Retinal Pigment Epithelial Cell Conditioned Medium Enhances the Yield of RPE Cells Differentiated from Human Embryonic Stem Cells
Received: 22-Jan-2015 / Accepted Date: 27-Jan-2015 / Published Date: 04-Mar-2015 DOI: 10.4172/2161-0681.1000216
Abstract
Geographic atrophy (GA) in patients with age-related macular degeneration (AMD) is characterized by the loss of retinal pigment epithelial (RPE) cells in the macular region. Human embryonic stem cells (hESC)-derived RPE have shown promise in cellular therapy for these diseases. However, the differentiation from hESC is a very lengthy process with an initially low RPE yield. The purpose of this study was to determine whether polarized hESC-RPE conditioned medium (CM) could improve the yield of RPE differentiated from hESC and to identify the mechanism of this effect. Two hESC lines, hES3 and H9, were differentiated into RPE by culturing in CM or regular medium for up to 8 weeks. We found substantially more pigmented cells in CM based on surface area quantification, and Q-RT PCR showed significantly higher expression of RPE-specific genes in the H9 and hES3 cells cultured in CM. The results indicated that CM increased the yield of RPE cells differentiated from H9 and hES3 lines via increased RPE colony formation and proliferation. These findings may be beneficial to the manufacturing process for future clinical studies.
Keywords: Age-related macular degeneration; Retinal pigment epithelial cell; Embryonic stem cells; Polarized RPE; Conditioned medium; Differentiation
312355Introduction
Retinal pigment epithelial (RPE) cells, located between the neural retina and the vascular choroid, support the health of the light sensitive photoreceptors. They are polarized, with an apical side which faces the photoreceptors and aids with phagocytosis of their shed outer segments, and a basal side that is lined by the Bruch’s membrane and faces the choroidal blood vessels [1]. RPE cells can transport nutrients, water, ions, and metabolic waste between the blood vessels and the photoreceptors [2]. Additionally, RPE cells are capable of secreting various types of growth factors which help maintain photoreceptors and blood vessels. The cone-rich macula is the central part of the retina that is responsible for high acuity vision [3].
Since the RPE is critical to the survival and function of the retinal photoreceptors, damaged or lost RPE is responsible for many types of vision loss. Stargardt macular degeneration (SMD), the most common macular degeneration in children, is characterized by mutations in the ABCA4 gene, which results in impaired transport of molecules across the photoreceptors and leads to photoreceptor death and secondary RPE degeneration [4]. Another hereditary retinal degeneration disease, Best vitelliform macula dystrophy (BVMD), is caused by a mutation in the Best1 gene. As a result of an impaired calcium chloride channel, there is a buildup of extracellular deposits below and within the RPE, which leads to macular degeneration and/or choroidal neovascularization [5]. In age-related macular degeneration (AMD), the leading cause of blindness in the elderly [6], the RPE become dysfunctional and die, resulting in degeneration of photoreceptors [7]. There are two types of advanced AMD: the wet form, identified by newly formed choroidal blood vessels growing through the Bruch’s membrane into the sub-RPE or subretinal space [8], and the dry form, which is characterized by geographic areas of central RPE loss associated with photoreceptor degeneration. While wet AMD can be treated with anti-vascular endothelial growth factor (VEGF) drugs, there is no treatment yet for dry AMD [9,10]. RPE cell replacement is one of the promising therapies for treatment of AMD as well as other retinal degenerative diseases. Human embryonic stem cells (hESC) are a good potential source of RPE, because they are pluripotent, can be self-renewed and expanded to generated almost an unlimited source of RPE cells [11,12]. Recent clinical trials are in progress testing subretinal implantation of cell suspensions of hESC-derived RPE to compensate for RPE loss in AMD [13].
Numerous methods for differentiating RPE cells from hESC have been published which yielded varying results. hESC can be cultured adherent to plates, or in suspension culture as embryoid bodies (EB) [14,15]. hESC can spontaneously differentiate into cells of neuroectodermal lineage [16-18] which can then be further differentiated into RPE [19]. For spontaneous differentiation, basic fibroblast growth factor (bFGF), which supports hESC renewal, is removed from the medium and RPElike cells spontaneously appear in 4-8 weeks showing as pigmented spots in culture dishes [14,15]. With EB spontaneous differentiation, it was found that only 1% of the differentiated cells were RPE [14,20]. Because RPE derivation is a time consuming and inefficient process, many groups have devised other methods in hopes of speeding up the generation and/or increasing the amount of RPE cells. The addition of Wnt and nodal antagonists, which help with forebrain development and induction of retinal progenitors [21,22], were found to result in 38% of cells being pigmented following 8 weeks of differentiation [23]. There was also improved RPE yield when nicotinamide, also known as vitamin B3, and activin A, a member of the TGF-β family of proteins known to direct RPE development [24], were added to differentiating floating hESC clusters [25]. Another group also showed further improvement in RPE yield in only 2 weeks of differentiation with the addition of several factors at various points in time [26].
In vivo RPE cells are already known to release several trophic molecules in order to maintain proper retinal integrity, such as angiogenic regulatory factors like VEGF, or the neuroprotective pigment epithelium-derived factor (PEDF) [27,28]. Additionally, RPE cells are known to release ciliary neurotrophic factor (CNTF), neurotrophin-3 (NT-3), and many other trophic factors to support photoreceptor cells and the choroid [29,30]. Knowing that some of these secreted molecules have been shown to play a role in RPE proliferation, survival, and differentiation, we wanted to determine if RPE conditioned medium (CM), which is readily available, can increase RPE yield from hESC and determine the mechanism of this effect.
Materials and Methods
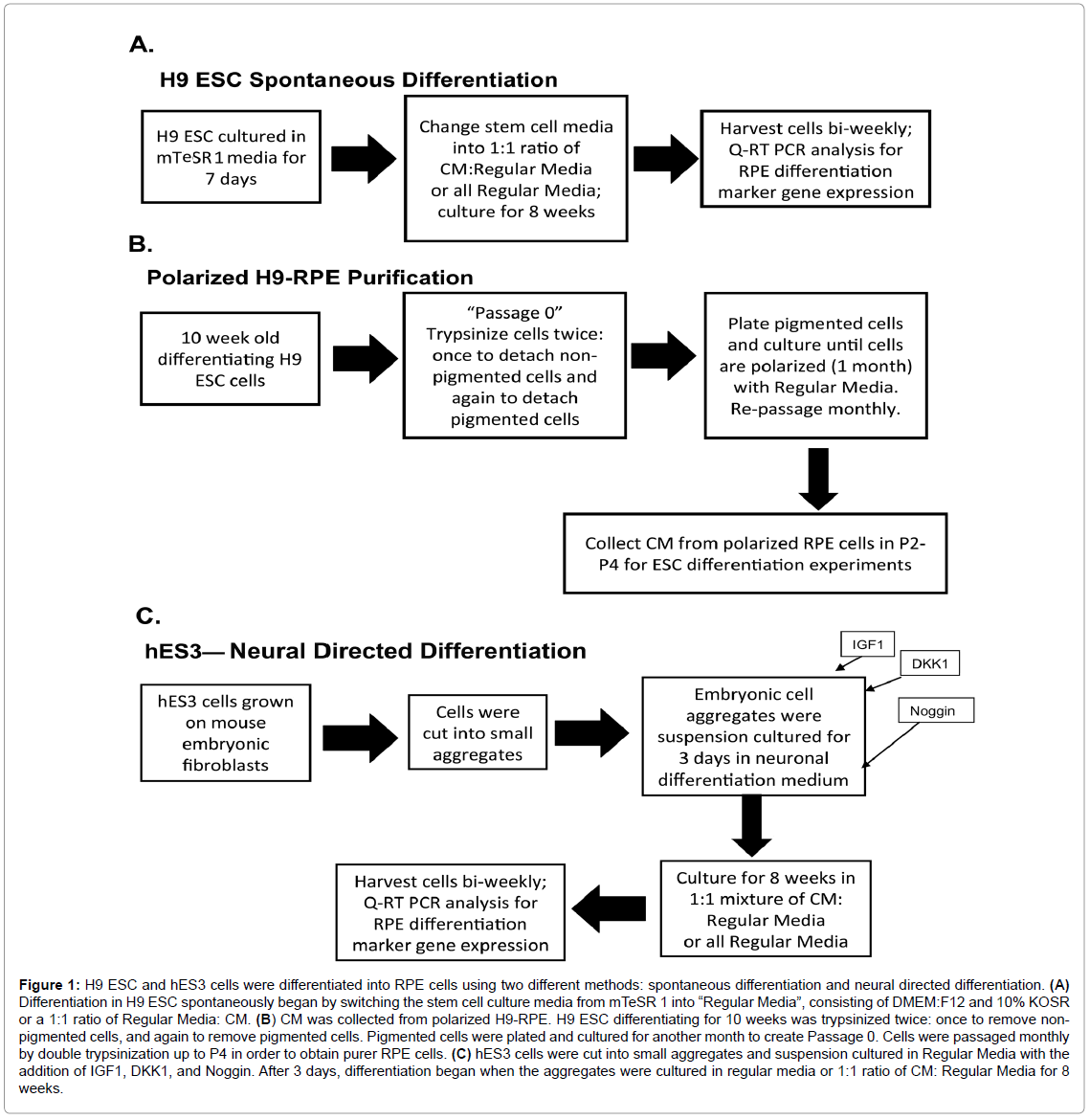
An overview of the differentiation process and experimental plan as described below is outlined in Figure 1. Regulatory approval for use of the H9 and hES3 ESC lines was obtained from the University of Southern California Stem Cell Research Oversight Committee (SCRO).
Figure 1: H9 ESC and hES3 cells were differentiated into RPE cells using two different methods: spontaneous differentiation and neural directed differentiation. (A) Differentiation in H9 ESC spontaneously began by switching the stem cell culture media from mTeSR 1 into “Regular Media”, consisting of DMEM:F12 and 10% KOSR or a 1:1 ratio of Regular Media: CM. (B) CM was collected from polarized H9-RPE. H9 ESC differentiating for 10 weeks was trypsinized twice: once to remove nonpigmented cells, and again to remove pigmented cells. Pigmented cells were plated and cultured for another month to create Passage 0. Cells were passaged monthly by double trypsinization up to P4 in order to obtain purer RPE cells. (C) hES3 cells were cut into small aggregates and suspension cultured in Regular Media with the addition of IGF1, DKK1, and Noggin. After 3 days, differentiation began when the aggregates were cultured in regular media or 1:1 ratio of CM: Regular Media for 8 weeks.
H9 cell culture and differentiation
H9 ESC cells (WiCell, Madison WI) were passaged every 5 days. Stem cell colonies were manually cut with the StemPro EZ Passage tool (Invitrogen, Carlsbad, CA), split 1:6 and cultured in mTeSR 1 medium (Stem Cell Technologies, Vancouver, BC, Canada) on 6-well plates coated with hESC-qualified, lactate dehydrogenase elevating virus (LDEV)-free Matrigel (BD Biosciences, San Jose, CA). Medium was changed daily with mTeSR 1 and spontaneous differentiation was initiated after 7 days of culturing by changing to “regular” medium (DMEM:F12, L-Glutamine, 15 mM HEPES (Corning Life Sciences, Tewksbury, MA) supplemented with 10% KOSR (Knockout Serum Replacement) (Life Technologies, Carlsbad, CA), or CM (50/50 mixture of regular medium and CM). Medium was changed every 3-4 days during the 8 week differentiation period.
RPE cell purification and culture
After 10 weeks of differentiation, RPE cells were purified from other differentiating cell types with a double-enzymatic method based on the fact that RPE cells are more adherent to the plate.
RPE cells were washed with Dulbecco’s phosphate-buffered saline without Ca2+ or Mg2+ (Corning Life Sciences) and then incubated with 0.05% trypsin, 0.1% EDTA in Hanks’ balanced saline solution (Corning Life Sciences) at 37°C for 5 minutes. The non-RPE cells were detached after the first trypsinization and pipetted out. The remaining putative RPE cells were again incubated with trypsin-EDTA for 5 minutes. The dissociated cells were counted and plated at a density of 1.3 × 105 cells per cm2 onto plates coated with phenol red-free, growth factorreduced, LDEV-free Matrigel (BD Biosciences). The cells continued to be cultured in the same differentiation medium as described above with the medium changed twice a week and were passaged every 4 weeks. Cells were used for experiments through passage 4.
Conditioned medium
Conditioned medium (CM) was collected twice a week from polarized H9-RPE [28] cultured in DMEM:F12 medium supplemented with 10% KOSR, 1 mM glutamine (Corning Life Sciences, Tewksbury, MA). The CM of cells from various passages (2-4) was pooled together. CM was obtained from RPE cells cultured for at least 1 month but no older than 2 months at the time of passage.
hES3 cell differentiation
Human embryonic stem 3 (hES3) cells were grown on mouse feeder cells and were obtained from the USC Stem Cell Core Facility. The cells were mechanically cut into small cell clusters with a 25-gauge ocular microsurgical knife and suspension cultured for 3 days in neuronal differentiation medium (DMEM:F12 1:1 with 10% KOSR, 1 × N2, 1 × B27, 2 mM glutamine, 1 ng/ml Noggin, 1 ng/ml DKK1, 5 ng/ml IGF1) to form EB. Following the 3 day suspension culture, the EB were plated on to Matrigel coated plates and cultured in the same differentiation medium as H9 cells described above.
RNA extraction
hES3 and H9 cells were harvested weekly for 8 weeks following differentiation. Cells were lysed according to kit instructions from RNeasy (Qiagen, Valencia, CA), and homogenized with Qiashredder (Qiagen, Valencia, CA). RNA extraction was performed following manufacturer’s instructions, and reconstituted in 30 μl nuclease-free water. 1 μl of RNA was converted to cDNA according to kit instructions from ImPromII (Promega, Madison, WI).
Q-RT PCR
Q-RT PCR was performed in duplicate following the instructions of the Light Cycler 480 SYBR Green I Master (Roche, Indianapolis, IN). CT values were obtained for the genes of interest and GAPDH housekeeping gene. Fold changes were calculated based on the average of 5 different biological samples. The primer sequences for the genes studied are listed as follows:
PAX6: Forward: CGCTAATGGGCCAGTGAGGAGC, Reverse: AAGGCCTCACACATCTGCGCG
PMEL: Forward: GGGCATCTTGCTGGTGTT, Reverse: GAGAAGTCTTGCTTCATAAGTCTGC
RPE65: Reverse: CCAGATGCCTTGGAAGAAGA, Reverse CTTGGCATTCAGAATCAGGAG
Bestrophin: Forward: TTCTATGTTGGCTGGCTGAA, Reverse: GCAGGTCCTGGTGCATCT
Beta 3 Tubulin: Forward: GCAACTACGTGGGCG ACT, Reverse: CGAGGCACGTACTTGTGAGA
Vimentin: Forward: GACCAGCTAACCAACGACAAAA, Reverse: GAAGCATCTCCTCCTGCAAT
PEDF: Forward: ACGCTATGGCTTGGATTCAG, Reverse: GGTCAAATTCTGGGTCACTTTT
GAPDH: Forward: CCCCGGTTTCTATAAATTGAGC, Reverse: CACCTTCCCCATGGTGTCT
Ki67: Forward: GAGAGTAACGCGGAGTGTCA, Reverse: TCACTGTCCCTATGACTTCTG
Oct4: Forward: GCTTCAAGAACATGTGTAAGCTG, Reverse: AGGGTTTCCGCTTTGCAT
Sox1: Forward: TACAGCCCCATCTCCAACTC, Reverse: GCTCCGACTTCACCAGAGAG
Cell viability studies
RPE cells were plated at 5 × 104 cells/cm2 cultured in regular medium or CM. For PrestoBlue (Life Technologies, Carlsbad, CA) analysis, the reagent was added to the cultures 2 days after plating according to manufacturer’s instructions, and incubated at 37°C for 2 hours before assaying for resazurin to resofurin reduction. Assay was performed obtaining the absorbance of the samples at 570 nm and 600 nm wavelengths using the Benchmark Plus Microplate Spectrophotometer (Biorad, Hercules, CA), and normalizing the 570 nm absorbance values to the 600 nm values. Percent reduction of PrestoBlue Reagent was calculated according to manufacturer’s instructions.
Pigmented cell quantification
Pigmented area percentage: The percentage of pigmented area was calculated by dividing the pigmented area by the total colony area. The colony area was determined by drawing a close-fitting rectangle around each individual colony using Powerpoint, which also indicated the length and width for area calculation. Six samples for each regular medium and CM were quantified for differentiating H9 and hES3 cells.
Pigmented colony quantification: The number of pigmented colonies per well of a 6 well plate were counted with N=6.
Immunohistochemistry
hES3, H9 ESC, and H9-RPE cells were fixed in 4% paraformaldehyde for 30 minutes, washed with PBS, and permeabilized with 0.1% Triton X-100 in PBS for 10 minutes. Cells were washed with PBS, and blocked with 1% BSA in PBS for 30 minutes. Primary antibody was incubated at 4°C overnight at the following concentrations: Bestrophin (Abcam, 1:500), Oct4 (Abcam, 1/1000), Sox1 (Abcam, 1/250). Cells were washed the following day with PBS, and secondary antibody, fluorescein antirabbit (Jackson ImmunoResearch) was added at a dilution of 1/100 and incubated at room temperature for 1 hour. Cells were washed again with PBS and mounted using Vectashield Mounting Medium containing DAPI (Vector Laboratories, Burlingame, CA).
Results
hESC grown in CM resulted in greater amount of pigmented cells
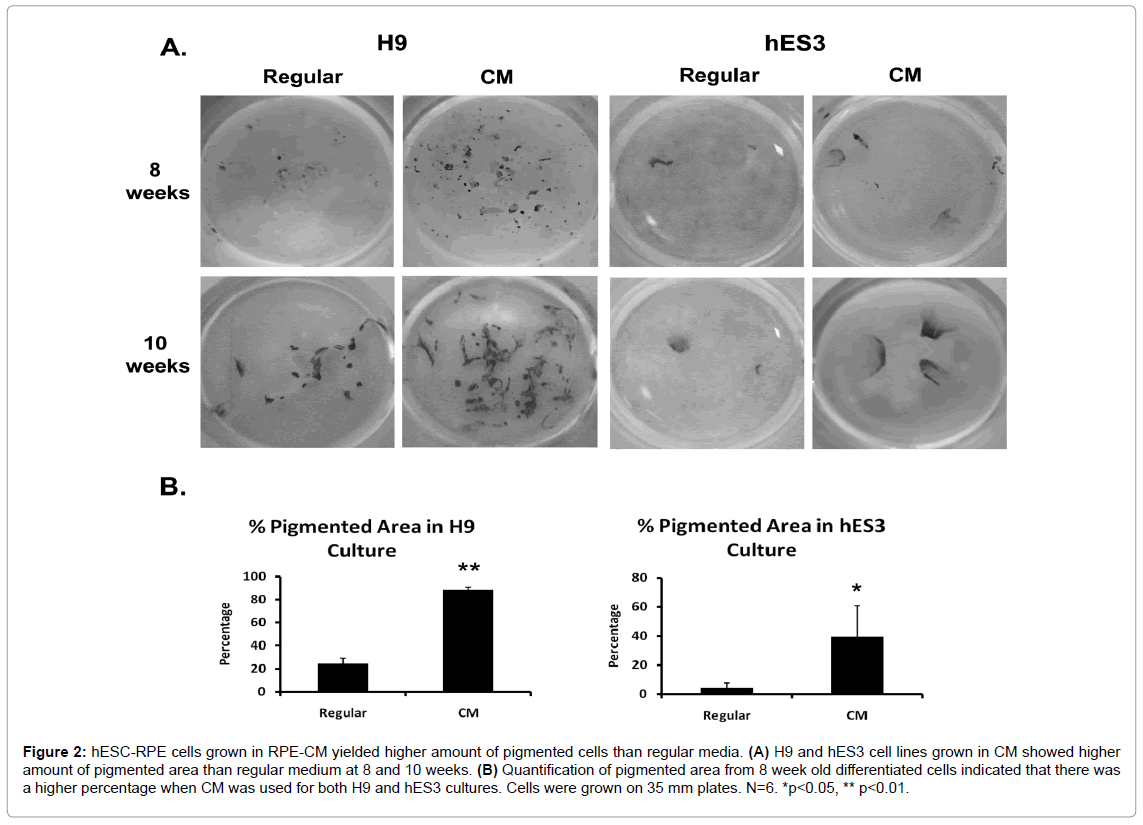
We tested the effect of CM on RPE differentiation in two different types of hESC lines, H9 and hES3. Here CM referred to the collected CM diluted with DMEM:F12 medium at a 1:1 ratio. Figure 2A shows that at 8 and 10 weeks of differentiation, CM-grown H9 and hES3 cells had more pigmented cells relative to cells grown in regular DMEM:F12 medium. For both cell lines, it appeared that CM yielded a greater number of pigmented colonies and the size of individual dark colonies was also larger. Interestingly, differentiating hES3-RPE cell colonies had a more fan-like appearance and appeared less pigmented than the darker, rod-like differentiating H9-RPE. The pigmented colony percentages were quantified in both cell lines (Figure 2B). H9 cells grown in CM resulted in 80% of the colony area being pigmented compared to 25% when cultured in regular medium. For hES3 cells grown in CM, approximately 40% of the colony area consisted of pigmented cells while regular medium yielded 5%. These results indicate that although H9 cells overall differentiated 5-6 times the amount of dark colonies relative to hES3, both lines cultured in CM still had higher amount of pigmented area compared to its corresponding regular medium
Figure 2: hESC-RPE cells grown in RPE-CM yielded higher amount of pigmented cells than regular media. (A) H9 and hES3 cell lines grown in CM showed higher amount of pigmented area than regular medium at 8 and 10 weeks. (B) Quantification of pigmented area from 8 week old differentiated cells indicated that there was a higher percentage when CM was used for both H9 and hES3 cultures. Cells were grown on 35 mm plates. N=6. *p<0.05, ** p<0.01.
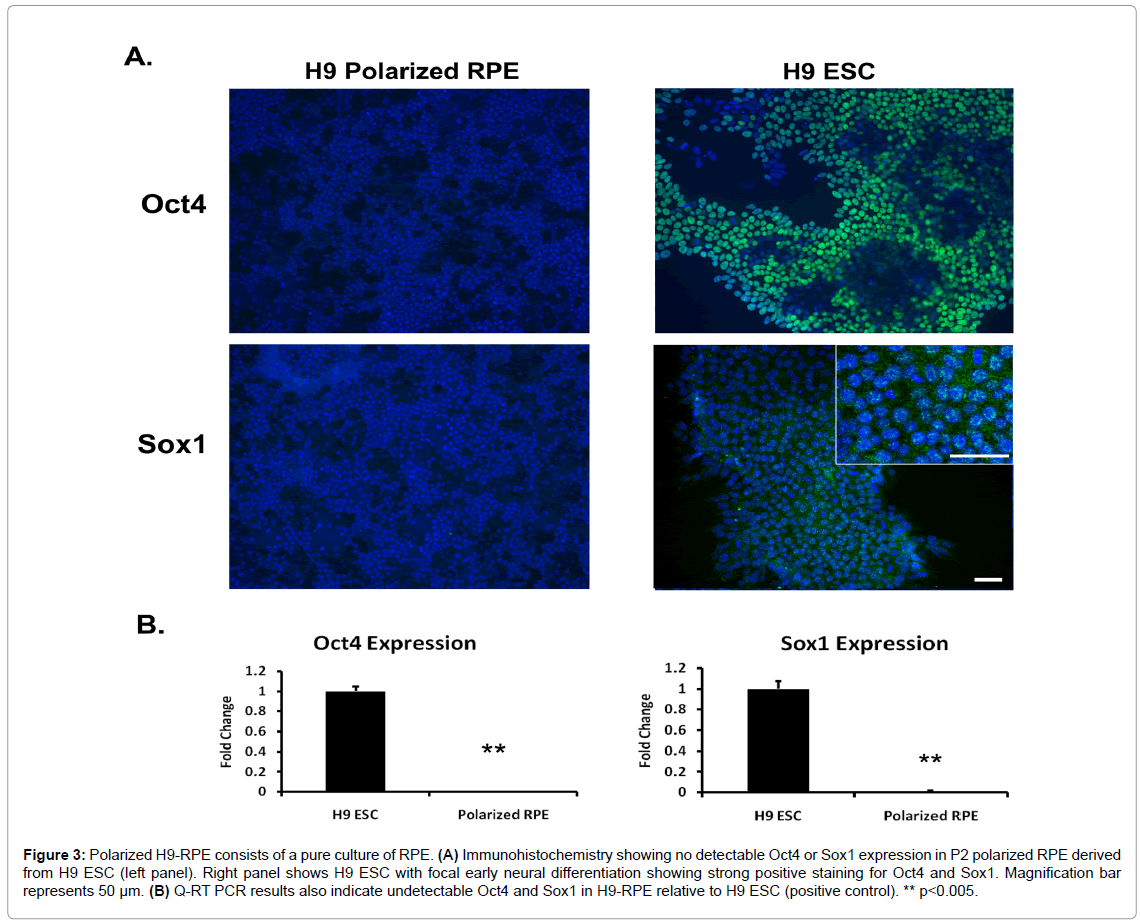
Polarized H9-RPE consists of a pure culture of RPE
The source of our CM, P2-P4 polarized H9-RPE, was confirmed to be a pure culture of RPE cells. Immunohistochemistry and Q-RT PCR showed no expression of stem cell marker Oct4 and early neural differentiation marker Sox1 in RPE, while positive control H9 hESC showed strong levels of expression (Figure 3).
Figure 3: Polarized H9-RPE consists of a pure culture of RPE. (A) Immunohistochemistry showing no detectable Oct4 or Sox1 expression in P2 polarized RPE derived from H9 ESC (left panel). Right panel shows H9 ESC with focal early neural differentiation showing strong positive staining for Oct4 and Sox1. Magnification bar represents 50 μm. (B) Q-RT PCR results also indicate undetectable Oct4 and Sox1 in H9-RPE relative to H9 ESC (positive control). ** p<0.005.
CM induced higher number of RPE differentiation from hESC
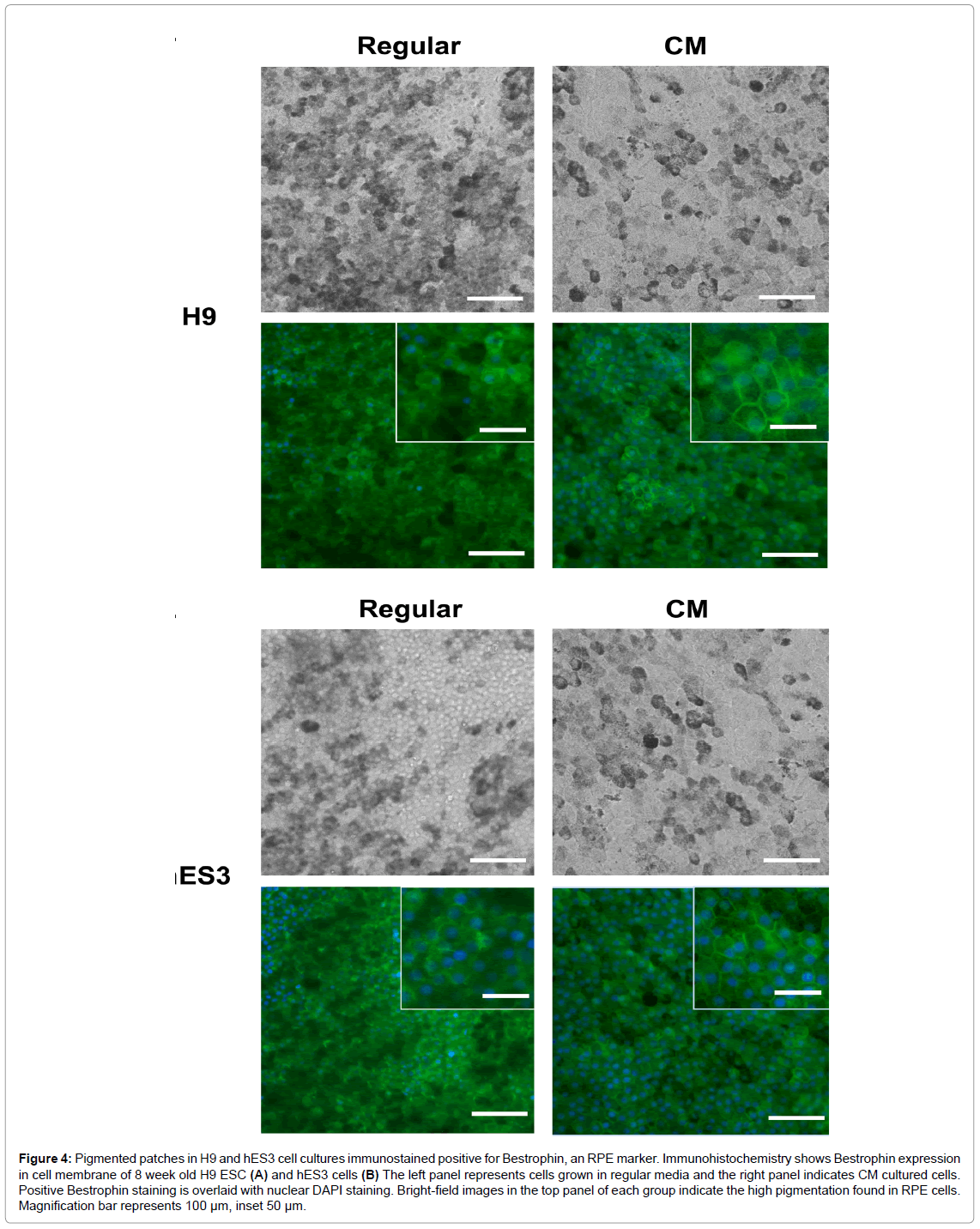
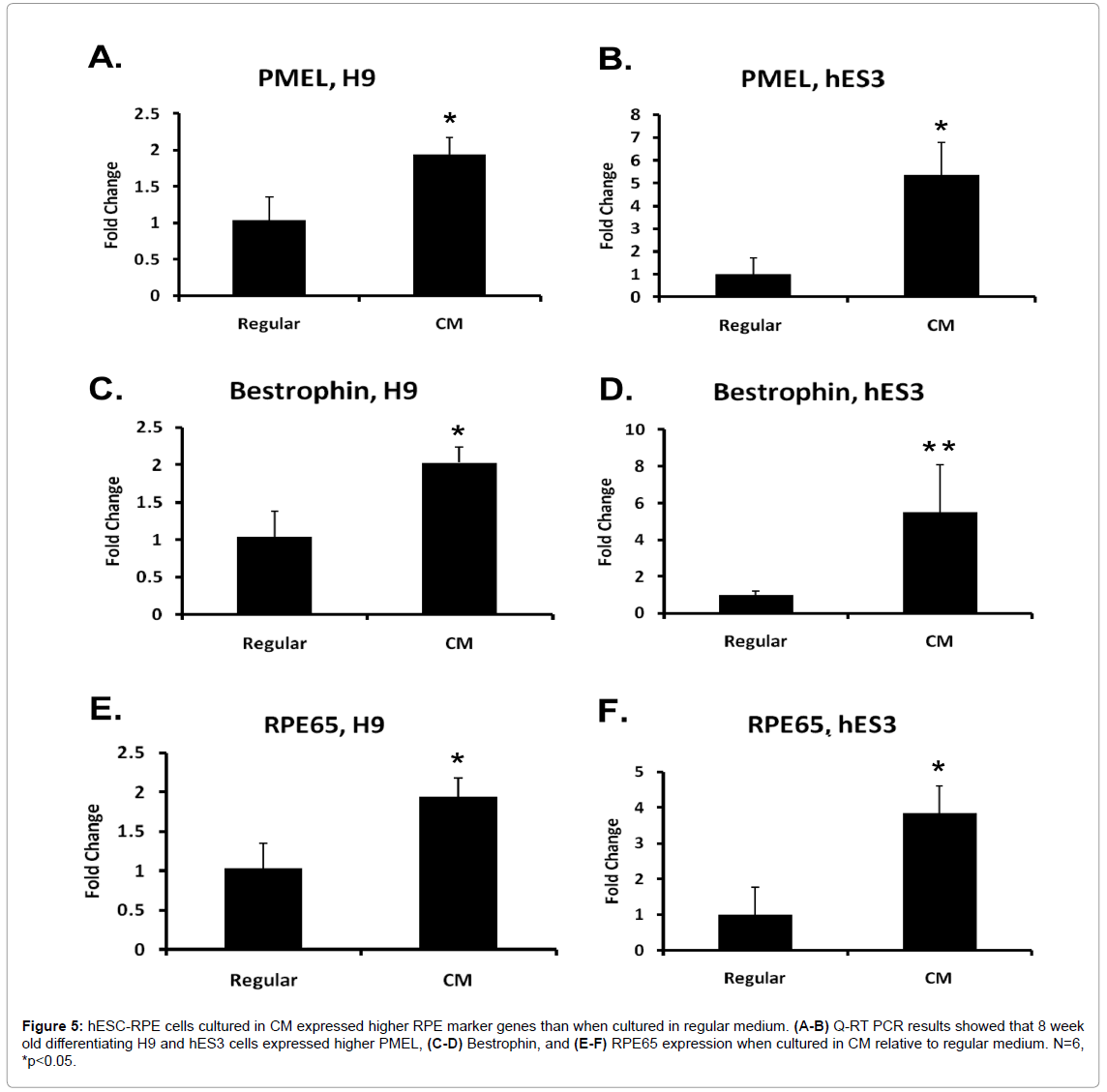
Next, we wanted to confirm that these pigmented cells are RPE cells. Cobblestone-like, pigmented cells in eight week old differentiating H9 and hES3 cultures grown in regular and CM immunostained positive for Bestrophin, an RPE-specific calcium-activated anion channel found in the cell membrane. There was no difference in the intensity level of Bestrophin between samples cultured in regular medium or CM (Figure 4). Q-RT PCR was used to measure the expression levels of selected RPE-specific marker genes. In addition to Bestrophin, these included premelanosome protein (PMEL) which is essential for pigmentation [31] and is expressed in middle-stage to mature RPE cells,as well as the mature RPE marker RPE65 which is a protein integral for regenerating photoreceptor visual pigment in the visual cycle. Figure 5 indicated higher expression in PMEL, Bestrophin and RPE65 in both lines differentiated in CM relative to regular medium. These results confirmed that the mix of differentiating cell types grown in CM contained higher amounts of RPE marker-expressing cells in both lines.
Figure 4: Pigmented patches in H9 and hES3 cell cultures immunostained positive for Bestrophin, an RPE marker. Immunohistochemistry shows Bestrophin expression in cell membrane of 8 week old H9 ESC (A) and hES3 cells (B) The left panel represents cells grown in regular media and the right panel indicates CM cultured cells. Positive Bestrophin staining is overlaid with nuclear DAPI staining. Bright-field images in the top panel of each group indicate the high pigmentation found in RPE cells. Magnification bar represents 100 μm, inset 50 μm.
Figure 5: hESC-RPE cells cultured in CM expressed higher RPE marker genes than when cultured in regular medium. (A-B) Q-RT PCR results showed that 8 week old differentiating H9 and hES3 cells expressed higher PMEL, (C-D) Bestrophin, and (E-F) RPE65 expression when cultured in CM relative to regular medium. N=6, *p<0.05.
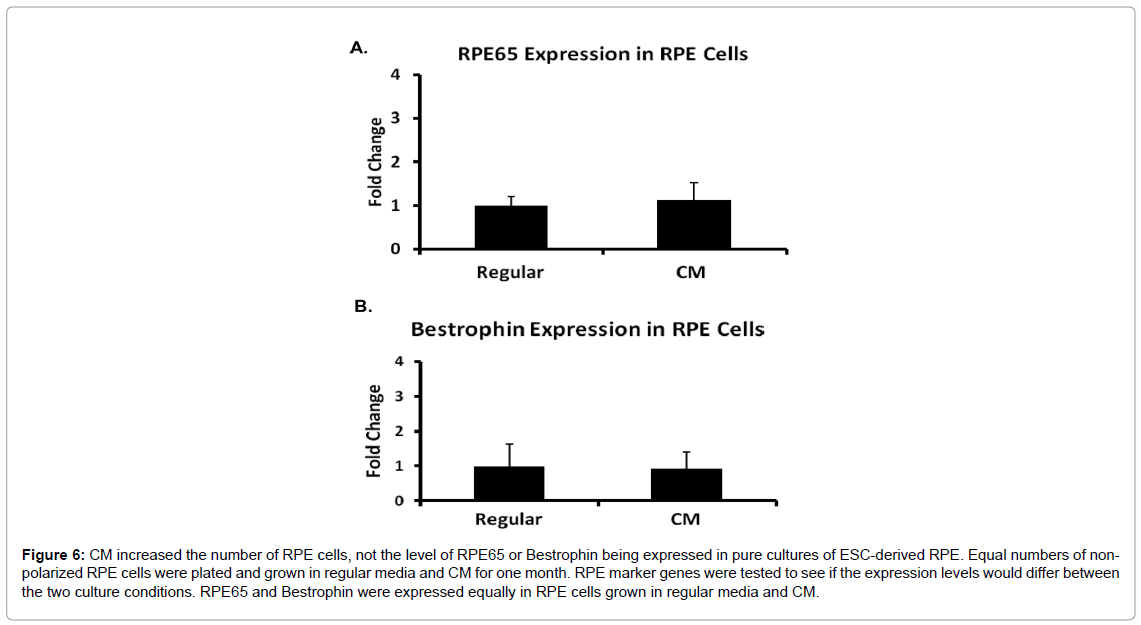
The elevated RPE marker gene levels of the hESC cultured in CM could have resulted from increased expression of those genes in each individual cell, as opposed to an increase in total number of RPE cells. To test this question, we plated equal numbers of RPE cells in regular medium and CM and cultured them for 1 month before harvesting. After 1 month, the cells in either medium were non-proliferative and polarized, and the cultures from CM or regular medium yielded almost the same amount of cells, which were approximately 8.0 × 106 cells per well of a 6-well plate. We expected the gene expression levels of RPE65 and Bestrophin would not be different between cells harvested from the two media with similar amount of cells if the increase in RPE marker genes was due to an increase in cell number. However, if there was an increase in RPE65 or Bestrophin levels in CM-cultured RPE cells, then we would conclude that CM caused an increase in RPE65 and Bestrophin expression in the cells themselves. Figure 6 showed that there was no difference in RPE65 and Bestrophin expression levels between cells grown in CM and regular media. The result indicated that the higher level of RPE marker genes in the differentiating hESC in CM was due to an increase in RPE cell number.
Figure 6: CM increased the number of RPE cells, not the level of RPE65 or Bestrophin being expressed in pure cultures of ESC-derived RPE. Equal numbers of nonpolarized RPE cells were plated and grown in regular media and CM for one month. RPE marker genes were tested to see if the expression levels would differ between the two culture conditions. RPE65 and Bestrophin were expressed equally in RPE cells grown in regular media and CM.
CM was not responsible for increasing the rate of RPE differentiation
The next question that needed to be addressed was how CM caused the increase of RPE cells in differentiating H9 ESC. We decided to look at the following possible mechanisms of whether CM increased (A) the speed of RPE differentiation, (B) proliferation of the differentiated RPE cells, and (C) the number of RPE colonies differentiated from hESC.
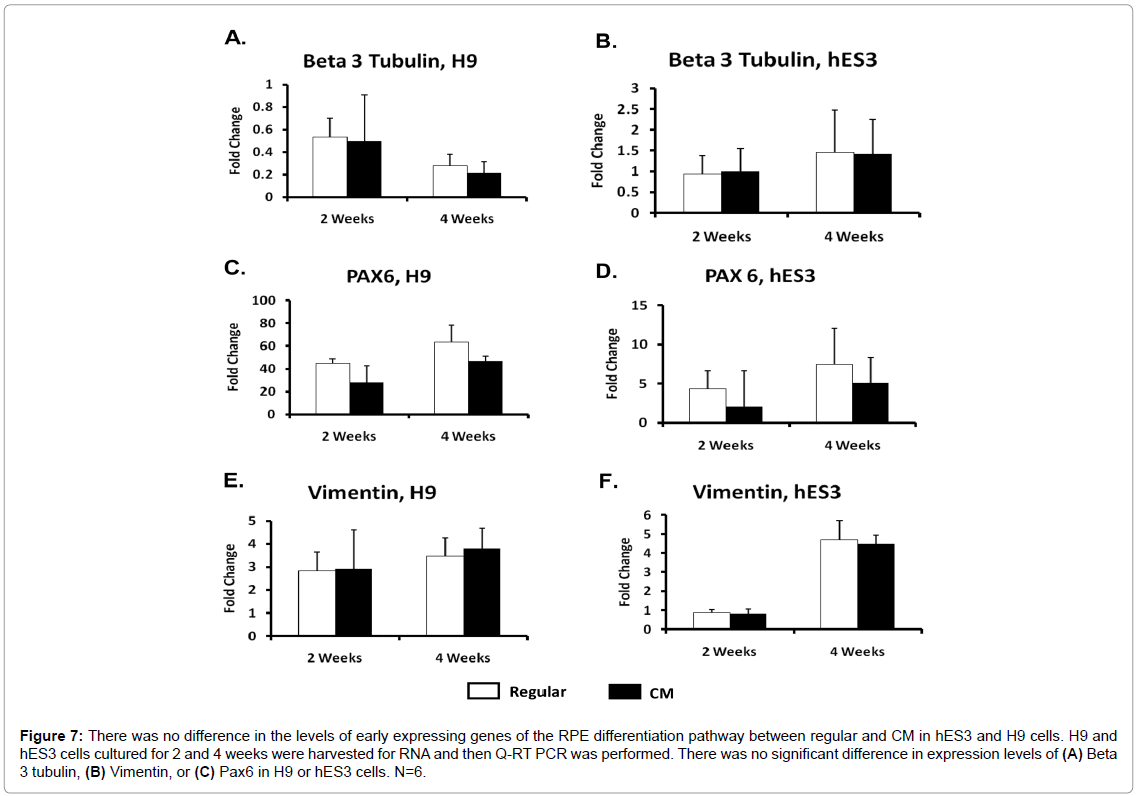
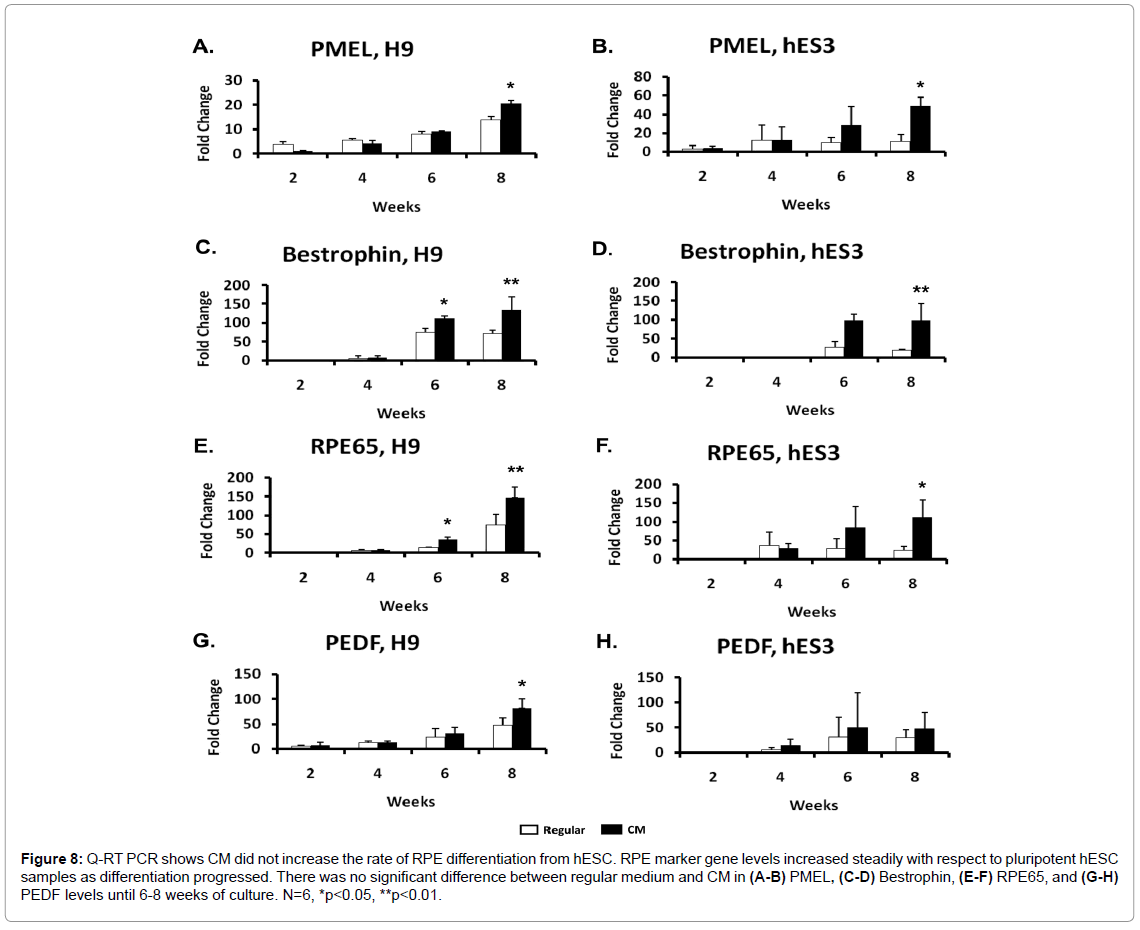
To determine whether CM increased the speed of RPE differentiation relative to regular medium, we tested the expression levels of early and late stage RPE signature genes. RNA was harvested from differentiating H9 and hES3 cells at 0, 2, and 4 weeks post-differentiation, using undifferentiated hESC as our Week 0 reference. We measured levels of Beta 3 tubulin and Pax6 which are expressed in the early stages of the eye field development, as well as vimentin, an intermediate filament which is also expressed in neural progenitors. There was no difference in expression levels in hESC cultured in CM and regular medium as early differentiation progressed (Figure 7). RNA was harvested from 2, 4, 6, and 8 weeks post-differentiation, and in addition to PMEL, which is expressed at the early stage of RPE differentiation, we also looked at mature RPE marker genes, RPE65, Bestrophin, and PEDF. There was no change in PMEL levels throughout the differentiation period until an increase at 8 weeks in CM (Figures 8A and 8B). Although Bestrophin and RPE65 first appeared in 4 week old differentiated hESC, there was no difference between the two culture media until weeks 6 and 8 in which hESC cultured in CM had higher Bestrophin expression relative to regular media (Figures 8C-8F). While H9 differentiating cells experienced higher PEDF expression levels at 8 weeks in CM differentiated cells, there was no significant difference between the two culture media in hES3 cells (Figures 8G and 8H). These results indicated CM does not speed up the appearance of RPE cells, but may play a role in increasing the amount of RPE in later stages of RPE differentiation.
Figure 7: There was no difference in the levels of early expressing genes of the RPE differentiation pathway between regular and CM in hES3 and H9 cells. H9 and hES3 cells cultured for 2 and 4 weeks were harvested for RNA and then Q-RT PCR was performed. There was no significant difference in expression levels of (A) Beta 3 tubulin, (B) Vimentin, or (C) Pax6 in H9 or hES3 cells. N=6.
Figure 8: Q-RT PCR shows CM did not increase the rate of RPE differentiation from hESC. RPE marker gene levels increased steadily with respect to pluripotent hESC samples as differentiation progressed. There was no significant difference between regular medium and CM in (A-B) PMEL, (C-D) Bestrophin, (E-F) RPE65, and (G-H) PEDF levels until 6-8 weeks of culture. N=6, *p<0.05, **p<0.01.
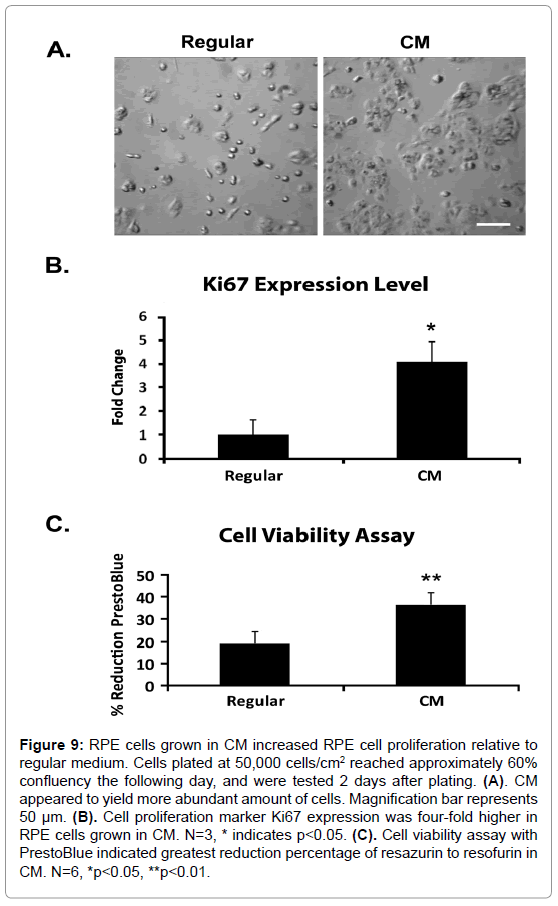
CM induced RPE cell proliferation
We noted the pigmented areas appeared larger in CM than in regular medium. Thus, we thought to investigate whether the addition of CM helped increase RPE cell proliferation. RPE cells were plated at a low density of 5×104 cells/cm2 and cultured for 2 days in regular medium or CM. Figure 9A showed CM having the higher density of RPE cells. The gene expression level of cell proliferation marker Ki67 correlated with the results shown in Figure 9A: Ki67 levels increased 4 fold in CM relative to regular medium (Figure 9B). Reduction levels of resazurin to resorufin using the PrestoBlue cell viability reagent measures cell proliferation. Figure 9C showed cells grown in CM had greater reduction of resazurin than the regular medium, with CM having the greatest reduction. These results indicated that one of the ways CM increased the amount of RPE in differentiating cells was by driving RPE cell proliferation.
Figure 9: RPE cells grown in CM increased RPE cell proliferation relative to regular medium. Cells plated at 50,000 cells/cm2 reached approximately 60% confluency the following day, and were tested 2 days after plating. (A). CM appeared to yield more abundant amount of cells. Magnification bar represents 50 μm. (B). Cell proliferation marker Ki67 expression was four-fold higher in RPE cells grown in CM. N=3, * indicates p<0.05. (C). Cell viability assay with PrestoBlue indicated greatest reduction percentage of resazurin to resofurin in CM. N=6, *p<0.05, **p<0.01.
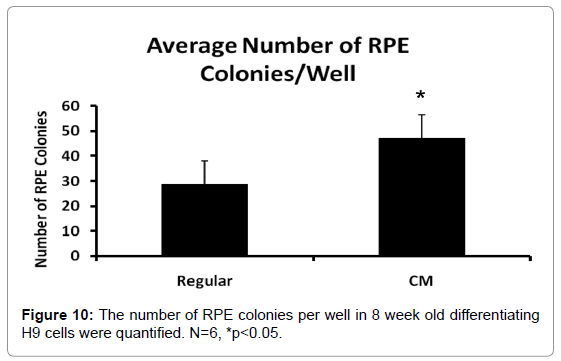
CM increased the number of RPE colonies during hESC differentiation
The RPE cell number rise in the CM-differentiated hESC could be attributed to increased number of differentiated RPE colonies. We counted the number of RPE colonies per well in the 2 different media conditions, with N=6 for each group. Individual pigmented bodies were counted as single colonies, regardless of size. CM cultured cells yielded the greater number of colonies (47) per well relative to regular medium (p<0.05) (29 colonies) (Figure 10). Additionally, the pigmented areas appeared slightly larger indicating that they could play dual roles in driving the number of differentiated RPE colonies as well as proliferation of those colonies
These results showed that although CM was able to increase the differentiation and proliferation of RPE, it did not appear to speed up the rate of RPE differentiation.
Discussion
RPE cells naturally secrete a large variety of growth factors in order to maintain proper retinal and choroidal morphology and function, as well as their own integrity. RPE CM has previously been shown to increase photoreceptor cell survival and differentiation, induce neurite growth, and even promote neuronal cell differentiation from retinoblastoma cells [28,29,32-34]. In this study, we investigated whether RPE CM has the additional ability to drive RPE differentiation from human embryonic stem cells. By using the medium collected from polarized H9-RPE cells, we determined CM could raise the amount of differentiated RPE by encouraging RPE proliferation and driving RPE colony formation from hESC. These findings provide another useful method to improve RPE differentiation from hESC.
While several groups have also shown good results with increasing the amount of RPE differentiation from hESC through the addition of different factors, the use of CM to generate hESC-RPE could be more advantageous and practical in clinical work. Vast amounts of hESCRPE grown in xeno-free conditions need to be generated and stored in cell banks for future patient use. CM, which can be obtained from animal-free hESC-RPE and was shown here to significantly increase RPE yield, could fulfill that requirement. Additionally, CM is composed of proteins that RPE cells naturally secrete. With CM, instead of a complicated protocol of adding specific growth factors at various points in time followed by the removal of other factors at later time points, the CM culturing method is simpler. The proliferative effect of CM on RPE was also shown to be heat stable and unaffected by extreme pH levels [35]. Furthermore, CM is easily obtainable and more economical, since media is being collected from existing polarized RPE cultures. On the other hand, the continuous supplementation of combinations of growth factors can be costly.
CM is also beneficial to RPE cell passaging. Following RPE differentiation from hESC, the cells need to be isolated and expanded for several passages before they are utilized. During cell passaging, RPE polarity is initially lost. The new passage needs to reach high confluency before pigmentation and tight junctions develop, which can take up to 4 weeks. We showed that CM increased the yield of differentiating RPE cells by driving RPE proliferation. However, the two RPE cell cultures resulting from the regular media and CM differentiation methods, had similar levels of Bestrophin and RPE65 expression. CM from cultured post-mortem human RPE was also shown to stimulate cell proliferation in RPE, astrocytes, and corneal fibroblasts [35]. By increasing cell proliferation, freshly split RPE cells initially grown in CM can reach confluency quicker than those grown in regular medium, thus lowering the amount of time required for RPE polarization. CM’s ability to enhance RPE proliferation could eventually shorten the amount of time needed in RPE polarization.
Both hES3 and H9 lines grown in CM showed a higher amount of pigmented cells compared to regular medium, but their relative abilities to differentiate RPE varied considerably. Even without the addition of CM, H9 cells were naturally able to produce a greater amount of RPE than hES3 cells. Interestingly, we showed here that neither CM nor regular media was observed to play a role in upregulating the early stage of RPE differentiation in either hES3 or H9 ESC. Other groups have also noticed varying responses in RPE differentiation between hESC lines. H9 and H1 cell lines have been shown to be some of the most efficient lines in RPE derivation [36] whereas another group noted that hES3 cells produced less RPE than H14 cells [37]. In our experience, we found that hES3 and H9 had distinctive responses to differentiation methods. H9 cells were spontaneously differentiated into RPE without the addition of any other factors, while hES3 cells experienced high cell death using this method and could not produce much RPE cells with spontaneous differentiation (data not shown). hES3 had to be grown in a suspension culture and supplemented with Noggin, DKK- 1, IGF1- to guide the cells toward a neural differentiation pathway, a precursor to RPE development before changing the medium to initiate differentiation [28,38]. This two-way differentiation method appeared to better prevent hES3 cell death than if they underwent spontaneous differentiation. On the other hand, even with this method to differentiate H9 cells, noticeably less RPE was yielded. Interestingly, CM’s effect of driving RPE differentiation was more profound in hES3 than H9 cells. This could be because H9 cells spontaneously produce higher amounts of RPE and the addition of CM had a relatively smaller effect. However, when hES3 cells, which produced smaller amounts of RPE, were cultured in CM, the relative increase in RPE was more striking. These findings signify the large variability in RPE production between hESC cell lines and their unique responses to various differentiation methods. Regardless, CM was still able to significantly enhance RPE differentiation between different hESC lines.
Our CM consisted of media pooled from passages 2-4 of polarized H9-RPE. Data from Kolomeyer’s study comparing CM obtained from polarized fetal RPE cultured for 30 days from Passage 2 and 4, suggested that there was little difference in secretion level in many of their tested trophic factors between passages [29]. However, when CM was obtained from Day 7 (likely non-polarized RPE) of Passage 2 and 4 there were noticeably different secretion levels of several trophic factors between passages. Similar to Kolomeyer’s presumed polarized RPE, we collected CM from our polarized H9-RPE at day 30 to 60 between passages 2-4; a time point in which we find similar growth factor secretion levels among the passages. Thus, use of pooled CM from polarized RPE from passages 2-4 should not create significant variations in hESC differentiation ability.
Although our results clearly indicate CM’s ability to induce increased RPE proliferation and yield, the particular trophic factors responsible for these capabilities are still undefined. Several growth factors known to be secreted by RPE have the potential to play a role in RPE proliferation or RPE differentiation including certain neurotrophins like CNTF and brain-derived neurotrophic factor. Other potential growth factors found in CM which could help enhance the amount of differentiated RPE include fibroblast growth factor (FGF) signaling molecules, such as basic FGF [29], members of the TGF-β family [25,26,39], as well as bone morphogenetic proteins [40]. Our group previously found PEDF to be secreted in high amounts in CM [28]. We tested its effect on RPE differentiation by adding recombinant PEDF (100 ng/ml) to differentiating hES3 cells for 8 weeks, but there did not appear to be any difference in pigmented cell levels relative to regular medium (data not shown). In future experiments the specific components of CM that mediate the increased RPE yield will be determined.
Conclusion
At the present time, CM is easily obtainable and economical with no additional growth factors needed, has previously been shown to be heat and pH stable [35], and was shown here to have the ability to increase RPE differentiation and proliferation. Additionally, with its ability to drive RPE proliferation, it can help with RPE cell expansion during passaging by cutting down the time needed for RPE polarization. The constituents found in CM hold promise in improving the manufacturing process of RPE differentiation from hESC.
Acknowledgements
The authors thank Ernesto Barron and Eric Barron for their help with figure design.
Grant Acknowledgements: This work was supported by California Institute for Regenerative Medicine (CIRM) grant DR1-01444 and by award number P30CA014089 from the National Cancer Institute. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Cancer Institute or the National Institutes of Health.
Author Contributions
Jamie Hsiung: Conception and design, collection and/or assembly of data, data analysis and interpretation, manuscript writing.
Danhong Zhu: Conception and design, data analysis and interpretation, manuscript writing.
Anthony Rodriguez: Assembly of data.
David R. Hinton: Conception and design, data analysis and interpretation, financial support, administrative support, manuscript writing, final approval of manuscript.
References
- Marmorstein AD (2001) The polarity of the retinal pigment epithelium. Traffic 2: 867-872.
- Strauss O (2005) The retinal pigment epithelium in visual function. Physiol Rev 85: 845-881.
- Handa J (2012) How does the macula protect itself from oxidative stress? Mol Aspects Med 33: 418-435.
- Radu RA, Yuan Q, Hu J, Peng JH, Lloyd M, et al. (2008) Accelerated accumulation of lipofuscin pigments in the RPE of a mouse model for ABCA4-mediated retinal dystrophies following Vitamin A supplementation. Invest Ophthalmol Vis Sci 49: 3821-3829.
- Allikmets R, Singh N, Sun H, Shroyer NF, Hutchinson A, et al. (1997) A photoreceptor cell-specific ATP-binding transporter gene (ABCR) is mutated in recessive Stargardt macular dystrophy. Nat Genet 17: 122.
- Gehrs KM, Anderson DH, Johnson LV, Hageman GS (2006) Age-related macular degeneration--emerging pathogenetic and therapeutic concepts. Ann Med 38: 450-471.
- Vugler A, Lawrence J, Walsh J, Carr A, Gias C, et al. (2007) Embryonic stem cells and retinal repair. Mech Dev 124: 807-829.
- Bressler NM, Bressler SB, Fine SL (1988) Age-related macular degeneration. Surv Ophthalmol 32: 375-413.
- Campa C, Harding SP (2011) Anti-VEGF compounds in the treatment of neovascular age related macular degeneration. Curr Drug Targets 12: 173-181.
- Bowes Rickman C, Farsiu S, Toth CA, Klingeborn M (2013) Dry age-related macular degeneration: mechanisms, therapeutic targets, and imaging. Invest Ophthalmol Vis Sci 54: ORSF68-80.
- Amit M, Carpenter MK, Inokuma MS, Chiu CP, Harris CP, et al. (2000) Clonally derived human embryonic stem cell lines maintain pluripotency and proliferative potential for prolonged periods of culture. Dev Biol 227: 271-278.
- Carr AJ, Smart MJ, Ramsden CM, Powner MB, da Cruz L, et al. (2013) Development of human embryonic stem cell therapies for age-related macular degeneration. Trends Neurosci 36: 385-395.
- Schwartz SD, Hubschman JP, Heilwell G, Franco-Cardenas V, Pan CK, et al. (2012) Embryonic stem cell trials for macular degeneration: a preliminary report. Lancet 379: 713-720.
- Klimanskaya I, Hipp J, Rezai KA, West M, Atala A, et al. (2004) Derivation and comparative assessment of retinal pigment epithelium from human embryonic stem cells using transcriptomics. Cloning Stem Cells 6: 217-245.
- Klimanskaya I (2006) Retinal pigment epithelium. Methods Enzymol 418: 169-194.
- Hemmati-Brivanlou A, Melton D (1997) Vertebrate embryonic cells will become nerve cells unless told otherwise. Cell 88: 13-17.
- Erceg S, LaÃnez S, Ronaghi M, Stojkovic P, Pérez-Aragó MA, et al. (2008) Differentiation of human embryonic stem cells to regional specific neural precursors in chemically defined medium conditions. PLoS One 3: e2122.
- Carpenter MK, Inokuma MS, Denham J, Mujtaba T, Chiu CP, et al. (2001) Enrichment of neurons and neural precursors from human embryonic stem cells. Exp Neurol 172: 383-397.
- MartÃnez-Morales JR, Rodrigo I, Bovolenta P (2004) Eye development: a view from the retina pigmented epithelium. Bioessays 26: 766-777.
- Levenstein ME, Ludwig TE, Xu RH, Llanas RA, VanDenHeuvel-Kramer K, et al. (2006) Basic fibroblast growth factor support of human embryonic stem cell self-renewal. Stem Cells 24: 568-574.
- Dhara SK, Stice SL (2008) Neural differentiation of human embryonic stem cells. J Cell Biochem 105: 633-640.
- Fuhrmann S (2008) Wnt signaling in eye organogenesis. Organogenesis 4: 60-67.
- Osakada F, Ikeda H, Mandai M, Wataya T, Watanabe K, et al. (2008) Toward the generation of rod and cone photoreceptors from mouse, monkey and human embryonic stem cells. Nat Biotechnol 26: 215-224.
- Fuhrmann S, Levine EM, Reh TA (2000) Extraocular mesenchyme patterns the optic vesicle during early eye development in the embryonic chick. Development 127: 4599-4609.
- Idelson M, Alper R, Obolensky A, Ben-Shushan E, Hemo I, et al. (2009) Directed differentiation of human embryonic stem cells into functional retinal pigment epithelium cells. Cell Stem Cell 5: 396-408.
- Buchholz DE, Pennington BO, Croze RH, Hinman CR, Coffey PJ, et al. (2013) Rapid and efficient directed differentiation of human pluripotent stem cells into retinal pigmented epithelium. Stem Cells Transl Med 2: 384-393.
- Sonoda S, Sreekumar PG, Kase S, Spee C, Ryan SJ, et al. (2009) Attainment of polarity promotes growth factor secretion by retinal pigment epithelial cells: relevance to age-related macular degeneration. Aging (Albany NY) 2: 28-42.
- Zhu D, Deng X, Spee C, Sonoda S, Hsieh CL, et al. (2011) Polarized secretion of PEDF from human embryonic stem cell-derived RPE promotes retinal progenitor cell survival. Invest Ophthalmol Vis Sci 52: 1573-1585.
- Kolomeyer AM, Sugino IK, Zarbin MA (2011) Characterization of conditioned media collected from cultured adult versus fetal retinal pigment epithelial cells. Invest Ophthalmol Vis Sci 52: 5973-5986.
- Vugler A, Carr AJ, Lawrence J, Chen LL, Burrell K, et al. (2008) Elucidating the phenomenon of HESC-derived RPE: anatomy of cell genesis, expansion and retinal transplantation. Exp Neurol 214: 347-361.
- Hellström AR, Watt B, Fard SS, Tenza D, Mannström P, et al. (2011) Inactivation of Pmel alters melanosome shape but has only a subtle effect on visible pigmentation. PLoS Genet 7: e1002285.
- Gaur VP, Liu Y, Turner JE (1992) RPE conditioned medium stimulates photoreceptor cell survival, neurite outgrowth and differentiation in vitro. Exp Eye Res 54: 645-659.
- Sheedlo HJ, Bartosh TJ, Wang Z, Srinivasan B, Brun-Zinkernagel AM, et al. (2007) RPE-derived factors modulate photoreceptor differentiation: a possible role in the retinal stem cell niche. In Vitro Cell Dev Biol Anim 43: 361-370.
- Tombran-Tink J, Johnson LV (1989) Neuronal differentiation of retinoblastoma cells induced by medium conditioned by human RPE cells. Invest Ophthalmol Vis Sci 30: 1700-1707.
- Bryan JA, Campochiaro PA (1986) A retinal pigment epithelial cell-derived growth factor(s). Arch Ophthalmol 104: 422-425.
- Liao JL, Yu J, Huang K, Hu J, Diemer T, et al. (2010) Molecular signature of primary retinal pigment epithelium and stem-cell-derived RPE cells. Hum Mol Genet 19: 4229-4238.
- Hu Q, Friedrich AM, Johnson LV, Clegg DO (2010) Memory in induced pluripotent stem cells: reprogrammed human retinal-pigmented epithelial cells show tendency for spontaneous redifferentiation. Stem Cells 28: 1981-1991.
- Lamba DA, Karl MO, Ware CB, Reh TA (2006) Efficient generation of retinal progenitor cells from human embryonic stem cells. Proc Natl Acad Sci U S A 103: 12769-12774.
- Tezel TH, Del Priore LV (1999) TGF beta secretion modulates the density-dependent growth of pig retinal pigment epithelium in vitro. Ophthalmic Res 31: 192-202.
- Adler R, Belecky-Adams TL (2002) The role of bone morphogenetic proteins in the differentiation of the ventral optic cup. Development 129: 3161-3171.
Citation: Hsiung J, Zhu D, Rodriguez A, Hinton DR (2015) Retinal Pigment Epithelial Cell Conditioned Medium Enhances the Yield of RPE Cells Differentiated from Human Embryonic Stem Cells. J Clin Exp Pathol 5:216. DOI: 10.4172/2161-0681.1000216
Copyright: © 2015 Hsiung J, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 16899
- [From(publication date): 4-2015 - Aug 29, 2025]
- Breakdown by view type
- HTML page views: 12153
- PDF downloads: 4746