Review Article Open Access
Role of Rhizobacteria in Reduction of Arsenic Uptake by Plants: A Review
| Suresh Jaiswal* | |
| Departrment of Medical Lab Science, School of Health and Allied Sciences, Pokhara University, Lekhnath-12, Kaski, Nepal | |
| Corresponding Author : | Dr. Suresh Jaiswal Lecturer, Department of Medical Lab Science School of Health and Allied Sciences, Pokhara University Lekhnath-12, Kaski, Nepal Tel: 00977 9843019151 E-mail: suuress@gmail.com |
| Received May 30, 2011; Accepted September 08, 2011; Published September 24, 2011 | |
| Citation: Jaiswal S (2011) Role of Rhizobacteria in Reduction of Arsenic Uptake by Plants: A Review. J Bioremed Biodegrad 2:126. doi: 10.4172/2155-6199.1000126 | |
| Copyright: © 2011 Jaiswal S. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Related article at Pubmed Pubmed  Scholar Google Scholar Google |
|
Visit for more related articles at Journal of Bioremediation & Biodegradation
Abstract
Heavy metal pollution of soil is a significant environmental problem and has its negative impact on human health and agriculture. Arsenic (As) distribution and toxicology in the environment is the serious issue worldwide. Rice is the major staple food for people in West Bengal and Bangladesh, is the biggest sufferers of arsenic contamination, and so protecting crops is of major importance and serious issues at these places. Bioremediation is the application of living organisms or products, or the application of biological principles, to ameliorate adverse environmental condition. Rhizosphere, as an important interface of soil and plant, plays a significant role in phytoremediation of contaminated soil by heavy metals, in which, microbial populations are known to affect heavy metal mobility and availability to the plant through release of chelating agents, acidification, phosphate solubilization and redox changes, and therefore, have potential to enhance phytoremediation processes. Phytoremediation strategies with appropriate heavy metal-adapted Rhizobacteria have received more and more attention. Microbes may influence our needs directly or indirectly. Directly by their detoxification potentials and may be the source of bioremediation and genetic engineering. Their exploitation in arsenic remediation would be simplest phenomenon to protect crops from arsenic, as it blocks the entry of heavy metals. This article deals with the identification, characterization and bioremediation advances in effect and significance of As-resistant Rhizobacteria in phytoremediation of heavy metal contaminated soils to prevent the transport arsenic metal from root to shoot to prevent the uptake by plants. There is also a need to improve our understanding of the mechanisms involved in the transfer and mobilization of heavy metals by Rhizobacteria and to conduct research on the selection of microbial isolates from rhizosphere of plants growing on heavy metal contaminated soils for specific restoration programmes.
| Keywords |
| Rhizobacteria; Phytoremediation; Arsenic; Mobilization |
| Introduction |
| Arsenic is a toxic metalloid of global concern. It is usually originated geogenically but can be intensified by human activities such as applications of pesticides and wood preservatives, mining and smelting operations, and coal combustion [1]. Consequentially, elevated levels of arsenic have been reported in soils and groundwater worldwide. The maximum concentration limit (MCL) recommended for drinking water by the World Health Organization (WHO) is 10 mg/L. Elevated levels of arsenic in drinking water can seriously impact human health and have been implicated in human diseases and mortality [2]. Around 6 million people in West Bengal and more than 46 million people in Bangladesh are estimated to be at risk from drinking water with arsenic above 50 mg/L [1,3]. |
| Remediation of arsenic contaminated soils and groundwater is necessary for providing safe drinking water. Bioremediation of arsenic contaminated soils and groundwater shows a great potential for future developments due to its environmental compatibility and possible cost-effectiveness. It relies on microbial activity to reduce, mobilize, or immobilize arsenic through sorption, biomethylation, complexation, and oxidation-reduction processes. Microbially mediated redox reactions involving organic carbon, Fe, Mn, and S are the basic underlying mechanisms affecting arsenic mobility. Microorganisms have evolved biochemical mechanisms to exploit arsenic oxyanions, either as an electron acceptor [e.g., As (V)] for anaerobic respiration, or as an electron donor [e.g., As(III)] to support chemoautotrophic fixation of carbon dioxide (CO2) into cell carbon [4-6]. A number of investigations have been performed to remediate arsenic contaminated soils and groundwater using biologically based methods. |
| Natural sources |
| Long before man’s activities had any effect on the balance of nature, arsenic was distributed ubiquitously throughout earth crusts, soil, sediments, water, air and living organisms. Arsenic naturally occurs in over 200 different mineral forms, of which approximately 60% are arsenates, 20% sulfides and sulfosalts and the remaining 20% includes arsenides, arsenites, oxides, silicates and elemental arsenic (As). The levels of arsenic in the soils of various countries are said to range from 0.1 to 40 mg kg−1 (mean 6 mg kg−1), 1 to 50 mg kg−1 (mean 6 mg kg−1) and mean 5 mg kg−1, but varies considerably among geographic regions [7]. Arsenic is present in soils in higher concentrations than those in rocks. The contents of arsenic in the soils of various countries are shown in (Table1) [8]. |
| Anthropogenic sources |
| These exceed natural sources in the environment by 3:1 [9]. Man in his utilization of natural resources releases arsenic into the air, water and soil. These emissions can ultimately affect residue levels in plants and animals. Arsenic may accumulate in soil through use of arsenical pesticides, application of fertilizers, and dust from the burning of fossil fuels, medicines, disposal of industrial, metallurgy and animal wastes. |
| Arsenic toxicity |
| Arsenic is a carcinogen, and is associated with animal and human skin, lung and bladder cancers [10]. The WHO agrees and says that over the next decade these cancers are likely to become the principle human health concern due to As. Both As(III) and As(V) are toxic, as such inorganic arsenic is regarded as a major environmental pollutant based on USEPA’s evaluation [11]. However, they interrupt biological functions in different manner. Inorganic arsenic arsenite binds to proteins with sulfhydryl groups, interfering with their functions. It inhibits respiration by binding to vicinal thiols in pyruvate dehydrogenase and 2-oxo-glutarate dehydrogenase. Arsenite does not act directly as a mutagen but induces intrachromosomal homologous recombination [12] and generates reactive oxygen species. As(V), on the other hand, interferes with oxidative phosphorylation and ATP synthesis [13]. |
| A 2007 case study also estimated that over 137 million people in probably 70 countries are associated with charactersticks of As poisoning (Associated Press, 2007). |
| Remediation technologies |
|
Heavy metals cannot be destroyed biologically (no “degradation”) change in the nuclear structure of the element, occurs) but are only transformed from one oxidation state or organic complex to another remediation of heavy metal contamination in soils is more difficult. Until now, methods used for their remediation such as excavation and land fill, thermal treatment, acid leaching and electroreclamation are not suitable for practical applications, because of their high cost, low efficiency, large destruction of soil structure and fertility and high dependence on the contaminants of concern, soil properties, site conditions, and so on. Thus, the development of phytoremediation strategies for heavy metals contaminated soils is necessary [14]. |
|
Effect of Rhizobacteria on phytoremediation |
| Plant-growth: Rhizosphere microorganisms, which are closely associated with roots, have been termed plant growth promoting Rhizobacteria (PGPR) [15]. Plant growth-promoting Rhizobacteria include a diverse group of free-living soil bacteria that can improve host plant growth and development in heavy metal contaminated soils by mitigating toxic effects of heavy metals on the plants shows effects of variations of PGPR on plant. Rhizosphere microorganisms, which are closely associated with roots, have been termed plant growth promoting Rhizobacteria (PGPR) [15]. Plant growth-promoting Rhizobacteria include a diverse group of free-living soil bacteria that can improve host plant growth and development in heavy metal contaminated soils by mitigating toxic effects of heavy metals on the plants shows effects of variations of PGPR on plant. |
| Arsenic remediation by bacteria |
| Plant growth promoting Rhizobacteria combat heavy-metal stress: Accumulation of heavy metals in the soil environment and their uptake by both plant growth promoting Rhizobacteria and plants is a matter of growing environmental concern. Unlike many other pollutants, which can undergo biodegradation and produce less toxic, less mobile and/ or less bio-available products, heavy metals are difficult to be removed from contaminated environment. These metals cannot be degraded biologically, and are ultimately indestructible, though the speciation and bioavailability of metals may change with variation in the environmental factors. |
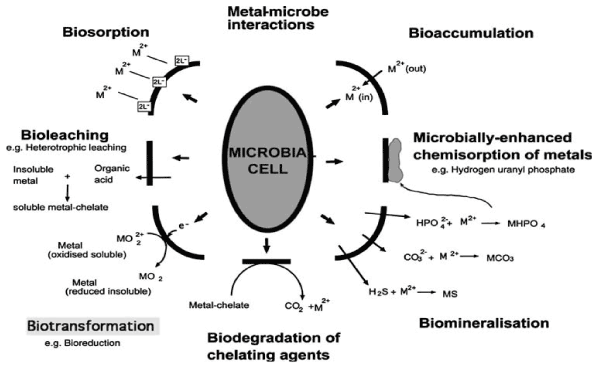
| The ability to grow even at high metal concentration is found in many rhizospheric microorganisms including symbiotic N2 fixing bacteria [16] and may be the result of intrinsic or induced mechanism [17] (Figure 1). Tolerance may be defined as the ability to cope with metal toxicity by means of intrinsic properties of the microorganisms, while resistance is the ability of microbes to survive in higher concentrations of toxic metals by detoxification mechanisms, activated in direct response to the presence of heavy metals. Toxic heavy metals therefore, need to be either completely removed from the contaminated soil, transformed or to be immobilized, producing much less or non-toxic species. However, in order to survive and proliferate in metal contaminated soils, tolerance has to be present both in microbes and their associative hosts. For survival under metal-stressed environment, plant growth promoting Rhizobacteria have evolved several mechanisms by which they can immobilize, mobilize or transform metals rendering them inactive to tolerate the uptake of heavy metal ions [18]. |
| These mechanisms include: |
| 1. Exclusion—the metal ions are kept away from the target Sites. |
| 2. Extrusion—the metals are pushed out of the cell through chromosomal/plasmid mediated. |
| 3. Accommodation —metals form complex with the metal binding proteins (e.g. metallothienins, a low molecular weight proteins) or other cell components |
| 4. Bio-transformation—toxic metal is reduced to less toxic forms and |
| 5. Methylation and demethylation. |
| The strategies might help to encounter the detrimental effect of AS: |
| • Removal of As from the environment. |
| • The development of safe crops that can be grown in the presence of As contamination. |
| • Reduce transport of arsenic from root to shoot in plants by the help of Rhizobacteria. |
| Arsenate (As(V)) is taken up by phosphate transporters, and As(III) is taken up by aquaglyceroporins (GlpF in E. coli, Fps1p in yeast and Aqp7 and Aqp9 in mammals) (Figure 2). In both E. coli and S. cerevisiae, arsenate is reduced to arsenite by the bacterial ArsC or yeast Acr2p enzymes. In both organisms, glutathione and glutaredoxin serve as the source of reducing potential. The proteins responsible for arsenate uptake and reduction in mammals have not yet been identified. In E. coli, arsenite is extruded from the cells by ArsB alone or by the ArsAB ATPase. In yeast Acr3p is a plasma membrane arsenite e\ux protein, and Ycf1p, which is a member of the MRP family of the ABC superfamily of drugresistance pumps, transports As(GS)3 into the vacuole. In mammals Mrp isoforms pump As(GS)3 out of cells. For example, Mrp2 extrudes As(III) into bile [20,21]. |
| Methodologies to reduce the uptake of arsenic metal by Rhizobacteria |
| For the study we can aim the work on three strategies. |
| A) As – microbe interaction. |
| B) As – microbe in plant growth promoting (Determination of Plant Growth Promoting characters for the fulfillment of (PGPB). |
| C) As – microbe – plant interaction (Study of metal tolerance in the rice plant in the presence and absence of As – microbe and their role in the plant growth promoting and reduce the uptake of arsenic by plants). |
| A) Arsenic - microbe interaction: |
| 1. Isolation of arsenic tolerant bacteria from rhizospheric regions of rice roots from arsenic contaminated areas from West Bengal. |
| 2. Determination of minimum inhibitory concentration, |
| 3. Characterization of the isolates (Biochemical and antibiotic seB) As – microbe (PGPR) in plant growth promoting:nsitivity behavior ), |
| 4. Heavy metal tolerance, |
| 5. Study of growth pattern. |
| B) As – microbe (PGPR) in plant growth promoting: |
| 1. Determination of ammonia production by the isolates, |
| 2. Determination of indole acetic acid production by the isolates, |
| 3. Determination catalase production test by the isolates, |
| 4. Determination of phosphate solubilization test by the isolates, |
| 5. Determination of HCN production test by the isolates, |
| 6. Determination of siderophore production test by the isolates. |
| C) As – microbe – plant interaction: |
| 1. Growth of rice plant (Oryza Sativa) under hydroponic culture system. |
| 2. To study the effect of different concentration of arsenic on the rice plant in the presence of As-microbes and their role in plant growth promoting. |
| 3. To study the role As – microbes in the tolerance of metal by the plants |
| Summary and Conclusion |
| Aim A |
| Arsenic is a toxic metal in biosphere and frequently reported to be an environment pollutant. The need of the hour is to enhance arsenic remediation by different external and internal inputs. Global cycling is affected by microorganism. The biogeochemical cycling of arsenic is significantly dependent but microbial transformation and immobilization which affect the distribution of mobility of arsenic species in the environment. Interaction among metals, microbes and plants have attracted attention because of biotechnological potential of microorganisms of metal removal directly from polluted soils or possible transfer of accumulated metals to higher plants and toxicity of metals towards microbial metabolism and growth. Further knowledge about the mechanism involved for arsenic detoxification or resistance will provide us with knowledge about the isolates in the role of bioremediation or in plant growth promotion in arsenic stress condition or its use in genetic engineering. |
| Aim B |
| The strains which will be obtained from the current study PGPR demonstrate that the application of plant growth promoting bacteria can be used for the enhancement of the plant growth promotion. These bacteria will show good effect in the increase of plant biomass, chlorophyll and their protein content. Comparative study of bacteria with PGPR and normal bacteria can be demonstrated for the study purpose. |
| Aim C |
| The present study to demonstrate that the application of arsenic tolerant bacteria having the character of plant growth promoting decrease as uptake by rice plants. The principal mechanism can be due to the biotransformation of arsenic metal and mobilization by as tolerant Rhizobacteria. |
References
|
Tables and Figures at a glance
| Table 1 |
Figures at a glance
 |
 |
 |
| Figure 1 | Figure 2 | Figure 3 |
Relevant Topics
- Anaerobic Biodegradation
- Biodegradable Balloons
- Biodegradable Confetti
- Biodegradable Diapers
- Biodegradable Plastics
- Biodegradable Sunscreen
- Biodegradation
- Bioremediation Bacteria
- Bioremediation Oil Spills
- Bioremediation Plants
- Bioremediation Products
- Ex Situ Bioremediation
- Heavy Metal Bioremediation
- In Situ Bioremediation
- Mycoremediation
- Non Biodegradable
- Phytoremediation
- Sewage Water Treatment
- Soil Bioremediation
- Types of Upwelling
- Waste Degredation
- Xenobiotics
Recommended Journals
Article Tools
Article Usage
- Total views: 15900
- [From(publication date):
September-2011 - Nov 20, 2025] - Breakdown by view type
- HTML page views : 10887
- PDF downloads : 5013
