Research Article Open Access
Thermostability Comparison of Two Solid States of Cefpiramide
Ming-juan Wang1, Wen-bo Zou1, Jing Xue1, Yan Wang1, Chang-qin Hu1*, Jos Hoogmartens2, Ann Van Schepdael2 and Erwin Adams2
1Department of Antibiotics, National Institutes for Food and Drug control, Beijing, P.R. China
2Katholieke Universiteit Leuven, Faculteit Farmaceutische Wetenschappen, Laboratorium voor Farmaceutische Analyse, Belgium
- *Corresponding Author:
- Chang-qin Hu
Department of Antibiotics
National Institutes for Food and Drug control
Beijing 100050, P.R. China
Tel: +86 1067095308
E-mail: hucq@nicpbp.org.cn
Received date: July 09, 2012; Accepted date: August 21, 2012; Published date: August 28, 2012
Citation: Wang Mj, Zou Wb, Xue J, Wang Y, Hu Cq, et al. (2012) Thermostability Comparison of Two Solid States of Cefpiramide. J Anal Bioanal Tech 3:142. doi: 10.4172/2155-9872.1000142
Copyright: © 2012 Wang Mj, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Analytical & Bioanalytical Techniques
Abstract
Cefpiramide is susceptible to many factors, including high temperature. Two solid states were observed in cefpiramide: a crystalline form and an amorphous one, and both the accelerated (40°C ± 2°C /75% ± 5% relative humidity (RH)) and stress (60°C ± 2°C / 75% ± 5% RH) stability study demonstrated that the crystalline form of cefpiramide was much more stable than its amorphous form, with related substances, high molecular mass polymers and assay as critical stability indicating parameters. Thermogravimetric analysis (TGA), differential scanning calorimetry (DSC) and water content results indicated that each molecule of crystalline cefpiramide may contain two molecules of crystal water, which can explain its high thermal stability. This study provides valuable information for cefpiramide and its drug products with respect to the improvement of its thermal stability through crystallization.
Keywords
Cefpiramide; Polymorphism; Thermostability; Stability study
Introduction
Cefpiramide (Figure 1), a semi-synthetic, third-generation cephalosporin, exhibits antibacterial activity against Gram-positive and Gram-negative bacteria, particularly Pseudomonas aeruginosa, which is resistant to several other antibiotics. It was originally produced by Sumitomo Pharmaceuticals (Tokyo, Japan) in the 1980s and used for the treatment of infections with glucose non-fermenting bacteria and community acquired infections. It functions by inhibiting bacterial cell wall biosynthesis via affinity for β-lactam-binding proteins [1-3].
Like other β-lactam antibiotics, cefpiramide is susceptible to hydrolysis across a wide range of pH values when in solution, especially under basic conditions, as observed in forced degradation tests. It is also sensitive to many other factors, such as high temperature, oxidation and light exposure. Therefore, to ensure the safety and efficiency of cefpiramide and its drug products, it is very important to design a proper package (e.g. protected from light) and recommend controlled storage conditions (e.g. storage temperature lower than 20°C) to minimize its degradation. As a consequence, it is required to perform stability studies to further diminish its risk in terms of safety and efficiency when administrated.
In many pharmaceuticals, polymorphism was observed (e.g. ceftezole sodium), and it was reported that the solid-state had an impact on its thermodynamic stability [4-11]. In our study, it was observed that cefpiramide had two solid state forms: crystalline and amorphous (Figure 2). With tests for related substances, high molecular mass polymers and assay of cefpiramide as critical stability indicating parameters, the stability of the two solid state forms of cefpiramide was compared under accelerated (40°C ± 2°C / 75% ± 5% relative humidity (RH)) and more stressed (60°C ± 2°C / 75% ± 5% RH) conditions to investigate the relationship between the solid state forms and thermodynamic stability. The test methods applied were all validated ones, described by the ministerial standard YBH05752008 (State Food and Drug Administration, P.R. China) [12]. The method for related substances was the same as that of the USP 35 [13].
Quality-by-design (QbD) has become an important paradigm in the pharmaceutical industry, which uses science and risk management approaches to gain product and process understanding and ultimately process control [14-19]. This study is expected to provide valuable information for the quality control of cefpiramide and its drug products.
Experimental Section
Reagents and samples
HPLC-grade methanol was obtained from Fisher Scientific (Fairlawn, NJ, USA). Analytical grade potassium dihydrogen phosphate (anhydrous), sodium hydroxide and anhydrous sodium carbonate were supplied by Sinopharm Chemical Reagent Co., Ltd (Beijing, China). Blue Dextran 2000 (Code No. 17-0360-01) was obtained from Pharmacia Biotech Company (Uppsala, Sweden). A milli-Q water purification system (Millipore, USA) was used to further purify glassdistilled water.
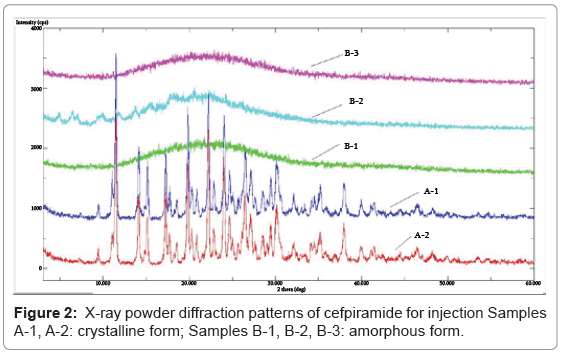
Cefpiramide (90.2%) and 1-methyl-1H-tetrazole-5-thiol (MTT) (99.0%) reference substances were provided by National Institutes for Food and Drug Control (Beijing, China). The crystalline samples of cefpiramide for injection (samples A-1, A-2) were obtained from Baiyunshan Pharmaceutical Company (Guangzhou, China) and the amorphous samples were from Ruiyang Pharmaceutical company (Shandong, China) (sample B-1), Zhongnuo Pharmaceutical Company (Hebei, China) (sample B-2) and Lukang Antibiotic Factory (Shandong, China) (sample B-3), respectively. The label contents of the above samples are all 1.0 g of cefpiramide per vial. Their X-ray powder diffraction (XRPD) patterns are shown in Figure 2.
Instrumentation
Assay and related substances tests were carried out on a Dionex Ultimate 3000 HPLC system (Sunnyvale, CA, USA) including ternary pump, an auto-sampler, a column heater and a diode array detector (DAD), using Chromeleon software (Dionex, CA, USA) for data acquisition.
Determination of high molecular mass polymers was performed on a SHIMADZU LC-10AT HPLC system (Kyoto, Japan) including a binary pumps, an auto-sampler and a UV detector (wavelength: 254 nm), using LC-solution software (Kyoto, Japan) for data acquisition.
The XRPD patterns were recorded on a Rigaku D/Max 2200 X-ray powder diffractometer equipped with a Cu Kα radiation tube (40 kV, 40 mV) and a scintillation counter (Rigaku Corporation, Tokyo, Japan). Thermogravimetric analysis (TGA) and differential scanning calorimetry (DSC) were performed on a Hi-Res TGA 2950 Thermogravimetric Analyzer and a DSC 2910 differential scanning calorimeter (TA instruments, New Castle, United States), respectively. Water contents were determined by a Mettler Toledo DL 18 titrator (Zurich, Switzerland).
Stability protocol
The stability protocol is shown in Table 1 In the protocol, related substances, high molecular mass polymers and assay were chosen as critical stability-indicating parameters due to the fact that the above parameters had great impact on the safety and efficiency of cefpiramide and were most likely to change during storage. At the beginning of the study (initially, t = 0 day), other tests including appearance, water content (Karl-Fischer titration), characterization of solid state forms (XRPD) and their thermostability properties (TGA, DSC) were also performed to provide other necessary information. Assay and related substances were evaluated at all time points. At the end of the accelerated and stress studies, XRPD was performed to evaluate the structural stability of the different forms, and high molecular mass polymers were also determined since β-lactam antibiotic polymerization is likely to occur under severe conditions (e.g. high temperature) and might cause anaphylaxis in clinical therapy [20-23].
| Study type | Storage conditions | Storage time (days) | |||||
| 0 | 10 | 20 | 30 | 40 | 60 | ||
| accelerated | 40 °C/75% RH | A | B | B | / | B | C |
| stress | 60 °C/75% RH | / | / | B | / | C | |
Table 1: Stability protocol for accelerated and stress storage of cefpiramide for injection
Test methods
The method for assay and related substances:The method for related substances was described by both the ministerial standard YBH05752008 and USP 35 [12,13]. In YBH05752008, the method for related substances was also applied to assay and for that purpose, the linearity in the concentration range of 0.125 ~1.25 mg×mL-1 was evaluated. The following condition was used:
The mobile phase consisted of methanol-30 mM potassium dihydrogen phosphate (pH 7.5, adjusted with 1 M sodium hydroxide) (22:78, v/v) at a flow rate of 0.8 mL.min-1. The XBridge Shield RP18 (size: 250×4.6 mm, 5 μm) (Waters Co., Dublin, Ireland) was maintained at 30°C. Sample solutions and reference solutions were prepared in mobile phase at a concentration of 0.5 mg.mL-1 and injected immediately (if stored at 4°C, they can be used for 4 hours). The injection volume was 20 μL. Ultraviolet detection was performed from 200 to 400 nm (extraction: 254 nm). Both analyses were performed as double determinations. The content of cefpiramide and the specified impurity MTT were determined by the external standard method, using peak areas for calibration, while the unspecified impurities were determined by calibration to cefpiramide (1.0% dilution), based on the assumption that their relative response factors to cefpiramide were 1.00. The assay results were calculated based on the label content (1.0 g of cefpiramide/ vial = 100%).
To verify the specificity of the method, the following forced degradation tests were performed to confirm the peak purities of cefpiramide and MTT under the above conditions by DAD analyses (acceptance criterion: no other peak (s) should be observed together with the target peak):
I. Acidic degradation:Transfer about 25 mg of cefpiramide reference substance to a 50 mL volumetric flask, add 1 mL of 1 M HCl, leave for 30 min at room temperature, then neutralize with 1 mL of 1 M NaOH and dilute to volume with mobile phase.
II. Basic degradation: Transfer about 25 mg of cefpiramide reference substance to a 50 mL volumetric flask, add 1 mL of 1 M NaOH, leave for 30 min at room temperature, then neutralize with 1 mL of 1 M HCl and dilute to volume with mobile phase.
III. Oxidative degradation: Transfer about 25 mg of cefpiramide reference substance to a 50 mL volumetric flask, add 0.5 mL of 30% (v/v) H2O2, leave for 30 min at room temperature, then dilute to volume with mobile phase.
IV. Thermal degradation: treat reference solution (0.5 mg×mL-1) at 60°C for 30 min (medium temperature) or at 95°C for 10 min (high temperature)
V. Light degradation: expose reference solution (0.5 mg×mL-1) to UV light (light intensities: 250-500 W/m2, according to ICH guideline) for 2 hours.
Limits of quantitation (LOQ) and limits of detection (LOD) of cefpiramide and MTT were also calculated to confirm the sensitivity of the method for related substances, based on the signal-to-noise ratios (S/NLOQ = 10, S/NLOD = 3) and further confirmed by replicate injection of the corresponding diluted reference solutions (acceptance criterion: the relative standard deviation (RSD) of cefpiramide/MTT peak areas is not more than 2.0%).
The method for high molecular mass polymers
I. Preparation of solutions:
(1) Blue Dextran 2000 solution: Dissolve 10 mg of Blue Dextran 2000 in water and dilute to 100.0 mL with water.
(2) Reference solution: Dissolve 10 mg of cefpiramide reference substance in 0.2 mL of 0.9% anhydrous sodium carbonate solution and dilute to 100.0 mL with water.
(3) Test solution: Dissolve 200 mg of the product to be examined in water and dilute to 10.0 mL with water. (Note: Sample solutions and reference solutions must be injected immediately after preparation).
II. Chromatographic conditions and System suitability requirements: Size exclusion chromatography (SEC) was applied to determine high molecular mass polymers (i.e. cefpiramide polymer) according to the ministerial standard YBH05752008 [12]. The Sephadex G-10 column (size: 300×15 mm, 40 ~120 μm) (Dalian, China) was maintained at room temperature. Mobile phase A is 0.01 Mammonium acetate. Mobile phase B is water. The flow rate is ~0.5 mL.min-1 (Note: column pressure must be lower than 1 MPa) and the UV detector was set at 254 nm. System suitability test: the test is not valid unless it meets the following requirements.
(1) Use mobile phase A and inject 200 μL of Blue Dextran 2000 solution into the SEC system. Do the same for mobile phase B. The theoretical plates are not less than 800, based on the Blue Dextran 2000 peak and their tailing factors are less than 2.0.
(2) The ratios of the retention time (tR) of Blue Dextran 2000 with mobile phase A to that of Blue Dextran 2000 with mobile phase B, tR of the peak of the reference solution with mobile phase B to that of Blue Dextran 2000 with mobile phase B, as well as tR of the high molecular mass polymer in the test solution with mobile phase A to that of Blue Dextran 2000 with mobile phase A should be within the 0.93-1.07 range.
(3) Use mobile phase A as the mobile phase and inject 200 μL of Blue Dextran 2000 solution five times. The RSD of the Blue Dextran 2000 peak area is not more than 5.0%.
III. Procedure: Use mobile phase B as the mobile phase and inject 200 μL of reference solution into the SEC system. Use mobile phase A as the mobile phase and inject 200 μL of test solution into the SEC system. Calculate the content of high molecular mass polymers in the sample to be examined by external standard method, using the peak areas for calibration*. The analysis is performed as double determinations. The content is not greater than 0.5%.
(*Calculation equation: (Apolymer, test solution/Acefpiramide, reference solution) × 1.0%, where Apolymer, test solution represents the peak area of high molecular mass polymer (if there are more than one peak, then sum the areas)in the test solution chromatogram while Acefpiramide, reference solution refers to the peak area of cefpiramide in the reference solution chromatogram.)
Methods for other tests: Appearance was tested by visual examination, which is very common in routine testing of pharmaceuticals. The water content was determined by Karl-Fischer titration, which is applied for many β-lactam antibiotics. Characterization of solid state forms was performed by XRPD and the thermal properties of the samples were evaluated by TGA and DSC, respectively.
Results and Discussion
Verification of the methods (linearity, specificity and sensitivity)
As described in the Method for Assay and Related Substances and the Method for High Molecular Mass Polymers, the test methods for assay, related substances and high molecular mass polymers were according to the ministerial standard YBH05752008 and the method for related substance was the same as that of the USP 35 [12,13]. To verify the suitability of the method for assay and related substances, linearity, specificity and sensitivity were evaluated:
(1) In the concentration range of 0.125 ~1.25 mg×mL-1 (injection volume: 20 μL), the correlation coefficient (r) of the regression line (y = peak area of cefpiramide, x = concentration of cefpiramide in reference solutions) was 1.000, indicating a linear relationship when the injection amount was in the 2.5 ~25 μg range. The standard deviation relative to specification (100%) was 0.6%.
(2) The forced degradation tests showed that cefpiramide was sensitive to many factors, especially to basic, oxidative and high temperature conditions (see The Method for Assay and Related Substances): after treating with 1 M NaOH for 30 min, cefpiramide degraded completely; when treated with 30% H2O2 for 30 min, only 10% of cefpiramide remained; after heating at 95°C for 10 min, only about 35% of original cefpiramide could be observed. Under other conditions being examined, such as medium temperature (60°C, 30 min), acidic condition (1 M HCl, 30 min) or UV light (2 hours), total related substances increased about 15%, 5% and 3%, respectively. The specified impurity MTT increased significantly under acidic, basic and high/medium temperature conditions. Therefore, in our following stability studies, the amount of MTT was chosen as a critical stability indicating parameter in the related substances test. By DAD peak purity analysis, it was demonstrated that both cefpiramide and MMT could be well-separated from (other) possible degradation products, which indicated that the specificity of the method could meet the requirements for assay and related substances.
(3) The calculated LOQs and LODs for cefpiramide/MTT were 0.03% /0.02% and 0.01% /0.006%, respectively, relative to the nominal test working concentration (0.5 mg×mL-1). RSD values on the areas of replicate injections of the LOQ concentrations were all below 2.0%. The results confirmed that the sensitivity of the method could satisfy our intended purposes for related substances.
Stability studies
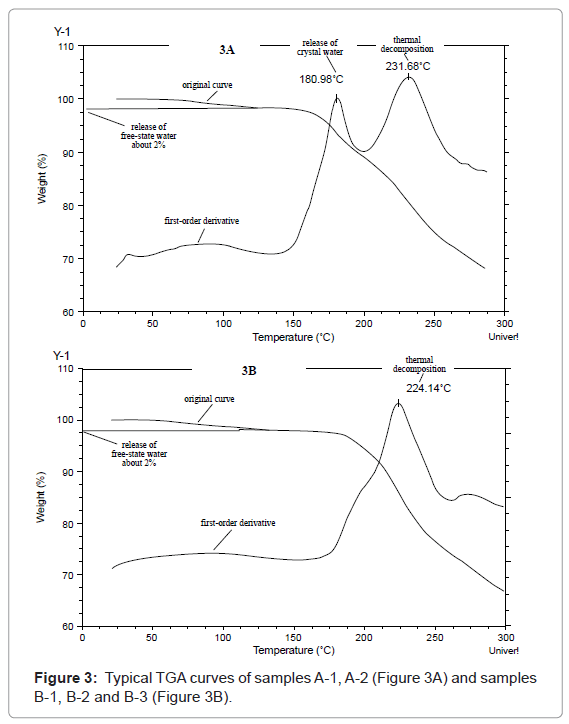
Comparison of initial quality properties: At time zero, all the samples being evaluated were compared in terms of appearance, water content, characterization of solid state form, TGA, related substances, high molecular mass polymers and assay, as shown in Table 2, Figure 2 and 3, respectively.
| Samples: | A-1 | A-2 | B-1 | B-2 | B-3 | |
| Appearance | white | white | light yellow | white | light yellow | |
| Water | 7.1% | 7.1% | 1.8% | 1.9% | 1.9% | |
| X-ray powder diffraction1 | Figure 2 | |||||
| TGA2 | Figure 3 | |||||
| Uniformity of mass (RSD) | 2.6% | 2.5% | 2.7% | 2.6% | 3.2% | |
| Related substances |
MTT | 0.16% | 0.14% | 0.31% | 0.38% | 0.89% |
| Total impurities | 0.41% | 0.46% | 1.31% | 2.37% | 2.50% | |
| High molecular mass polymers | 0.10% | 0.09% | 0.27% | 0.31% | 1.06% | |
| Assay3 | 105.7% | 106.3% | 97.2% | 94.4% | 90.8% | |
1 Samples A-1, A-2: crystalline form; Samples B-1, B-2, B-3: amorphous form.
2 Samples A-1 and A-2 had similar TGA curves (Figure 3-A) while samples B-1, B-2, B-3 had similar TGA curves (Figure 3-B).
3 Assay was based on its labeled amount of cefpiramide (1.0 g/ vial).
Table 2: Comparison of initial quality parameters (t = 0 day).
By comparison, it can be obviously observed that the initial quality properties of the crystalline form of cefpiramide (samples A-1, A-2) were much better than those of the amorphous form (samples B-1, B-2 and B-3), whether demonstrated by related substances or high molecular mass polymers. All the related substances observed in the samples (relative retention times (RRT) = 0.30 (MTT), 0.54, 0.63, 0.91 and 1.37, respectively) were from degradation. Sodium benzoate (RRT = 0.36) was used as preservative in samples B-1 and B-3. The major impurity observed in commercial cefpiramide for injection was either MTT or the impurity with RRT of 0.91. Both of the above degradation products were produced at even medium temperature, as well as those with RRT of 0.54 and 1.37. The above results indicated that manufacturers and related health service organization (e.g. hospitals, drugstores) should strictly control the storage temperature (≤ 20°C) and take necessary measures to minimize the degradation of cefpiramide to ensure its safety and efficiency.
The water contents of the crystalline samples were about 7%, much higher than those of the amorphous ones (1.8 ~1.9%). The TGA curves (Figure 3) showed that before 150°C, both solid states lost about 2% of their weights. Based on their water content results, it was presumed that the above loss was due to release of free-state water, while the remaining 5% of water in the crystalline samples was released at about 181°C, indicating that it may exist as crystal water. DSC results confirmed the presence of crystal water which showed an absorption peak near 181°C due to the release of crystal water. Based on the relative molecular mass of cefpiramide, it is supposed that each molecule of cefpiramide might contain two molecules of crystal water. To confirm the proposed structure of the crystalline form of cefpiramide, single crystal analysis will be needed.
Stability comparison demonstrated by assay : As shown in Table 3, the content of the amorphous samples decreased more than 8.6% after storage at 40°C / 75% RH for 60 days and even more than 23.7% after storage at 60°C / 75% RH for 60 days, while the crystalline samples degraded not more than 2.3% at the above conditions, demonstrating the better stability of the crystalline form. Results obtained at upside and downside positions were similar, which proved the compatibility of the containers with cefpiramide and further confirmed the above results.
| Storage conditions | Storage time(days) | Assay (based on the labeled amount of cefpiramide) | ||||
|---|---|---|---|---|---|---|
| crystalline form | amorphous form | |||||
| A-1 | A-2 | B-1 | B-2 | B-3 | ||
| Initial (0 day) | 105.7% | 106.3% | 97.2% | 94.4% | 90.8% | |
| 40°C/75% RH | 10 days | 106.1% | 105.8% | 93.4% | 90.5% | 87.6% |
| 20 days | 105.0% | 105.1% | 91.7% | 91.0% | 84.3% | |
| 40 days | 105.0% | 104.6% | 91.4% | 89.6% | 83.2% | |
| 60 days | 103.5% | 104.1% | 88.4% | 85.8% | 82.1% | |
| Maximum change | -2.2% | -2.2% | -8.8% | -8.6% | -8.7% | |
| 60°C/75% RH | 30 days | 103.1% | 104.0% | 82.8% | 77.6% | 75.7% |
| 60 days | 103.4% | 104.0% | 73.5% | 64.2% | 66.3% | |
| Maximum change | -2.6% | -2.3% | -23.7% | -30.2% | -24.5% | |
Table 3: Stability data of assay for up to 60 days at 40°C/75% RH and 60°C/75% RH, respectively.
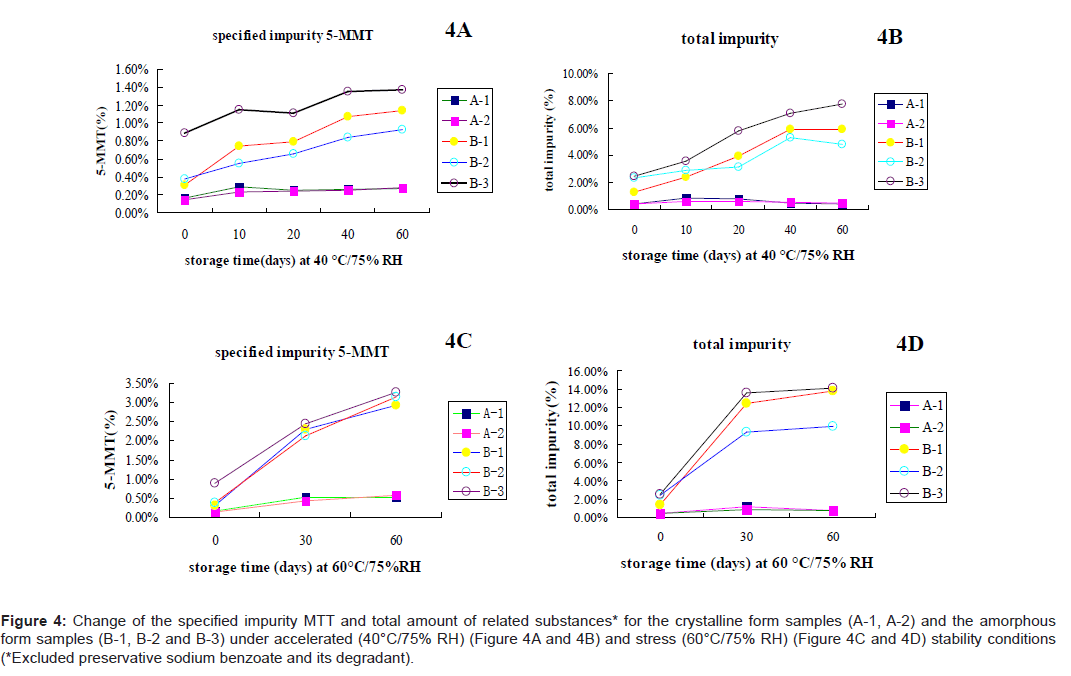
Stability comparison demonstrated by related substances: Figure 4 shows that the degradation rate of the crystalline samples (A-1, A-2) was much slower than that of the amorphous samples (B-1, B-2 and B-3) under accelerated condition (40°C/75% RH), in terms of both MTT and total related substances. During 60 days of storage, the specified impurity MTT in the crystalline samples only increased 0.1%, while MTT in the amorphous samples increased 0.9 ~1.4% (Figure 4A). The total amount of related substances in the crystalline samples almost remained unchanged (+0.2 ~0.4%), while the total related substances in the amorphous samples increased 2.9 ~5.3% (Figure 4B). Under stress condition (60°C / 75% RH), the stability difference between the crystalline form and the amorphous form was more obvious (Figure 4C and 4D). The specified impurity MTT and the total amount of related substances in the amorphous samples increased 2.4 ~2.8% and 7.6 ~12.5%, respectively. Meanwhile MTT and the total amount of related substances in the crystalline samples only increased 0.4% and 0.6%, respectively (Figure 4B) , which clearly demonstrated that the crystalline form of cefpiramide was more stable than its amorphous form.
Figure 4: Change of the specified impurity MTT and total amount of related substances* for the crystalline form samples (A-1, A-2) and the amorphous form samples (B-1, B-2 and B-3) under accelerated (40°C/75% RH) (Figure 4A and 4B) and stress (60°C/75% RH) (Figure 4C and 4D) stability conditions (*Excluded preservative sodium benzoate and its degradant).
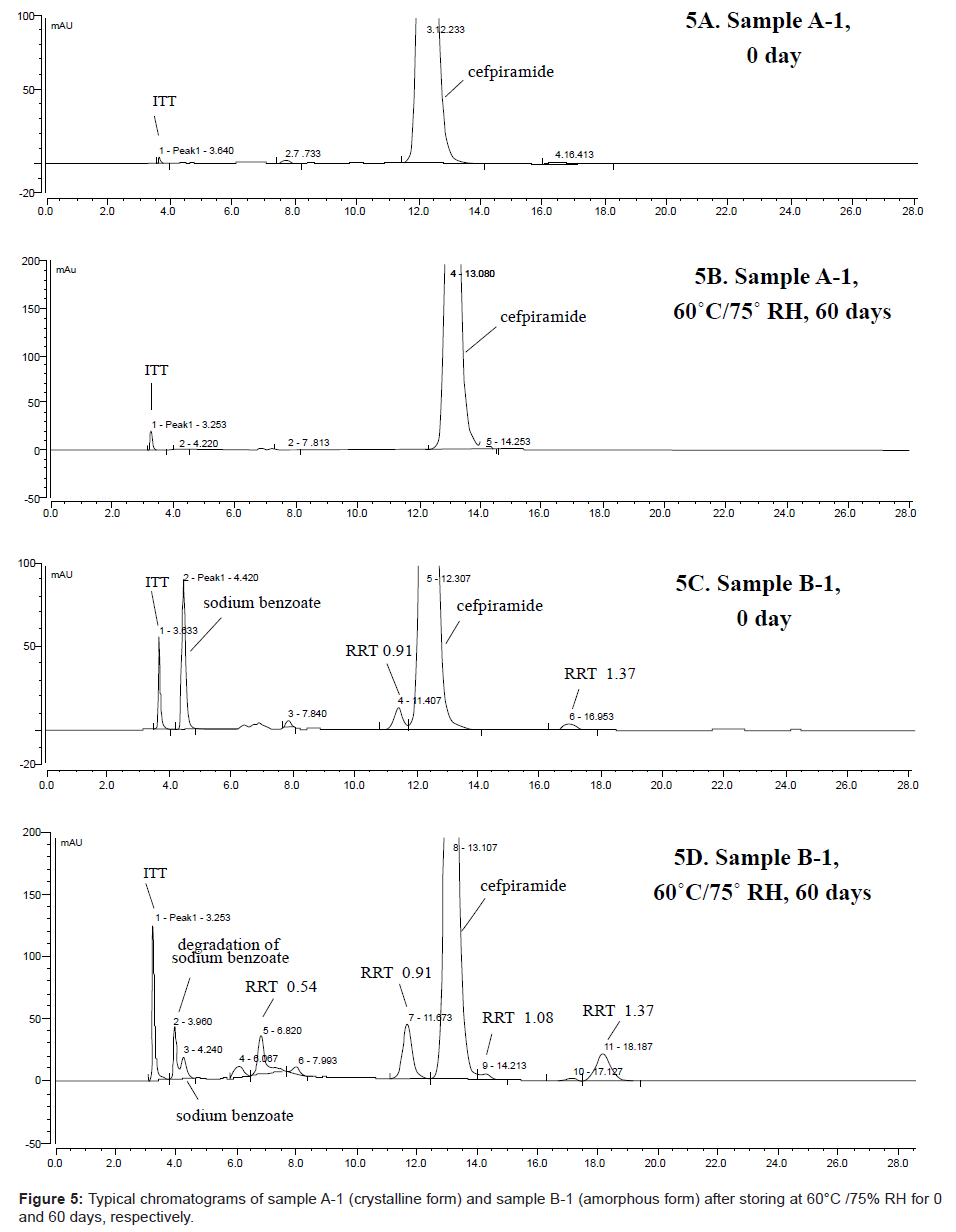
As expected, besides MTT, other thermal degradation products (e.g. RRT 0.54, 0.91 and 1.37) also increased significantly and several new impurities (e.g. RRT = 0.30 (degradant of sodium benzoate), 1.08) were observed under stress stability condition (60°C / 75% RH), as shown in Figure 5, which further indicated the necessity of taking proper measures (such as control of the storage temperature below 20°C) to minimize degradation of cefpiramide.
Stability comparison demonstrated by high molecular polymers: It is of common knowledge that the polymers of β-lactam antibiotics have great impact on the safety of β-lactam antibiotics since they might induce anaphylaxis in clinical therapy [20-23]. Therefore, at the end of the stability studies, high molecular mass polymers in the samples being examined were analyzed by a validated SEC method (see The Method for High Molecular Mass Polymers) and results are shown in Table 4. As it is shown, after storage at 40°C / 75% RH and 60°C / 75% RH for 60 days, the high molecular mass polymers in the amorphous samples (B- 1, B-2 and B-3) increased 0.72 ~0.99% and 2.98 ~3.82%, respectively; while the high molecular mass polymers in the crystalline samples (A-1, A-2) only increased 0.08 ~0.09% and 0.21 ~0.24%, respectively, which further confirmed that the crystalline form of cefpiramide is much more stable.
| Solid state | Samples | Initial (0 day) |
40 °C/75%RH�? 60 days |
Change (%) | 60 °C/75%RH�? 60 days |
Change (%) |
|---|---|---|---|---|---|---|
| Crystalline form | A-1 | 0.10% | 0.18% | +0.08% | 0.31% | +0.21% |
| A-2 | 0.09% | 0.18% | +0.09% | 0.33% | +0.24% | |
| Amorphous form | B-1 | 0.27% | 1.06% | +0.79% | 3.25% | +2.98% |
| B-2 | 0.31% | 1.30% | +0.99% | 4.13% | +3.82% | |
| B-2 | 1.06% | 1.78% | +0.72% | 4.23% | +3.17% |
Table 4: Comparison of results for high molecular mass polymers.
The decrease in content is not fully compensated by the increase in related substances and high molecular mass polymers. This may be due to the fact that the degradants produced absorb much less at the detection wavelength. However, the stability trend demonstrated by either assay or related substances and high molecular mass polymers was consistent: the crystalline form of cefpiramide was much more stable than its amorphous form at both the accelerated and stress stability conditions.
Structural stability of different solid states of cefpiramide: At the end of the stability study, XRPD was performed to verify the structural stability of different solid states of cefpiramide (crystalline or amorphous). No significant change was observed in the XRPD patterns of crystalline samples or amorphous ones when compared to their initial patterns, indicating that there was no transformation between the two solid states under the stability conditions being examined.
Conclusion
Two solid states forms were observed for cefpiramide: crystalline and amorphous. With related substances, high molecular mass polymers and assay of cefpiramide as critical stability-indicating parameters, the thermal stability of the two solid states was compared and it was demonstrated that the crystalline form of cefpiramide was much more stable than its amorphous form at both the accelerated and stress stability conditions. Both of the solid states were stable under the conditions being examined.
The forced degradation tests showed that cefpiramide was sensitive to many factors, especially to basic, oxidative and high temperature conditions. All the related substances observed in the samples being examined were from degradation and the major impurity observed in commercial cefpiramide for injection was either MTT or the impurity with RRT of 0.91, both of which could be produced at even medium temperature conditions. The above results indicated that manufacturers and related health service organization (e.g. hospitals, drugstores) should strictly respect the storage temperature (≤ 20°C) and take other necessary measures to minimize the degradation of cefpiramide to ensure its safety and efficiency when administrated.
This study investigated the relationship between solid state forms of cefpiramide and their thermodynamic stability. Such information and the results of forced degradation tests provided valuable approaches for the quality control of cefpiramide and its drug products.
References
- Muraoka I, Hasegawa T, Nadai M, Wang L, Haghgoo S, et al. (1995) Biliary and renal excretions of cefpiramide in Eisai hyperbilirubinemic rats. Antimicrob Agents Chemother 39:70-74.
- Wang H, Yu Y, Xie X, Wang C, Zhang Y, et al. (2000) In-vitro antibacterial activities of cefpiramide and other broad-spectrum antibiotics against 440 clinical isolates in China. J Infect Chemother 6:81-85.
- Powell AJ, Tomberg J, Deacon AM, Nicholas RA, Davies C (2009) Crystal structures of penicillin-binding protein 2 from penicillin-susceptible and -resistant strains of Neisseria gonorrhoeae reveal an unexpectedly subtle mechanism for antibiotic resistance. J Biol Chem 284:1202-1212.
- Hu CQ, Cheng SH, Lu L (2002) Approach to the crystalline characteristics of ceftezole sodium. Yao Xue Xue Bao 37: 275-279.
- Hickey MB, Peterson ML, Manas ES, Alvarez J, Haeffner F, et al. (2007) Hydrates and solid-state reactivity: a survey of beta-lactam antibiotics. J Pharm Sci 96:1090-1099.
- Hu CQ, Yin LH, Liang YN (2008) Physical and chemical characteristics of a new cefazolin sodium hydrate crystal. Yao Xue Xue Bao 43:868-872.
- Qiang GF, Lv Y, Du GH (2009) The present situation and development direction of the research for polymorphic drugs in China. Chin J New Drugs 18:1196-1200.
- Liu SY, Sun YJ (2010) Studies on preparation and stability of polymorphs of cefathiamidine. Chin J Antibiot 35:760-762.
- Wenging Q, Army W, Kun PK, Ang ZQ (2010) Research on adjusting crystallization technology to improve the quality of amoxicillin. Hebei Chem Eng Ind 33:27-31.
- Zhao YN, Chen XQ, Zhu K, Li G (2011) Thermostability and crystal structure of anticancer drug dasatinib. Yao Xue Xue Bao 46:1104-1107.
- Sohn YT, Park SH (2006) Crystal form of cephradine. Arch Pharm Res 29:178-182.
- FDA (2008) FDA Fact Sheet. P. R. China.
- USP 35 – NF 30. The pharmacopeia of the United States of America; Mack Publishing Company: Easton, pp2565.
- International Conference on Harmonization of technical requirements for registration of pharmaceuticals for human use (2008) ICH harmonized tripartite guideline, draft Step 4. Pharmaceutical Development Q8 (R1).
- International Conference on Harmonization of technical requirements for registration of pharmaceuticals for human use (2009) ICH harmonized tripartite guideline, Pharmaceutical Development Q8 (R2).
- International Conference on Harmonization of technical requirements for registration of pharmaceuticals for human use (2008) ICH harmonized tripartite guideline, Quality Risk Management Q9.
- International Conference on Harmonization of technical requirements for registration of pharmaceuticals for human use (2008) ICH harmonized tripartite guideline, Pharmaceutical Quality System Q10.
- Lee SL, Raw AS, Yu L (2008) Significance of drug substance physiochemical properties in regulatory quality by design. In drugs and the pharmaceutical sciences 78 (Preformulation in solid dosage form developmemt), London: Informa Healthcare. pp 571-583.
- Mhatre R, Rathore AS (2009) Quality by design: an overview of the basic concepts. In quality by design for biopharmaceuticals. New York: Wiley. pp1-8.
- Ahlstedt S, Kristoffersson A, Svärd PO, Thor L, Ortengren B (1976) Ampicillin polymers as elicitors of passive cutaneous anaphylaxis. Int Arch Allergy Appl Immunol 51:131-139.
- Roets E, De Pourcq P, Toppet S, Hoogmartens J, Vanderhaeghe H, et al. (1984) Isolation and structure elucidation of ampicillin and amoxicillin oligomers. J Chromatogr A 303:117-123.
- Hu CQ, Jin S, Wang K (1994) The chromatographic behaviour of cephalosporins in gel filtration chromatography, a novel method to separate high molecular weight impurities. J Pharm Biomed Anal 12:533-541.
- Cai SY, Hu CQ (2005) Chromatographic determination of polymerized impurities in meropenem. J Pharm Biomed Anal 37:585-589.
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 15037
- [From(publication date):
August-2012 - Aug 20, 2025] - Breakdown by view type
- HTML page views : 10343
- PDF downloads : 4694