Thermodynamic and Kinetic Investigations for Biosorption of Chromium(VI) With Green Algae (Pithophora oedogonia)
Received: 14-Sep-2017 / Accepted Date: 30-Sep-2017 / Published Date: 06-Oct-2017 DOI: 10.4172/2155-6199.1000414
Abstract
Heavy metals are the conservative pollutants which cannot be degraded by bacterial attack and are permanent addition to marine environment. Their conservation usually exceeds the allowable levels usually found in water ways and soil. They locate their way up the food chain and due to their accumulation, can disrupt biological process. Recently, biosorption has come up as effective tool in which biomass of various organisms have been tested. In the present study, the biosorptive potential of algae biomass of green algae (Pithophora oedogonia) has been investigated for removal of toxic heavy metal, such as chromium (VI) ions. Various physico-chemical factors have been optimized for biosorptive capacities of sorbates by sorbents. Optimum pH was found to be 4 and optimum temperature was 30°C for Cr (VI). Various adsorption models were elucidated to data, such as Langmuir, Freundlich and Temkin isotherms whereas Freundlich model was found to be fittest showing multilayer sorption. Pseudo-second order kinetic model was also found to fit for this study with regression coefficient value of 0.97. Additionally, Fourier Transform Infra-red Spectroscopic studies (FTIR) indicated various electronegative functional groups on the surface of green algae (Pithophora oedogonia) which could possibly offer the binding sites for cations under investigation.
Keywords: Heavy metals; Biosorption; Algae; Kinetics; FTIR
Introduction
The natural balance of biogeochemical cycles within ecosystem is the prime operators of the biosphere and biodiversity. However urbanization and industrialization in recent time have burdened the natural input of the environment in recent times. In these lines, a variety of industrial pollutants are discharged daily into the environment, containing heavy metal pollutants, just like manganese (Mn+2), lead (Pb+2), mercury (Hg+2), cadmium (Cd+2), chromium (Cr+6), etc. This problem is worldwide and the investigated number of contaminated places with heavy metals is magnifying regularly [1]. Main sources of heavy metals came from metal finishing industries and electroplating, chemical manufacturing, battery manufacturing, fertilizer industries, pigment manufacturing industries, tannery operations metallurgical industries, mine drainage, leakages from contaminated ground water and landfills [2-4]. The heavy metal, chromium (Cr), is a grey solid with greater melting point (1907°C) and an estimated atomic weight of 51.996 g/mol. Chromium has oxidation phase ranging from divalent [Cr (II)] to hexavalent [Cr (VI)] form of Cr [5]. Chromium produces a large number of compounds, in both (hexavalent) and trivalent Cr3+ and chromium [Cr (VI)] forms. Chromium compounds are unwavering in the trivalent state, with the hexavalent form being the second most secure state [5]. A number of studies in humans and animals have been reported to show the effect of Cr (VI). For example in humans exposure to Cr (VI) via inhalation may result in the complication during pregnancy and child birth [5]. Oral studies have reported severe developmental effects in mice, such as gross abnormalities and reproductive effects including decrease little size, reduced sperm count and degeneration of the outer cellular layer of the seminiferous tubules [5] Epidemiological studies of workers have clearly established that inhaled chromium is human carcinogen, resulting in an increased risk of lung cancer. Although chromium exposed workers were exposed to both Cr3+ and Cr (VI) compounds only Cr (VI) has been found to be carcinogenic in animal studies, thus EPA has concluded that only Cr (VI) should be classified as a human carcinogen [5,6]. Animal studies have shown Cr (VI) to cause lung tumors via inhalation exposure [5]. EPA has classified Cr (VI) as a group A, known human carcinogen [6]. Adsorption is a phenomenon which attracted attention of many scientists. Synthetic and natural adsorbents are used for this purpose. Many natural oxides are being used for elimination of heavy metals such as oxides of Aluminum [7-11]. Silica gel (synthetic polymer) that contains silanol groups on its surface position that allocates metal adsorption [12,13]. Though many of synthetic and natural adsorbents can help in removing the heavy Metals many of them have disadvantages such as they may be costly or they may be less efficient etc. The biosorption technique has emerged as a significant tool in the live of removal of toxic metals in the past decade. Biosorption is different from bioaccumulation. In biosorption dead biomass is used for the elimination of heavy metal pollutants whereas in bioaccumulation, heavy metals pollutants are metabolically uptaken by living organisms [14]. Biochemical structure or composition of cell wall of biomass is responsible for the quantity of heavy metal ions which can attach on the surface of the biomass. In all living organisms, bioaccumulation and biosorption can occur naturally [15]. But under experimental conditions, biosorption has been found to be 13-20% higher with dead Organism than with living organism). Contrary to other forms of biomass, algal biomass has been found to show promising results for biosorption of heavy metal ions [16-18]. The underlying fact is that the cell wall of algae may possess various electronegative functional groups which may be responsible for their binding to cations, such as toxic metal ions. Exploitation of green algae in waste water treatment technology may hence be cost-effective. Adsorptive elimination of heavy metals from effluents which have received much consideration in latest years is generally attained by using activated alumina or carbon [19,20]. Activated carbon is a spongy matter with an exceptionally large surface area and fundamental adsorption to different chemicals. It is only able to eradicate almost 30-40 mg/g of Zn, Cd and Cr in water and is non-regeneratable, which is pretty expensive to wastewater treatment. A number of allied processes are operating in this process, such as reverse osmosis, electro-dialysis, ultra-filtration, precipitation, etc. But most of these are expensive as far as removal of heavy metal ion is concerned. This created a need to consider other options. Researchers have diverted attention to living organisms in the recent decades and the capacity of biological tool in this context has been tested in a number of rent studies which are abundantly available and are reproducible [21]. The biological materials when employed in sorption procedures are referred to as biosorbents capable to adsorb sorbates, such as heavy metal ions. The term biosorption may hence be coined. Biosorption is the metabolically passive process which does not require energy [22]. In the present study, green algae, Pithophora oedogonia has been selected for its evaluation for the biosorption of Cr (VI) ions. A number of physico-chemical parameters have been studied which have been found to affect this process. Additionally, various adsorption models and pseudo-second order model was been employed to the data obtained. The surface of the biosorbent has been examined by performing FTIR to explore the possible binding sites of Cr (VI) ions.
Materials And Methods
Green algae, Pithophora oedogonium was used as biosorbent in the present study P. oedogonium was collected from ponds of GCU Botanical Gardens, Lahore, Pakistan. The samples were identified by following the key (British fresh-water algae, exclusive of desmidieae and diatomaceae by M.C Cooke, MALLDALS Vol. II, Plates). The samples were brought to the laboratory at Department of Zoology, Government College University Lahore. Algal biomass was washed thoroughly with tap water and the debris and soil particles were removed subsequently. Biomass was treated with 0.02M HNO3 to remove any possible cations etc. already adsorbed on the surface. Then algae were dried at 60°C and finally dried biomass was ground by nylon mesh of 300 nm [21]. A stock solution of 100 ppm of chromium (VI) was prepared by dissolving 512.5 mg of K2Cr2O7 in 1000 mL of distilled water. Thereafter, by serial dilutions, 100, 200, and 300 ppm solutions were prepared. Erlenmeyer flasks of 125 mL were used for this experiment. Before use, all the flasks were washed with HNO3 followed by a thorough wash with distilled water.100 mL of Cr solution were taken in 250 mL Erlenmeyer flasks. The solutions were set at the pH of 4.0 for Cr by using 0.1 N HNO3 and 0.1 N NaOH. 10 mg, 20 mg and 30 mg of algal biomass (biosorbent) were separately taken in the metal ion solution of this metal. Thereafter all the flasks were set on rotator shaker at 25°C at 180 rpm (rotation per minute) for 2 hours. Samples (each of 5 mL) were periodically collected after every 30 minutes. The samples for each metal ion solution were analyzed at PIXE (proton Induced X-ray Emission), Model, 6SDH2. This study was performed at Centre of Advanced Studies in Physics (CASP), GC University Lahore, Pakistan.
Results
Biosorption of chromium by Pithophora oedogonia
The efficiency of the biosorption is greatly affected by the physicochemical properties of the solutions, such as temperature, pH, initial concentration, etc. The biosorption capacity of P. oedogonia was studied under variable conditions.
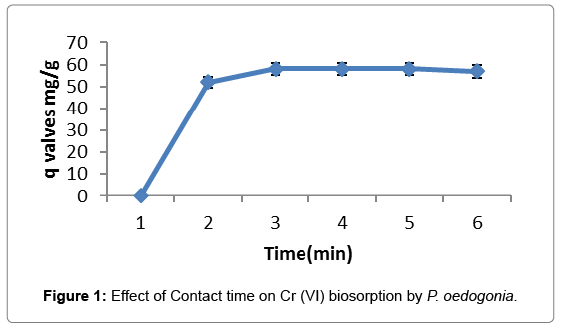
Effect of contact time
The efficiency of biosorption increased in the contact time during the first 30 minutes, followed by slower uptake up to 90 min and thereafter equilibrium was obtained because of no significant uptake of Cr (VI) ions after 90 minutes (biosorbent concentration at 10 mg/L; initial sorbate concentration at 100 mg/L; pH 4.0) (Figure 1).
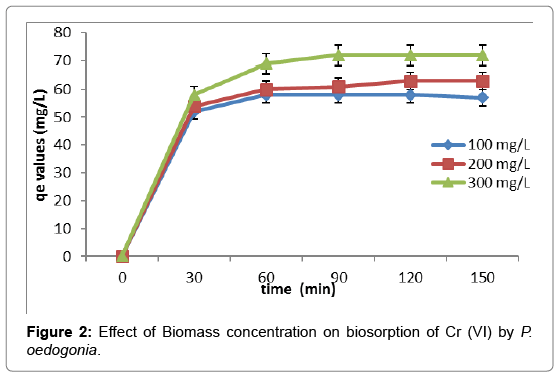
Effect of biomass quantity
Effect of biosorbent dosage on uptake of the Cr (VI) was observed by using different concentration of the algal biomass, 10, 20, 30 mg/L. The concentration of the algal biomass was found to be directly proportional to the Cr (VI) biosorption. But the maximum uptake of Cr (VI) ion was observed at the minimum exposure of algal biomass like 10 mg/L (Figure 2).
Mathematical modeling
Freundlich and Langmuir isotherm were used to interpret the data of biosorption as well as parameters of kinetic and thermodynamic studies were also used to evaluate mechanistic aspect.
Equilibrium isotherm
Equilibrium data is associated with the help of Langmuir isotherms.
The Langmuir equation is given below:
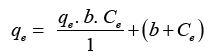
Here, Ce=Equilibrium conc. Of heavy metal ion; qe=Equilibrium const. of metal ion on biosorbent surface (mg/g); K2=saturation const. (mg/L).
Values of Ce, qe and K2 were obtained by calculation of linear plot slop and intercept of 1/Ce versus 1/qe. The value of distribution coefficient (K) between aqueous solution and sorbent could be found out by following equation:
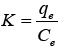
Adsorption intensity among sorbate and adsorbent could also be found out by Freundlich isotherm was used to evaluate multi-layered adsorbent. The equation given below is used for this isotherm:
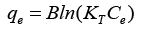
Kf=Freundlich empirical const./sorption capacity
1/n=empirical constant/sorption Intensity
1/n and Kf values could be calculated from slope and intercept respectively bya linear plot of lnCe versus lnqe.
The equation given below was used for assessment of adsorption process which is Temkin isotherm:
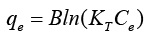
KT=equilibrium binding const. toward the optimum binding energy; B=const. correlated to heat of adsorption.
Kinetic studies
“Pseudo-second order kinetic model (Ho and McKay) was employed to evaluate the mechanistic aspect of biosorption of heavy metal by using Pithophora oedogonium. Pseudo-second order kinetic model considers that the rate of occupation of biosorption sites is proportional to the square of the number of unoccupied sites.
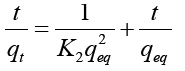
Where, t (min)=Time; qt (mg g−1)=Uptake capacity at time ‘t’; K2 (gmg−1 min−1)=Equilibrium rate constant of pseudo-second-order adsorption. After being integrated and rearranged, following expression could be achieved:
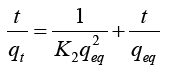
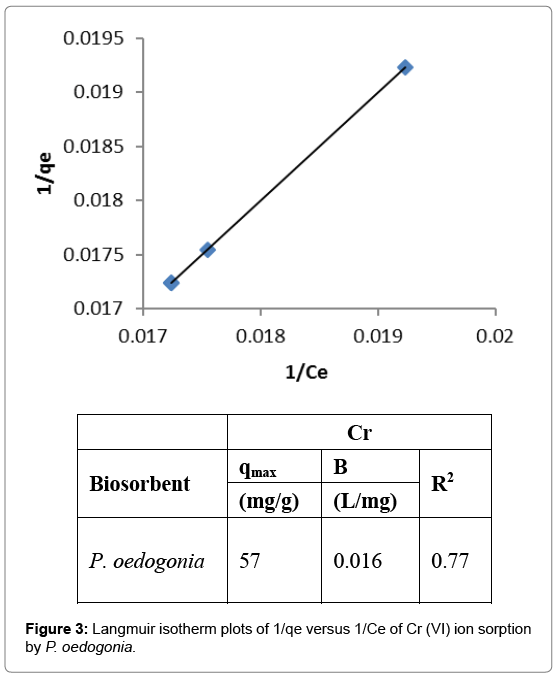
Langmuir isotherm
Plots of inverse of the equilbrium concentration of Cr (VI) ions biosorption by the sorbent (1/qe), versus inverse of equilbrium concentration of the Cr (VI) in the aqueous solution (1/Ce) at the various ion concentration in the solution were drawn. Maximum adsorption capacity (qmax) of Cr (VI) biosorption by Pithophora oedogonia was found to be 57 mg/g; “b” as 0.016, and R2 as 0.77 (Figure 3).
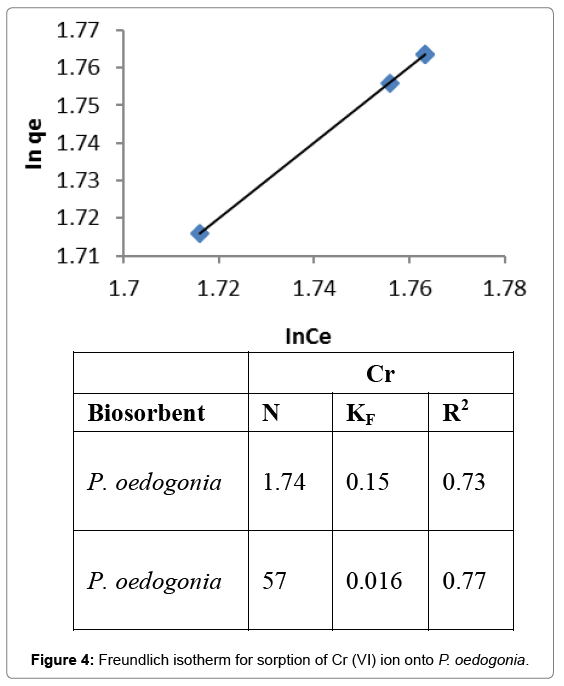
Freundlich isotherm
This isotherm was used to determine the adsorption intensity of the sorbent towards the heavy metal ions. A linear plot of lnqe versus lnCe was drawn and values of intercept and slope were determined to determine Freundlich’s constants. The value of N for Cr (VI) biosorption by Pithophora oedogonia was found to be 1.74, KF (saturation constant) was 0.15, and R2 (regression coefficient) was 0.73 (Figure 4).
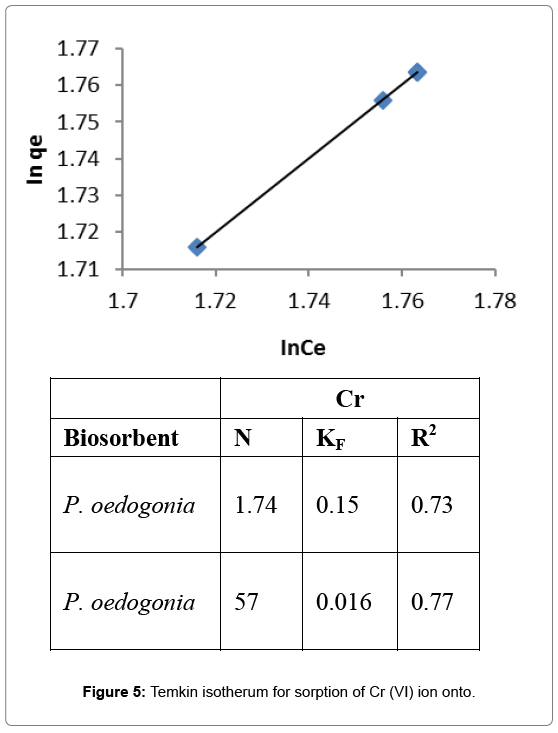
Temkin isotherm
Linear plot of qe versus lnCe was drawn and the values of intercept and slope were calculated. Values of Temkin’s constant determined thereafter. Value of KT (equilbrium binding constant for Cr (VI) by Pithophora oedogonia was found to be 0.055, B (Temkin’s constant) was 134.5, and R2 (regression coefficient) was found 0.72 for Cr (VI) (Figure 5).
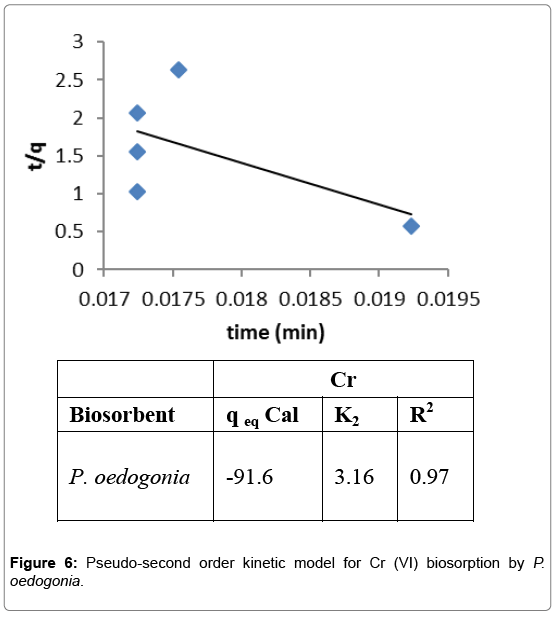
Pseudo-second order kinetic model
Pseudo-second order model was employed for the biosorption of Cr (VI) by Pithophora oedogonia and was found to be well fitted for the data obtained from the experiment. A linear plot of t/qeq versus t was drawn and intercept and slope of the data was calculated to determine the constants of pseudo-second order kinetic model. Value of qeq was found to be -91.6, K2 was 3.16, and R2 as 0.97 (Figure 6).
FTIR spectroscopy
FTIR spectroscopy shows the results in peaks of 3674 (unloaded samples) to 3669 (loaded samples for Cr), 1671 (unloaded samples) to 1684 (loaded samples for Cr) and 2893 (unloaded samples) to 2883 (loaded samples for Cr). These shifts are the characteristic electronegative functional groups, such as carbonyl (-C=O), amine (-N-H), and alkyl (-C=H2) respectively.
Discussion
Biosorption of heavy metal, chromium by Pithophora oedogonia has been taken under investigation at different pH values i.e., 3.0, 4.0, 5.0, P. oedogonia shows maximum uptake of heavy metals such as Cr (VI) at optimum pH 4. this can be explained by the fact that at pH above this, viz: pH 6, 7, 8, 9, 10 etc., the cations tend to participate, and eventually the biosorption is affected; whereas, at pH lower than the optimum pH i.e., 3.0, 2.0, and 1.0 the aqueous solution tends to get protonated and the test cations have to compete with these protons. As a result, lower biosorption has been recorded at lower pH optimum time period required to achieve equilbrium in the aqueous solution was found to be 90 for Cr (VI). The most perfect explanation is that binding sites located on the surface of algal biomass are fully free at the start of the experiment; they then start getting saturated when exposed to aqueous solution of metal ions. In the first 30 minutes, quick uptake of metal ions has been observed. The following 60 minutes (up to 90 minutes of contact time) relatively slower uptake of Cr (VI) was observed by Pithophora oedogonia. This is the time period when cations have to compete with each other as well as hydrogen ions (H+), naturally existing in the aqueous solution. Up to 60 minutes of contact time, almost all the binding sites are fully saturated and hence equilbrium is achieved. Therefore, no further biosorption was recorded after 60 minutes of contact time. Time required to attain qmax in the present study (90 minutes) did not vary significantly in the present study to those reported previously conducted similar studies. Gupta and Rastogi observed that maximum adsorption took place within first 30. The possible explanation of this could be the variation in cell wall biochemistry of the biosorbents used in the above mentioned reports. Aqueous solution is another parameter which is affecting the process of biosorption. The metal uptake was recorded directly proportional to the biosorbent concentration. However, of the three concentrations used in the present study,10 mg/L of biomass concentration has showen optimum biosorption capacity (qeq) values followed by 20 mg/L and 30 mg/L respectively. It can be inferred that the adsorption of cations per unit mass is maximum in case of 10 mg/L. Moreover, equilibrium was found to achieve relatively quickly at biosorbent concentration of 10 mg/L as compared to 20 mg/L and 30 mg/L. similar trend was suggested by the previously performed similar studies [23]. Optimum biomass dosage for biosorption was found to be 1 mg/L. The most probable reason may be the difference of biosorbent used for biosorption. Of the various adsorption isotherm models employed to the experimental data. Langmuir’s isotherm was found to be best fitted followed by Temkin isotherm whereas Freundlich isotherm was not found to be fitted as good as the others. Experimental values were in agreement with the values obtained from Langmuir isotherm, thus showing monolayer sorption. This finding is in agreement with the previously performed similar studies [23]. Pseudo-second order kinetic model was found to be suitable for the study of kinetics of biosorption of Cr (VI) by Pithophora oedogonia. The values of Pseudo-second order constants, such as K2, R2 , and qeq Cal are showing good values. Similar findings have also been reported by the other researchers of biosorption [24]. The cell wall of filamentous algae, such as P. oedogonia, etc. may consist of macromolecules, such as cellulose in many (b-1, 4-glucopyroside) [25]. Moreover storage compounds, such as amylase and amylopectin may also be present on the cell wall. These compounds possess many functional groups, such g as carboxyl, hydroxyl, carbonyl, etc. These functional groups are the biosorptive binding sites for the cations, such as Cr (VI). Various mechanisms have been proposed as mechanism of the studies of biosorption by algae, such as complexation, chelation, coordination, ion exchange, precipitation, reduction etc. However, in the present study some of these seem to be more plausible discussed as follows. Ion exchange is a reversible chemical reaction where an ion within a solution is replaced by a similarly charged ion attached onto an immobile solid particle. In general; the ion exchange mechanism can be represented by the following equation:
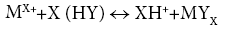
Here HY corresponds to the number of acid sites on the solid surface, MX+ is metal ion, and MYX is the sorbed MX+. By considering the above equation, the ion-exchange equilibrium constant can be determined [23]. Hence, filamentous green alga P. oedogonium which has shown promising results in the present study is expected to contribute to the bioremediation of polluted water. Future studies of biosorption of toxic heavy metal cations may also help to eliminate the mechanism of removal of heavy metals using algal biomass. Further studies based on commercial usage of Pithophora oedogonia in biosorption technology needs special emphasis on mechanistic studies, such as kinetics, thermodynamics, etc. FTIR spectra clearly show the presence of electronegative functional groups, the peaks of which seems to shift, when loaded with Cr (VI) ion. These findings are in agreement with previously performed similar studies [23]. The cell wall of filamentous algae, such as P. oedogonia, consists of macromolecules, such as cellulose and other compounds, such as b-1, 4-glucopyroside [26-29]. Moreover, storage compounds, such as amylase and amyl pectin may also be present on the cell wall [30,31]. These compounds possess many functional groups, such as carboxyl, hydroxyl, carbonyl, etc. These functional groups are the biosorptive binding sites for the cations, such as Cr (VI). Various mechanisms may hence be proposed as mechanism of the studies of biosorption by algae, such as complication, chelating, coordination, ion exchange, precipitation, reduction etc. However, in the present study some of these seem to be more plausible discussed as follows.
References
- Cairney T (1993) Contaminated land problems and solutions. Chapman-Hall, Glasgow,UK, pp:1-140
- Faisal M,Hasnain S (2004)Microbia conversion of Cr (VI) into Cr (iii) in industrial effluent. African JBiotechnol 110:610-617.
- Mclaughlin MJ, Tller RG, Naidu R,Steven DP (1996) Review: the behavior and environmental impact of contaminants in fertilizers. Aust J Soil Res 34: 1-54.
- Reed BF, Arnnachalam S, Thoma B (1994)Removal of lead and cadmium from aqueous waste streams using Granular activated carbon (GAC) columns. Environ Prog 13: 60-65.
- ATSDR (1998) Toxicological profile for Chlorinated Dibenzo-p-Dioxins. US Department of Health and Human Services. Atlanta GA.
- US EPA (1999)Storm water technology fact sheet: Bioretention. EPA USEPA Washington DC
- Gupta DC,Tiwari UC (1985)Aluminium oxide as adsorbent for removal of hexavalent chromium from aqueous waste. Indian J EnvHlth27: 205-215.
- Srivastava SK, Tyagi R, Pant N, Pal N (1989) Studies on the removal of some toxic metal ions: part II (Removal of lead and cadmium by montmorillonite and kaolinite). EnvironTechnol Lett 10: 275-282.
- Bailey RP, Bennett T,Benjamin MM (1992) Sorption onto and recovery of Cr (VI) using iron-oxide coated sand. Water Sci Technol 26: 1239-1244
- Potgieter JH, PotgieterV, Kalibantonga PD (2006) Heavy metals removal from solution by palygorskite clay. Minerals Engineering 19: 463-470
- Kikuchi A, Sanuki N, Higashi K, Koshiba T,Kamada H,et al. (2006) Abscisic acid and stress treatment are essential for the acquisition of embryogenic competence by carrot somatic cells. Plant 223: 637-645.
- Ricordel S, Taha S, CisseI, Dorange G (2001) Heavy metals removal by adsorption onto peanut husks carbon: characterization kinetics study and modeling.SepPurifTechnol 24: 389-401
- Michard G, Viollier E, zequel D,Sarazin G (1994) Geochemical study of a crater lake: Pavin Lake, France – Identification, location and quantification of the chemical reactions in the lake.ChemGeol 115: 103-115
- Seki H, Suzuki A (1998) Biosorption of heavy metal ions to brown algae, Macrocystispyrifera, Kjellmaniellacrassiforiaand Undariapinnatifida. JColloid InterfaceSci 206: 297-301
- Chojnacka K, Chojnacki A,Górecka H (2005) Biosorption of Cr3+, Cd2 + and Cu2 +ions by blue-green algae Spirulina spKinetics, equilibrium and the mechanism of the process. Chemosphere59: 75-84.
- Kapoor A,Viraraghavan T (1997)Heavy metal biosorption sites in Aspergillus niger. BioresourceTechnol 61: 221-227.
- Aksu Z,Donmez G (2001) Comparison of copper (II) biosorption properties of live and treated Candida sp.JEnviron Sci. Health 36: 367-381
- Mehta SK, Gaur JP (2005) Use of algae for removing heavy metal ions from wastewater: progress and prospects. CritRevBiotechnol 25: 113-152.
- Faust SD, Aly OM (1987)Adsorption processes for water treatment. Butterwort Publishers. Boston, pp: 108-113
- Ralph TY, Richard QL, Joel P (1999) Adsorbents for Dioxins: A new technique for sorbent screening for low volatile organics. IndEngChemRes 38: 2726-2731
- Yaqub A, Mughal MS, Adnan A, Khan WA (2012)Biosorption of hexavalent chromium (Cr VI) by Spirogyra Sp. Equilibrium, kinetics and thermodynamics. JAPS 22: 408-415
- VelásquezL,Dussan J (2009)Biosorption and bioaccumulation of heavy metals on dead and living biomass of Bacillus sphaericus. J Hazard Mater 167: 713-716
- Arief VO, Trilestari K, Sunarso J,Ismadji NS (2008)Recent progress on heavy metals from liquids using low cost biosorbents: charterization, biosorption parameters and mechanism studies. Clean36: 937-962
- Pahlvanzadeh H, Keshtkar AR, Safdari J,Abadi Z (2010) Biosorption of nickel (II) from aqueous solution by brown algae: Equilibrium, dynamic and thermodynamic studies. J Hazard Mater 175: 304-310.
- Davis JA, Volesky B,Vierra RHSF (2000)Sargassum Seaweed as biosorbent for heavy metals. Water Res 34:4270-4278.
- Huang CP,Fu PLK (1984) Treatment of arsenic (v) containing water by the activated carbon process. J Water pollu. Control Fed 56: 233-237.
- Jackson AP,Alloway BT(1991)The bio-availability of cadmium to lettuce and cabbage in soils previously treated with sewage sludges. Plant Soils 132: 179-186
- WHO (World Health Organization) (1987) Report of the 30th Meeting of the Joint FAO/WHO. Expert Committee on Food Additives. Geneva and Rome, World Health Organization.
- USEPA (US Environmental Protection Agency) (1998) Toxicological Review of Trivalent Chromium Environmental Protection Agency. Washington DC, USA.
- Shahzad S, Yaqub A, Mazhar NA, Hussain M (2016)Optimization of Conditions for Biosorption of Chromium and Cadmium by Shell of Andontoidesferussacianus. World Journal of Pharmaceutical and Medical Research 5: 1-10.
Citation: Suleman S, Noman S, Atif Y, Faizan N, Arooj M (2017) Thermodynamic and Kinetic Investigations for Biosorption of Chromium (VI) With Green Algae (Pithophora oedogonia). J Bioremediat Biodegrad 8: 414 DOI: 10.4172/2155-6199.1000414
Copyright: © 2017 Suleman S, et al. This is an open-a ccess article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 5668
- [From(publication date): 0-2017 - Dec 20, 2025]
- Breakdown by view type
- HTML page views: 4679
- PDF downloads: 989