Research Article Open Access
Oxidative Absorption of Hydrogen Sulfide using Iron-chelate Based Process: Chelate Degradation
Girish M Deshmukh*, Aparna Shete and Deepali M Pawar
Department of Petrochemical Engineering, Dr. Babasaheb Ambedkar Technological University, Lonere (Raigad), India
- *Corresponding Author:
- Girish M Deshmukh
Department of Petrochemical Engineering
Dr. Babasaheb Ambedkar Technological University
Lonere (Raigad) - 402103, India
Tel: +91-2140-275142, +91-9422383731
Fax: +91-2140-275142
E-mail: gmdeshmukh7@rediffmail.com
Received date: June 05, 2012; Accepted date: July 16, 2012; Published date: July 21, 2012
Citation: Deshmukh GM, Shete A, Pawar DM (2012) Oxidative Absorption of Hydrogen Sulfide using Iron-chelate Based Process: Chelate Degradation. J Anal Bioanal Tech 3:138. doi: 10.4172/2155-9872.1000138
Copyright: © 2012 Deshmukh GM, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Analytical & Bioanalytical Techniques
Abstract
Oxidative absorption of hydrogen sulfide into a solution of ferric chelate is studied in stirred cell glass reactor. The experiments are performed to investigate degradation of chelates viz., NTA, EDTA and DTPA at 313 K temperature, pH 6, Iron concentration 10000 ppm and Fe-Chelate molar ratio 1:2. Oxidative absorption of hydrogen sulfide into a solution of Fe-NTA was found better. Therefore, further experiments with 10%, 50% and 100% concentrations of hydrogen sulfide are performed. It is observed that this process is applicable for removal of low to high concentrations of hydrogen sulfide. Effect of antioxidant using sodium thiosulphate is also studied in order to minimize degradation of NTA. The kinetic parameters are studied and it is observed that the reaction appeared to be first order in ferric chelate and Rate constant values for 100%, 50% and 10% hydrogen sulfide concentration are 0.035 hr-1, 0.013 hr-1 and 0.019 hr-1 respectively.
Keywords
Hydrogen sulfide; absorption; ferric chelate; chelate degradation; nitrilotriacetic acid (NTA)
Introduction
Many commercial processes are available for the removal of hydrogen sulfide from gaseous streams. Most of the processes use gasliquid contactors in which hydrogen sulfide is contacted with a reagent to give either another dissolved sulfide containing components (for e.g. alkanol-amin or hydroxide based processes) or elemental sulphur as a precipitate. Important representatives of the latter type are so called iron chelate based processes. The desulphurization technique of bifunctional redox iron chelate process can be used for treating natural gas or petroleum gas. The desulphurization and cleaning processes can minimize the atmospheric emission of gases like SOx, NOx and CO. Here the principle of redox reaction of metal chelate with hydrogen sulfide is used for desulphurization.
The reaction sequence for oxidative absorption of hydrogen sulfide in iron chelate solutions is mentioned below:
H2S(g) ↔ H2S(aq) (1)
The overall chemistry is simple and consists of two sets chemical reactions.
Absorption:
H2S(aq) + 2 Fe3+ Chelantn- → S↓ + 2 H+ + 2 Fe2+ Chelantn- (2)
Regeneration:
O2 (aq) + 4 Fe2+Chelantn- + 2 H2O → 4 Fe3+ + Chelantn- + 4 OH- (3)
The function of the chelant is to prevent the formation of insoluble iron compounds without interfering with the ability of the iron to undergo reduction and oxidation.
The number ‘n’ denotes the charge of the chelant anion. In the absorption step, H2S comes in contact with a liquid containing a soluble ferric iron chelate that is the active ferric chelate (Fe3+chelantn-) is converted to inactive ferrous chelate ( Fe2+ chelantn-). The H2S forms sulfide and hydrosulfide ions which are selectively oxidized to form elemental sulfur and the ferric chelate, Fe3+chelant* is reduced to the corresponding ferrous chelate, Fe2+ chelant*.
In the regeneration step, the ferrous chelate, Fe2+chelant*, is reoxidized with air, back to ferric chelate, Fe3+chelant*. The regenerated solution is then recycled to the absorption section.
The set of chemical reactions given below summarizes the chelant free radical-induced oxidative degradation chemistry and is considerably more complex:
Fe2+chelant* + O2 + 2H+ → Fe3+chelant* + H2O2 (4)
Fe2+chelant* + H2O2 → Fe3+chelant*
The iron chelate can be regarded as a pseudo catalyst in the reaction of hydrogen sulfide with oxygen. The sulfur that is produced is easily recoverable from the slurry. Another advantage of iron chelate-based processes is that they essentially operate at ambient conditions, as illustrated in Table 1 [1].
| Temperature | 20-60 °C |
| pH | 4-8 |
| Important chelants | EDTA, HEDTA, NTA, DTPA |
| Iron concentration | 1000 to 10000 ppm |
| Chelant/iron ratio (M) | 1.1-2 |
Table 1: Typical Operating conditions of Iron Chelate Based Processes.
The most of the study has been done on oxidative degradation of chelate during regeneration of ferrous chelate. Chen et al. [2] used HPLC techniques to study the degradation of NTA during regeneration of ferrous chelate. It is recommended to use of chelating agents that are less reactive toward the hydroxyl radical as Fe3+/Fe2+ redox catalysts for absorption of hydrogen sulfide to sulfur. Some experimental studies reporting on oxidative absorption of hydrogen sulfide by ferric chelates are available.
Neumann and Lynn [3] examined the oxidative absorption of hydrogen sulfide by O2-saturated iron NTA solutions (0.1 mol dm-3, pH=4) in a small co-current wetted wall column. The overall system was proven effective with an overall efficiency reaching 99% for inlet hydrogen sulfide loadings of 40000-90000 ppm. Wubs and Beenackers [1] had established the reaction kinetics of hydrogen sulfide with ferric chelates of EDTA and HEDTA in a stirred cell reactor for temperature range of 21-60°C and pH range of 4-10. It was observed that pH plays significant role in oxidative absorption of hydrogen sulfide in ferric chelates. Piche et al. [4] studied the oxidative absorption of hydrogen sulfide in ferric chelate of CDTA in a counter current laboratory column randomly packed with 15 mm plastic Ralu rings. The maximum observed hydrogen sulfide conversion in the scrubber approached 91% for 10.5 pH and hydrogen sulfide concentration of 70 ppm.
As per the literature available, it is clear that the chelated iron solution is modified in such a way to be the best solution for desulphurization of petroleum gases with minimum drawbacks. The use of liquid based redox process using chelated iron can be reinvestigated here for desulphurization of gases having low to moderate ppm of hydrogen sulfide to minimize further degradation or remove the other drawbacks.
The present experimental work includes study for oxidative absorption of hydrogen sulfide using ferric chelates of NTA, EDTA and DTPA, 10%, 50% and 100% concentrations of hydrogen sulfide in Fe-NTA solution and use of sodium thiosulphate in order to minimize degradation of NTA.
Experimental
Materials
The following materials were obtained in the highest purity available and were used without purification.
Reagents: Ferric chloride hexahydrate (Merck), sodium salt of Nitrilotriacetic acid (Merck), ethylenedinitrilo tetraacetic acid disodium salt (EDTA) and Diethylene Triamine Penta Acetic Acid (DTPA) (Baker Analysed Reagents), Sulfuric acid (Merck), HPLC grade acetonitrile (Merck), HPLC water (Merck), Nitric acid (Merck), copper nitrate (Thomas Baker), Sodium thiosulphate (Merck), Ferrous Sulfide (Merck). The double distilled water was used throughout the study for experimentation and HPLC water was used for analysis
Gases: Hydrogen sulfide was produced in Kipp’s apparatus. Air was supplied through air pump.
Analytical equipment
Analysis of samples was performed using High Performance Liquid Chromatography (HPLC) equipped with a quaternary HP 1050 Pump and HP 1050 series variable wavelength UV/Visible detector. The manual injector is equipped with a sample loop of 20 μl. Nucleosil 100- 5 RP-C18 column (CHEMITO, Mumbai), 250 mm in length and 4.0 mm in diameter with a particle size of 5 μm.
Chemical preparation
The Fe3+ NTA, Fe3+ EDTA and Fe3+ DTPA respectively, solution were prepared by diluting a predetermined amount of NTA, EDTA and DTPA respectively, in double distilled water (150 ml). The appropriate amount of FeCl3•6H2O (Fe:NTA ratio = 1:2 mol/mol, Fe:EDTA ratio = 1.1:2 mol/mol and Fe:DTPA ratio = 1.1:2 mol/mol respectively) were added to the NTA, EDTA and DTPA respectively. The solution was diluted with double distilled water to adjust the total volume to 1000 ml, giving clear solution with a pH of about 6.
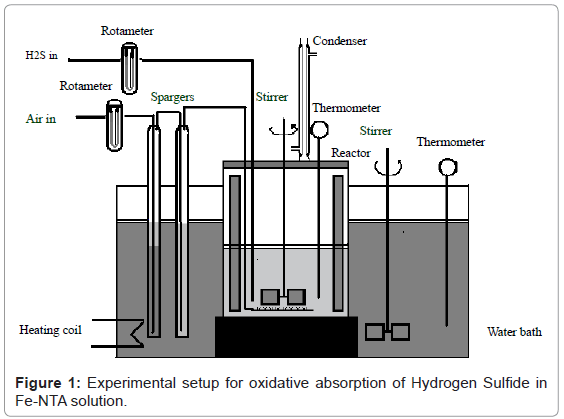
Experimental setup
The kinetic experiments for oxidative absorption of hydrogen sulfide into FeNTA aqueous solution were carried out batch wise in a thermo stated stirred cell reactor made up of glass having internal diameter 8 cm and volume 300 ml. A four blade glass turbine impeller of 1.5 cm blade diameter was used to stir the mixture as shown in Figure 1. Four symmetrically mounted glass baffles increased the effectiveness of stirring and prevented the formation of a vortex. The constant temperature water bath with constant stirring regulates the temperature of the liquid mixture during the experiments. The speed of agitation was maintained at 1000 rpm. The calibrated thermometer, tachometer, digital pH meter and rotameter were used to observe the temperature of solution, speed of stirrer, pH of the sample and flow rate respectively. A Kipp’s apparatus of 1 liter volume was used to produce hydrogen sulfide gas.
Experimental procedure
The experimental procedure includes the following steps:
1. Charging the Fe3+ NTA solution, of known volume and initial NTA concentration.
2. Liquid heating up to the temperature of 313 K.
3. Producing hydrogen sulfide gas in Keep’s apparatus by the reaction of ferrous sulfide sticks and sulfuric acid.
4. Initiating the reaction by carrying air and H2S. The H2S flow rate was maintained at 0.1 LPM and air flow rate was 1 LPM. Reduction of ferric ions to ferrous is started due to reaction with hydrogen sulfide and elemental sulfur is precipitated. Ferrous is converted again to ferric by oxygen present in air.
5. Steady state operation for 10 hours to study oxidative absorption of hydrogen sulfide for various reaction conditions. The samples were collected after an interval of one hour to measure the concentration of NTA.
Results and Discussion
There are several iron chelate based processes for the removal of hydrogen sulfide from gas streams have been developed over the years and commercialized. The most important difference between these processes concerned the liquid formulation and the way the degradation of chelate is suppressed. Obtaining kinetic data for oxidative absorption of hydrogen sulfide in iron chelate complex and the influence of the antioxidants on hydrogen sulfide removal, if applied in this sulfur removal process, will give the improved designs for desulphurization unit. This work dealt with these subjects and contributes to the development of a firm basis for designing of desulphurization unit.
Degradation of various chelates for oxidative absorption of hydrogen sulfide
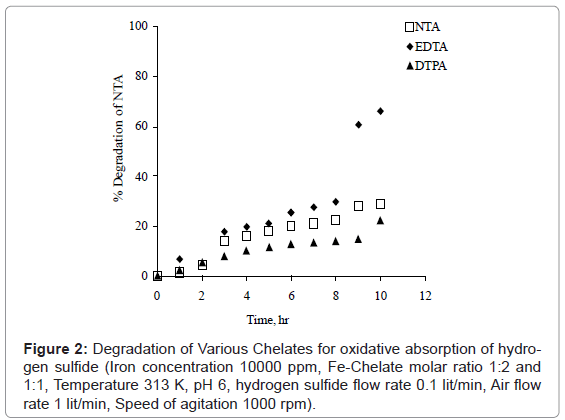
The degradation of EDTA, HEDTA, NTA and other aminopolycarboxylate chelating agents for oxidation of Fe-NTA complex was reviewed by Chen et al. [2]. The percentage degradation of HEDTA, EDTA and NTA were 80% within 40 hrs, 80 hrs and 100 hrs respectively. Pekss and Salmas also studied the oxidative degradation of chelates at the pH of 3-6, temperature range 30 to 60°C. The degradation of EDTA, DTPA and NTA for oxidative absorption of hydrogen sufide in iron-chelate complex was studied at temperature 313 K and pH 6 as shown in figure 2. The ferric chelate ratio was taken as 1:1 and 1:2 as per the stability of ferric with the particular chelates and the initial concentration of ferric was taken 10000 ppm. The concentrations of chelates were determined after different time interval using HPLC. The percentage degradation of NTA, DTPA and EDTA were 30%, 22% and 66% observed in 10 hours respectively. The order of degradation was found to be: EDTA > NTA > DTPA. It was also observed that hydrogen sulfide was absorbed only into iron-NTA solution. Ferric chelates of EDTA and DTPA was found unsatisfactory as there was no separation of elemental sulfur but degradation was observed. It is found that NTA is far superior ligand for better absorption of hydrogen sulfide in Fe (III) chelate system.
Degradation of NTA for oxidative absorption of various hydrogen sulfide concentrations
The effect of hydrogen sulfide concentration also plays most important role in NTA degradation due to reduction of ferric into ferrous during absorption process. Most of the investigators have reported removal of hydrogen sulfide from biogas using iron chelate solution and found suitable for removal of hydrogen sulfide from biogas. Horikawa et al. [5] have studied purification of a biogas by chemical absorption of hydrogen sulfide in an iron-chelated solution catalyzed by Fe/EDTA using continuous counter-current absorber. It is reported that for a biogas flow-rate of 1000 ml/min and 2.2 kg/cm2 pressure, approximately 90% and 70% of the H2S can be removed with the catalytic solution flowing at 83 and 68 ml/min, respectively. This means that above a flow-rate of 83 ml/min or with an appropriate ratio of gas contacting phases, it is possible to achieve total removal of H2S.
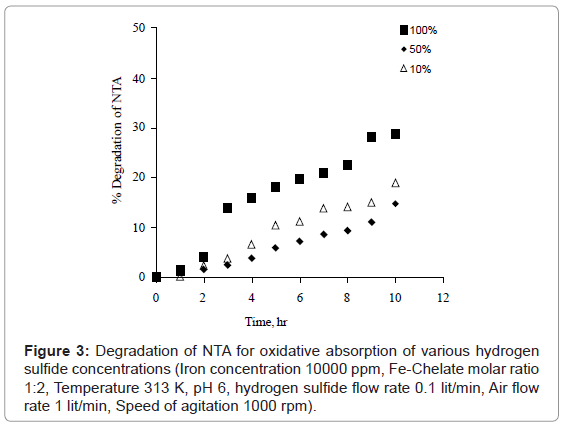
Biogas contains 2-5% concentration of hydrogen sulfide. However, concentration of hydrogen sulfide may be higher in industrial waste gases. The effect of hydrogen sulfide concentration for different hydrogen sulfide percentage viz. 10%, 50% and 100% on NTA degradation was studied at optimized experimental conditions. The various hydrogen sulfide concentrations (10% and 50%) were prepared by diluting nitrogen gas in pure hydrogen sulfide. It was observed that degradation of NTA for 10% and 50% hydrogen sulfide concentrations is less than 20% and slightly higher for 100% concentration as shown in Figure 3. This indicates that the Fe-NTA solution with optimized condition may suitable for removal of hydrogen sulfide from gases containing low to high concentrations.
Effect of antioxidant on degradation of NTA for oxidative absorption of hydrogen sulfide
Formation of the hydroxyl ion during redox reaction of Fe-NTA may be responsible for the degradation of NTA. A number of additives that function as hydroxyl scavengers have been used to slow down or prevent radical-induced oxidative degradation. The added material reacts with hydroxyl ions or free radicals, thus preventing them from attacking, hydroxylation, and thus degrading the organic ligand. There are many such antioxidants that may be used viz. ethylene glycol, potassium citrate, sodium thiocynate, 1-butanol and sodium thiosulphate as antioxidants.
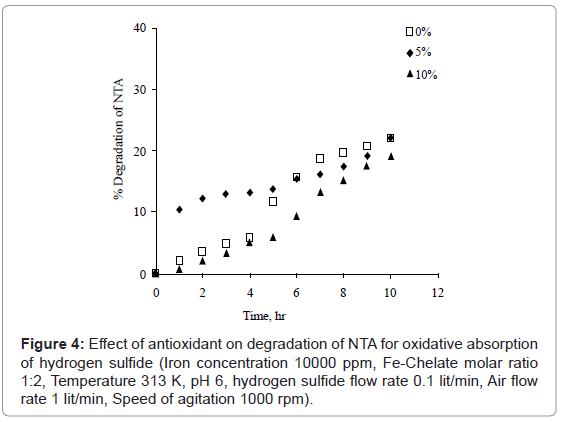
Chen et al. [2] found 1M NaCl as well as 5% (mol %) of sodium thiosulphate may greatly reduce the degradation of NTA. Hence, sodium thiosulphate was used as an antioxidant in order to study degradation of NTA for oxidative absorption of hydrogen sulfide. The effect of various sodium thiosulphate concentrations viz. 0%, 5% and 10% (mol% based on NTA concentration) was studied with optimum conditions. It was observed that the addition of sodium thiosulphate minimizes the degradation of NTA as shown in figure 4. It is found that 10% (mol %) concentration of sodium thiosulphate shows minimum degradation of NTA and is optimum concentration.
Kinetics of NTA degradation for oxidative absorption of hydrogen sulfide
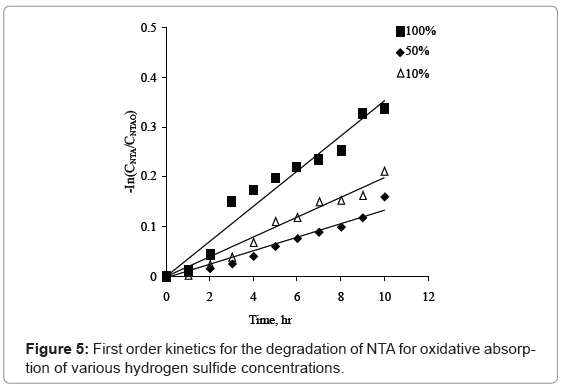
Wubs and Beenackers [1] had found that reaction of hydrogen sulfide is first order in hydroxyl ferric chelate. The kinetic experimental data was obtained for degradation of NTA at different hydrogen sulfide concentrations range from 10% to 100%. The reported data was analyzed to identify the order of the reaction. The regression coefficient values for zero order, first order and second order reaction at different hydrogen sulfide were in the range of 0.94 to 0.97, 0.95 to 0.97 and 0.94 to 0.96 respectively as shown in figure 5. The reaction order with respect to NTA is found to be first order as it is best fitting for all of the three hydrogen sulfide concentrations. Rate constant values for 100%, 50% and 10% hydrogen sulfide concentration are 0.035 hr-1, 0.013 hr-1 and 0.019 hr-1 respectively.
Conclusion
The oxidative absorption of hydrogen sulfide in Fe-chelate was carried out in stirred cell glass reactor with four blade turbine impeller and four leg baffle. NTA was found as best suitable chelate compared to EDTA and DTPA for oxidative absorption of hydrogen sulfide. Also, the degradation of NTA was minimized using 10% (mol %) concentration of sodium thiosulphate. The order of the reaction obtained from kinetic experimental data is first order in Fe-NTA. This process is applicable for desulfurization of gases containing low to high concentration of hydrogen sulfide.
Acknowledgements
We are grateful to the University Grants Commission, India for providing financial support to this research work.
References
- Wubs HJ, Beenackers AACM (1994)Kinetics of H2S absorption into aqueous ferric solutions of EDTA and HEDTA. AIChE Journal 40: 433-444.
- Chen D, Martell AE, McManus D (1995) Studies on the mechanism of chelate degradation in iron-based, liquid redox H2S removal processes. Can J Chem 73: 264-274.
- Neumann DW, Lynn S (1984) Oxidative absorption of H2S and O2 by iron chelate solutions. AIChE Journal 30: 62-69.
- Piche S, Ribeirs N, Bacaoui A, Larachi F (2005) Assessment of redox alkaline/iron Chelate absorption process for the removal of H2S in air emission. Chem Eng Science 60: 6452- 6461.
- Horikawa MS, Rossi F, Gimenes ML, Costa CMM, da Silva MGC (2004) Chemical absorption of H2S for biogas purification. Braz J Chem Eng 21: 415-422.
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 20279
- [From(publication date):
July-2012 - Dec 20, 2025] - Breakdown by view type
- HTML page views : 15001
- PDF downloads : 5278