The Genetic Polymorphisms of TERT-CLPTM1L and Lung Cancer Susceptibility: A Meta-Analysis
Received: 17-Jul-2015 / Accepted Date: 24-Dec-2015 / Published Date: 30-Dec-2015 DOI: 10.4172/2161-1165.1000215
Abstract
Objective: To assessed the loci of the association between the single three polymorphisms (SNPs) (rs2736100, rs402710 and rs401681) and lung cancer risk. Methods: We performed a comprehensive meta-analysis of 19 publications with a total of 32,319 lung cancer cases and 5, 0529 controls via Medline, PubMed, Elsevier and Web of Science. We assessed the loci of the association between the single three polymorphisms (SNPs) (rs2736100, rs402710 and rs401681) and lung cancer risk using dominant model and performed subgroup analysis by cancer pathology and ethnicity. Pooled odds ratio (OR) with 95% confidence interval (CI) was calculated using review manager 5.2 package and Stata12.0 package. Results: Overall, significant associations were observed for rs2736100 and rs401681 polymorphisms of TERT-CLPTM1L with increased lung cancer risk (for rs2736100, OR=1.32, 95% CI=1.27-1.36, P<0.00001; for rs401681, OR=1.19, 95%CI=1.12-1.26, P<0.00001). When stratified by ethnicity, rs2736100 polymorphism was associated with increased risk of Asians (OR=1.42, 95% CI=1.36-1.49, P=0.06); When stratified by pathology, there was no significant associations were found between rs2736100, rs401681 and lung cancer and lung adenocarcinoma. Conclusions: TETT-CLPTM1L is a candidate susceptibility gene for lung cancer risk in Caucasian and Asian populations. Increased cancer risk was found in rs2736100CC and rs401681CC and homozygous variant genetic model.
Keywords: Gene; Polymorphism; TERT-CLPTM1L; Lung cancer; Meta-analysis
164220Introduction
Lung cancer is the most common cancer type and the leading cause of cancer deaths in world-wide [1].
Although most etiological factors of lung cancer are environmental, including smoking, asbestos and air pollution [1], only a small percentage of individuals exposed to hazardous environmental factors develop lung cancer, which suggest that the genetic factors, may predispose individuals to develop lung cancer.
In recent years, more than 40 susceptible genetic regions for lung cancer have been identified by GWAS analysis, including 5p15, 6p21 and 15q25 in Caucasian and Asian populations. The 5p15 contains two independent association signals, which map to TERT and CLPTM1L.
Several variants in the TERT-CLPTM1L have been identified by GWAS to be associated with melanoma [1], skin cancer [2], bladder cancer [3,4], pancreatic cancer [5] and breast cancer [6,7].
The telomerase gene TERT is a reverse transcriptase that controls telomere length and is critical for telomere replication and stabilization [8].
CLPTM1L is named based on its homology with cleft lip and palate transmembrane protein 1, which was identified as disrupted in a family with cleft lip and palate. CLPTM1L plays a role in recognition and repair of DNA damage.
Several studies showed that the rs2736100, rs402710 and rs401681 polymorphism of TERT-CLPTM1L was strongly associated with several cancer susceptibility [9], including lung cancer [10] and suggested that TERT-CLPTM1L polymorphisms may play an important role in cancer pathogenesis.
However, a single study might have been underpowered to detect the overall effects. A quantitative synthesis of the accumulated data from different studies is helpful to provide evidence on the association of TERT-CLPTM1L polymorphism with lung cancer risk.
Thus, in this study, we conducted a pooled analysis of data from 19 studies of lung cancer on a total of 32319 cases and 50529controls of both from Caucasian and Asian ancestry.
Materials and Methods
Search strategy
A systematic literature search was performed using Medline, PubMed, Elsevier and Web of Science (updated to 10 January 2015). The search strategy was based on combinations of ‘‘TERT-CLPTM1L”, or “5p15.33,’’ “rs2736100” or “rs401681” or “rs402710” and ‘‘lung cancer’’, ‘‘lung diseases’’ or “lung carcinoma’’.
Finally, 53 references of the retrieved articles were scanned. Reviews, comments, and letters were also checked for additional studies.
Inclusion and exclusion criteria
Articles that meet the following criteria were included:
(a)case-control or nested case-control study; (b) assess the association between rs2736100, rs402710 or rs401681and lung cancer risk; (c) genotypes in controls being in Hardy-Weinberg equilibrium ; (d) sufficient data for genotype frequency of rs2736100, rs402710 or rs401681in cases and controls; (e) published studies.
Exclusion criteria were:
(a)investigations in subjects with family cancer risks or cancer-prone disposition; (b) no relation to the three SNPs; (c) no genotype of the three SNPs; (d) unpublished studies.
Statistical analysis
With 95% CIs was used to assess the strength of association between the TERT-CLPTM1Lpolymorphism and Lung cancer risk Heterogeneity between studies was evaluated by the Q test.
A fixed effect model was used to pool the data when the p value of the Q test was <0.05; otherwise, random effects model was selected.
Hardy-Weinberg equilibrium was assessed using the x2test. Publication bias was assessed using Begg’s test.
All statistical analyses were performed in Review Manager 5.2 software (v.5.2; Oxford, England) and Stata12 Analysis System software (v.12.0; SAS Institute, Cary, NC, USA).
Characteristics of all included studies
As shown in Figure 1, 53 eligible studies were selected after a comprehensive literature research up to10 January 2015.
However, after reading the abstracts, 16 studies were excluded; 7 studies was a meta-analysis, 5 studies did not investigate the relationship between the three SNPs and lung cancer, 3 studies didn’t refer to the three SNPs, 18 studies were excluded; 16 studies did not offer sufficient genotype, and two studies did not accord with Hardy-Weinberg equilibrium.
Final data pool is consisted of publications with 19 data sets consisting of 32,319 cases and 50,529 controls were relevant to the association between TERT-CLPTM1L rs2736100, rs402710, and rs401681 and lung cancer.
Quantitative analysis results
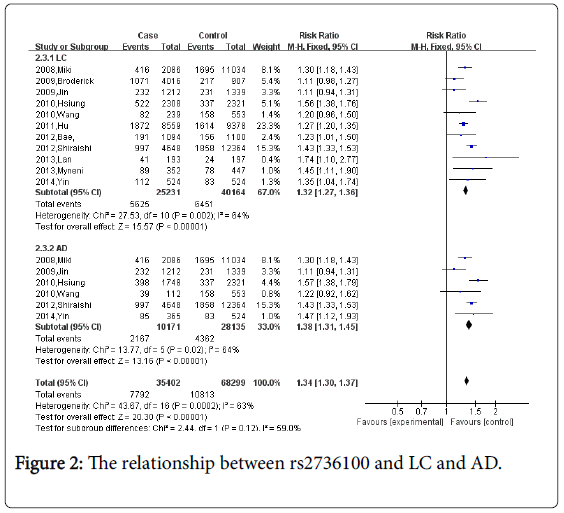
For rs2736100, eleven studies which including 25,231cases and 40,164 controls were identified. Overall, the meta-analysis revealed that rs2736100 was associated with an increased risk of lung cancer (CC vs. AA+AC: OR=1.32, 95% CI=1.27-1.36, P<0.00001).
Figure 2 When stratified by pathology, 6 studies concerned rs2736100 and lung adenocarcinoma (AD), and a significant association was observed (CC vs. AA+AC: OR=1.38, 95% CI=1.31-1.45, P<0.00001).
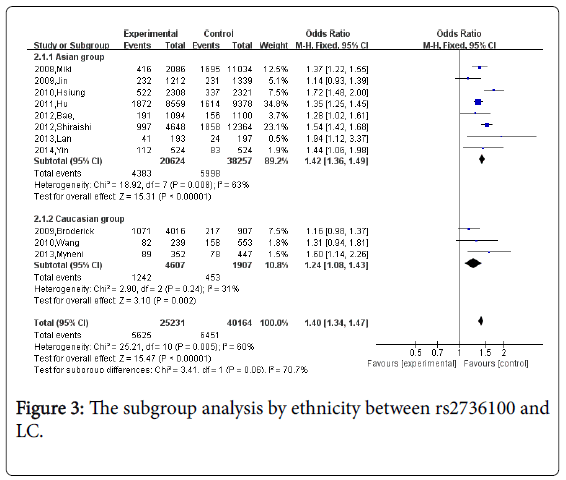
Figure 3 We also conducted a subgroup analysis by ethnicity, significant association between rs2736100 and lung cancer risk was found in both Asian group and Caucasian group (OR=1.42, 95%CI=1.36-1.49; OR=1.24; 95%CI=1.08-1.43, separately), so the SNP is more singnicant in Asian. (Psubgroup=0.06).
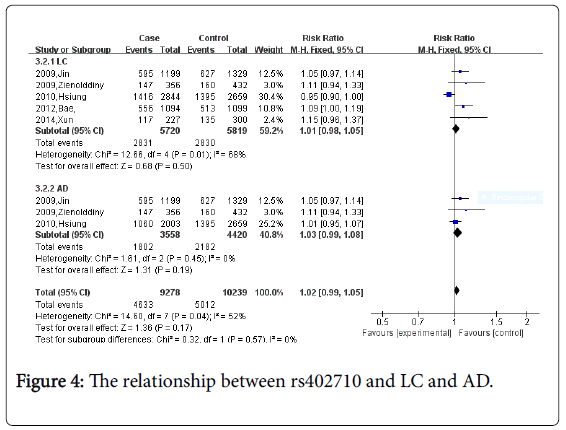
For rs402710, five studies which including 5,720 cases and 5,819 controls were identified.
The meta-analysis demonstrated no significant association between rs402710 and lung cancer and lung adenocarcinoma risk (Figure 4, CC vs. AA+AC: OR=1.01, 95% CI=0.98-1.05 P=0.50; OR=1.03; 95%CI=0.99-1.08, P=0.57 separately).
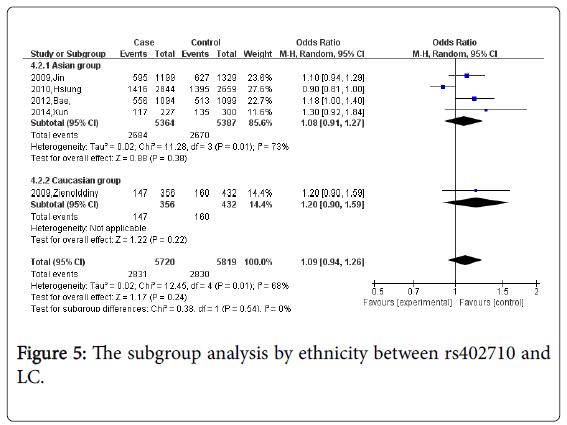
Figure 5 When stratified by pathology and ethnicity, there were also no associations observed (P=0.54).
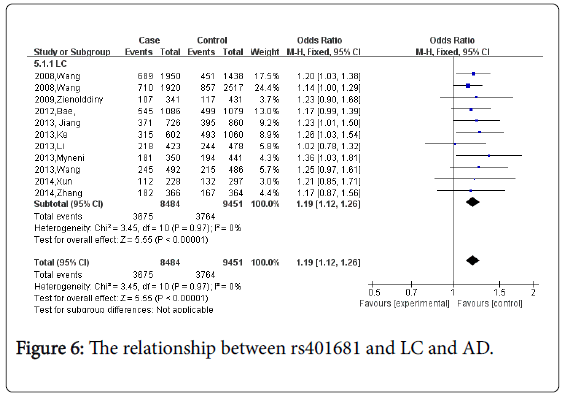
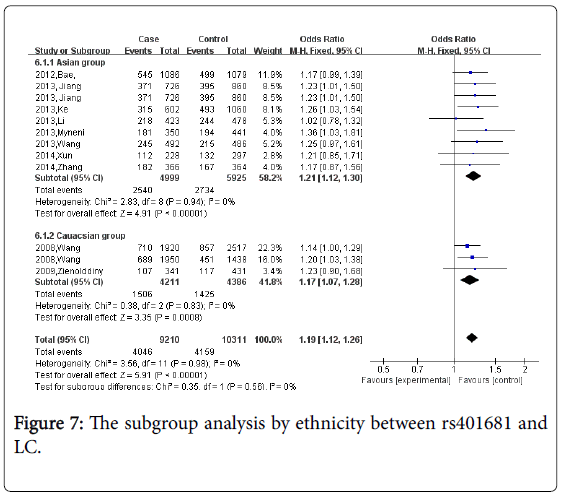
For rs401681, ten studies which including 8,484 cases and 9,451 controls were identified. Overall, Figure 6 the meta-analyses demonstrated a significant association between rs401681 and lung cancer risk (CC vs. AA+AC: OR=1.19, 95% CI=1.12-1.26 P<0.0001). Figure 7 When stratified by ethnicity, the significant risk was observed both in Asians and Caucasians (OR=1.24, 95%CI=1.11-1.38; in Asian group; OR=1.17, 95%CI=1.01-1.28, in Caucasian group). No statistical difference in the subgroup analysis of ethnicity. (Psubgroup=0.56).
Heterogeneity and sensitivity analysis
Next, we adopted the dominant model in heterogeneity comparison analysis. There was significant heterogeneity in the studies of rs2736100 or rs402710 (rs2736100: Pheterogrmeity=0.002, I2=64%; rs402710: Pheterogrmeity=0.01, I2=68%, separately), however, there was no significant heterogeneity for rs401681 (Pheterogrmeity=0.97, I2=0%). We proceeded with subgroup analysis for rs2736100 and rs402710 by ethnicity and pathology. For rs2736100, there was significant heterogeneity in Asian and lung cancer in subgroup analysis (Pheterogrmeity=0.008, I2=63% in Asian; Pheterogrmeity=0.01, I2=68% in lung cancer), while there was no heterogeneity in Caucasian and Lung adenocarcinoma (Pheterogrmeity=0.24, I2=31% in Caucasian; Pheterogrmeity=0.45, I2=0% in Lung adenocarcinoma).
In addition, we conducted a sensitivity analysis. For rs2736100 site, after removing two studies (2009Hsiung [11]), the results were stable and homogeneous (Pheterogrmeity=0.11, I2=38%). For rs402710, there was no significant heterogeneity in lung adenocarcinoma in subgroup analysis (Pheterogrmeity=0.45, I2=0%). Finally elimination of any individual study made no qualitative difference on pooled OR values, so the results of this meta-analysis were relatively stable and reliable.
Publication bias
Begg’s funnel plot and Egger’s test were performed to assess the publication bias of the literatures. The shape of the funnel plots showed that no significant evidence of obvious asymmetry in the overall meta-analysis (for rs2736100, P=0.251; for rs402710, P=0.703; for rs401681, P=0.967,, separately). Also, the results showed no obvious evidence of publication bias in the subgroup analysis.
Discussion
By pooling summary results from 19 studies, we provided additional evidence for inherited genetic predisposition to lung cancer that the TERT rs2736100, CLPTM1L rs401681 SNPs were significantly associated with lung cancer risk.
The rs2736100A/C SNP is located in intron 1 of TERT, which has been reported to be associated with lung cancer risk in both Caucasians and Asians. McKay et al. did not report heterogeneity with respect to histology, gender or smoking status [12]. However recent reports indicate that the association between rs2736100 and lung cancer risk is more evident for adenocarcinoma, females, nondrinkers, nonsmokers and cooking oil fume exposure [10,13-16]. Myneni et al. reported that TERT was more prominently associated with lung cancer in participants who were older than 60 years, exposed to low indoor air pollution and adenocarcinoma and squamous cell carcinoma [17]. This suggests that TERT may play an important role in the pathogenesis of AD and that telomerase activity may interact with smoking to infiuence the development of lung cancer. It is possible that rs2736100 is in high linkage disequilibrium (LD) with other potential functional or causal SNPs, contributing to the development of lung cancer. To date, the functional significance of the SNP rs2736100 remained unclear. In our meta-analysis, rs2736100A/C individuals from Asian and Caucasian subgroups displayed increased lung cancer risk, especially for AD.
CLPTM1L contains two obvious SNPs (rs402710 and rs401681). The SNP rs402710 A/C is located in intron 4 of the CLPTM1L gene, was found to be associated with higher DNA adduct formation in tumor adjacent lung tissue and may enhance the formation and persistence of DNA adducts McKay et al. found that rs402710 showed relatively little LD with rs2736100 (r2=0.026) [12]. In contrast, rs401681 was in strong LD with rs402710 [17]. So far, only Xun et al, they found rs402710 may decreased risk of lung cancer [18]. Zienolddiny et al. reported that the genotype of rs402710 did not show statistical difference in terms of gender [19]. The SNP rs401681 is localized in intron 13 of CLPTM1L. The relationship between rs401681 and lung cancer is controversial. For instance, Wang H. et al. reported that rs401681 was associated with non-small cell lung cancer but not with small cell lung cancer in the Chinese mainland population [20]. Two Chinese studuies found that the SNP affect the susceptibility of lung cancer, especially adenocarcinoma in women nonsmokers [21,22]. While some study acknowleged the susceptibility of smoking and Alcohol Drinking people [18,23]. Myneni et al. reported rs401681 has stronger association in participants exposed to low indoor air pollution and squamous cell carcinoma [17]. However, Bae et al. found that the two SNPs significantly affected the risk of lung cancer for only adenocarcinoma [24]. In our meta-analysisrs402710 was not associated with lung cancer risk, however, rs401681 was associated with lung cancer risk and when stratified by ethnicity, significant associations was observed both in Asians group and Caucasians group.
There are some limitations that need to be addressed. Firstly, although no publication bias was shown in the funnel plots and Egger’s tests, selection bias may still exist as non-English literatures were excluded. Secondly, due to insufficient genotype frequencies, we were unable to calculate the subgroup analyses of gender, smoking state, age and histology. Thirdly, there was a serious imbalance in ethnic disparity as 11 studies referred to Asian,especially to Chinese. Fourthly, ORs without adjustment were pooled together; although there was no substantial change between the two kinds of ORs in this synthesis, it may be a considerable source of heterogeneity.
In summary, our data suggested that TETT-CLPTM1L is a candidate susceptibility gene for lung cancer risk in Caucasian and Asian populations. Increased cancer risk was found in rs2736100CCand rs401681CC and homozygous variant genetic model. However as rs2736100and rs401681 are located in the introns of the TERT and CLPTM1L genes, the association between these SNPs and lung cancer may be due to LD with other functional variants. Further functional studies and well-designed studies are necessary to clarify the mechanisms by which TETT-CLPTM1L influences susceptibility to lung cancer.
Conflicts of Interest
The authors declare that they have no conflict of interest.
References
- Stefanaki I, Panagiotou OA, Kodela E, Gogas H, Kypreou KP, et al. (2013) Replication and predictive value of SNPs associated with melanoma and pigmentation traits in a Southern European case-control study. PLoS One 8: e55712.
- Nan H, Qureshi AA, Prescott J, De Vivo I, Han J (2011) Genetic variants in telomere-maintaining genes and skin cancer risk.Hum Genet 129: 247-253.
- Golka K, Selinski S, Lehmann ML, Blaszkewicz M, Marchan R, et al. (2011) Genetic variants in urinary bladder cancer: collective power of the "wimp SNPs".Arch Toxicol 85: 539-554.
- Gago-Dominguez M, Jiang X, Conti DV, Castelao JE, Stern MC, et al. (2011) Genetic variations on chromosomes 5p15 and 15q25 and bladder cancer risk: findings from the Los Angeles-Shanghai bladder case-control study. Carcinogenesis32: 197-202.
- Petersen GM, Amundadottir L, Fuchs CS, Kraft P, Stolzenberg-Solomon RZ, et al. (2010) A genome-wide association study identifies pancreatic cancer susceptibility loci on chromosomes 13q22.1, 1q32.1 and 5p15.33. Nat Genet42: 224-228.
- Beesley J, Pickett HA, Johnatty SE, Dunning AM, Chen X, et al. (2011) Functional polymorphisms in the TERT promoter are associated with risk of serous epithelial ovarian and breast cancers.PLoS One 6: e24987.
- Niture SK, Gnatt A, Jaiswal AK (2013) Oncogene PKCε controls INrf2-Nrf2 interaction in normal and cancer cells through phosphorylation of INrf2.J Cell Sci 126: 5657-5669.
- Lan Q, Cawthon R, Gao Y, Hu W, Hosgood HD, et al. (2013) Longer telomere length in peripheral white blood cells is associated with risk of lung cancer and the rs2736100 (CLPTM1L-TERT) polymorphism in a prospective cohort study among women in China. PLoS One 8: e59230.
- Mocellin S, Verdi D, Pooley KA, Landi MT, Egan KM, et al. (2012) Telomerase reverse transcriptase locus polymorphisms and cancer risk: a field synopsis and meta-analysis.J Natl Cancer Inst 104: 840-854.
- Li C, Yin Z, Wu W, Li X, Zhou B (2013) Genetic variants in TERT-CLPTM1L genetic region associated with several types of cancer: a meta-analysis.Gene 526: 390-399.
- Hsiung CA, Lan Q, Hong YC, Chen CJ, Hosgood HD, et al. (2010) The 5p15.33 locus is associated with risk of lung adenocarcinoma in never-smoking females in Asia.PLoS Genet 6.
- McKay JD, Hung RJ, Gaborieau V, Boffetta P, Chabrier A, et al. (2008) Lung cancer susceptibility locus at 5p15.33.Nat Genet 40: 1404-1406.
- Jin G, Xu L, Shu Y, Tian T, Liang J, et al. (2009) Common genetic variants on 5p15.33 contribute to risk of lung adenocarcinoma in a Chinese population.Carcinogenesis 30: 987-990.
- Chen XF, Cai S, Chen QG, Ni ZH, Tang JH, et al. (2012) Multiple variants of TERT and CLPTM1L constitute risk factors for lung adenocarcinoma.Genet Mol Res 11: 370-378.
- Lan Q, Hsiung CA, Matsuo K, Hong YC, Seow A, et al. (2012) Genome-wide association analysis identifies new lung cancer susceptibility loci in never-smoking women in Asia. Nat Genet 44: 1330-1335.
- Yin Z, Cui Z, Ren Y, Zhang H, Yan Y, et al. (2014) Genetic polymorphisms of TERT and CLPTM1L, cooking oil fume exposure, and risk of lung cancer: a case-control study in a Chinese non-smoking female population.Med Oncol 31: 114.
- Myneni AA, Chang SC, Niu R, Liu L, Ochs-Balcom HM, et al. (2013) Genetic polymorphisms of TERT and CLPTM1L and risk of lung cancer--a case-control study in a Chinese population.Lung Cancer 80: 131-137.
- Xun X, Wang H, Yang H, Wang H, Wang B, et al. (2014) CLPTM1L genetic polymorphisms and interaction with smoking and alcohol drinking in lung cancer risk: a case-control study in the Han population from northwest China.Medicine (Baltimore) 93: e289.
- Zienolddiny S, Skaug V, Landvik NE, Ryberg D, Phillips DH, et al. (2009) The TERT-CLPTM1L lung cancer susceptibility variant associates with higher DNA adduct formation in the lung. Carcinogenesis30: 1368-1371.
- Wang H, Zhao Y, Ma J, Zhang G, Mu Y, et al. (2013) The genetic variant rs401681C/T is associated with the risk of non-small cell lung cancer in a Chinese mainland population.Genet Mol Res 12: 67-73.
- Li C1, Yin Z, Wu W, Li X, Ren Y, et al. (2013) Genetic variations in TERT-CLPTM1L genes and risk of lung cancer in Chinese women nonsmokers.PLoS One 8: e64988.
- Jiang M, Wu H, Qin C (2013) Genetic variant rs401681 at 5p15.33 modifies susceptibility to lung cancer but not esophageal squamous cell carcinoma.PLoS One 8: e84277.
- Zhang Y, Zhao M, Shen L, Ren Y, Su L, et al. (2014) Genetic polymorphisms of TERT and CLPTM1L and risk of lung cancer: a case-control study in northeast Chinese male population.Med Oncol 31: 18.
- Bae EY, Lee SY, Kang BK, Lee EJ, Choi YY, et al. (2012) Replication of results of genome-wide association studies on lung cancer susceptibility loci in a Korean population. Respirology17: 699-706.
Citation: Cheng S, Wang F, Song B, Liu J (2015) The Genetic Polymorphisms of TERT-CLPTM1L and Lung Cancer Susceptibility: A Meta- Analysis. Epidemiology (sunnyvale) 5:215. DOI: 10.4172/2161-1165.1000215
Copyright: © 2015 Cheng S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 11058
- [From(publication date): 12-2015 - Aug 20, 2025]
- Breakdown by view type
- HTML page views: 10188
- PDF downloads: 870