Research Article Open Access
Screening and Isolation of Laccase Producers, Determination of Optimal Condition for Growth, Laccase Production and Choose the Best Strain
| Amutha C* and Abhijit M | |
| Department of Animal Behaviour and Physiology, School of Biological Sciences, Madurai Kamaraj University, Madurai, India | |
| Corresponding Author : | Amutha C Department of Animal Behaviour and Physiology School of Biological Sciences Madurai Kamaraj University, Madurai India Tel: 9942551479 E-mail: amuthaji@gmail.com |
| Received April 24, 2015; Accepted May 27, 2015; Published May 29, 2015 | |
| Citation: Amutha C, Abhijit M (2015) Screening and Isolation of Laccase Producers, Determination of Optimal Condition for Growth, Laccase Production and Choose the Best Strain. J Bioremed Biodeg 6:298. doi:10.4172/2155-6199.1000298 | |
| Copyright: ©2015 Amutha C, et al. This is an open-a ccess article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Related article at Pubmed Pubmed  Scholar Google Scholar Google |
|
Visit for more related articles at Journal of Bioremediation & Biodegradation
Abstract
This study aimed to find out appropriate laccase producing fungi help to greener the environment by decolourising industrial dyes and degrade BPA, besides removing heavy metal (Cd, Cr, Ni and Pb), viability among organisms and functional group activation in presence of heavy metal after comparison with some fungal isolates from terrestrial environment. In this study an important enzyme laccase is identified because it is not only common but easily extractable from fungus and having wide range of industrial and environmental applications. By this reason the best strain among all fungi from different area were compared and optimized its growth along with laccase production in modified Kirk and Farrell media and effect of economical agro wastes on laccase production also checked.
| Keywords |
| Bio-remediation; Endocrine disrupting chemicals; Bisphenol-A |
| Introduction |
| Scientific developments, innovations and achievements not only facilitate human life style but invariably pollute the environment differently. In recent times different types of toxic chemicals enter into the insitu environment due to industrial development. It is well known to us that endocrine-system is one of the important and complicated system in our body which can control vital hormonal secretion, can regulate different types of major functions in our body, but recently EDC’s (endocrine disrupting chemicals) are the most important threat to us, as well as for all living organisms due to blocking the endocrine function in different ways and it may come from any other sources like our daily life, it would be a tough challenge for us to restrict their devastating activity using our scientific concepts [1,2]. Not only EDC’s, there are several other types of environmental threats having different ways of function and effect on environment, like heavy metal toxicity it is a serious issue for environment in recent time [3]. In another side industrial wastes mainly, toxic industrial dyes also a biggest problems for water toxicity and have several negative effects on living organisms as well as on environment [4]. In most cases these kinds of threats are from our domestic discharges which are route cause for some of our serious issues as well as for environment. Several techniques are there to restrict these kinds of problems but either most of them are complicated or costly or non effective, but for this aspect it is the time to find out some effective, economical and simplest ways. It is very well known that microbes having massive diversity in several aspects, metabolic activity on several types of chemicals one of them, because they can use those chemicals (toxic /non toxic) for their basic source like C, N, P etc and after utilization several toxic chemicals lost their toxicity completely [5-7]. Now our most important duty is to use microbial metabolic activity for the degradation of toxic chemicals, so bioremediation will be an important phenomenon in future [8]. Side by side we have to extract the metabolic enzymes from microbes by which they can degrade toxic chemicals and we can use them in several types of industrial and environmental aspects. Some of microbial enzymes are there having wide variety of positive effect, like laccase [8- 10] most of the cases fungus are the major source of these enzyme but some bacteria have this kind of laccase production ability. This laccase is widely used in different aspects, not only for industrial development, it has lots of positive ability to make the environment green. |
| Materials and Methods |
| Sampling and positive isolates selection |
| Samples were collected from different regions of India like Kanpur, Kharagpur, Burdwan, Howrah, Pondicherry, Madurai, Meghamalai etc. Serially diluted soil samples added into potato dextrose broth in presence of 1% (w/v) benomyl 0.01% (w/v) chloramphenicol, 0.01% (w/v) tetracycline and kept it in room temperature for minimum seven days [11]. All the laccase producers are screened after benomyl selection on the basis of laccase specific substrates like guaiacol, tannic acid, 2,2-azino-bis (3-ethylbenzthiazoline-6-sulfonic acid) diammonium salt(ABTS), Syringaldazine and α- napthal etc [12-17]. Positivity of the isolates was determined from colour formation in presence of different substrates in both liquid, solid medium in presence of organism and using their crude extracts. |
| ¨ |
| Growth determination |
| All the positive isolates growth kinetics carried out after taking lyophilized dry biomass weight for the identification of fast grower among all positive isolates isolated from different parts of India [18,19]. In this comparison, study all positive isolates growth is very significant because enzyme production is directly proportional to the growth. |
| Decolourization study of commonly used industrial dyes |
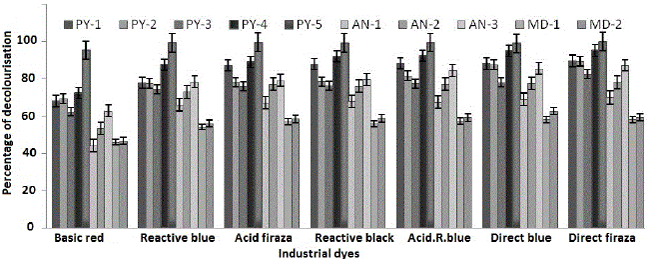
| All the positive isolates obtained were tested against some widely and commonly used industrial dyes collected from some different parts of India like, reactive black, basic red, direct firaza, acid firaza, acid reactive blue, reactive R. blue, direct blue etc [20,21]. In every set of experiment 5000 ppm concentration of industrial dyes used for decolourization percentage determination in minimal salt medium. All the decolourization percent determined from the 15th day treated sample using this formula [22-25]. |
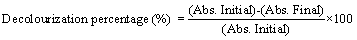 |
| Abs. initial=absorbance on 1st day; Abs. final=absorbance on 15th day |
| Enzyme activity determination |
| Crude extracts from all positive isolates was passed through Whatman No: 1 filter paper and 0.22 μm of syringe filter. Enzyme activity determined in presence of guaiacol using spectrophotometer at 470 nm [26] Lowry method followed for protein estimation [27] for enzyme activity determination the formula used [14,28,29]. |
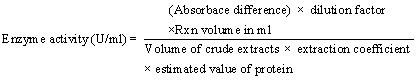 |
| Removal of heavy metals and Determination of functional groups activation in presence of heavy metals |
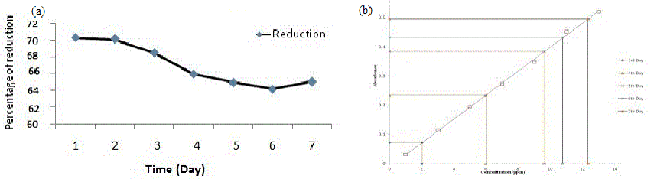
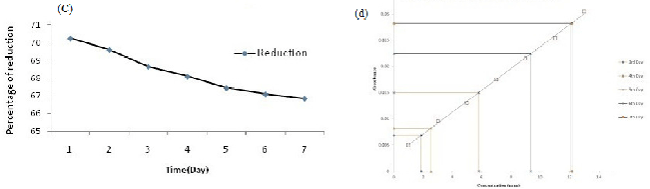
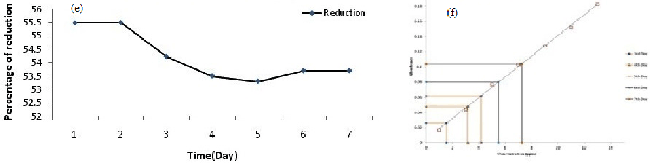
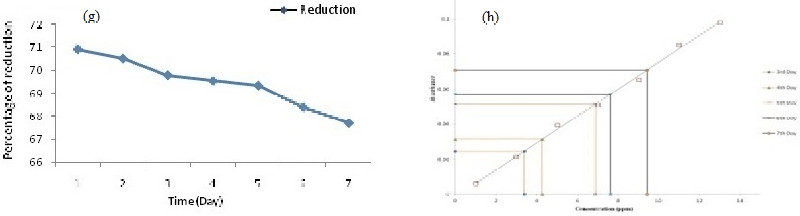
| All the positive isolates grew in presence of 500 ppm concentration of four different heavy metals, like cadmium, chromium, nickel, lead known for most wide range of toxicity in all over India and viability of strains tested using alamar blue. Heavy metal removal tested with lyophilized dry biomass of positive strains after taking samples from day interval up to seven days from heavy metal treated fungus culture with the help of atomic absorption spectrophotometer (AAS) (VARIAN AA240); during sampling after each day 4 ml samples took from culture flask, centrifuged and dried. The dried biomass lyophilized and kept in dust free condition in room, before going to take AAS, lyophilized treated biomass (2 mg) dissolved in concentrated nitric acid (8 ml), perchloric acid (2 ml) and kept it in 120°C in hot air oven for overnight until the solution become clear, if the solution still turbid after two consecutive days, then solution should pass through filter paper then only it is possible to run through AAS [30,31]. In this experiment all the test samples diluted four times according to machine efficiency. Standard curve prepared using 1, 3, 5, 7, 9, 11, 13 ppm concentration of known heavy metal concentration solution and concentration of unknown samples determined after extrapolating the absorbance value of 0th to 7th day samples to the ‘X’ axis. Cd, Cr, Pb, Ni removal measured by the help of AAS using absorption spectra 283 nm, 228 nm, 357 nm, and 223 nm respectively also individual day samples treated with alamar blue and absorbance spectra was taken at both 570 nm and 600 nm range to calculate the percent difference in reduction of the dye because it is directly proportional to the viability of the organism (fungus). A pinch from lyophilized heavy metal treated biomass used for the determination of functional group activation using FT-IR (SHIMADZU IRAFFINITY-1) with the help of potassium bromide (KBr) pellet method. In KBr pellet method, sample and KBr mixed in 1:100 ratio and maintained 7-8 ton pressure for 1 to 2 minutes, after getting the pellet was placed for IR treatment. Activation of functional groups determined after comparing with control samples (without heavy metal treated samples). Viability of organisms indirectly examined after determination of percent difference in reduction of alamar blue and the formula has given below Table 1 [32]. |
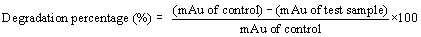 |
| Where |
| εox: Molar extinction coefficient of alamar blue oxidized form (BLUE) |
| εRED: Molar extinction coefficient of alamar blue reduced form (RED) |
| A: Absorbance of test wells. |
| A’: Absorbance of negative control well |
| A°: Absorbance of positive growth control well |
| λ1: 570 nm |
| λ2: 600 nm |
| Degradation study of Bis phenol-A (BPA) |
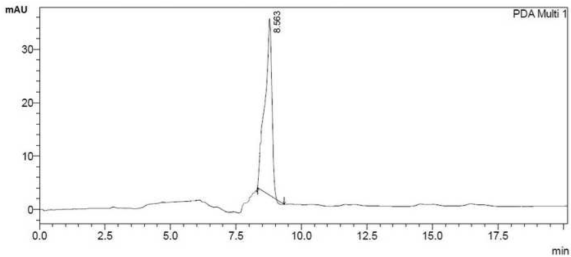
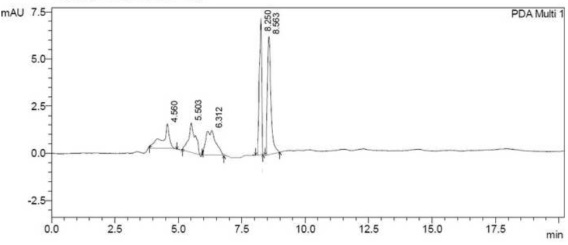
| All the positive isolates grew in presence of 200 ppm concentration of BPA in minimal salt medium, normal room temperature and 150 rpm (rotation per minute) maintained up to seven days. Control and seventh day samples were extracted using L.R grade ethyl acetate using separating funnel. The organic part from separating funnel collected very carefully and treated with anhydrous sodium sulphate for making the organic solution completely water free. Organic part completely evaporated using vaccum evaporator (Concentrator 5301-Eppendorf). Precipitated samples dissolved in HPLC grade methanol and run through reverse phase C-18 column of HPLC (SHIMADZU-CBM- 20A) with 35 percent acetonitrile used as a mobile phase [33-36]. |
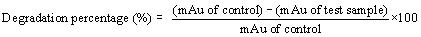 |
| mAu=miliampire |
| Optimization of fungal growth medium and laccase production |
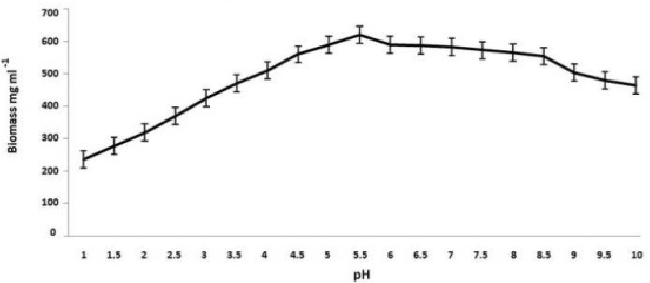
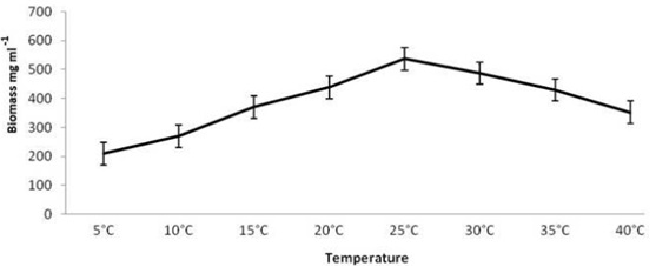
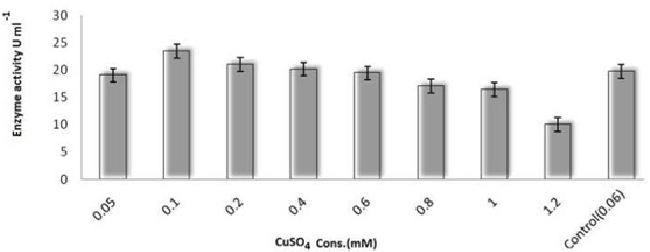
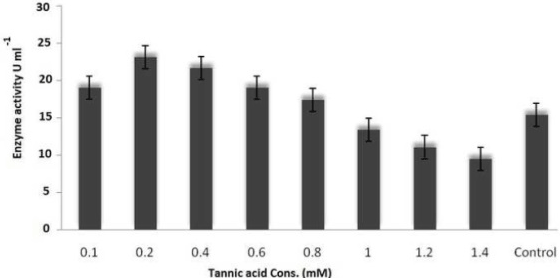
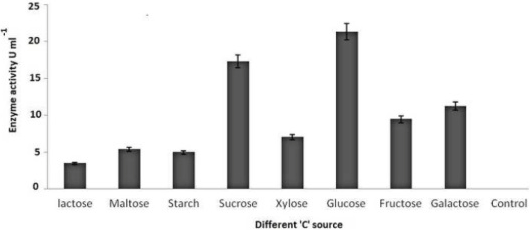
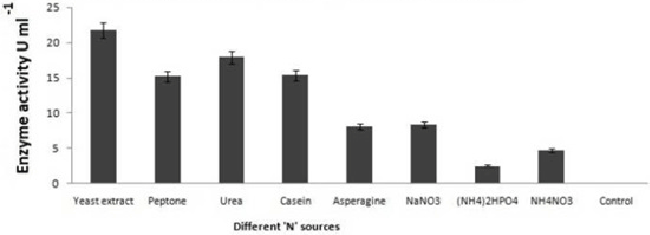
| Potato dextrose broth (PDB), Sabourand dextrose broth (SDA), Malt extract broth (MEB), Yeast glucose broth (YGB), Nutrient broth (NB), Czapek dox broth (CDB), Kimmig fungi broth (KFB) used for the determination of best medium for the growth of the best laccase producing isolates along with optimum Temperature and pH [37,38].Modified Kirk and Farrell medium [39] used for the optimization of laccase production. For optimization of laccase production temperature (28°C), pH (5.8), C-source, N-source, CuSO4, tannic acid concentration, organic solvents used to identify optimal condition [40- 43]. |
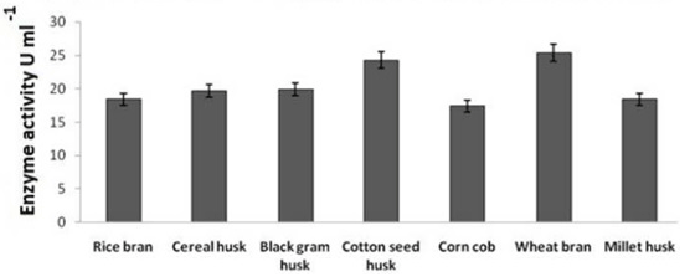
| Effect of agro wastes in laccase production |
| 30 gram of agro wastes moistened by distilled water in 250 ml of conical flask and sterilized by autoclaving maintained 120°C and 15 psi for 15 minutes. After autoclaving, cooled it inside laminar hood and a piece of YGA with fresh culture took using sterile sharp knife and inoculated inside the conical flask containing sterile agro wastes and incubated for 10 days in 28°C, after 10 days sterilized sodium acetate buffer 100 mM (pH 5.5) 100 ml added inside that conical flask and centrifuged at 120 rpm, 28°C for overnight. From this culture the liquid part used as crude extract containing laccase enzyme. This crude extracts used for enzyme activity determination to compare with other media in respect to enzyme activity. Guaiacol used as substrate for enzyme activity determination using UV-vis spectrophotometer at 450 nm [23,44,45]. |
| Identification of best strain |
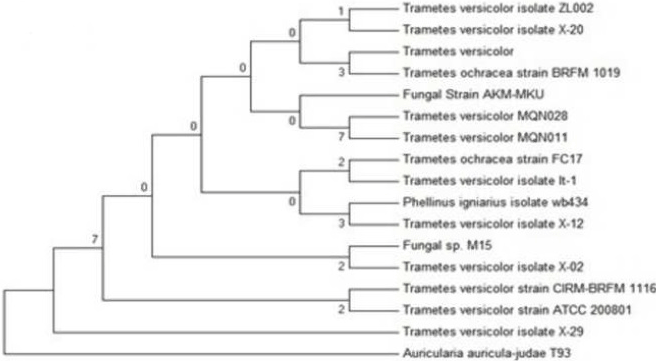
| After isolation of genomic DNA from the PY-5 (best isolate), the genomic DNA was amplified using universal primer ITS-1&4 (internal transcribe spacer) and run through 0.8% agarose in presence of 1 kb marker. Amplified product purified using PCR purification kit (sigma). Send the purified PCR product for sequencing. After sequencing, the strain identification was made by BLAST (basic local alignment search tool) and the phylogenetic relationship determined by MEGA 6 software using boot strap neighbour joining method. Based on the high growth rate, decolorization of industrial dye, enzyme activity (laccase), heavy metal removal and degradation of BPA, the best isolate was identified |
| Results and Discussion |
| Positive strains |
| Collection of all these seventy two isolates from eight different places in India, among these strains only fifteen isolates shows positive result i.e. laccase producing activity. These exhibit positive result by imparting appropriate colour formation in presence of specific substrate. Colour intensity varies due to variability in the concentration of laccase production. Different positive strains, their intensity of response in presence of specific substrate are given in Table 2. |
| Change in Biomass production |
| After fifteen days of incubation, biomass (mg l-1) was measured; during this study growth was determined to indirectly correlate enzyme production. The increase of organisms growth increase the laccase production (i.e.) here more biomass means more amount of laccase enzyme production which is more effective in environmental aspect. Bio mass production increment of all positive isolates along with maximum grower (PY-5) is given (Table 3). |
| Decolourisation percentage (%) |
| Decolourisation percentage was calculated from fifteenth day treated sample and it shown maximum decolourisation, after fifteenth day change in decolourisation percentage was almost constant. Fifteenth day decolourisation percentage data has given in Figure 1. |
| Enzyme activity |
| Enzyme activity of all positive isolates grown in some minimal salt medium like Olga and modified Kirk and Farrell medium was used to determined from the crude extract, in both medium strain PY-5 shown greater enzyme activity when compare to others isolates , also the modified Kirk and Farrell medium shown more enzyme production than Olga medium. The comparison of enzyme activity level has given in Figure 2 |
| Heavy metal removal and functional group activation |
| AAS data clearly reveals that the isolate PY-5 removes heavy metals more efficiently with respect to other positive isolates, after seven day PY-5 removes 12.34 ppm, 9.4 ppm, 12.13 ppm, 7.23 ppm of cadmium, nickel, chromium, lead respectively. During measurement through AAS all the four heavy metal treated samples diluted four times and simultaneously the viability was determined in presence of alamar blue because viability is indirectly related with heavy metal removal by absorption and adsorption. Adsorption is common for dead and alive cells but absorption is possible through channel or carrier protein and it is possible when cells are alive. The percent difference in reduction of alamar-blue in case of PY-5 presence of cadmium showed 65.10% of differences in reductions so remaining 34.90% of cells are inhibited or dead. The heavy metal removal, percent difference in reduction of alamar blue due to variable viability of the different positive isolates and their comparison has given in Figures 3a-3h respectively. Cadmium, chromium, nickel and lead treated biomass samples examined for the functional group activity determination with the help of FT-IR. From the FT-IR peak it is very clear that in presence of cadmium the hydroxyl group peaks became sharp to broad respect to control because of direct contact with the cadmium and hydroxyl groups from first day onwards, also same kind of trend happened in presence of chromium but in presence of chromium the peak broadening in seventh day respect to first day showed a significant peak broadening respect to control, it is due to slow effect of chromium on hydroxyl group respect to cadmium but in other metal treated samples there is no stretching or bending or broadening of any other peaks along with hydroxyl group peak. FT-IR result has given in Figure 4a-4d along with their individual control. |
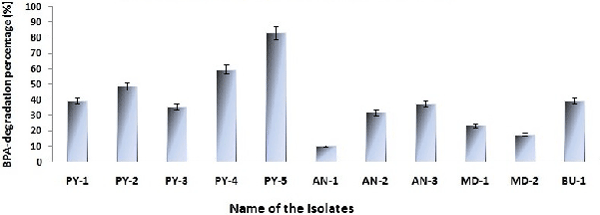
| BPA-degradation percentage (%) |
| Degradation of BPA in presence of all positive isolates calculated from the HPLC data and after 7th day the degradation percentage (%) of each individual positive isolates has given in Figure 5. From the Figure 5 it is very easy to detect that degradation of BPA by the isolate PY-5 is maximum. BPA degradation by isolate PY-5 detected through HPLC and the degradation identification with respect to control and 7th day treated sample has given in Figure 6a and 6b. |
| Optimization of fungal growth medium and laccase production |
| On employing different media and based on growth and laccase production, it is very clear that yeast glucose broth is best for the growth of PY-5 at pH-5.5 and temperature 28°C. Best media and its growth condition have given in Figures 7-9) respectively. Modified Kirk and Ferrell media used for the optimization of enzyme production in presence of pH-5.5 and 28°C of temperature, after optimization of media components and their concentration like CuSO4, organic solvents, tannic acid, ‘C’ and ‘N’ sources of already modified media it is now clear that the enzyme production became higher in remodified media (KF-2) respect to previous one (KF-1). Variation in the concentration of different components with variation in enzyme activity, optimal solvent concentration and difference between KF-1 and KF-2 in terms of enzyme activity has given in Figures 7-14. |
| Effect of agro wastes in laccase production |
| This experiment showed some significant result, that solid phase fermentation is more effective than liquid phase fermentation and economically affordable also, agro wastes are more effective for not only laccase production but also effective for other economically important enzyme production having positive role in environmental aspect (Figure 15). |
| Identification |
| Sequence of ITS-1&4 compared with database and it showed the 100% sequence similarity with Trametes versicolor. So the isolate PY-5 isolated from Pondicherry shown best environment friendly activity with respect to other positive isolates is Trametes versicolor. The dendrogram Figure 16 reveals the evolutionary relation of the best strain Trametes versicolor of this study. |
| Discussion |
| This comparative study shows among all positive isolates (PY-5) is the best respect to others positive strain in all aspect what criteria used in this study, only the best result showing strain sequencing has done, others strains sequencing yet to be done. In this experiment laccase production is one of the important criteria due to its broad range of activity, due to this reason growth interms of biomass production was the first criteria took in this study and laccase producion is dirrectly proportional to growth, from this growth curve PY-5 showed significant high growth rate respect to all other positive strains not only that, the growth started in case of PY-5 more earlier than others and growth increased in almost logarithmic manner. Next criteria was decolourization percentage of widely used industrial dyes having toxic effect on both aquatic,terrestrial living organisms, during this experiment all the dyes used were degraded above 95% after fifteen day of exposure only by PY-5 which is extremely higher than all other positive strains. In next experiment PY-5 showed the as usual higher enzyme activity respect to all other positive strains in both Olga and modified Kirk and Farrell medium. These three criteria are almost inter-related, decolourization depends on laccase activity, laccase production dirrectly proportional to growth and growth maximum means higher laccase production and enzyme activity will be more. In next study all the positive organisms grew in presence of heavy metals like Cr, Cd, Ni, Pb to detect their removal having wide range of toxicity and their toxicity is very common in all over India. From the comparative study , PY-5 showed removal (adsorption and absorption) of Cd, Ni, Cr, Pb are 12.34, 9.40, 12.13, 7.23 ppm respectively and these concentration will be four times more because during AAS study all the samples diluted four times,from this comparison and data, it showed that the removal of heavy metal in case of PY-5 is more than other positive strains and these amount of removal is more effective for environment because those removal concentrations are higher than those specific heavy metal’s environmental toxicity level. Along with heavy metal removal the viability testing is more important because removal can be possible through absorption and adsorption,adsorption is common for both dead and alive cells but absorption possible when the cells are alive. In heavy metal removal study, viability is related to absorption , these study showed in presence of Cd the percent reduction change of alamar blue gradualy reducing and from seventh day it was stabilised and increased, it indicates that decreasing order of percent reduction change due to decreasing of cellular activity and it is indirrectly proportional to either cell death or inhibition of cellular function, from seventh day exposure percent reduction change increase means activity again increased and in presence of Cd after some days of adaptation PY-5 survived and increased its growth. Same kind of incidents happened in case of lead, the percent reduction change from begining decreased but from seventh day onwards the percent reduction change became constant means after some exposure this strain also adapted and survived, but in case of Ni and Cr ,decrease of the percent reduction change lowered from fifth day onwards, it indicates that PY-5 after exposing in presence of Ni and Cr going to be adapted and survive slowly. Functional group activation also having some significant role with heavy metal interaction, from FTIR spectra of PY-5 in presence of Cd showed broadening and streatching of hydroxyl group from first day onwords to seventh day respect to control sample but other functional group unchanged, it indicates hydroxyl groups plays important role to interact with Cd but in case of Cr the hydroxyl group streatching and broadening gradually increased from first to seventh day. In case of Pb and Ni respect to control no change recorded in functional group streatcing and bending respect to control. Finaly comparison was done on the basis of the degradation of BPA, from this study PY-5 exhibited its higer quantity of BPA degradation 83.33%, which is more than others strain degradation activity. This study concentrated on BPA because of its silent threat, it is producing from epoxy rasin made water jar and after deposition in cells it can create several cronic disease in living system. After the comparison the best strain PY-5’s growth temperature, pH was optimized in presence of YGB after media detection, also the modified Kirk and Farrell medium again modifeid for enhancement of laccase production and results showed again modification is effective for laccase production. Due to high cost of cemicals economicaly low cost agrowastes can be better option than any other media, laccase production in presence of agro wastes mainly wheat bran showed significantly higher production of laccase. After identification of PY-5 it showed hundread percent sequence similarity with Trametes versicolor. From this study it is clear that Trametes versicolor is effective in bioremediation activity, specialy Cd toxicity and laccase production with the help of agro wastes can be very helpful for both industrial and environmental aspect. |
| Conclusion |
| From the above comparative study it is very clear that white rot fungus Trametes versicolor is effective among all fungal isolates isolated (in these comparative studies) from terrestrial region in respect of bioremediation as well as environmentally friendly activity. Comparatively easily available fungus Trametes versicolor can play a key role in making environment green not only that, easily available laccase enzyme from Trametes versicolor can play a crucial role in various industries due to its easy and wide level of availability, activity. |
| Acknowledgement |
| This research was supported by Department of Biotechnology-Interdisciplinary programme in Life science (DBT-IPLS, India). We thank to Dr. R. Ramachandran, Ellai raja for FT-IR analysis and Mr. Chinnasamy for providing AAS facility. We are thankful to our parents, well wisher, friends and almighty GOD. |
References
|
Tables and Figures at a glance
| Table 1 | Table 2 | Table 3 |
Figures at a glance
 |
 |
 |
 |
 |
 |
| Figure 1 | Figure 2 | Figure 3ab | Figure 3cd | Figure 3ef | Figure 3gh |
 |
 |
 |
 |
 |
 |
| Figure 4ad | Figure 5 | Figure 6a | Figure 6b | Figure 7 | Figure 8 |
 |
 |
 |
 |
 |
 |
| Figure 9 | Figure 10 | Figure 11 | Figure 12 | Figure 13 | Figure 14 |
 |
 |
 |
| Figure 15 | Figure 16a | Figure 16b |
Relevant Topics
- Anaerobic Biodegradation
- Biodegradable Balloons
- Biodegradable Confetti
- Biodegradable Diapers
- Biodegradable Plastics
- Biodegradable Sunscreen
- Biodegradation
- Bioremediation Bacteria
- Bioremediation Oil Spills
- Bioremediation Plants
- Bioremediation Products
- Ex Situ Bioremediation
- Heavy Metal Bioremediation
- In Situ Bioremediation
- Mycoremediation
- Non Biodegradable
- Phytoremediation
- Sewage Water Treatment
- Soil Bioremediation
- Types of Upwelling
- Waste Degredation
- Xenobiotics
Recommended Journals
Article Tools
Article Usage
- Total views: 20021
- [From(publication date):
July-2015 - Aug 24, 2025] - Breakdown by view type
- HTML page views : 14551
- PDF downloads : 5470
