Research Article Open Access
Potential Effect of Two Bacillus spp on Decolorization of Azo dye
| Maulin P Shah*, Patel KA, Nair SS, Darji AM | |
| Industrial Waste Water Research Laboratory, Division of Applied and Environmental Microbiology Lab, Enviro Technology Limited, Plot No: 2413/14, GIDC, Ankleshwar-393002, Gujarat, India | |
| Corresponding Author : | Maulin P Shah Applied and Environmental Microbiology Lab Enviro Technology, Limited (CETP) Ankleshwar- 393 002, Gujarat, India Tel: +91-90 999 65504 Fax No: +91-2646-250707 E-mail: shahmp@uniphos.com |
| Received July 15, 2013; Accepted August 20, 2013; Published August 22, 2013 | |
| Citation: Shah MP, Patel KA, Nair SS, Darji AM (2013) Potential Effect of Two Bacillus spp on Decolorization of Azo dye. J Bioremed Biodeg 4:199. doi: 10.4172/2155-6199.1000199 | |
| Copyright: © 2013 Shah MP, et al. This is an open-a ccess article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Related article at Pubmed Pubmed  Scholar Google Scholar Google |
|
Visit for more related articles at Journal of Bioremediation & Biodegradation
Abstract
Decolorization of azo dye by two bacterial species Bacillus megaterium and Bacillus cereus has been analyzed using mineral effluent, consisting of known concentration of the dye in ZZ medium. Physico chemical parameters such as carbon and nitrogen source, temperature, pH and inoculum volume are optimized for the decolorization process by changing one parameter at a time. Optimal condition for Bacillus cereus was found to be 1% sucrose, 0.25% peptone, pH 7, temperature 37°C and 8% inoculum and that for Bacillus megaterium was found to be glucose 1%, 0.25% yeast extract, pH 6, temperature 37°C and 10% inoculum. Extent of decolorization recorded by Bacillus cereus under ideal conditions was 95% and that by Bacillus megaterium was 98%. The study has confirmed the potential of Bacillus cereus and Bacillus megaterium in the decolorization of Azo dye and opened scope for future analysis of their performance in the treatment of textile effluent.
| Keywords |
| Azo dye; Bacillus; Decolorization; Inoculum |
| Introduction |
| Rapid industrialization has necessitated the manufacture and use of different chemicals in day to day life [1,2]. The textile industry is one of them which extensively use synthetic chemicals as dyes. Wastewaters from textile industries pose a threat to the environment, as large amount of chemically different dyes are used. A significant proportion of these dyes enter the environment via wastewater [1]. Approximately 10,000 different dyes and pigments are used industrially and over 0.7 million tons of synthetic dyes are produced annually, worldwide [3]. Pollution due to textile industry effluent has increased during recent years. Moreover, it is very difficult to treat textile industry effluents because of their high BOD, COD, heat, color, pH and the presence of metal ions [4]. The textile finishing generates a large amount of waste water containing dyes and represents one of the largest causes of water pollution [5], as 10-15% of dyes are lost in the effluent during the dyeing process [6]. The new closed-loop technologies such as the reuse of microbial or enzymatic treatment of dyeing effluents could help reducing this enormous water pollution [7]. Azo dyes have been used increasingly in industries because of their ease and cost effectiveness in synthesis compared to natural dyes. However, most azo dyes are toxic, carcinogenic and mutagenic [8]. Azo bonds present in these compounds are resistant to breakdown, with the potential for the persistence and accumulation in the environment [9]. However, they can be degraded by bacteria under aerobic and anaerobic conditions [10]. Several physico-chemical techniques have been proposed for treatment of colored textile effluents. These include adsorption on different materials, oxidation and precipitation by Fenton’s reagent, bleaching with chloride or ozone photo degradation or membrane filtration [11]. All these physical or chemical methods are very expensive and result in the production of large amounts of sludge, which creates the secondary level of land pollution. Therefore, economic and safe removal of the polluting dyes is still an important issue. Bioremediation through microorganisms has been identified as a cost effective and environment friendly alternative for disposal of textile effluent [12,13]. In recent years a number of studies have focused on some microorganisms capable of degrading and absorbing dyes from wastewater. A wide variety of microorganisms are reported to be capable of decolonization of dyes [12,14-26]. The current study has evaluated the potential of two bacterial strains isolated from textile effluent for their decolorization efficiency of the textile dye, under “in vitro” conditions and optimization of the factors influencing the process. |
| Materials and Methods |
| Textile dye azo red and effluent sample were collected from a dying industry located at Ankleshwar, GIDC. The sample was collected from the effluent disposal site of the industry. All microbiological media and medium ingredients were purchased from Hi-Media Laboratories (Mumbai, India). The dye decolorizing bacteria were isolated from the effluent by serial dilution and plating appropriate dilutions on modified Zhou and Zimmermann (ZZ) agar medium containing (yeast extract-5 gL-1, glucose-5 gL-1, (NH4)2 SO4-0.5 gL-1, KH2PO4-2.66 gL-1, Na2HPO4-4.32 gL-1, agar-20 gL-1 and azo dye - 100 mgL-1) and pH 7.0. All the isolated colonies were studied by inoculating them in effluent basal medium containing (yeast extract-5 gL-1, (NH4)2 SO4-0.5 gL-1, KH2PO4-2.66 gL-1, Na2HPO4- 4.32 gL-1, glucose-5 gL-1 and azo dye- 100 mgL-1) at pH 7.0. The inoculated medium was incubated at 30°C under shaking culture conditions. Dye degrading isolates were identified on the basis of morphological and biochemical tests according to Bergey’s Manual of Systematic Bacteriology [27]. The isolates showing more decolorization of the azo dye selected for further studies. The dye decolorizing activity of bacteria was evaluated using a modified method of Arun Prasad and Bhaskara Rao [28]. Decolorization was performed in 100 ml of ZZ medium containing 0.02g of azo dye [called as mineral effluent hereafter] and 10% (v/v) inoculum of each isolate separately. Mineral effluent without inoculums served as control. Inoculated medium and control were incubated at 30°C for four days under shake culture conditions. About 2 ml of the samples aseptically from experimental and control media and centrifuged at 7,500 RPM for 15 minutes. The clear supernatant was used for measuring absorption at 600 spectrophotometer (Shimadzu UV-1800, Japan). The percent decolorization of the mineral effluent was determined by the following formula: |
| D=[(A0-A1) /A0]×100 |
| Where, D=% of decolorization; A0=initial absorbance; A1=Final absorbance |
| Decolorization of azo dye by the bacterial isolates was optimized with respect to the effect of carbon source (sucrose, glucose, mesoinositol), nitrogen source (Beef extract, peptone, yeast extract), temperature, pH and inoculum volume Decolourization under different culture conditions was done by changing the factors one at a time, The basic conditions of culture being 30°C, pH 7.0 under shaking conditions (130 RPM), 10%, (v/v) inoculum in mineral effluent. Duration of the experiment was for four days. In all the exp effluent without culture inoculum was served as control four days the samples were withdrawn and analyzed for percent decolorization of the dye. The time course of decolorization was carried out under optimum conditions obtained from above studies and the optimum conditions are: for Bacillus cereus (1% sucrose, 0.25% peptone, pH 7, 37°C and 8% inoculums), for B. megaterium (1% glucose, 0.25% yeast extract, pH 6, 37°C and 10% inoculum). Flasks were incubated up to 96 h at their respective temperature and samples were removed after every 24 h and analyzed for decolorization activity as described above. |
| Results and Discussion |
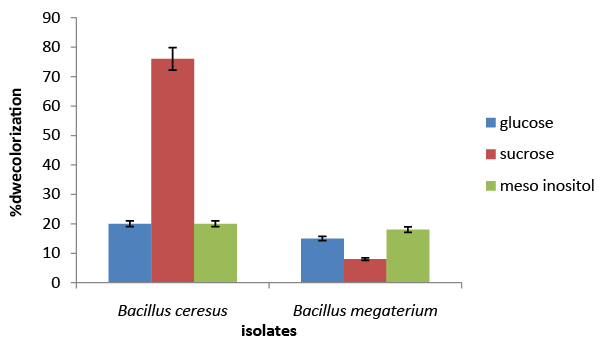
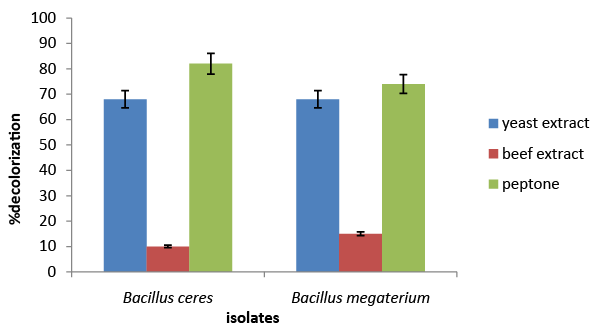
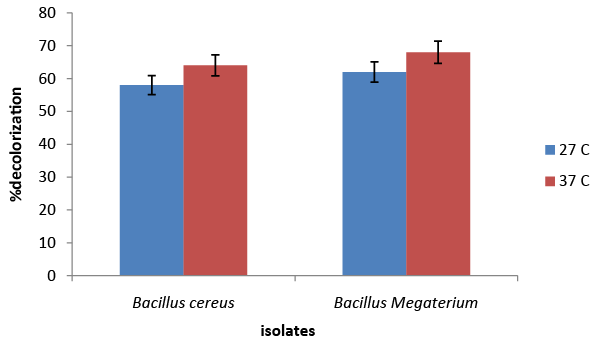
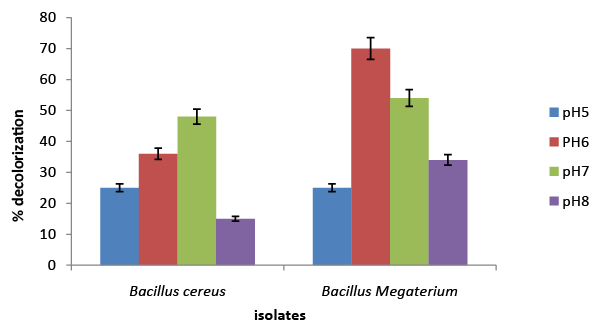
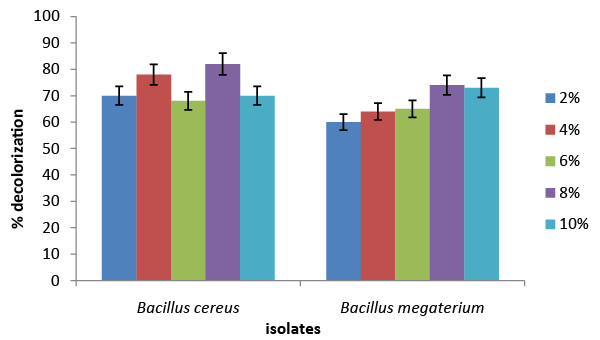
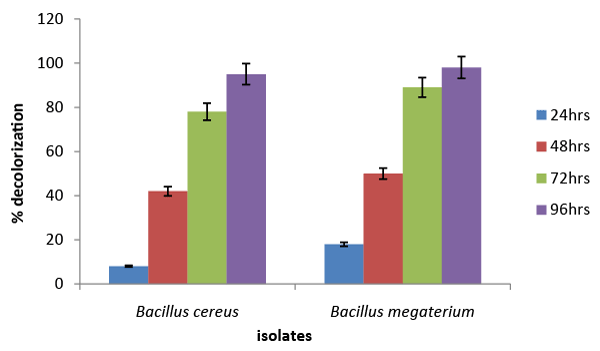
| The results of the screening and identification of bacteria isolated from the effluent samples confirmed by using standard microbiological procedures, API 20E and API 20NE. Figure 1 illustrate the effect of different carbon sources on decolorization of azo dye by Bacillus cereus and Bacillus megaterium. Percentage decolorization of the dye recorded for Bacillus cereus when glucose was used as carbon source was 20% and that for Bacillus megaterium was 15%. The percentage of decolorization of azo dye with sucrose as carbon source was found to be 76% and 8%, respectively for B. cereus and Bacillus megaterium. When meso-inositol was used as carbon source 20% and 18% decolorization of the dye was recorded in cultures of Bacillus cereus and Bacillus megaterium, respectively. From the above data it is observed that Bacillus cereus is more efficient in decolorizing azo dye than Bacillus megaterium and sucrose is the ideal carbon source for its activity under “in vitro” condition. Figure 2 illustrates the effect of different nitrogen sources on decolorization of azo dye by Bacillus cereus and Bacillus megaterium. Percentage decolorization of the dye recorded for Bacillus cereus when beef extract was used as nitrogen source was 10% and that for Bacillus megaterium was 15%. The percentage of decolorization of azo dye with peptone as nitrogen source was found to be 82% and 74%, respectively for Bacillus cereus and Bacillus megaterium. When the yeast extract was used as nitrogen source 68% decolorization of the dye was recorded with both the bacterial species. From the above data it is observed that Bacillus cereus is more efficient in decolorizing azo dye than Bacillus megaterium and peptone is the ideal nitrogen source for its activity under in vitro condition. Figure 3 illustrates the effect of different temperature on decolorization of azo dye by Bacillus cereus and Bacillus megaterium. Percentage decolorization of the dye recorded for Bacillus cereus at 27°C was 58% and that for Bacillus megaterium was 62%. The percentage of decolorization of azo dye at 37°C was found to be 64% and 68%, respectively for Bacillus cereus and Bacillus megaterium. From the above data it is observed that Bacillus megaterium is more efficient in decolorizing azo dye than Bacillus cereus and 37°C is the ideal temperature for its activity under “in vitro” condition. Figure 4 illustrates the effect of different pH on decolorization of azo dye by Bacillus cereus and Bacillus megaterium. Percentage decolorization of the dye recorded for both Bacillus cereus and Bacillus megaterium at pH 5 was 25%. Decolorization of azo dye at pH 6 was found to be 36% and 70%, respectively for Bacillus cereus and Bacillus megaterium. The percentage of decolorization of azo dye at pH 7 was found to be 48% and 54%, respectively for B. cereus and Bacillus megaterium. When the cultures were maintained at pH 8, 15% and 34% decolorization of the dye was recorded in cultures of Bacillus cereus and Bacillus megaterium, respectively. From the above data it can be inferred that Bacillus megaterium is more efficient in decolorizing azo dye than Bacillus cereus and pH 6 is the ideal for its activity under “in vitro” condition. Figure 5 illustrates the effect of different volume of inoculums on decolorization of azo dye by Bacillus cereus and Bacillus megaterium. Percentage decolorization of the dye recorded for B. cereus for 2% inoculum was 70% and that for Bacillus megaterium was 60%. The percentage decolorization of azo dye with 4% inoculum was found to be 78% and 64%, respectively for Bacillus cereus and Bacillus megaterium. The percentage decolorization of azo dye with 6% inoculum was found to be 68% and 65%, respectively for Bacillus cereus and Bacillus megaterium. The percentage decolorization of the dye recorded for Bacillus cereus with 8% inoculum was 82% and that for Bacillus megaterium was 74%. When 10% inoculums were used 70% and 73% decolorization of the dye was recorded in cultures of Bacillus cereus and Bacillus megaterium, respectively. From the above data it is observed that Bacillus cereus is more efficient in decolorizing azo dye than Bacillus megaterium and 8% inoculum is the ideal volume for its activity under “in vitro” condition. Figure 6 illustrates the Time course of dye decolorization of azo dye under optimum conditions by Bacillus cereus and Bacillus megaterium. Percentage decolorization of the dye recorded for Bacillus cereus was 95% at 1% sucrose 0.25% peptone, pH 7, 37°C and 8% inoculum and that for Bacillus megaterium was 98% at glucose 1%, 0.25% yeast extract, pH 6, 37°C and 10% inoculum. The outcomes of this experiment indicated that Bacillus megaterium performed the decolorization process to a better extent than Bacillus cereus under a combination of the ideal levels of all factors influencing the process. However, both the species of bacteria can be inferred as good agents for the degradation of azo dye red. Azo dyes represent one of the recalcitrant chemicals not being degraded by conventional effluent treatment processes [29]. Bacteria have been recognized as an important and efficient agent for the degradation and decolorization of textile dyes [10-12]. Bacillus cereus has been reported to decolorize different azo dyes from textile effluent. Modi et al. [30] have reported maltose and peptone as the ideal carbon and nitrogen sources, respectively for efficient decolorization of Reactive Red 195 by B. cereus, recording 97% reduction in color of the dye in the effluent. In another study Ola et al. [31], have reported decolorization of Cibacron red P4B and Cibacron black PSG to the levels 81% and 75%, respectively by this species. Further the study [31] has reported requirement of different carbon and nitrogen sources for maximum decolorization of the two dyes. The current study has revealed 95% decolorization of azo dye red with sucrose and peptone as ideal carbon and nitrogen sources, respectively. From the above outcomes of different studies, it can be inferred that the metabolic flux of B. cereus alters with the type of carbon and nitrogen sources available in the surrounding environment and efficiency of decolorization of the azo dyes is greatly dependent on these factors. Degradation and decolorization of the wide range of azo dyes has been reported by different researchers [32-35] who confirmed the role of the bacterial enzyme azoreductase in cleavage of the dye leading to its decolorization. In another study Tripathi and Srivastava [29] have confirmed 94.4% decolorization of another azo dye, orange G by Bacillus megaterium and predicted decolorization at ideal conditions using statistical tools, Central Composite Design (CCD). The current study has demonstrated the decolorization of azo dye red by this species and the level of decolorization was found to be 98% at optimum conditions of key parameters analyzed. Therefore, it can be concluded that Bacillus megaterium is a highly potential bacterial species capable of degrading and decolorizing wide range of azo dyes. |
| Conclusion |
| Current investigation has confirmed the decolorization of Azo dye red by the bacteria B. cereus and Bacillus megaterium under “in vitro” conditions. Extent of decolorization recorded by Bacillus cereus under ideal conditions was 95% and that by Bacillus megaterium was 98%. Thus the study has confirmed the potential of B. cereus and Bacillus megaterium in the decolorization of the dye indicating their possible application for treatment of textile effluents. |
| Acknowledgements |
| Authors are highly grateful to the management of Enviro Technology Limited., Ankleshwar, Gujarat, India for allowing us to carry out such a noble work for the sustainable environment. |
References
|
Figures at a glance
 |
 |
 |
| Figure 1 | Figure 2 | Figure 3 |
 |
 |
 |
| Figure 4 | Figure 5 | Figure 6 |
Relevant Topics
- Anaerobic Biodegradation
- Biodegradable Balloons
- Biodegradable Confetti
- Biodegradable Diapers
- Biodegradable Plastics
- Biodegradable Sunscreen
- Biodegradation
- Bioremediation Bacteria
- Bioremediation Oil Spills
- Bioremediation Plants
- Bioremediation Products
- Ex Situ Bioremediation
- Heavy Metal Bioremediation
- In Situ Bioremediation
- Mycoremediation
- Non Biodegradable
- Phytoremediation
- Sewage Water Treatment
- Soil Bioremediation
- Types of Upwelling
- Waste Degredation
- Xenobiotics
Recommended Journals
Article Tools
Article Usage
- Total views: 16038
- [From(publication date):
August-2013 - Dec 21, 2025] - Breakdown by view type
- HTML page views : 11171
- PDF downloads : 4867
