.08) | 1.06 (1.04-1.08) | 1.05 (1.02-1.08) | 1.05 (1.03-1.07) |
| Marijuana use1 | ||||
| Hypertension | 1.06 (1.03-1.09) | 1.09 (1.04-1.15) | 1.08 (1.04-1.11) | 1.06 (1.03-1.10) |
| Marijuana use1 | ||||
| Hyperglycemia | 1.04 (1.00-1.08) | 1.04 (1.00-1.09) | 1.04 (1.00-1.08) | 1.04 (1.00-1.08) |
| Marijuana use1 | ||||
| Hyperinsulinemia | - | 1.03 (0.98-1.08) | 1.04 (1.00-1.07) | - |
| Marijuana use1 | ||||
| High OGTT | - | 1.07 (1.05-1.10) | 1.07 (1.07-1.10) | - |
| Marijuana use1 | ||||
| Hypertriglyceridemia | 1.05 (1.03-1.07) | 1.03 (1.01-1.06) | 1.03 (1.01-1.08) | 1.05 (1.03-1.07) |
| Marijuana use1 | ||||
| Low HDL-C | 1.03 (1.00-1.06) | 1.00 (0.97-1.03) | 1.03 (1.00-1.05) | 1.03 (1.00-1.06) |
| Marijuana use1 | ||||
| Abdominal Obesity | 1.03 (1.00-1.07) | - | 1.07 (1.05-1.10) | 1.03 (1.00-1.06) |
| Marijuana use1 | ||||
| Obesity | - | 1.03 (1.01-1.05) | - | - |
| Marijuana use1 | ||||
| High Albumin/Creatinine Ratio | - | 0.99 (0.96-1.02) | - | - |
| Marijuana use1 | ||||
Table 2a: Unadjusted analysis of having metabolic syndrome and risky levels of components by the different criteria for each year of marijuana use, bold values indicate significance at α<0.05, Marijuana use1 - each year of marijuana use among regular or non-regular marijuana users.
The relationship with cigarette use shows increases in odds for MetS which is only significant by ATP III. For hypertension, hyperglycemia and high OGTT levels a significant increase in odds is demonstrated by all the criteria (Table 2b).
| Variable | Diagnostic Criteria | |||
|---|---|---|---|---|
| ATP III | WHO | EGIR | IDF | |
| Metabolic Syndrome | 1.02 (1.01-1.05) | 1.02 (0.98-1.05) | 1.01 (0.99-1.04) | 1.01 (0.99-1.03) |
| Cig smoking1 | ||||
| Hypertension | 1.03 (1.00-1.06) | 1.07 (1.03-1.12) | 1.08 (1.04-1.11) | 1.03 (1.00-1.06) |
| Cig smoking1 | ||||
| Hyperglycemia | 1.02 (1.00-1.03) | 1.02 (1.00-1.04) | 1.04 (1.00-1.08) | 1.02 (1.00-1.03) |
| Cig smoking1 | ||||
| Hyperinsulinemia | - | 1.03 (0.99-1.07) | 1.04 (1.00-1.07) | - |
| Cig smoking1 | ||||
| High OGTT | - | 1.02 (1.00-1.05) | 1.07 (1.05-1.10) | - |
| Cig smoking1 | ||||
| Hypertriglyceridemia | 1.02 (1.01-1.04) | 1.03 (1.00-1.05) | 1.03 (1.01-1.06) | 1.02 (1.01-1.04) |
| Cig smoking1 | ||||
| Low HDL-C | 1.01 (0.99-1.02) | 0.97 (0.94-1.01) | 1.02 (1.00-1.05) | 1.01 (0.99-1.02) |
| Cig smoking1 | ||||
| Abdominal Obesity | 1.00 (0.98-1.02) | - | 1.07 (1.05-1.10) | 1.00 (0.98-1.02) |
| Cig smoking1 | ||||
| Obesity | - | 1.00 (0.98-1.02) | - | - |
| Cig smoking1 | ||||
| High Albumin/Creatinine Ratio | - | 1.01 (0.99-1.03) | - | - |
| Cig smoking1 | ||||
Table 2b: Unadjusted analysis of having metabolic syndrome and risky levels of components by the different criteria for every year of cigarette smoking, bold values indicate significance at α<0.05, Cig smoking1-each year of cigarette use among regular or non-regular marijuana users.
Multivariate analysis
For every year of marijuana use, adjusted odds ratio (AOR) for having MetS (controlling for years of smoking, gender, age, marriage, education, country of birth, PIR, having health insurance, participating in at least moderate physical activity, weekly alcohol intake, other illicit drug use and undergoing rehabilitation) by ATP III and IDF criteria was 1.05 (95% CI: 1.02, 1.08). By WHO, AOR was 1.08 (95% CI: 1.04, 1.13) and by EGIR criteria, AOR was 1.06 (95% CI: 1.01, 1.11) (Table 3a).
| ATP III | WHO | EGIR | IDF | |
|---|---|---|---|---|
| Marijuana use1 | 1.05 (1.02, 1.08) | 1.08 (1.04, 1.13) | 1.06 (1.01, 1.11) | 1.05 (1.02, 1.08) |
| Cigarette smoking1 | 1.01 (0.98, 1.04) | 0.99 (0.95, 1.04) | 1.00 (0.96, 1.04) | 1.00 (0.97, 1.03) |
| Age 25+ | 2.70 (0.66, 11.10) | 0.67 (0.12, 3.78) | 0.92 (0.19, 4.45) | 1.01 (0.37, 2.78) |
| Males | 0.78 (0.29, 2.09) | 2.54 (0.98, 6.63) | 1.29 (0.45, 3.64) | 0.71 (0.27, 1.90) |
| Asians | 1.37 (0.18, 10.19) | 1.72 (0.27, 10.82) | 0.98 (0.19, 5.14 ) | 1.41 (0.24, 8.19) |
| Blacks | 0.74 (0.33,1.66) | 0.63 (0.13, 2.96) | 0.59 (0.20, 5.08 ) | 0.81 (0.36, 1.83) |
| M Americans2 | 0.63 (0.22, 1.80) | 1.54 (0.55, 4.27) | 2.02 (0.96, 4.23 ) | 1.16 (0.31, 4.28) |
| Other Hispanics | 0.22 (0.02, 2.55) | 1.44 (0.33, 6.22) | 1.01 (0.20, 5.08 ) | 2.39 (0.40, 14.24) |
| Other Race | 0.99 (0.22, 4.48) | 0.89 (0.18, 4.46) | 0.69 (0.20, 2.40 ) | 1.45 (0.39, 5.47) |
| Born in USA | 3.87 (0.44, 34.28) | 3.04 (0.60, 15.29) | 4.08 (0.84, 19.75) | 5.53 (1.39, 22.08) |
| Education | 0.97 (0.59, 1.59) | 1.26 (0.94, 1.71) | 1.39 (0.80, 2.40) | 0.98 (0.61, 1.58) |
| PIR3 | 0.93 (0.78, 1.10) | 1.01 (0.65, 1.57) | 0.91 (0.76, 1.09) | 0.99 (0.81, 1.21) |
| Insured | 1.25 (0.68, 2.27) | 0.52 (0.25, 1.04) | 0.66 (0.38, 1.15) | 1.00 (0.52, 1.93) |
| Married | 1.17 (0.53, 2.58) | 0.83 (0.30, 2.33) | 1.75 (0.63, 4.83) | 1.78 (0.54, 2.58) |
| Moderate PA4 | 1.17 (0.54, 2.51) | 0.74 (0.31, 1.76) | 0.85 (0.40, 1.79) | 1.16 (0.53, 2.53) |
| Alcohol Intake5 | 1.84 (1.10, 3.08) | 0.74 (0.28, 1.99) | 0.95 (0.52, 1.74) | 1.69 (1.04, 2.74) |
| Other drug use | 0.92 (0.45, 1.89) | 0.49 (0.21, 1.18) | 0.39 (0.23, 0.64) | 0.99 (0.46, 2.14) |
| Rehabilitation | 1.01 (0.44, 2.33) | 1.33 (0.50, 3.57) | 1.66 (0.58, 4.77) | 1.23 (0.55, 2.72) |
Table 3a: Multivariate analysis of metabolic syndrome with years of marijuana use by different criteria controlling for cigarette smoking and other variables, 1-Each year increase in marijuana use or cigarette smoking; 2-Mexican American; 3-Family Income-to-Poverty Ratio; 4-At least moderate physical activity (recreational); 5-Weekly, Bold values indicates significant at α<0.05; ATP III-National Cholesterol Examination Panel, Adult Treatment Panel III; EGIR: European Group for the Study of Insulin Resistance; IDF: International Diabetes Federation; WHO: World Health Organization.
Each year of marijuana use showed AORs for hypertension as: 1.07 (95% CI: 1.03, 1.12) by ATP III and IDF, 1.05 (95% CI: 1.01, 1.09) by WHO and 1.08 (95% CI: 1.03, 1.12) by EGIR All the applicable criteria show increased odds for abdominal obesity: 1.06 (95% CI: 1.00, 1.11) by ATP III, 1.09 (95% CI: 1.05, 1.14) by EGIR and 1.07 (95% CI: 1.01, 1.13) by IDF. For obesity the AOR was 1.03 (95% CI: 1.01, 1.06) according to WHO. The AOR for having a high oral glucose tolerance test level was 1.12 (95% CI: 1.07, 1.18) by WHO and EGIR.
Every year increase in smoking cigarette by this model, was associated with AOR of 1.05 (95% CI: 1.01, 1.09) for hypertension by WHO criteria. For abdominal obesity, the AOR was 0.97 (95% CI: 0.95, 0.99) by IDF criteria.
Discussion
Duration of marijuana use seems to be a significant factor associated with MetS. Its effect is small, but of the same order of magnitude or possibly greater than the effect of years of cigarette smoking. It must be noted that conventionally, cigarette smoking status has been used in analysis, but we attempted assessing the effect of years of smoking cigarette on MetS. Although current studies on marijuana use and MetS show a protective effect of marijuana on glycemic factors, this may be the result of not considering the years of using marijuana in cross sectional analysis. All criteria, demonstrate that every year increase in marijuana use is associated with at least 5% increase in odds of having MetS. In relation to components of MetS, a general increase in odds is observed with progress in years of using marijuana, however they vary by significance.
We observed different strengths in AORs for MetS with each year of marijuana use based on the different criteria for Mets, but the same direction of associations. Even though literature has discussed the possibility of a common definition for MetS [15-17], this suggests that the different criteria for metabolic syndrome may be comprehensive and can produce unified relationships with respect to marijuana use. This is irrespective of the fact that WHO and EGIR set predefined risk factors on glucose or insulin impairment. However, WHO and EGIR criteria however, showed marked reductions in prevalence. This is primarily due to the prequalifying criteria for glucose/insulin impairment.
Metabolic syndrome is a powerful tool for identifying people at risk for CVD and diabetes [15]. It is important that research on marijuana demonstrate the true relationship with MetS and it components. These findings could provide a behavioral path to preventive and therapeutic interventions for CVD and diabetes [15] in relation to marijuana use.
Criteria of MetS is not settled, neither is the definition of marijuana use. Metabolic syndrome is a complex condition and there may be more factors intrinsic and extrinsic to MetS and marijuana use that need attention. This study finds that prolonged use of marijuana is a likely associated factor for MetS, glucose intolerance and hypertension. Increased years of marijuana use are also associated with hypertriglyceridemia but are significant using ATP III and IDF criteria. Even though all the criteria use a plasma triglyceride cut off ≥ 150 mg/dl, WHO and EGIR do not account for the use of cholesterol lowering medications and this could be a factor in the difference in significance. The recreational use of marijuana may ultimately threaten public health gains in the area of cardiovascular disease prevention. A longitudinal study of the relationship between recreational marijuana use and MetS concerning clinical factors and biological markers for all the four core attributes of MetS: insulin resistance, visceral obesity, atherogenic dyslipidemia and endothelial dysfunction [15] are exigent (Table 3b).
| ATP III | WHO | EGIR | IDF | ||
|---|---|---|---|---|---|
| Hypertension | |||||
| Marijuana use1 | 1.07 (1.03, 1.12) | 1.05 (1.01, 1.09) | 1.08 (1.03, 1.12) | 1.07 (1.03, 1.12) | |
| Cigarette smoking1 | 1.02 (0.98, 1.05) | 1.05 (1.02, 1.09) | 1.02 (0.99, 1.05) | 1.02 (0.98, 1.05) | |
| Hyperglycemia | |||||
| Marijuana use1 | 1.02 (0.98, 1.07) | 1.02 (0.98, 1.07) | 1.02 (0.98, 1.07) | 1.02 (0.98, 1.07) | |
| Cigarette smoking1 | 1.00 (0.97, 1.03) | 1.00 (0.97, 1.03) | 1.00 (0.97, 1.03) | 1.00 (0.97, 1.03) | |
| Hyperinsulinemia | |||||
| Marijuana use1 | - | 1.01 (0.92, 1.11) | 1.04 (0.99, 1.11) | - | |
| Cigarette smoking1 | - | 1.00 (0.97, 1.03) | 1.00 (0.96, 1.04) | - | |
| High Oral Glucose Tolerance Test level | |||||
| Marijuana use1 | - | 1.12 (1.07, 1.18) | 1.12 (1.07, 1.18) | - | |
| Cigarette smoking1 | - | 0.99 (0.96, 1.03) | 0.99 (0.96, 1.03) | - | |
| Hypertriglyceridemia | |||||
| Marijuana use1 | 1.04 (1.00, 1.07) | 1.02 (0.98, 1.05) | 1.02 (0.98, 1.05) | 1.04 (1.00, 1.07) | |
| Cigarette smoking1 | 1.01 (0.99, 1.03) | 1.01 (0.98, 1.05) | 1.01 (0.98, 1.05) | 1.01 (0.99, 1.03) | |
| Low HDL-C | |||||
| Marijuana use1 | 1.03 (0.99, 1.07) | 1.02 (0.98, 1.07) | 1.04 (0.99, 1.08) | 1.03 (0.99, 1.07) | |
| Cigarette smoking1 | 0.99 (0.97, 1.02) | 0.97 (0.92, 1.02) | 0.98 (0.93, 1.03) | 0.99 (0.97, 1.02) | |
| Abdominal obesity | |||||
| Marijuana use1 | 1.06 (1.00, 1.11) | - | 1.09 (1.05, 1.14) | 1.07 (1.01, 1.13) | |
| Cigarette smoking1 | 0.99 (0.97, 1.02) | - | 0.97 (0.95, 0.99) | 0.95 (0.95, 1.01) | |
| Obesity by (BMI) | |||||
| Marijuana use1 | - | 1.03 (1.01, 1.06) | - | - | |
| Cigarette smoking1 | - | 0.99 (0.97, 1.01) | - | - | |
| High Albumin/Creatinine ratio | |||||
| Marijuana use1 | - | 0.96 (0.91, 1.01) | - | - | |
| Cigarette smoking1 | - | 1.01 (0.97, 1.05) | - | - | |
Table 3b: Multivariate analysis of components of metabolic syndrome with each year of marijuana use and cigarette smoking by different criteria (the controlled factors in the model are shown in Table 3a).
The active constituent of marijuana, delta-9-tetrahydrocanabinol (D-THC), acts on the endocannabinoid system (ECS), primarily CB1 receptors and CB2 receptors. The ECS plays a role in regulation of appetite and metabolism [18]. Modulation of the ECS affects the four core attributes of MetS [19]. These effects are being studied for management of obesity [20,21], dyslipidemia [21], atherosclerosis [22] and insulin resistance [20,23]. Cannabinoids or cannabis extracts may have therapeutic indications but, because absorption is erratic, the pharmacodynamics is still under active investigation for therapeutic purposes [24]. Arguments for recreational use of marijuana based on research for therapeutic use may need re-evaluation.
In Figure 1, initial reductions in blood pressure and glucose values change to increases after about five years of use. This shows a probable eventual deleterious effect on blood pressure and glycemic levels. However, after about twenty years of using marijuana, low levels of HDL-C tend to increase, which may elude an ultimate beneficial effect on HDL-C. This further stresses the complex relationship between cannabinoids and metabolic processes. All the applicable criteria show that increased years of marijuana use is associated with abdominal obesity. Active investigation of marijuana in long term metabolic derangements is important. Criteria by IDF show higher odds for abdominal obesity than ATP III. This is because IDF uses racial-ethnic specific waist circumference.
In this study, non-significant varying relationships are observed with each year of cigarette smoking and the different criteria for MetS. This relationship could be explained by the combination of nonsmokers and past smokers. For marijuana use, all participants had at least used marijuana before. The association of cigarette smoking status with hypertension is established knowledge [25,26]. Increased years of cigarette smoking was also associated with increased odds for high OGTT levels (WHO and EGIR). Research has shown that diabetic patients who continue to smoke have uncontrolled glucose levels even with treatment [27]. Studies have long shown that nicotine from cigarette smoking impairs glucose metabolism [28-30] and reflects as high proportions of glycated hemoglobin, high OGTT and high fasting insulin [31,32].
Strengths and limitations
Demographic, lifestyle, clinical, laboratory parameters and a large nationally representative sample was obtained from NHANES data, however this cross-sectional study estimates associations not risks. Marijuana use was self-reported and the study may have a reporting bias especially with information on illicit substance use as marijuana. We initially controlled for the quantity of marijuana used but this did not significantly affect the results and was excluded from the model. We did not control for diet, an important factor for MetS, however, we controlled for factors important in dietary and health decisions as income to poverty ratio (PIR), alcohol use, physical activity, health insurance and education. For ethnic-specific waist circumference by IDF, we classified NHBs as Europids based on ancestral genesis [33] that the ancestry of Blacks or African-American are predominantly Niger-Kordofanian (~71%), European (~13%) or other African (~8%) populations [34]. All NHWs were classified using values for Americans since distinctions based on the ethnic classification were unavailable. We however controlled for place of birth to possibly account for these differences.
Conclusion
Irrespective of the criteria for metabolic syndrome, each year of marijuana use showed increased odds of having metabolic syndrome, hypertension or high oral glucose tolerance test levels. Extended duration of marijuana use could possibly increase the risk for the development of metabolic syndrome. Longitudinal studies can show this risk. Irrespective of the criteria for MetS, we estimated increased odds of MetS with each year marijuana use. This may constitute an important pathway between marijuana use and cardiovascular disease in later life. The impact of duration of marijuana use should be considered in assessing the relationships with MetS.
Longitudinal research is required to define the true relationship between marijuana use and metabolic syndrome. If a cardiovascular risk is established, a good understanding of the pathogenesis of metabolic syndrome and metabolic pathways of marijuana metabolites should be laid out. This will help address any risk factors which may initiate and facilitate CVD progression among marijuana users.
Conflict of Interest
We declare no conflict of interest in processes associated with this study. All authors contributed equally in the research. Grants or Financial Support: The authors received no financial support for the study.
Acknowledgement
The authors thank the National Centre for Health Statistics (NCHS, NHANES III); Dr. Micheal Eriksen, Sc.M, Sc.D, Founding Dean, Georgia State University, Atlanta, Tobacco Center of Regulatory Science; Dr. Italia V. Rolle, PhD, Senior Epidemiologist, Epidemiology Branch, Office of Smoking and Health, Centers for Disease Control and Prevention, Atlanta, GA, USA; David Yankey, Mathematical Statistician, Immunization Services Division, Centers for Disease Control and Prevention, Atlanta, GA, USA, Georgia State University, Department of Mathematics and Statistics, Atlanta, Georgia.
References
- Galston WA, Dionne E Jr (2013) The new politics of marijuana legalization: Why opinion is changing. Governance Studies at Brookings, pp: 1-17.
- Cena H, Fonte ML, Turconi G (2011) Relationship between smoking and metabolic syndrome. Nutr Rev 69: 745-753.
- Aguilar M, Bhuket T, Torres S, Liu B, Wong RJ (2015) Prevalence of the metabolic syndrome in the United States, 2003-2012. JAMA 313: 1973-1974.
- Organization WHO (2014) Global status report on non-communicable diseases 2014: World Health Organization.
- Vidot DC (2016) Metabolic syndrome among marijuana users in the United States: An Analysis of national health and nutrition examination survey data. Am J Med 129: 173-179.
- Lakka HM, Laaksonen DE, Lakka TA, Niskanen LK, Kumpusalo E, et al. (2002) The metabolic syndrome and total and cardiovascular disease mortality in middle-aged men. JAMA 288: 2709-2716.
- Grundy SM (2004) Definition of metabolic syndrome report of the National Heart, Lung and Blood Institute/American Heart Association Conference on scientific issues related to definition. Circulation 109: 433-438.
- Alberti KGMM, Zimmet P, Shaw J (2006) Metabolic syndrome-a new world-wide definition. A consensus statement from the international diabetes federation. Diabetic Med 23: 469-480.
- Penner EA, Buettner H, Mittleman MA (2013) The impact of marijuana use on glucose, insulin and insulin resistance among US adults. Am J Med 126: 583-589.
- Baker D, Pryce G, Giovannoni G, Thompson AJ (2003) The therapeutic potential of cannabis. Lancet Neurol 2: 291-298.
- Latimer W, Zur J (2010) Epidemiologic trends of adolescent use of alcohol, tobacco and other drugs. Child Adolesc Psychiatr Clin N Am 19: 451-464.
- SAMHSA (2015) Behavioral health trends in the United States: Results from the 2014 national survey on drug use and health. Substance Abuse and Mental Health Services Administration: Rockville, MD, USA.
- Trinidad DR (2011) A nationwide analysis of US racial/ethnic disparities in smoking behaviors, smoking cessation and cessation-related factors. American J Public Health 101: 699-706.
- Ferdinand KC (2007) The cardiometabolic syndrome and cardiovascular disease in racial and ethnic minorities: New areas of research and intervention. J Cardiometab Syndr 2: 235-237.
- Huang PL (2009) A comprehensive definition for metabolic syndrome. Dis Model Mech 2: 231-237.
- Eckel RH, Grundy SM, Zimmet PZ (2005) The metabolic syndrome. Lancet 365: 1415-1428.
- Zimmet P, Magliano D, Matsuzawa Y, Alberti G, Shaw J (2005) The metabolic syndrome: A global public health problem and a new definition. J Atheroscler Thromb 12: 295-300.
- Boyd ST (2006) The endocannabinoid system. Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy 26: 218S-221S.
- Perkins JM and SN Davis (2008) Endocannabinoid system overactivity and the metabolic syndrome: Prospects for treatment. Curr Diab Rep 8: 12-19.
- Blüher M, Engeli S, Klöting N, Berndt J, Fasshauer M, et al. (2006) Dysregulation of the peripheral and adipose tissue endocannabinoid system in human abdominal obesity. Diabetes 55: 3053-3060.
- Després JP, Golay A, Sjöström L, Rimonabant in obesity-lipids study group (2005) Effects of rimonabant on metabolic risk factors in overweight patients with dyslipidemia. N Engl J Med 353: 2121-2134.
- Steffens S, Veillard NR, Arnaud C, Pelli G, Burger F, et al. (2005) Low dose oral cannabinoid therapy reduces progression of atherosclerosis in mice. Nature 434: 782-786.
- Osei-Hyiaman D (2008) Hepatic CB1 receptor is required for development of diet-induced steatosis, dyslipidemia and insulin and leptin resistance in mice. J Clin Invest 118: 3160-3169.
- Le Foll B, Gorelick DA, Goldberg SR (2009) The future of endocannabinoid-oriented clinical research after CB1 antagonists. Psychopharmacology (Berl) 205: 171-174.
- Talukder MH (2011) Chronic cigarette smoking causes hypertension, increased oxidative stress, impaired NO bioavailability, endothelial dysfunction, and cardiac remodeling in mice. Am J Physiol Heart Circ Physiol 300: H388-H396.
- Ingall TJ (1991) Predictors of intracranial carotid artery atherosclerosis: Duration of cigarette smoking and hypertension are more powerful than serum lipid levels. Arch Neurol 48: 687.
- Gerber P (2013) Smoking is associated with impaired long-term glucose metabolism in patients with type 1 diabetes mellitus. Nutr Metab Cardiovasc Dis 23: 102-108.
- Morgan TM (2004) Acute effects of nicotine on serum glucose insulin growth hormone and cortisol in healthy smokers. Metabolism 53: 578-582.
- McCormick W (1935) The rôle of the glycemic response to nicotine tobacco smoking and blood sugar. Am J Epidemiol 22: 214-220.
- Rafalson L, Donahue RP, Dmochowski J, Rejman K, Dorn J, et al. (2009) Cigarette smoking is associated with conversion from normoglycemia to impaired fasting glucose: The Western New York health study. Ann Epidemiol 19: 365-371.
- Janzon L, Berntorp K, Hanson M, Lindell SE, Trell E (1983) Glucose tolerance and smoking: A population study of oral and intravenous glucose tolerance tests in middle-aged men. Diabetologia 25: 86-88.
- Persson PG, Carlsson S, Svanström L, Ostenson CG, Efendic S, et al. (2000) Cigarette smoking, oral moist snuff use and glucose intolerance. J Intern Med 248: 103-110.
- Waters MC (1991) The role of lineage in identity formation among Black Americans. Qual Sociol 14: 57-76.
- Tishkoff SA (2009) The genetic structure and history of Africans and African Americans. Science 324: 1035-1044.
Relevant Topics
- Addiction Recovery
- Alcohol Addiction Treatment
- Alcohol Rehabilitation
- Amphetamine Addiction
- Amphetamine-Related Disorders
- Cocaine Addiction
- Cocaine-Related Disorders
- Computer Addiction Research
- Drug Addiction Treatment
- Drug Rehabilitation
- Facts About Alcoholism
- Food Addiction Research
- Heroin Addiction Treatment
- Holistic Addiction Treatment
- Hospital-Addiction Syndrome
- Morphine Addiction
- Munchausen Syndrome
- Neonatal Abstinence Syndrome
- Nutritional Suitability
- Opioid-Related Disorders
- Relapse prevention
- Substance-Related Disorders
Recommended Journals
Article Tools
Article Usage
- Total views: 7280
- [From(publication date):
specialissue-2017 - Jul 16, 2025] - Breakdown by view type
- HTML page views : 6133
- PDF downloads : 1147