Short Communication Open Access
Strategies to Reduce Infantile Cerebral Palsy
Kazuo Maeda*
Department of Obstetrics and Gynaecology (Emeritus), Tottori University Medical School, Yonago, Japan
- *Corresponding Author:
- Kazuo Maeda
Department of Obstetrics and Gynaecology (Emeritus)
Tottori University Medical School, Yonago, Japan
Tel: 81859226856
Email: maedak@mocha.ocn.ne.jp
Received Date: June 13, 2016; Accepted Date: June 17, 2016; Published Date: June 20, 2016
Citation: Maeda K (2016) Strategies to Reduce Infantile Cerebral Palsy. J Preg Child Health 3:263. doi:10.4172/2376-127X.1000263
Copyright: © 2016 Maeda K. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use,distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Pregnancy and Child Health
Abstract
Aim: To prevent cerebral palsy by avoiding fetal brain damage. Methods: 1. Emergency early delivery timing is before the loss of FHR variability, which is such severe fetal brain damage as anencephaly, 2. FGR caused by the fibrin deposit in placental intervillous space should be soluted to prevent severe hypoxia. 3. Preterm brain periventricular echo density (PVE) should be rejected to prevent PVL and CP, 4. Neonatal hypoxic ischemic encephalopathy (HIE) should be prevented by emergency C-section in sudden continuous bradycardia, and Developed HIE is treated by hypothermia. Anti-glutamate drug should be established. 5. Neonatal respiratory distress syndrome (RDS) is predicted by GLHW ultrasonic tissue characterization to treat by steroid in the fetus and artificial surfactant in neonate 6. Preeclampsia is treated by anti-sympathicotonic therapy to prevent placental infarction, hypoxia and CP, 7. Developed CP is treated by suitable stem cell therapy. Results: Effect to reduce infantile CP is shown in some strategies, while most of them should be established in the future.
Keywords
CP; Fetus; New-born; Preterm birth; Brain damage; Hypoxia; Ultrasound; PVL; PVE; NRFS; Fetal monitoring; FGR; Fibrin deposit; Loss of acceleration; Loss of variability
Introduction
The cerebral palsy (CP) is an infantile motility disability caused by various congenital or acquired brain damages. Its symptoms are paraplegia, quadriplegia, hemiplegia, spastic plegia, athetosis, ataxia and chorea, mostly without intelligential damage. Although maternal and perinatal infants’ mortalities were dramatically reduced in the past 100 years in Japan [1], world CP production rate has not been changed even after introduction of detailed fetal monitoring, despite some studies on fetal heart rate monitoring reported the reduction of CP in Japan [2,3]. Strategies to prevent CP will differ according to the situation and developing process of CP.
Methods, Results and Discussion
Rapid delivery in cases before the loss of fetal heart rate long term variability
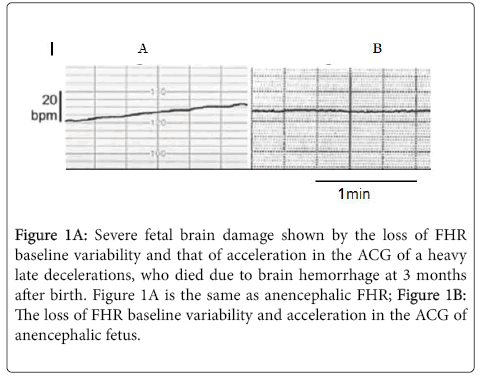
Although it was recommended to perform rapid delivery after the loss of fetal heart rate (FHR) long term variability, the strategy will reduce fetal death, but the CP prevention will be unable, because the FHR variability develops by the loss of fetal brain response to minor fetal movements in the most severe hypoxia after the loss of FHR acceleration in, which was the same as the complete loss of variability and acceleration against fetal movement in ACG of fetal anencephaly [4], namely, fetal state after the loss of variability will be the same as anencephaly, that is total loss of brain function, thus, the CP will not reduce. Therefore, early delivery with C-section should be done before the loss of variability. The developmental process was studied by the analysis of ACG, which recorded FHR and fetal movements, where the loss of FHR variability will be caused by the most severe hypoxic fetal brain damages, which may cause infantile CP (Figure 1).
Figure 1A: Severe fetal brain damage shown by the loss of FHR baseline variability and that of acceleration in the ACG of a heavy late decelerations, who died due to brain hemorrhage at 3 months after birth. Figure 1A is the same as anencephalic FHR; Figure 1B: The loss of FHR baseline variability and acceleration in the ACG of anencephalic fetus.
Therefore, the authors recommend to perform the rapid delivery with C-section before the loss of variability [5-9].
The strategy to deliver the fetus before the loss of variability will be the artificial births in cases of the loss of acceleration, which precedes the loss of variability, in the decreased long term variability, severe FHR changes but preserving variability, or in high hypoxia index, which may be tentatively 20-24 points, which was lower than the index in cases of the loss of variability, that was 25-26 [6], Hypoxia index is (the sum of fetal bradycardia duration [min] × 100)/the lowest FHR in the bradycardia lower than 110 pm. Further studies are required on the hypoxia index.
Since the loss of variability caused by the brain damage was as severe as anencephaly, thus it may develop at least one in 5,000 deliveries, therefore, new management will reduce 200 CP cases in 1,000.000 births in a year in Japan, which will be 5% or more of CP cases developed in Japan in a year, if the CP development rate is 0.2% (2/1,000) of births. The assumption may be adopted to the world deliveries.
The solution of placental intervillous fibrin deposit
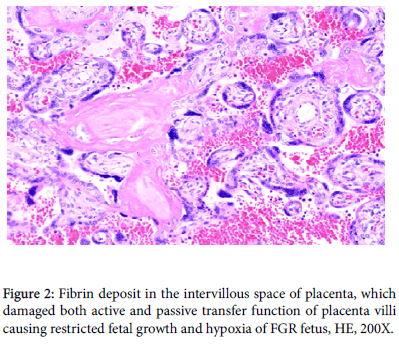
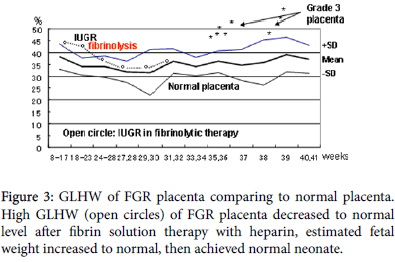
Fibrin deposits in intervillous space damage active transfer of fetal nutritive material from maternal blood to the fetus initially developing fetal growth restriction (FGR), and it progresses to damage passive villous transfer of oxygen in advanced FGR producing fetal hypoxia and fetal asphyxia then fetal death in cardiolypin antigen positive pregnancy. The fibrin deposit is diagnosed by the high tissue characterization value of gray-level histogram width (GLHW), and treated with 5000 iu heparin administration to the pregnant woman, where GLHW reduces, estimated fetal weight increases and fetal asphyxia was treated [5] (Figures 2 and 3).
Management of preterm neonates of periventricular echo density (PVE)
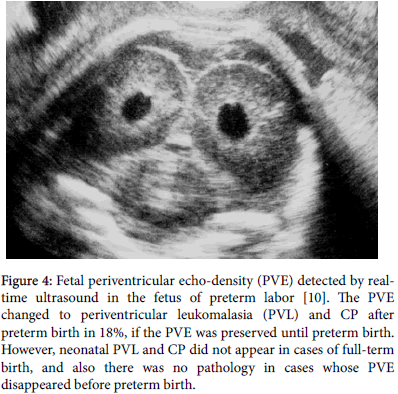
Infantile brain damage was frequent in preterm neonates [7]. The 18% of preterm neonates, who preserved PVE until preterm birth, which was high echogenicity of periventricular tissue in B-mode image (Figure 4), which showed larger gray-level histogram width (GLHW) value than normal brain in tissue characterization, and developed neonatal PVL and CP, which was around 0.2% of total birth, while the PVE vanished preterm birth or full term births did not develop neonatal PVL and CP [10].
Figure 4: Fetal periventricular echo-density (PVE) detected by realtime ultrasound in the fetus of preterm labor [10]. The PVE changed to periventricular leukomalasia (PVL) and CP after preterm birth in 18%, if the PVE was preserved until preterm birth. However, neonatal PVL and CP did not appear in cases of full-term birth, and also there was no pathology in cases whose PVE disappeared before preterm birth.
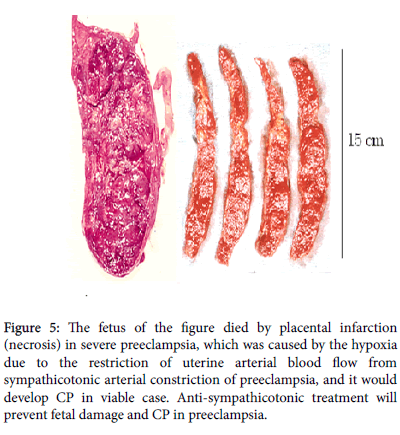
Therefore, the strategies will (1) Pharmacological tocolysis of preterm labour until full term births. or (2) Sedation of preterm labour until full term birth by anaesthesia or paralysis of uterus-brain nerve, which is the part of positive feed-back loop to produce regular preterm labour contraction [11-15]. or (3) Ultrasonic study of preterm infant’s brain immediately after preterm births to detect the PVE before developing PVL and the PVE will be treated until its disappearance by treatments with growth factor, hydrocortisone, erythropoietin etc., while their effect has not been established (Figure 5).
Figure 5: The fetus of the figure died by placental infarction (necrosis) in severe preeclampsia, which was caused by the hypoxia due to the restriction of uterine arterial blood flow from sympathicotonic arterial constriction of preeclampsia, and it would develop CP in viable case. Anti-sympathicotonic treatment will prevent fetal damage and CP in preeclampsia.
References
- Maeda K (2014) highly improved perinatal states in Japan. JOGR 40: 1968-1977.
- Takeshita K, Ando Y, Ohtani K, Takashima S, et al. (1989) Cerebral palsy in Tottori, Japan. Neuroepidemiology 8: 184-92.
- Tsuzaki T, Sekijima K, Morishita K, Takeuchi Y, Mizuta M, et al. (1990) The survey on the perinatal variables and the incidence of cerebral palsy for 12 years before and after the application of the fetal monitoring system. JpnActaObstetGinecol 42: 99-105.
- Maeda K (2014) Quantitative analysis of fetalactocardiogram: update. J Health Med Inform.
- Maeda K, Utsu M, Kihaile PE (1998) Quantification of sonographicechogebesity with grey-level histogram width: a clinical tissue characterization. Ultrasound Med Biol 24: 225-234.
- Maeda K (2014) Modalities of fetal evaluation to detect fetal compromise prior to the development of significant neurological damage. J ObstetGynecol Res.
- Ishii N, Kono Y, Yonemoto N, Kusuda S, Fujimura M (2013) Outcome of infants born at 22 and 23 weeks’ gestation. Paediatrics 132: 62-71.
- Maeda K (2014) Origin of the long-term variability and acceleration of FHR studied for the prevention of cerebral palsy in fetal hypoxia and general insults. J Perinat Med 42: 401-403.
- Maeda K (2013) Actocardiographic studies of fetal hypoxia detected by the bradycrdia, loss of fetal heart rate acceleration and long term variability. J Health Med Inform.
- Yamamoto N, Utsu M, Serizawa M, Ohki S, Murakoshi T, et al. (2000) Neonatal periventricular leukomalacia preceded by fetal periventricular echo density. FetalDiagTher 15: 198-208.
- Maeda K (2013) Uterine contractions in normal labor developed by a positive feedback and oscillation. J Health Med Informat.
- Choi DW, Koh J (1988) Peters S Pharmacology of glutamate neurotoxicity in cortical cell culture: attenuation by NMDA antagonists. J Neuroscience 8: 185-186.
- Serizawa M, Maeda K (2010) Non-invasive fetal lung maturity prediction based on ultrasonic gray level histogram width. Ultrasound Med Biol 36: 1998-2003.
- Maeda K (2013) Preeclmpsia is caused by continuous sympathetic center excitation due to an enlarged pregnant uterus. J Perinat Med 42: 233-237.
- Wang S, Cheng H, Dai G, Wang X, Hua R, et al. (2013) Umbilical cord mesenchymal stem cell transplantation significntly improves neurological function in patients with sequelae of traumatic brain injury. Brain Res 1532: 76-84.
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 14490
- [From(publication date):
June-2016 - Aug 30, 2025] - Breakdown by view type
- HTML page views : 13395
- PDF downloads : 1095