Research Article Open Access
The Use of Biological ‘Infill’ Materials for the Treatment of Recto/Ano-vaginal Fistulae-A Systematic Review
Nuha A Yassin1*, Alan Askari1, John T Jenkins1, Ahmad Uraiqat1, Omar D Faiz1, Ailsa L Hart2 and Robin K S Phillips11Department of Colorectal Surgery, St Mark’s Hospital and Academic Institute, Watford Road, Harrow, HA1 3UJ, United Kingdom
2The IBD Unit, St Mark’s Hospital and Academic Institute, Watford Road, Harrow, HA1 3UJ, United Kingdom
- *Corresponding Author:
- Nuha A Yassin
St Mark’s Hospital and Academic Institute
Watford Road, Harrow
HA1 3UJ, United Kingdom
Tel: 0208 2354251
E-mail: nayassin@gmail.com
Received date: September 18, 2015; Accepted date: August 19, 2016; Published date: August 25, 2016
Citation: Yassin NA, Askari A, Jenkins JT, Uraiqat A, Faiz OD, et al. (2016) The Use of Biological ‘Infill’ Materials for the Treatment of Recto/Anovaginal Fistulae-A Systematic Review. J Gastrointest Dig Syst 6:465. doi: 10.4172/2161-069X.1000465
Copyright: © 2016 Yassin NA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License; which permits unrestricted use; distribution; and reproduction in any medium; provided the original author and source are credited.
Visit for more related articles at Journal of Gastrointestinal & Digestive System
Abstract
Aim: Recto-vaginal fistulae are distressing. They are most commonly caused by obstetric injury, a failed pelvic anastomosis, or Crohn’s disease. Biological infill materials have been used in their management. We assessed the efficacy of biological infill materials when recto-vaginal fistulae. Methods: We applied the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. Results: Twenty-five articles yielded a pooled total of 105 patients; 27% had Crohn’s disease. Fifty-six percent of patients had treatment with glue, 31% with plug, 6% with permacol paste and 7% with stem-cells. For Crohn’s, 50% were treated with glue, 39% with a fistula plug and 11% with stem cells. The overall healing rate for all infill materials was 41%, with the highest reported rate amongst the stem cell group (71%) and the lowest (32%) for glue. For Crohn's, the overall healing rate was 36%, glue was the lowest (14%), followed by the anal fistula plug (55%). Stem cells had a healing rate of 67%. Conclusions: Overall, the use of infill materials to treat Crohn’s and non-Crohn’s related recto-vaginal fistulae is associated with healing in approximately 41% of cases. Crohn’s related recto-vaginal fistulae represent a more challenging group of patients to treat.
Keywords
Rectovaginal fistula; Anovaginal fistula; Proctology; Glue; Plug; Infill material
Introduction
Fistulae to the vagina distress and stigmatise women and their management remains a challenge [1]. Some may arise from intraperitoneal organs, such as the appendix, terminal ileum or sigmoid colon, where they usually communicate with the vaginal fornix. Treatment involves surgical resection of the origin of fistulation. Rectovaginal fistulae (RVF) are more complicated to manage. They will usually be found in the posterior midline of the vagina. Those that are obstetric-related may have a concomitant anal sphincter injury. Several factors are implicated in the success of any surgical repair including anatomical aspects and aetiology. The rectovaginal septum may be thin and poorly vascularised. Many cases of local repair result in failure. Other confounders are medical comorbidities, inflammatory bowel disease, persistent infection and the presence of scar tissue [2,3].
The most common cause is obstetric injury [4], followed by Crohn’s disease [5]. Less common causes include malignancies, for example of the cervix, vagina, uterus or rectum. In addition, recto-vaginal fistulae can result after radiotherapy or as postoperative complications, typically after a low anterior resection with stapled anastomosis, hysterectomy or rectocele repair. The diagnosis is mainly clinical by direct observation supported by examination under anaesthetic. Radiology is frequently disappointing, but in some case anal ultrasound, MRI or contrast radiology may be useful [6].
Surgical options include extended anal sphincter repair (applicable when there is an associated anal sphincter injury), transanal advancement flap (made easier when perineal descent/internal intussusception are present), transvaginal or transperienal approaches (frequently with an adjunct of either a Martius flap or a biological material) or an abdomino-anal pull-through operation [7]. Other techniques and adjuncts include omentum and local muscle flaps, suchs as gracilis.
Reported success rates of clinical healing vary from 41-100% for non-IBD fistulae [8-17] and 40-67% with Crohn’s [18-27]
Novel surgical techniques include ligation of the intersphinteric fistula tract (LIFT) [28] and the use of biological infill material [29]. The added benefit of these infill materials is unclear. We systematically reviewed the role of biological infill materials (not including biological meshes) when treating rectovaginal fistulae of all aetiologies.
Methods
This review was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [30].
Search strategy
Two independent reviewers searched PubMed (January 1966 to August 2013), the Cochrane Database and EMBASE (January 1980- August 2013). The keywords used were: rectovaginal, fistula, anovaginal, glue, paste, plug, bioprosthetic and stem cell. Searches were limited to English language articles. The abstracts of all potentially relevant studies were consulted to identify studies suitable for inclusion. The full texts of all eligible articles were retrieved. Additional strategies included searching the cited references of selected articles.
Selection criteria
All studies investigating biological infill material as a treatment for rectovaginal fistulae were included. We did not include studies assessing biological mesh as an adjunct to surgical repair of rectovaginal fistula.
Data collection and analysis
We determined study design, patient demographics, fistula aetiology, previous seton drainage, previous anal surgery, perioperative management, management of the internal fistula opening, fistula healing, faecal incontinence, follow-up duration and all complications. Data were independently extracted by two reviewers (NY and AA). Quality assessment of the included articles was performed using criteria from the Cochrane Reviewers' Handbook and the Newcastle-Ottawa scale [31] (for non-randomised studies). Disagreement was resolved according to the predefined strategy using consensus and arbitration.
As a consequence of the methodological nature of the studies, quantitative data analyses were only performed for eligible studies with adequate data. Due to study heterogeneity, small patient numbers and dearth of comparative studies, meta-analysis was not suitable; therefore data were pooled without formal statistical analysis.
Results
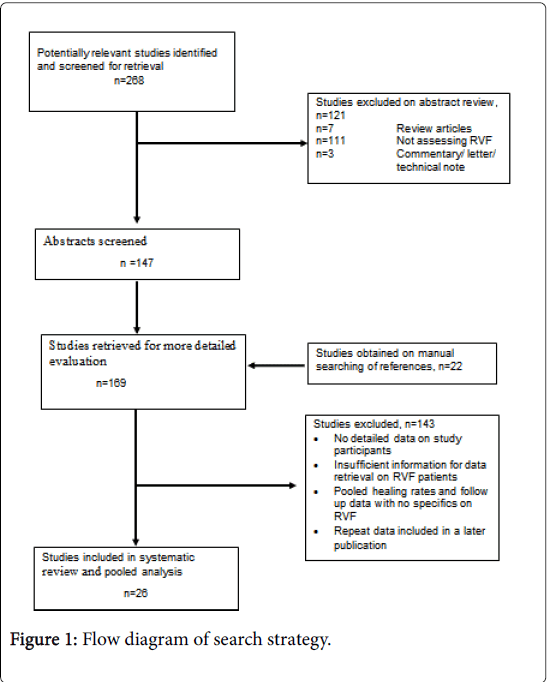
Two-hundred and sixty-eight studies were identified, of which 121 were excluded: reviews (n=7), letters or commentaries (n=3), or those that did not specifically assess rectovaginal fistulae (n=111). 147 studies were assessable for abstract screening. Manual searching of references provided a further 22 studies, giving 169 studies for full evaluation. Of these, 143 were subsequently excluded as they did not satisfy the inclusion criteria and failed to provide adequate detail for data retrieval. Twenty-six studies were included in the systematic review.
Study characteristics
Of the 26 eligible studies, 14 were prospective studies [32-45] and 12 were retrospective [46-57]. A total of 18 studies were either case reports or case series [32-34,40,44-57], four were non-randomised clinical trials [35-38), one was a multi-centre case series [42] and one was a single multi-centre randomised controlled trial [41] (Table 1 and Figure 1).
| Author | Year | Country | Type of Study | Prospective or Retrospective | Infill Material | F/U in months [mean] | F/U in months [range] | Follow up in months [median] | F/U in months [range] |
|---|---|---|---|---|---|---|---|---|---|
| Abel [32] | 1993 | USA | case series | Prospective | Glue | 7.2 | - | - | - |
| Venkatesh [33] | 1999 | USA | case series | Prospective | Glue | - | Sep-57 | - | - |
| Aitola [34] | 1999 | Finland | case series | Prospective | Glue | 6 | - | - | - |
| Cintron [35, 36] | 2000 | USA | clinical trial - non randomised | Prospective | Glue | 12 | - | - | - |
| Park [36] | 2000 | USA | clinical trial - non randomised | Prospective | Glue | 6 | - | - | - |
| Halverson [46] | 2001 | USA | case series | Retrospective | Glue | - | - | 4 | Jan-25 |
| Buchanan [37] | 2003 | UK | clinical trial - non randomised | Prospective | Glue | - | - | 14 | - |
| Loungnarath [47] | 2004 | USA | case series | Retrospective | Glue | - | - | 23 | Jun-36 |
| Garcia-Olmo [38] | 2005 | Spain | clinical trial - non randomised | Prospective | Stem Cells | 22 | Dec-30 | - | - |
| Singer [39] | 2005 | USA | RCT | Prospective | Glue | - | 23-28 | - | - |
| Ellis [48] | 2008 | USA | case series | Retrospective | Plug & Mesh | 12 | Jun-22 | - | - |
| Alexander [40] | 2008 | Netherlands | case series | Prospective | Glue | - | - | - | - |
| Garcia-Olmo[41] | 2009 | Spain | Multi-centre RCT | Prospective | Glue & Stem Cells | 12 | - | - | - |
| Thekkinkattil [42] | 2009 | UK & Netherlands | Multi-centre case series | Prospective | Plug | - | Mar-18 | - | - |
| Safar [49] | 2009 | USA | case series | Retrospective | Plug | 4 | - | - | - |
| El-Gazzaz [50] | 2010 | USA | case series | Retrospective | Glue & Plug | - | - | 44.6 | 13.1-79.1 |
| El-Gazzaz [51] | 2010 | USA | case series | Retrospective | Glue | 45.8 | - | - | - |
| Grimaud [43] | 2010 | France | Multi-centre RCT | Prospective | Glue | 2 | 01-Mar | - | - |
| De Parades [52] | 2010 | France | case series | Retrospective | Glue | 11.7 | 0.2-33.5 | - | - |
| Gaertner [53] | 2011 | USA | case series | Retrospective | Glue & Plug | 38.6 | - | - | - |
| Gajsek [44] | 2011 | UK | case series | Prospective | Plug | - | - | 118 | 81.5-129 |
| Kleif [54] | 2011 | Denmark | case series | Retrospective | Plug | - | 02-Aug | - | - |
| Choi [55] | 2012 | USA | case series | Retrospective | Glue | 9.2 | - | - | - |
| Garcia [56] | 2012 | USA | case report | Retrospective | Glue | 6 | 6 | - | - |
| Sileri [45] | 2012 | Italy | case series | Prospective | Permacol Paste | - | Mar-36 | - | - |
| Binda [57] | 2013 | Italy | case report | Retrospective | Glue | 12 | - | - | - |
Table 1: Study characteristics.
Patient age ranged from 17-76 years with the mean follow up reported between 2 and 46 months. Most studies reported glue as the infill material under assessment either alone (n=16) or in combination with stem cells (n=1), or in comparison with a fistula plug (n=2). Four studies used plugs alone and one study compared fistula plug and mesh. Only one study reported the use of stem cells alone as the chosen infill material (Table 1).
Patient characteristics
In total, 105 patients underwent treatment for RVF using infill material, of which 28 patients (27%) had Crohn’s Disease. Overall, 59 (57%) patients had treatment with glue, 33 (31%) with a plug, six (6%) with permacol paste and seven (7%) with stem cells. Within the Crohn’s group, the majority were treated with glue (n=14; 50%), in 11 a fistula plug was used (n=11/28, 39%) and three had stem cells (n=3/28, 11%).
Peri-operative management
Antibiotic therapies: Significant differences existed in the selected studies regarding peri-operative management and surgical technique (Table 2).
| Author | n= | Prior Seton drainage (Yes/No) | Bowel Prep (Yes/No) | Antibiotics | Fistula Tract Management | Peri-Operative Management | Name of Material Used |
|---|---|---|---|---|---|---|---|
| Abel | 5 | - | Yes | Pre-Operative | Curettage | Post-Operative oral intake restriction | Thrombinar |
| Choi | 1 | No | - | - | - | - | Fibrin Sealant |
| El-Gazzaz | 2 | - | - | Pre-Operative | - | - | Glue |
| El-Gazzaz | 1 | - | - | Pre-Operative | - | - | Plug |
| El-Gazzaz | 7 | - | - | Pre-Operative | - | - | Glue |
| Ellis | 7 | 05-Jul | No | Pre-Operative | Nil | post operative laxatives, reduction in physical activity and sitz baths | Surgisis AFP - Cook |
| Gaertner | 8 | Yes | - | Peri-Operative | - | - | Glue |
| Gaertner | 4 | Yes | - | Peri-Operative | - | - | Plug |
| Gajsek | 9 | Yes | Yes | Pre-Operative & Post-Operative | Brush, Hydrogen Peroxide & Saline | post operative antibiotics, laxatives and restriction to physical activity and intercourse | Surgisis Biodesign Button Plug - Cook |
| Garcia | 1 | - | Yes | - | - | post operative low residue diet and no intercourse | BioGlue®, CryoLife, |
| Halverson | 3 | - | Yes | Yes | Curettage | - | Tisseel |
| Cintron | 3 | - | No | No | Curettage | nil | ViGuard-FS ® |
| Garcia-Olmo | 4 | - | - | - | Curettage | nil | Tissucol Duotm, Baxter |
| Garcia-Olmo | 4 | - | - | - | Curettage | - | ASC, Cellerix, Spain; company code, Cx401 |
| Garcia-Olmo | 3 | - | - | - | - | nil | ASC, Cellerix, Spain; company code, Cx401 |
| Buchanan | 1 | - | No | Pre-Operative | Curettage, Hydrogen Peroxide & Saline | nil | Tisseel™ |
| Loungnarath | 3 | - | Yes | Pre-Operative | Curettage | Liquid diet and no driving or sitting for 2 weeks | Tisseel® |
| Grimaud | 3 | - | Yes | - | Curettage | 2 weeks of sexual abstinence | Beriplast + Trasylol |
| Alexander | 1 | - | Yes | Pre-Operative | Curettage | 5 day IV Metronidozole, immobilisation and liquid diet | BioGlue ® |
| Venkatesh | 8 | - | Yes | Pre-Operative | Curettage | 48 hours liquids, 7 day antibiotics | Autologous fibrin |
| Thekkinkattil | 9 | - | Yes | Post-Operative | Hydrogen Peroxide & Saline | Antibiotics and laxatives for 1 week, restrict activity | Surgisis® AFP™ |
| Park | 3 | - | - | - | Curettage | - | ViGuard-F.S |
| Singer | 1 | - | Yes | No | Curettage | Not to have sitz baths, given laxatives | Tissucol® |
| Sileri | 6 | Yes | Yes | Yes | Curettage & Saline | - | Permacol (TM) Injection, Covidien plc. |
| Kleif | 1 | Yes | Yes | Pre-Operative | Brush, Hydrogen Peroxide & Antibiotics | Bed rest 24hrs, restrict activity and no intercourse 2 weeks | Surgisis AFP |
| Safar | 1 | - | Yes | Pre-Operative | Hydrogen Peroxide | Restrict activity, topical antibiotics for 10 days | Surgisis AFP |
| De Parades | 5 | Yes | Yes | Pre-Operative | Curettage & Saline | Immobilisation in theatre for 15mins, post-operative antibiotics, restrict activity and no intercourse | Tissucol |
| Binda | 1 | Yes | - | Post-Operative | Curettage & Hydrogen Peroxide | - | Gore BioA |
Table 2: Perioperative management.
Eleven studies used a single dose of Pre-operative antibiotics at induction of anaesthesia, one used antibiotics during the perioperative period, and two used antibiotics Post-operatively. One study used antibiotics both pre and Post-operatively.
A further two studies used antibiotics but did not specify at what time period, whilst two studies did not use antibiotics at all and six studies did not mention their use.
Tract management: Over half of the studies (n=15/26) used curettage of the fistula tract, either alone or in conjunction with either saline or hydrogen peroxide. Two studies used a brush in the tract in conjunction with hydrogen peroxide. In one of these studies antibiotics were also applied to the fistula tract.
Primary outcomes
Fistula healing was assessed clinically only in all but two studies. In these two studies, MRI was used to determine resolution of the tract in addition to clinical healing [36,56]. The overall fistula “healing” rate with all available infill materials was 41% (n=43/105) with the highest rate being amongst those receiving stem cells therapy (n=5/7, 71%), followed by permacol paste (67%, n=4/6) although it is important to note that the number of included patients is small (Tables 3 and 4). Fibrin glue was associated with the poorest healing rate of 32% (n=19/59). Within the Crohn’s population, the overall healing rate was lower (n=10/28, 36%). Glue had the lowest healing rates with 2/14 patients healed (14%), followed by the anal fistula plug (n=6/11, 55%). The use of stem cells yielded a healing rate of 67% (n=2/3), although it is important to note the small number of patients included in the Crohn’s group.
| Author | Year | Material | n= | Age Range | IO management | All patient's healed (n=) | All patients healed (%) | Crohn's (n=) | Crohn's Healed (n=)* | Crohn's Healed (%) |
|---|---|---|---|---|---|---|---|---|---|---|
| Abel | 1993 | Glue | 5 | 25-48 | - | 04-May | 80 | 1 | 0/1 | 0% |
| Venkatesh | 1999 | Glue | 8 | 22-69 | - | 06-Aug | 75 | 6 | 0/6 | 0% |
| Cintron | 2000 | Glue | 3 | 30-50 | - | 01-Mar | 33 | - | - | - |
| Park | 2000 | Glue | 3 | 28-53 | - | 01-Mar | 33 | - | - | - |
| Halverson | 2001 | Glue | 3 | 27.4-46.6 | 2 MAF, 1 suture | 01-Mar | 33 | - | - | - |
| Buchanan | 2003 | Glue | 1 | 44 | - | 0/1 | 0 | - | - | - |
| Loungnarath | 2004 | Glue | 3 | 20-76 | - | 01-Mar | 33 | - | - | - |
| Singer | 2005 | Glue | 1 | 41 | - | 0/1 | 0 | - | - | - |
| Garcia-Olmo | 2005 | Stem Cells | 3 | 35-40 | suture + MAF | 2 | 66 | 3 | 01-Mar | 33% |
| Alexander | 2008 | Glue | 1 | 46 | suture + MAF | 0/1 | 0 | - | - | - |
| Ellis | 2008 | Plug | 7 | 17-48 | suture | 06-Jul | 86 | 5 | 4 | 80% |
| Garcia-Olmo | 2009 | Glue | 4 | 33-51 | suture | 03-Apr | 75 | - | - | - |
| Garcia-Olmo | 2009 | Glue & Stem Cells | 4 | 41-53 | suture | 0/4 | 0 | - | - | - |
| Thekkinkattil | 2009 | Plug | 9 | 18-65 | suture | 02-Sep | 22 | - | - | - |
| Safar | 2009 | Plug | 1 | 46 | suture | 0/1 | 0 | 1 | 0 | 0% |
| El-Gazzaz | 2010 | Glue | 2 | 40-44 | - | 01-Feb | 50 | 2 | 1 | 50% |
| El-Gazzaz | 2010 | Glue | 1 | 40-44 | - | 0/1 | 0 | 1 | 0 | 0% |
| El-Gazzaz | 2010 | Glue | 7 | 32.8-52.8 | suture | 03-Jul | 43 | - | - | - |
| Grimaud | 2010 | Glue | 3 | 27-41 | - | 0/3 | 0 | 3 | 0 | 0% |
| De Parades | 2010 | Plug | 5 | 22.8-69.1 | - | 0 | 0 | - | - | - |
| Kleif | 2011 | Permacol Paste | 1 | 44 | suture + MAF | 0/1 | 0 | - | - | - |
| Gaertner | 2011 | Plug | 4 | 39 | - | 02-Apr | 0 | 4 | 02-Apr | 50% |
| Gaertner | 2011 | Glue | 8 | 39 | - | 0/8 | 50 | 8 | 0 | - |
| Gajsek | 2011 | Glue | 9 | 32-45 | suture | 04-Sep | 44 | 9 | 4 | 44% |
| Choi | 2012 | Glue | 1 | 33 | - | 0/1 | 0 | - | - | - |
| Garcia | 2012 | Glue | 1 | 45 | - | 01-Jan | 100 | - | - | - |
| Sileri | 2012 | Plug | 6 | 22-75 | EAF | 04-Jun | 67 | - | - | - |
| Binda | 2013 | Glue | 1 | 46 | suture | 01-Jan | 100 | - | - | - |
Table 3: Patient demographics and outcomes.IO=Internal opening (of fistula tract), MAF=Mucosal advancement flap, EAF= Endorectal advancement flap, -Blank spaces denote information unavailable from the papers, *Data on concomitant medical therapy is not available for all studies.
| Pooled healing rates - all patients | |||
|---|---|---|---|
| Material | Total (n=) | Healed (n=) | Healed (%) |
| Glue Healing | 59 | 19 | 32% |
| Plug Healing | 33 | 15 | 46% |
| Permacol Healing | 6 | 4 | 67% |
| Stem Cell Healing | 7 | 5 | 71% |
| TOTAL | 105 | 43 | 41% |
| Crohn's subgroup healing rates | 5 | ||
| Material | Total (n=) | Healed (n=) | % Healed |
| Glue | 14 | 2 | 14% |
| Plug | 11 | 6 | 54% |
| Stem Cells | 3 | 2 | 67% |
| Total | 28 | 10 | 36% |
Table 4: Pooled analysis of healing rates, overall and in the Crohn’s subgroup.
Discussion
Many studies have reported the outcomes using infill materials in the management of anal fistulae but few have considered rectovaginal fistulae. The difficulty in reproducing the early high anal fistula healing rates using fibrin glue and the anal fistula plug has been reiterated in reports assessing their role in rectovaginal fistula. To date, the overall healing rate when all infill materials are considered has been around 41%. In the Crohn’s patients with recto-vaginal fistulae, the overall healing rate was 36%.
The reasons for treatment failure are multifactorial. Fistula aetiology clearly plays an important role. Crohn’s fistulae are more refractory to therapies. El Gazaz et al identified smoking and steroid use to be associated with lower healing rates, whereas immunomodulator use assisted healing [50]. The same group published in a separate study on obstetric and cryptoglandular rectovaginal fistulae that a higher body mass index (BMI), in addition to the number of previous attempts at repair, reduced fistula healing rates. Patient age, smoking status and other comorbidities exerted no obvious effect upon fistula healing [51].
Some of the limitations of this systematic review are the small numbers included in some of the studies, which make positive healing results seem significant. Researchers have to bear in mind that the results of pilot and feasibility studies although encouraging, should be taken with the limitations of small sample numbers in mind. Healing was seen in 67% and 71% of the permacol and stem cell groups respectively for all patients [41,45,38]. In the Crohn’s cohort, 67% of patients healed using stem cells. However, as highlighted above, the sample numbers were rather small for these studies and therefore the results should be interpreted with that limiting factor in mind.
The existing literature has significant limitations, including variability in follow up duration, which has been as short as two months, and the assumption that clinical healing alone reflects true healing without verification using suitable imaging modalities. Only two studies have reported the use of MRI as part of the assessment of healing of anal fistulae [36,56]. Although radiological healing rates of rectovaginal fistulae were not specifically addressed in those studies, it was reported for transphincteric fistulae that skin healing was identified as early as two weeks after treatment in 77% of patients, yet only 3% of those patients remained asymptomatic at 14 months or healed at 16 months [36].
In order to optimise fistula healing rates, surgeons may find it beneficial to consider each component of the fistula tract and the underlying pathology, and even employ a multimodal approach. The internal opening, the fistula tract itself, and any secondary extensions all need management. Some argue that removing granulation tissue, epithelial lining and surrounding fistula tract tissue might favour wound healing.
The operative management of the internal opening and fistula tract varied between the studies in this systematic review. A simple ‘figure of eight’ suture was placed before or after the instillation of the chosen infill materials. Some studies combined this with the use of a mucosal advancement flap and in one study an endorectal advancement flap was used. A number of studies were excluded if more extensive surgery was reported, as it is unclear whether the reported healing rates were due to the use of a specific infill material or due to the surgical management of the internal opening. Similarly, the fistula tract management varied widely, with some surgeons advocating curettage whereas others advised against it; some used a brush to deal with the granulation tissue and others washed with either saline or hydrogen peroxide. No consensus was found regarding bowel preparation, perioperative antibiotics, restriction in physical activity and sexual intercourse, or the use of sitz baths. The evidence is limited by the diversity of study designs and the low quality of the published literature in this area.
An appropriate and durable biological material has the potential to fill in the defect and act as a scaffold for the delivery of cells, be they fibroblasts or stem cells. Animal studies show that fibrin glue is an unsuitable material for use and as an alternative collagen paste may act as a better infill material as well as a vehicle for the delivery of cells and drugs [45,57]. Rectovaginal fistulae pose an additional challenge regarding the choice of infill material, as the tracts are short and more difficult to manage with a lower chance of success. Nevertheless, the use of the anal fistula plug was supported in the Anal Fistula Plug (AFP) consensus statement in 2007. A Consensus conference was held in Chicago to develop uniformity of opinion from 15 experienced surgeons with first-hand experience of using the AFP, five of whom had success rates as high as 85% in 50 or more cases each. The expert panel felt that transphincteric fistulae were the optimal indication for the use of the plug. For ano/recto-vaginal fistulae, it was felt that due to the short tract it was less likely that the plug would be successful. However, the plug was still felt to be a reasonable alternative to other operative options. It is also important to note that all 15 surgeons acknowledged that they had received full payment and honoraria by the AFP designers to produce such a consensus statement [58].
Management of fistulae with autologous stem cells represent a novel approach which might yield promising results in the future. Only one group have published in this area [57], with very small numbers of rectovaginal fistulae, but encouraging healing rates. Whether a combination of stem cells and an infill material as a vehicle alters outcome remains to be determined.
Another potentially novel scaffold for fistula repair and drug delivery material is the poly-porous microsphere. Such microspheres have yet to be used in the treatment of rectovaginal fistulae, and their retention at the fistula site may pose difficulties, but animal models have provided promising results. An animal model using poly-porous (D,L-lactide-co-glycolide) microspheres containing either antibacterial silver-releasing degradable phosphate glass or metronidazole has shown that they may be a potentially useful scaffold for guided tissue regeneration and for local drug delivery [59].
Fistula trials, such as the FIAT [60] randomised controlled trial, are currently recruiting to assess the safety and efficacy of the Surgisis® anal fistula plug in high transphincteric anal fistulae. The outcome measures include fistula healing rates, faecal incontinence, quality of life and cost effectiveness. Conceivably, a similar study that includes radiological verification of healing and quality of sexual function might be designed for the much rarer recto-vaginal fistulae so that we can better evaluate the role of infill materials in this challenging condition.
This systematic review summarises the evidence with regards the use of infill materials as management options when treating rectovaginal fistulae. Patients are more aware of the variety of surgical options available, and may choose simpler options over more invasive alternatives [61]. Patients with Crohn’s disease have limited options including medical treatment in the form of immunomodulators +/- anti-TNF therapy, but some may still require proctectomy [62]. Overall healing rates of 41% for rectovaginal fistula patients, or 36% for the patients with Crohn’s disease, may be an acceptable option when the alternative could be major surgery or a stoma.
References
- Bangser M (2006) Obstetric fistula and stigma. Lancet 367: 535-536.
- Fenner DE, Genberg B, Brahma P, Marek L, DeLancey JO (2003) Fecal and urinary incontinence after vaginal delivery with anal sphincter disruption in an obstetrics unit in the United States. Am J Obstet Gynecol. 189: 1543-1549.
- Signorello LB, Harlow BL, Chekos AK, Repke JT (2000) Midline episiotomy and anal incontinence: retrospective cohort study. BMJ 320: 86-90.
- Venkatesh KS, Ramanujam PS, Larson DM, Haywood MA (1989) Anorectal complications of vaginal delivery. Dis Colon Rectum 32: 1039-1041.
- Cohen JL, Stricker JW, Schoetz DJ Jr, Coller JA, Veidenheimer MC (1989) Rectovaginal fistula in Crohn's disease. Dis Colon Rectum 32: 825-828.
- Shobeiri SA, Quiroz L, Nihira M (2009) Rectovaginal fistulography: a technique for the identification of recurrent elusive fistulae. Int Urogynecol J Pelvic Floor Dysfunct 20:571-573.
- Tozer PJ, Balmforth D, Kayani B, Rahbour G, Hart AL, et al. (2013) Surgical management of rectovaginal fistula in a tertiary referral centre: many techniques are needed. Colorectal Dis 15: 871-877.
- Rahman MS, Al-Suleiman SA, El-Yahia AR, Rahman J (2003) Surgical treatment of rectovaginal fistula of obstetric origin: a review of 15 years' experience in a teaching hospital. Journal of obstetrics and gynaecology.J Obstet Gynaecol 23:607-610.
- Ayhan A, Tuncer ZS, Dogan L, Pekin S, Kisnisci HA (1995) Results of treatment in 182 consecutive patients with genital fistulae. Int J Gynaecol Obstet 48:43-47.
- Tancer ML, Lasser D, Rosenblum N (1990) Rectovaginal fistula or perineal and anal sphincter disruption, or both, after vaginal delivery. Surg Gynecol Obstet 171:43-46.
- Mazier WP, Senagore AJ, Schiesel EC (1995) Operative repair of anovaginal and rectovaginal fistulas. Dis Colon Rectum 38: 4-6.
- Watson SJ, Phillips RK (1995) Non-inflammatory rectovaginal fistula. Br J Surg 82: 1641-1643.
- Chew SS, Rieger NA (2004) Transperineal repair of obstetric-related anovaginal fistula. Aust N Z J Obstet Gynaecol 44: 68-71.
- Lowry AC, Thorson AG, Rothenberger DA, Goldberg SM (1988) Repair of simple rectovaginal fistulas. Influence of previous repairs. Dis Colon Rectum 31: 676-678.
- Tsang CB, Madoff RD, Wong WD, Rothenberger DA, Finne CO, et al. (1998) Anal sphincter integrity and function influences outcome in rectovaginal fistula repair. Dis Colon Rectum 41: 1141-1146.
- Khanduja KS, Yamashita HJ, Wise WE Jr, Aguilar PS, Hartmann RF (1994) Delayed repair of obstetric injuries of the anorectum and vagina. A stratified surgical approach. Dis Colon Rectum 37: 344-349.
- Zimmerman DD, Gosselink MP, Briel JW, Schouten WR (2002) The outcome of transanal advancement flap repair of rectovaginal fistulae is not improved by an additional labial fat flap transposition. Techniques in coloproctology 6:37-42.
- Pearl RK, Andrews JR, Orsay CP, Weisman RI, Prasad ML, et al. (1993) Role of the seton in the management of anorectal fistulas. Dis Colon Rectum 36: 573-577.
- White RA, Eisenstat TE, Rubin RJ, Salvati EP (1990) Seton management of complex anorectal fistulas in patients with Crohn's disease. Dis Colon Rectum 33: 587-589.
- Scott HJ, Northover JM (1996) Evaluation of surgery for perianal Crohn's fistulas. Dis Colon Rectum 39: 1039-1043.
- Williams JG, Rothenberger DA, Nemer FD, Goldberg SM (1991) Fistula-in-ano in Crohn's disease. Results of aggressive surgical treatment. Dis Colon Rectum 34: 378-384.
- Sher ME, Bauer JJ, Gelernt I (1991) Surgical repair of rectovaginal fistulas in patients with Crohn's disease: transvaginal approach. Dis Colon Rectum 34: 641-648.
- Sonoda T, Hull T, Piedmonte MR, Fazio VW (2002) Outcomes of primary repair of anorectal and rectovaginal fistulas using the endorectal advancement flap. Dis Colon Rectum 45: 1622-1628.
- Sangwan YP, Schoetz DJ Jr, Murray JJ, Roberts PL, Coller JA (1996) Perianal Crohn's disease. Results of local surgical treatment. Dis Colon Rectum 39: 529-535.
- Michelassi F, Melis M, Rubin M, Hurst RD (2000) Surgical treatment of anorectal complications in Crohn's disease. Surgery 128: 597-603.
- Hyman N (1999) Endoanal advancement flap repair for complex anorectal fistulas. Am J Surg 178: 337-340.
- Makowiec F, Jehle EC, Becker HD, Starlinger M (1995) Clinical course after transanal advancement flap repair of perianal fistula in patients with Crohn's disease. Br J Surg 82: 603-606.
- Yassin NA1, Hammond TM, Lunniss PJ, Phillips RK (2013) Ligation of the intersphincteric fistula tract in the management of anal fistula. A systematic review. Colorectal Dis 15: 527-535.
- Lewis R1, Lunniss PJ, Hammond TM (2012) Novel biological strategies in the management of anal fistula. Colorectal Dis 14: 1445-1455.
- Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, et al. (2003) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ 339:b2700.
- Wells BS, O'Connell D, Peterson J, Welch V, Losos M, et al. (2000) The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta- analyses. 3rd Symposium on Systematic Reviews: Beyond the Basics3-5.
- Abel ME, Chiu YS, Russell TR, Volpe PA (1993) Autologous fibrin glue in the treatment of rectovaginal and complex fistulas. Dis Colon Rectum 36: 447-449.
- Venkatesh KS, Ramanujam P (1999) Fibrin glue application in the treatment of recurrent anorectal fistulas. Dis Colon Rectum 42: 1136-1139.
- Aitola P, Hiltunen KM, Matikainen M. Fibrin glue in perianal fistulae-a pilot study. Ann Chir Gynaecol 88:136-138.
- Cintron JR, Park JJ, Orsay CP, Pearl RK, Nelson RL, et al. (2000) Repair of fistulas-in-ano using fibrin adhesive: long-term follow-up. Dis Colon Rectum 43: 944-949.
- Park JJ, Cintron JR, Orsay CP, Pearl RK, Nelson RL, et al. (2000) Repair of chronic anorectal fistulae using commercial fibrin sealant. Arch Surg 135: 166-169.
- Buchanan GN, Bartram CI, Phillips RK, Gould SW, Halligan S, et al. (2003) Efficacy of fibrin sealant in the management of complex anal fistula: a prospective trial. Dis Colon Rectum 46: 1167-1174.
- García-Olmo D, García-Arranz M, Herreros D, Pascual I, Peiro C, et al. (2005) A phase I clinical trial of the treatment of Crohn's fistula by adipose mesenchymal stem cell transplantation. Dis Colon Rectum 48: 1416-1423.
- Singer M, Cintron J, Nelson R, Orsay C, Bastawrous A, et al. (2005)Treatment of fistulae-in-ano with fibrin sealant in combination with intra-adhesive antibiotics and/or surgical closure of the internal fistula opening. Dis Colon Rectum 48:799-808.
- Alexander SM, Mitalas LE, Gosselink MP, Oom DM, Zimmerman DD, et al. (2008) Obliteration of the fistulous tract with BioGlue adversely affects the outcome of transanal advancement flap repair. Tech Coloproctol 12: 225-228.
- Garcia-Olmo D, Herreros D, Pascual I, Pascual JA, Del-Valle E, et al. (2009) Expanded adipose-derived stem cells for the treatment of complex perianal fistula: a phase II clinical trial. Dis Colon Rectum 52:79-86.
- Thekkinkattil DK, Botterill I, Ambrose NS, Lundby L, Sagar PM, et al. (2009) Efficacy of the anal fistula plug in complex anorectal fistulae. Colorectal Dis 11: 584-587.
- Grimaud JC, Munoz-Bongrand N, Siproudhis L, Abramowitz L, Sénéjoux A, et al. (2010) Fibrin glue is effective healing perianal fistulas in patients with Crohn's disease. Gastroenterology 138: 2275-2281, 2281.
- Gajsek U, McArthur DR, Sagar PM (2011) Long-term efficacy of the button fistula plug in the treatment of Ileal pouch-vaginal and Crohn's-related rectovaginal fistulas. Dis Colon Rectum 54: 999-1002.
- Sileri P, Boehm G, Franceschilli L, Giorgi F, Perrone F, et al. (2012) Collagen matrix injection combined with flap repair for complex anal fistula. Colorectal Dis 14 Suppl 3: 24-28.
- Halverson AL, Hull TL, Fazio VW, Church J, Hammel J, et al. (2001) Repair of recurrent rectovaginal fistulas. Surgery 130: 753-757.
- Loungnarath R, Dietz DW, Mutch MG, Birnbaum EH, Kodner IJ, et al. (2004) Fibrin glue treatment of complex anal fistulas has low success rate. Dis Colon Rectum 47: 432-436.
- Ellis CN (2008) Outcomes after repair of rectovaginal fistulas using bioprosthetics. Dis Colon Rectum 51: 1084-1088.
- Safar B, Jobanputra S, Sands D, Weiss EG, Nogueras JJ, et al. (2009) Anal fistula plug: initial experience and outcomes. Dis Colon Rectum 52: 248-252.
- El-Gazzaz G, Hull T, Mignanelli E, Hammel J, Gurland B, et al. (2010) Analysis of function and predictors of failure in women undergoing repair of Crohn's related rectovaginal fistula. J Gastrointest Surg 14:824-829.
- El-Gazzaz G, Hull TL, Mignanelli E, Hammel J, Gurland B, et al. (2010) Obstetric and cryptoglandular rectovaginal fistulae: long-term surgical outcome; quality of life; and sexual function J Gastrointest Surg 14:1758-1763.
- de Parades V, Far HS, Etienney I, Zeitoun JD, Atienza P, et al. (2010) Seton drainage and fibrin glue injection for complex anal fistulas. Colorectal Dis 12: 459-463.
- Gaertner WB, Madoff RD, Spencer MP, Mellgren A, Goldberg SM, et al. (2011) Results of combined medical and surgical treatment of recto-vaginal fistula in Crohn's disease. Colorectal Dis 13: 678-683.
- Kleif J, Hagen K, Wille-Jørgensen P (2011) Acceptable results using plug for the treatment of complex anal fistulas. Dan Med Bull 58: A4254.
- Choi JM, Nguyen V, Khavari R, Reeves K, Snyder M, et al. (2012) Complex rectovaginal fistulae after pelvic organ prolapse repair with synthetic mesh: a multidisciplinary approach to evaluation and management. Female Pelvic Medicine & Reconstructive Surgery 18:366-371.
- Garcia S, Dissanaike S (2012) Case report: Treatment of rectovaginal fistula with Bioglue(®). Int J Surg Case Rep 3: 327-329.
- Binda GA, Piscitelli A, Longhin R (2013) Treatment of high ano-vaginal fistula with GORE BIO-A ® Fistula Plug in an immunocompromised patient. Tech Coloproctol 17: 609-611.
- García-Olmo D, García-Arranz M, Herreros D, Pascual I, Peiro C, et al. (2005) A phase I clinical trial of the treatment of Crohn's fistula by adipose mesenchymal stem cell transplantation. Dis Colon Rectum 48: 1416-1423.
- (2008) The Surgisis AFP anal fistula plug: report of a consensus conference. Colorectal Dis 10: 17-20.
- Blaker JJ, Pratten J, Ready D, Knowles JC, Forbes A, et al. (2008) Assessment of antimicrobial microspheres as a prospective novel treatment targeted towards the repair of perianal fistulae. Aliment Pharmacol Ther 28:614-622.
- Jayne D (2010) The Fistula-In-Ano Trial comparing Surgisis® anal fistula plug versus surgeon's preference (advancement flap, fistulotomy, cutting seton) for transsphincteric fistula-in-ano.
- Ellis CN, Rostas JW, Greiner FG (2010) Long-term outcomes with the use of bioprosthetic plugs for the management of complex anal fistulas. Dis Colon Rectum 53: 798-802.
- Hellers G, Bergstrand O, Ewerth S, Holmström B (1980) Occurrence and outcome after primary treatment of anal fistulae in Crohn's disease. Gut 21: 525-527.
Relevant Topics
- Constipation
- Digestive Enzymes
- Endoscopy
- Epigastric Pain
- Gall Bladder
- Gastric Cancer
- Gastrointestinal Bleeding
- Gastrointestinal Hormones
- Gastrointestinal Infections
- Gastrointestinal Inflammation
- Gastrointestinal Pathology
- Gastrointestinal Pharmacology
- Gastrointestinal Radiology
- Gastrointestinal Surgery
- Gastrointestinal Tuberculosis
- GIST Sarcoma
- Intestinal Blockage
- Pancreas
- Salivary Glands
- Stomach Bloating
- Stomach Cramps
- Stomach Disorders
- Stomach Ulcer
Recommended Journals
Article Tools
Article Usage
- Total views: 14621
- [From(publication date):
August-2016 - Sep 02, 2025] - Breakdown by view type
- HTML page views : 13634
- PDF downloads : 987