Research Article Open Access
Stabilization of Pb in Pb Smelting Slag-Contaminated Soil by Compostmodified Bio Chars and their Effects on Maize Plant Growth
| Ogundiran MB1*, Lawal OO1, Sifau A1 and Adejumo2 | |
| 1Analytical/Environmental Unit, Department of Chemistry, University of Ibadan, Ibadan, Nigeria | |
| 2Department of Crop Protection and Environmental Biology, University of Ibadan, Ibadan, Nigeria | |
| Corresponding Author : | Ogundiran MB Analytical/Environmental Unit Department of Chemistry University of Ibadan Ibadan, Nigeria Tel: +2348023309861; E-mail: mbogundiran@yahoo.com |
| Received April 17, 2015 ; Accepted May 27, 2015 ; Published May 29, 2015 | |
| Citation: Ogundiran MB, Lawal OO, Sifau A, Adejumo (2015) Stabilization of Pb in Pb Smelting Slag-Contaminated Soil by Compost-modified Bio Chars and their Effects on Maize Plant Growth. J Bioremed Biodeg 6:297. doi:10.4172/2155-6199.1000297 | |
| Copyright: © 2015 Ogundiran MB, et al. This is an open-a ccess article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Related article at Pubmed Pubmed  Scholar Google Scholar Google |
|
Visit for more related articles at Journal of Bioremediation & Biodegradation
Abstract
Compost has been used to stabilize lead (Pb) in soil. However, compost contains a high level of dissolved organic matter (DOM) which may make Pb bioavailable in plant and thereby limiting its effectiveness and application. Addition of bio char to compost can reduce this effect. Rice husk (RH) and Cashew nut shell (CNS) bio chars and compost-modified bio chars were used in comparison to compost for stabilizing Pb in lead smelting slag (LSS)- contaminated soil (Pb=18,300 mg/kg) in Nigeria. Efficiency of Pb stabilization in control and amended soils was assessed using CaCl2 batch leaching experiment and plant performance. In pot experiments, maize plant was grown on the contaminated soil and on soil treated with minimum and optimum doses of the amendments singly and in combination for 6 weeks. Agronomical and chemical parameters of the plants were measured. CaCl2-extractable Pb in the untreated soil was reduced from 60 mg/kg to 0.55 mg/kg in RHB amended soils and non-detectable in other amended soils. RH-bio char/compost increased plant height, number of leaf and leaf area more than the others. Similarly, at minimum rate, it reduced root and shoot Pb by 91 and 86.0% respectively. Compost-modified rice husk bio char stabilized Pb in lead smelting slag contaminated soil, reduced Pb plant uptake and improved plant growth. Lead stabilization through the use of rice husk bio char with compost may be a green method for remediation of lead smelting slag-contaminated soil.
| Keywords |
| Bio char; Compost-modified bio char; Lead smelting slag; Pb stabilization. |
| Introduction |
| High concentrations of lead in soil that is contaminated by lead slag is dangerous to man and the ecosystem. The level of Pb above background concentrations in soil affects soil fertility, plant and animal health. High levels of Pb in soil can alter biomass of soil microbes and their activities in nutrient recycling. They can also cause plant physiological disturbances such as oxidative stress and reduction in plant yield [1]. Furthermore, a high concentration of Pb in soil is capable of migrating to other environmental media such as air and water bodies where it equally becomes poisonous to living organisms. Lead accumulation in human systems has been associated with neurological, reproductive, developmental, metabolic and behavioral changes [2]. |
| There are reports of soil contamination with Pb in many parts of the world that are known with mining and lead-acid battery recycling activities [3-5]. To circumvent bioavailability of Pb in contaminated soil, various methods of soil remediation have been employed. The commonly utilized methods include containment, biological treatments, in situ solidification/stabilization and immobilization [6,7]. However, organic wastes (such as compost and bio char) stabilization of heavy metals in contaminated soil is gaining ground over the conventional technologies [8-12]. This is because compost and bio char have strong binding power for heavy metals and have potentials to improve soil conditioning properties for plant growth [8,12]. |
| Studies have shown that remediation of Pb-impacted soil using compost stabilized heavy metals, restored soil fertility and promoted plant health on the remediated soils [8-10]. Bio char remediation of heavy metal-contaminated soil is an emerging soil reclamation method which employs biological residues combusted under low oxygen conditions, resulting in a porous, low density carbon rich material to decontaminate polluted soil [12]. Bio char contains high CEC which can retain essential plant macronutrients such as ca, Mg and K in soil. |
| Stabilization of heavy metals in soil may occur via complexation due to alkaline pH and surface organic functional groups of bio char [13]. It has been documented that bio char amendment improved soil Physicochemical properties such as particle size distribution, water holding capacity, soil organic carbon, pH, CEC, soil biological activities, and plant growth in addition to stabilization of heavy metals in soil [12,14]. Additionally, unlike compost, bio char has characteristic potential to remain long in soils with reduced bio-decomposition [15]. However, the energy required for pyrolysis of bio char production may an issue to be considered. Remediation technology that combines compost and bio char will be more environment friendly and less expensive in terms of reduction in fast decomposition of compost and energy required to produce bio char. |
| Previous research on heavy metals stabilization in soil by organic wastes was mainly with the use of compost [9,10] and bio char of different types [12]. But, not much information is available on the synergistic use of compost, rice husk and cashew nut bio chars as amendments for heavy metals stabilization in contaminated soils. Therefore, this study was designed to investigate the efficacy of compost (C), rice husk bio char (RHB), cashew nut shell (CNSB) bio char and compost-modified bio chars (CNSBC and RHBC) to stabilize Pb in lead smelting slag (LSS)-contaminated soil in Nigeria. The effectiveness was measured using CaCl2 batch leaching experiment, yield and Pb uptake by maize plant. |
| Experimental Soil Collection |
| The soil sample for this study was randomly collected from an abandoned lead smelting slag contaminated site in Ibadan, Nigeria. The site lies between longitude 7°24�?�?N and latitude 4°00�?�?E at an elevation of 174 m above sea level. The large expanse of agricultural land in this area has been made unproductive due to the impact of the LSS illegally dumped on the land several years ago. Soils were sampled at different points at 15 cm depth and mixed to form composite sample which was transported to the laboratory. |
| Amendments Preparation |
| The bio chars were produced from rice husk and cashew nut shell. These agricultural residues were chosen due to their abundance in Nigeria and in other places in the world. Their use as bio char sources will minimize the problem of managing their waste. Compost was prepared from wild sunflower (Tithonia diversifolia) and poultry liter in ratio 3:1 of sunflower to poultry manure for 12 weeks [9]. The different agricultural residues (rice husk and cashew nut shell) collected were pyrolysed locally in the presence of low oxygen using a simple, low cost two barrel charcoal retort method [16] at approximate pyrolysis temperature between 450 and 500°C. The produced chars were ground to fine powder and sieved through a 2 mm sieve. |
| Soil Physico-chemical Characterization |
| The soil samples were air-dried at room temperature for two weeks, mechanically ground and sieved to <2 mm. The <2 mm fraction soil samples were analyzed for physicochemical parameters. Organic carbon was determined using potassium dichromate method [17]. PH was measured in water at soil: water ratio of 1:1 using pH meter. Particle size analysis was determined by hydrometer method. To determine the total environmentally available Pb concentrations in the soil and amendments, one gram of soil was acid digested with 10 ml. 2 M HNO3 for 2 hours at 90-100 °C [5]. In order to solubilize the minerals in the bio chars and compost there is need to destroy the organic matter contents. To achieve this, one gram each of bio chars and compost was ashed in a muffle furnace for 6 hours at a temperature between 450- 500 °C. The ash was dissolved in 10 ml of 2 M HNO3 [18]. The total environmentally available Pb concentration in soil and amendments digests was determined by atomic absorption spectrophotometer (Buck scientific model 210A) with air-acetylene flame. Exchangeable cations (Ca, Mg, Na, K) were extracted using 0.2M Silver thiourea solution [19]. Potassium and Na were determined with a flame photometer (Jenway, PFP7) while Ca and Mg were measured using atomic absorption spectrophotometer. The cation exchange capacity was determined by the sum of the exchangeable bases including Ca, Mg, K and Na [20]. Kjedahl’s method was used to determine nitrogen content [21]. Phosphorus was determined by Mehlich III method [22]. The results of physicochemical properties of the soil, bio chars and compost are reported in Table 1. |
| Optimization Experiment |
| 0.01M CaCl2 batch leaching experiment was conducted to optimize Pb stabilization potentials of bio chars, compost and compost-modified bio chars in terms of the amount added to the contaminated soil. 0.0 g (contaminated soil without amendment), 0.05 g, 0.1 g, 0.2 g, 0.4 g of each bio char and compost was added singly and in combination with compost to 1.00 g of contaminated soil in 50 ml polycarbonate centrifuge tube. 5 ml of 0.01M CaCl2 solution was added and the solution was shaken for 2 hrs at 160 rpm using an orbital shaker, after which the solution was filtered using Whatman filter paper. The filtrates were analyzed for Pb concentration using atomic absorption spectrophotometer (Buck scientific model 210A). The percent stabilization efficiencies of the amendments were calculated. |
| Greenhouse Experiment |
| Pot experiment was set up in a greenhouse to study the effect of bio chars and compost both singly and in combination (equal proportion by weight using the minimum dose) on soil Pb stabilization, maize plant growth and Pb accumulation. For comparison, the most effective dose (0.4 g) and the minimum dose (0.05 g) of the amendments from the batch leaching test were selected for the pot experiment. The amount of bio chars and compost required were calculated with regards to the amount of soil used for planting (500 g) based on the results of the optimisation. This was equivalent to 25 g (50 g for combination) and 200 g of amendment per 500 g of soil to represent 0.05 and 0.4 g amendment/g soil rates respectively. Altogether there were 9 treatments and each treatment was performed in duplicate, giving a total of 18 bags. No extra macronutrient treatment from external source was applied to the soils. |
| The amendments were thoroughly mixed with the soils and transferred into 30 cm diameter polyethylene bags. Each bag was placed in a plastic bowl to prevent loss of leachate from the system and water was applied to each bag until soil reached 70% water holding capacity after which they were allowed to equilibrate for one week. During the period of one week the bags were watered every two days (approximately 100-200 mL). After one week, the soil was sown with two maize seeds but the survival of the seedlings to the end of the experiments was dependent on the type and rates of the amendments. Watering of the plants was done at two days intervals. The leachate collected in each saucer was also returned to the experimental soil when necessary to prevent loss of the metal. At six weeks of planting, agronomical parameters such as plant height, number of leaves and leaf area were taken. After 6 weeks, plants were harvested. The roots of the maize plants were washed with distilled water to remove soil, the plant were separated into shoot and root and oven-dried for 3 days at 80 °C. The dry biomass was determined. After biomass determination, the oven-dried plant parts were ground. Depending on the root dry weight of the plant ≤1.00 g of each was ashed in a muffle furnace for 6 hours at a temperature between 450-500°C for Pb determination [18].The experimental soils were also analysed for the total environmentally available Pb [5] |
| Results and Discussion |
| Physico-chemical characteristics of soil and the amendments |
| Table 1 presents the Physico-chemical properties of the soil, bio chars and compost. The pH of unamended soil was 5.7 which show that the soil was acidic. The pH of CNSB is slightly acidic (6.30) compared to the pH of rice RHB and compost which are alkaline. The acidic pH of CNSB can be attributed to its anacardic acids content [23]. The organic carbon of the soil was as low as 1.80%, which could be attributed to the high content of the slag which prevented vegetation of the soil. The concentration of Pb in the untreated soil was 18,300 mg/kg. Compost, RHB and CNSB have low Pb contents of 1.50, 0.90 and 1.2 mg/kg respectively, suggesting that there is no considerable contribution of Pb level when these materials are applied to the soil. The bio chars and compost have high levels of Ca, Mg, N, P, K and CEC which was expected to improve the soil condition and enhance plant growth on application. The Physico-chemical parameters present the proposed amendments as non-hazardous, as potential soil nutrients sources and as heavy metal stabilizers. |
| Effect of amendment dose on 0.01 M CaCl2 extractable Pb |
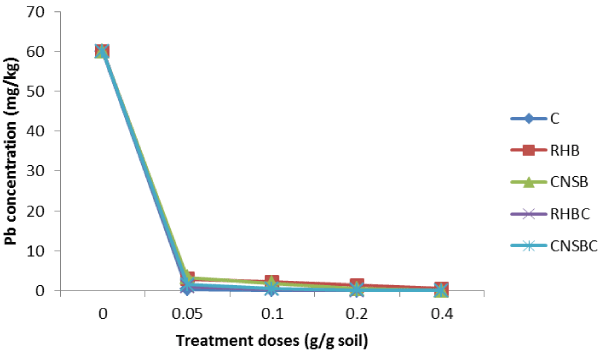
| The control and the amended soils at the different doses were subjected to 0.01 M CaCl2 solution extraction to determine extractable Pb. The extractable Pb was highly dependent on the dose and the amendment type. The results are summarized in Figure 1. As can be observed, increase in amendments doses from 0.05 to 0.4 g/g soil decreased the extractable Pb concentrations from 60 mg/kg in the control soil to non-detectable levels in the Compost, CNSB, Compostmodified bio chars and to 0.55 mg/kg in the RHB treated soils. |
| Effect of treatments on maize plant growth |
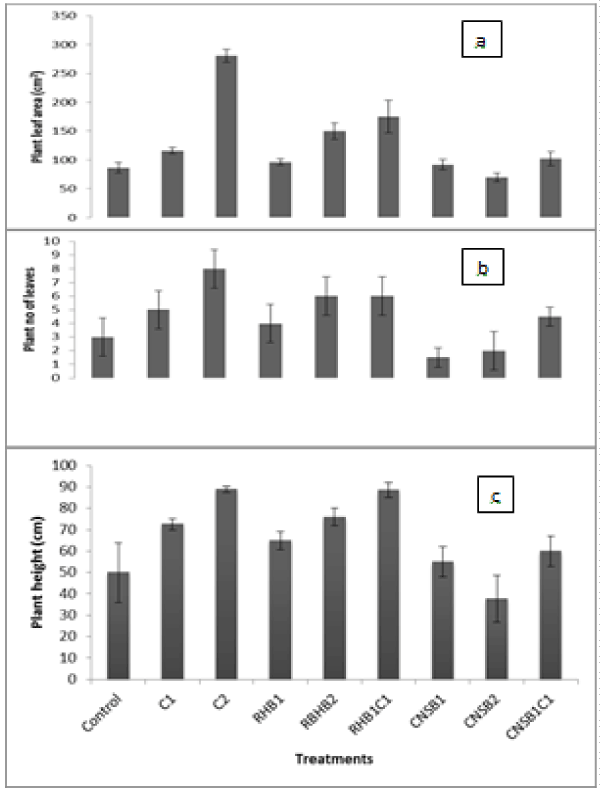
| To evaluate the efficiency of the treatments on the stabilization of Pb in the soil and bioavailability in plant, maize plants were grown for six weeks on the untreated and treated soils. It must be noted that although maize plant would not generally be considered as appropriate plant to evaluate treatment efficiency, it was used because it is a known lead accumulator [24]. This implies that if reduction in bioavailable Pb levels could be achieved with an accumulator through the application of the amendments on the contaminated soil, then it will possibly be better with other plants and the remediation plan will be judged successful. The growth parameter, that is, plant height, number of leaves and leaf area for the plants grown on the control and amended soils were measured and the results are presented in Figure 2. Maize growth was enhanced by the amendments except with higher rate of CNSB. There are variations in the mean plant height, number of leaves and leaf area ranging from 40 ± 11 to 90 ± 1 cm, 2 ± 1 to 8 ± 1 and 70 ± 8 to 280 ± 11 cm2 respectively among the plants grown on the soils. From the results, evidence of stunted growth perhaps due to Pb toxicity and the acidic nature of the soil were observed with plants grown on the control soil and the soil treated with higher rates of CNSB. |
| Plants grown on the soil that was amended with higher rate of CNSB showed mean height of 40 cm compare with 50 cm in the control, 2 leaves compared with 3 leaves in the control and an area of 70 cm2 compared with 86 cm2 in the control. The condition did not improve with addition of compost at this rate. CNS bio char is acidic as shown in Table 1. This likely had negative effect on the growth of the plant. On the other hand, compost, RHB and compost-modified RHB thrived with the three rates. The plants grown on them displayed healthy outlook and taller height with higher number of leaves and leaf areas. Plant parameters performed better with mixture of compost and the bio chars at lower rates of 50 g/500 g soil than when the two bio chars were used singly at very high rates of 200 g/500 g soil. The reason for this observation may be due to the combined effect of CEC and alkalinity impacted on the soil by the compost-modified RHB. As can be observed in Table 1, compost had the highest CEC followed by RHB and then CNSB. Cation exchange capacity of the amendments reflected their potential nutrient holding capacity. The combining effects of nutrient holding capacities of compost and RHB on making the contaminated soil fertile yielded healthier plants. |
| Effects of treatments on maize plant dry biomass |
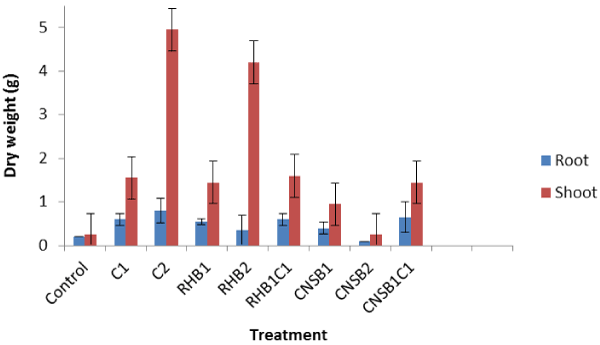
| The response of maize plant in terms of root and shoot dry biomass is presented in Figure 3. In view of the amendment type and rate, compost alone promoted the highest root and shoot biomass. For all other treatments, with the exception of CNSB at the rate of 200 g/500 g the root and shoot biomass yields were higher than those of the control plant. Cashew nut shell bio char at the highest rate (200 g/500 g) produced the least root (0.1) and shoot (0.25) biomass values. However, addition of compost to the CNSB even at a lower rate (50 (1:1) g/500 g) improved the performance of the bio char with 0.65 root and 1.45 shoot biomass values. The observed less performance of the CNSB may be associated with its acidic nature, the condition which is not favourable for plant growth. Unlike CNSB, the pH values of both compost (8.70) and RHB (7.40) are alkaline. The observed desirable performance of compost-modified CNSB may also be connected to this alkaline nature of compost which increased the pH of CNSB and therefore favours plant growth. This suggests that pH of the bio char is an important factor to consider when developing bio char remediation of heavy metals contaminated soils. |
| Effects of amendment on post-harvest soil Pb concentration |
| The concentrations of Pb measured in soil, maize plant root and shoot after harvesting are shown in Table 2. Total soil Pb content was reduced from 15,340 mg/kg in the control soil after harvesting to concentrations which ranged from 8,250 (C2) to 12750 mg/kg (CNSB1) in the treated soils. Similarly, total root Pb content was reduced from 2,260 mg/kg to concentrations which ranged from 200 (RHB1/C1) to 950 mg/kg (C1). Furthermore, total shoot Pb content was reduced from 530 mg/kg to concentrations which ranged from 74 (RHB1/C1) to 390 mg/kg (C1). The results demonstrate that compost performed best in terms of stabilization of Pb in soil while the compost-modified bio chars particularly mixture of rice husk bio char and compost at lower rate performed better at reducing Pb available in maize plant. This was followed by the two bio chars when used singly. The soil Pb levels in soil amended with higher rates (200 g/500 g) of compost and amendments were the least. This perhaps was due to dilution effect which high rate of amendment could produce on the contaminant in soil [25]. However, this high application rate may not be applicable in real field conditions. Overall, application of compost-modified RHB performed best in terms of both stabilization of Pb in soil and reduction in plant available Pb. |
| The observed decrease in the concentration of Pb in the soil, plant root and shoot may be associated with some inherent factors of the different amendments which changed the chemistry of the soil. The factors among others include the pH, organic matter and the chemical components of the amendments. |
| Sabilisation of Pb in soil can be achieved through adsorption, complexation and precipitation which are influenced by the pH, dissolved organic carbon (humic substances) and mineral components of the amendments and soil respectively. At lower pH of the soil, there is an increase in the H+ ion concentrations and it is expected that H+ion will compete with adsorption of Pb on the surface of the amended soil, thus decreasing stabilization of Pb in the soil. At high pH, OHion increases which favours soil stabilization of Pb. The acidic to near neutral pH (6.30) of CNSB coupled with acidity (pH=5.70) of the soil implies competition of H+ with Pb on the surface of the CNSB amended soil, hence less Pb stabilization. On the other hand, addition of compost and RHB to the soil was expected to increase the pH of the soil due to their alkaline pH, implying an increase in the negative sites of the soil which favoured Pb stabilization. |
| Compost and bio chars contain dissolved organic matter (humic and fulvic acids) which also possibly enhanced stabilization of Pb in soil. High ability of Pb to form more stable coordination complexes with humic acids has been documented [26]. Previously reported Fourier Transform Infrared (FTIR) studies on the organic constituents of compost and rice husk derived residues show that they contain organic functional groups such as alcohols, phenols, carboxylic acids and esters [27-29]. These functional groups would have become ionized at high pH and formed negatively charged ions which stabilized Pb. The degree of stabilization depends on the availability of the organic compounds in soil. Organic matter in bio chars breaks down slowly to form dissolved organic matter (humic substances). This may explain the lower stabilization of Pb in bio char amended soil when compared with compost amended soils. The release of dissolved organic matter in compost is faster due to the action of microorganisms, which implies higher concentrations in soil with more binding rates with soil Pb. This probably accounts for enhanced Pb stabilization in compost amended soil (Table 2). |
| Inorganic chemical composition of the amendments perhaps also played an important role in the stabilization of Pb in the soil and uptake by plant. As presented in Table 1, compost had high phosphorus content. Phosphorus reacts with Pb in soil to form pyromorphite [Pb5 (PO4)3OH], the most stable compound of Pb in the environment [30]. Xu et al. [28] identified peaks that were associated with CO3 2- and PO4 3- in FTIR spectra of Rice husk-derived bio char. This suggests that stabilization of Pb through precipitation of lead as carbonate and phosphate is a possibility. Typical XRF chemical characterization of RH ash revealed that it contains up to 92% SiO2 [31]. The result of this study also showed that RHB contained a high amount (9780 mg/kg) of Ca (Table 1). In a study by Wu et al. [27], XRD pattern of rice husk-derived bio char also indicates the presence of inorganic components such as CaCO3, KCl and SiO2. Following these obtained and documented results, Pb stablisation in the RHB amended soils may also be attributed to encapsulation of Pb in the calcium silicate hydrate (C-S-H) gel formed from the pozzolanic reaction of Ca with the silica content of RHB in an alkaline environment of the soil [32]. Thus, this prevented Pb from becoming available in compost-RHB amended soil. Nevertheless, further investigations are required to justify or reject these assumptions. |
| Effect of treatments on uptake of Pb by maize plant |
| The results of uptake of Pb in maize plant grown both on the control and amended soils are shown in Table 2. The root and shoot Pb concentrations in the untreated soil were substantially higher than those in the treated soils. In the plants that were grown on the control soil, Pb concentrations of 2260 and 530 mg/kg were observed in the root and shoot respectively. All the amendments reduced maize plant Pb uptake, although to different extents. The least Pb uptake by maize was observed in the soil treated with compost-modified RHB followed by compost-modified CNSB, RH-bio char, CNS-bio char and then compost. Compost-modified RH-Bio char at the rates of 50 g/500 g soil lowered the root concentrations by 91% similarly it reduced the shoot Pb from 530 mg/kg to 74 (86.0%). The results suggest that compost-modified bio chars performed better than the amendments being used singly. It is interesting to note that stabilization of Pb in soil amended with compost is better than stabilization of Pb in bio char amended soils. However, uptake by plant is higher in compost treated soils. This may be attributed to higher humic substances (humic and fulvic acids) contained in the compost. Fulvic acids (FAs) are smaller than humic acids (HAs) and are more chemically reactive than HAs. Due to their smaller size, they can enter plant roots and shoots readily thereby carrying along heavy metals. As cited by Boruvka and Drabek [26], heavy metals bound on insoluble humic substances are relatively immobile. On the other hand, binding on smaller organic molecules may increase metal mobility and bioavailability. Considering the results of this pot experiments, it can be deduced that remediation of Pb contaminated soil with compost-modified bio char has great potential to reduce bioavailable Pb. |
| Conclusions |
| The Physico-chemical parameters of compost, rice husk bio char and to an extent cashew nut shell bio char present them as non-toxic, potential soil fertilizers and heavy metal immobilizing agents. There are substantial observable reductions in extractable Pb concentrations in Pb smelting slag contaminated soil, maize plant root and shoot following the application of compost, rice husk and cashew nut shell bio chars and compost-modified bio chars. The combined effects of nutrient holding capacities of compost and RHB on making the contaminated soil fertile yielded healthier plants. The extent of stabilization might have been controlled by pH, organic matter and the chemical components of the amendments. Application of compost was the best in lowering Pb in soil while compost-modified RH- bio char was the best in lowering uptake by maize plant. Cashew nut shell performed less both in stabilization of Pb in soil and plant uptake especially when applied singly. The observed less performance of the CNSB may be associated with its acidic nature, the condition which is unfavourable to plant growth. This suggests that the pH of bio char is an important factor to consider when developing bio char remediation of heavy metals contaminated soils. Combination of compost with bio char could be a very effective way of decontaminating and revegetating lead smelting slag-contaminated soil. There is need for further studies on optimization of the use of compost modified-bio char in the contaminated soil, its effects on other minor quantities of heavy metals that may be contained in the soil, its mechanism of heavy metals stabilization in soil and field applications. |
References
|
Tables and Figures at a glance
| Table 1 | Table 2 |
Figures at a glance
 |
 |
 |
| Figure 1 | Figure 2 | Figure 3 |
Relevant Topics
- Anaerobic Biodegradation
- Biodegradable Balloons
- Biodegradable Confetti
- Biodegradable Diapers
- Biodegradable Plastics
- Biodegradable Sunscreen
- Biodegradation
- Bioremediation Bacteria
- Bioremediation Oil Spills
- Bioremediation Plants
- Bioremediation Products
- Ex Situ Bioremediation
- Heavy Metal Bioremediation
- In Situ Bioremediation
- Mycoremediation
- Non Biodegradable
- Phytoremediation
- Sewage Water Treatment
- Soil Bioremediation
- Types of Upwelling
- Waste Degredation
- Xenobiotics
Recommended Journals
Article Tools
Article Usage
- Total views: 14246
- [From(publication date):
July-2015 - Sep 01, 2025] - Breakdown by view type
- HTML page views : 9609
- PDF downloads : 4637
