Research Article Open Access
Tracing the Variability of Dissolved Organic Matter Fluorescence in the East China Sea in the Red Tide Season with use of Excitation-emission Matrix Spectroscopy and Parallel Factor Analysis
Weihong Zhao*, Lisha Lv and Hui MiaoKey Laboratory of Ecology and Environmental Sciences, Institute of Oceanology, Chinese Academy of Sciences, Qingdao, 266071, China
- *Corresponding Author:
- Weihong Zhao
Key Laboratory of Ecology and Environmental Sciences
Institute of Oceanology
Chinese Academy of Sciences
Qingdao, 266071, China
Tel: 86-532-82898611
E-mail: klmees@qdio.ac.cn
Received date: September 04, 2013; Accepted date: December 23, 2013; Published date: December 30, 2013
Citation: Zhao W, Lv L, Miao H (2013) Tracing the Variability of Dissolved Organic Matter Fluorescence in the East China Sea in the Red Tide Season with use of Excitation-emission Matrix Spectroscopy and Parallel Factor Analysis. J Marine Sci Res Dev 4:144. doi:10.4172/2155-9910.1000144
Copyright: © 2013 Zhao W, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Marine Science: Research & Development
Abstract
From the end of March to the end of May, 2011, five cruises were carried out to survey the red tide occurrence in the Zhejiang coast of the East China Sea where the red tides occurred each spring and there was a trend for community succession from diatoms to dinoflagellates. Using Excitation Emission Matrix Spectrum(EEMs) combined with Parallel Factor Analysis (PARAFAC) examine the fluorescent components feature of dissolved organic matter (DOM) sampled from the East China Sea in the red tide season. Three fluorescent components were identified by PARAFAC, including tyrosine-like component C1(230,280/320), tryptophan-like component C2(240,305/355) and humic-like component C3(270,340/480). The result showed that the fluorescence intensity of C1 was relatively high and changed along with the succession of red tides, besides, the weak correlation coefficient with salinity and the particularity of its source suggested that phytoplankton activity was the important factor in fluorescence intensity change of C1. The fluorescence intensities of component C2 and C3 were relatively low and changed not very significant, but its good linearity with salinity indicated that the terrestrial input was the important sources of two components during the algae dispersion. Lower Fluorescence Index (FI) (<1.4) also tested the terrestrial distribution.
Nevertheless, correlation coefficient with salinity was slightly decreasing showed the effects of biological activity had increased during the outbreak of dinoflagellate. Higher (>0.8) Biological Index (BIX) and lower Humification Index (HIX) (<2) inferred that biological activity intensively in the red tide season in the East China Sea would contribute the CDOM in the water.
Keywords
Dissolved organic matter; Excitation-emission matrix spectroscopy; Parallel factor analysis (PARAFAC); The East China Sea
Introduction
Chromophoric dissolved organic Matter < CDOM > is an important part of the dissolved organic matter (DOM) and plays an important role in marine photoreactions [1-6] and the biogeochemistry of biogenic elements [7]. Spectroscopic techniques can provide information about the source and composition of the DOM present in a natural abundance, thereby eliminating the need for isolating or concentrating it prior to analysis [8,9]. There are notable differences in CDOM composition in different water environments. The highest humic-like CDOM concentrations in estuarine and coastal areas are usually observed in freshwater and estuaries due to river runoff [10], and decreases in coastal and offshore waters [11]. Amount of proteinlike CDOM were monitored in the algae bloom water and changed with the bloom process [12,13].
The East China Sea is the red tide-frequent-occurrence area and its eutrophication water area is the first of the four China Sea area, which threatens fisheries, public health and economic. According to statistics, since the 1990s, the number of the red tides in this region accounted for over 50% of the total in China, and the frequency increased. Meanwhile, red tides in East China Sea also presented the characteristic of the duration lengthening and area expanding. Its occurring time is concentrated in the April and May annually [14-16].
The red tide and subsequent succession are resulted from a combination of biological, chemical and physical factors. Chromophoric dissolved organic Matter < CDOM > plays an important role in the biogeochemistry of biogenic elements. In this paper, using EEMs combined with PARAFAC traced the fluorescent components variation of dissolved organic matter. The type, distribution and origin of the fluorescence dissolved organic matter were discussed. Besides, the relationship between organic matter and red tide succession was examined, which provided theoretical basis for a progressive research on red tide occurrence mechanism.
Materials and Methods
Study area
The investigation area located at 28-30.5°N and 122-123.5°E in the East China Sea. This water area is greatly influenced by the Changjiang dilute water (CDW), the East China Sea Coastal Current (ECSCC), the Taiwan Warm Current (TWC) from Taiwan Strait, and a branch of the Kuroshio Current [17]. The Yangtze River (or Changjiang River) is the fifth largest river in the world with huge amounts of freshwater discharge (9.24×1011 yr-1). The fresh water discharge has significant seasonal variation with a minimum in May to October and a maximum in November to April. The CDW is divided into two branches out of the estuary; one extends southward along the coast of Zhejiang province and another northeastward directing at the Cheju Island [18,19]. The relative strength of these two branches varies seasonally. The southward branch was stronger in autumn and winter, and the northward branch was stronger in spring and summer.
Sample collection and preparation
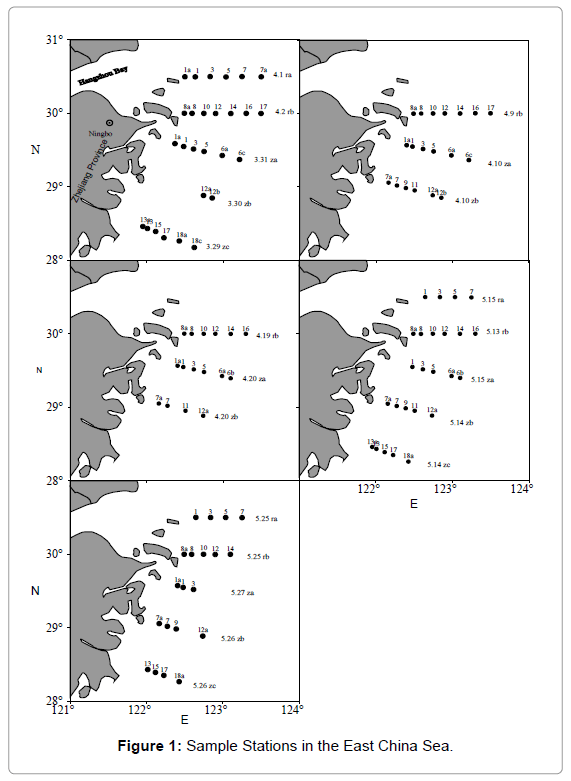
Samples were collected in 5 cruises from sampling stations and sample times are shown in Figure 1. Diatom bloom occurred during March 31 to April 2, then diatom began to dispersion and 8 um size unknown algae appeared in part of the survey area in April 9 to April 10. Afterwards, most of the diatom dispersion and about 8um size unknown algae dominated in April 19 to April 20. Soon, dino flagellate began to bloom in May 13 to May 15 and out broke in May 25 to May 27. Surface, middle water samples which chlorophyll a was maximal, and bottom water samples (30-60m) were collected. The water samples were filtered using a vacuum pump through GF/F filters (Whatman, U.K, φ=25 mm, pre-combusted at 450°C for 4 h). The filtrate was stored in 60 ml brown glass bottle (pre-combusted at 450°C for 4 h) frozen at -20°C in the dark for later organic matter analysis.
Measurement of dissolved organic matter fluorescence, DOC and salinity
The EEMs fluorescence of the DOM was measured using a Hitachi F-4500 fluorescence spectrophotometer, with normal operating conditions of 5 nm slit widths of excitation and 10 nm slit widths of emission, a PMT voltage of 700 V. Fluorescence measurements were made by scanning emission spectra from 250 to 650 nm with 5 nm intervals, excitation wavelengths from 200 to 500 nm with 5 nm.The EEMs were normalized to quinine sulfate units using 0.01 mgL-1 quinine sulfate monohydrate in a solution of 0.05 mol L-1 H2SO4. The EEMs of each sample was Raman calibrated and subtracted from a Raman normalized Mill-Q water EEMs. Set the data which influenced by Rayleigh scattering to zero. EEMs were converted to R.U. by correcting with the area under the water Raman Peak at 350 nm excitation [20]. DOC was measured using a Shimadzu TOC-V total organic carbon analyser. Salinity was determined by CTD.
PARAFAC modeling
A total of 552 EEMs were subject to the multivariate modeling technique PARAFAC [21] using MAT-LAB 2010a with “the N-way toolbox for MATLAB’. The number of components was validated by split-half analysis.
HIX, FI and BIX calculation
The degree of DOM humification is an indicator of a material’s age and recalcitrance within a natural system [22,23]. The humification index (HIX) determined from the ratio of two integrated regions of an emission scan (sum from λEm 435–480 nm divided by the sum from λEm 300–345 nm) collected with excitation at 255 nm as a method for comparing the relative humification of DOM samples [22]. Fluorescence intensity ratios can be used to infer the relative contributions from autochthonous and allochthonous OM in natural waters. Fluorescence index (FI) was the ratio of the fluorescence intensity at emission 450 and 500 nm at afixedexcitation wavelength of 370 nm [24]. The biological/autochthonous index or BIX was used to assess the relative contribution of autochthonous DOM and is calculated from the ratio of emission intensities at λEm 380 nm and λEm 430 nm wavelength using a fixed excitation (λEx310 nm) [24].
Results and Discussion
The fluorescence characteristic of DOM
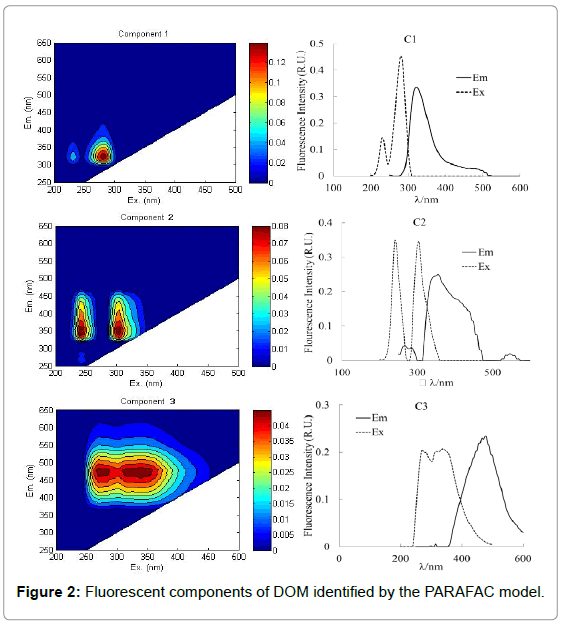
Three fluorescent components were identified using PARAFAC (Figure 2 and Table 1): C1 (230 nm, 280/320 nm), C2 (240 nm, 305/355 nm) and C3 (270 nm, 340/480 nm).
| Fluorescence Components | Peak position λEx/Em(nm) | Literature reported | ||
|---|---|---|---|---|
| Description | Ex/Em (nm) | Reference | ||
| C1 | 230,280/320 | tyrosine-like | 230/325-350 | [1] |
| 280/320-350; | [25] | |||
| 270-280/320-350; | [26] | |||
| C2 | 240,305/355 | tryptophan-like | <250�?290�?/360 | [27] |
| <250 (290)/360 | [28] | |||
| C3 | 270,340/480 | Terrestrial humic-like | 230-260/380-480 | [29] |
| 320-360/420-480 | ||||
Table 1: Positions of the fluorescence maxima of the three components.
The EEMs spectral characteristics of C1 was composed of two peaks with 320 nm emission maxima wavelength at excitation maxima wavelength 230 nm and 280 nm which was similar to a tyrosine-like fluorescent compound previously observed [1,25,26]. The spectral features were also similar to the high fluorescence intensities tyrosinelike fluorescent component observed in red tide water in the East China Sea [12].
The C2 component with emission maxima wavelength at 355nm and two excitation maxima wavelength at 240 nm and 305 nm were categorized as the previously defined autochthon-nous tryptophan-like fluorescence peak. This component was also similar to the previously reported PARAFAC components of amino acids that are free or bound in proteins [27,29,30].
The C3 component was also composed of two peaks with excitation maxima wavelength at 270 nm and 340 nm at 480 nm emission maxima wavelength that were similar to the terrestrial humic-like fluorescence peak A and peak C. The spectral features were also similar to those reported for terrestrial-derived humic-like PARAFAC components [31].
Homology analysis of components
To investigate whether the three components had the same sources, we calculated the correlation coefficient between three components in different red tide period, as listed in Table 2. It showed that they were positive related significantly between three components (R>0.72, P<0.01). Particularly, tryptophan-like component C2 had higher correlation (R>0.8) with tyrosine-like C1 and humic-like component C3, which illustrated that they had some connection or difference in source or structure.
| Time | RC1/C2 | RC1/C3 | RC2/C3 | N |
|---|---|---|---|---|
| 3.31-4.2 | 0.8019 | 0.7456 | 0.9526 | 77 |
| 4.9-4.10 | 0.9213 | 0.9161 | 0.9533 | 50 |
| 4.19-4.20 | 0.9599 | 0.7265 | 0.8345 | 45 |
| 5.13-5.15 | 0.8764 | 0.7365 | 0.8652 | 72 |
| 5.25-5.27 | 0.8731 | 0.7941 | 0.9722 | 57 |
| All above | 0.8578 | 0.7446 | 0.9085 | 448 |
Table 2: Correlation coefficient between three components C1, C2, C3, (P <0.01).
The vertical distribution of the three components fluorescence intensities
Table 3 showed that the fluorescence intensities of three components in surface, middle and bottom layers in different red tide period. Chen and Bada [32] reported that fluorescence intensity of protein-like fluorescence was the highest in the surface water, decreasing with the increase of depth. But the result of our study was different. No matter in surface water or in middle water, fluorescence intensity of protein like components C1 and C2 were almost the same, sometimes it was higher in the middle water than the surface, but both were higher than the bottom which was in accordance with biological activity. Similarly, humic-like component C3 showed the same distribution that the fluorescence intensity was similar in surface and middle, but higher than the bottom. It was not consistent with previously views that fluorescence intensities of humic-like increased with the depth [1,33].
| Component | water layer | FI3.31-4.2 | FI4.9-4.10 | FI4.19-4.20 | FI5.13-5.15 | FI5.25-5.27 |
|---|---|---|---|---|---|---|
| C1 | surface | 0.0965 ± 0.0602 | 0.0835 ± 0.054 | 0.1132 ± 0.0857 | 0.0833 ± 0.0314 | 0.0973 ± 0.0415 |
| middle | 0.0753 ± 0.0337 | 0.0834 ± 0.0393 | 0.1467 ± 0.1289 | 0.0884 ± 0.0355 | 0.0978 ± 0.0409 | |
| bottom | 0.0577 ± 0.0522 | 0.0747 ± 0.0482 | 0.0911 ± 0.1004 | 0.0479 ± 0.0367 | 0.0981 ± 0.055 | |
| mean | 0.0762 ± 0.0459 | 0.0807 ± 0.0465 | 0.1199 ± 0.1076 | 0.0714 ± 0.0389 | 0.0977 ± 0.0455 | |
| C2 | surface | 0.0303 ± 0.0187 | 0.0246 ± 0.0116 | 0.0315 ± 0.0154 | 0.0241 ± 0.008 | 0.0275 ± 0.0117 |
| middle | 0.0249 ± 0.0131 | 0.0262 ± 0.0131 | 0.0368 ± 0.0251 | 0.0265 ± 0.0105 | 0.0268 ± 0.0114 | |
| bottom | 0.0179 ± 0.0147 | 0.0242 ± 0.015 | 0.0238 ± 0.0163 | 0.0146 ± 0.008 | 0.0246 ± 0.0139 | |
| mean | 0.0242 ± 0.0164 | 0.0251 ± 0.0129 | 0.0315 ± 0.0201 | 0.0211 ± 0.0102 | 0.0262 ± 0.0122 | |
| C3 | surface | 0.0283 ± 0.0149 | 0.0261 ± 0.0106 | 0.0286 ± 0.0122 | 0.0236 ± 0.008 | 0.0306 ± 0.0105 |
| middle | 0.0261 ± 0.0105 | 0.0265 ± 0.0114 | 0.0324 ± 0.0195 | 0.0239 ± 0.0064 | 0.0298 ± 0.0094 | |
| bottom | 0.0197 ± 0.0144 | 0.0258 ± 0.0138 | 0.024 ± 0.0127 | 0.0159 ± 0.0064 | 0.0269 ± 0.0141 | |
| mean | 0.0244 ± 0.013 | 0.0261 ± 0.0117 | 0.0288 ± 0.0155 | 0.0209 ± 0.0079 | 0.0289 ± 0.0115 |
Table 3: The fluorescence intensities of three components in surface, middle and bottom layers in different red tide period (R.U.).
The horizontal distribution of PARAFAC components
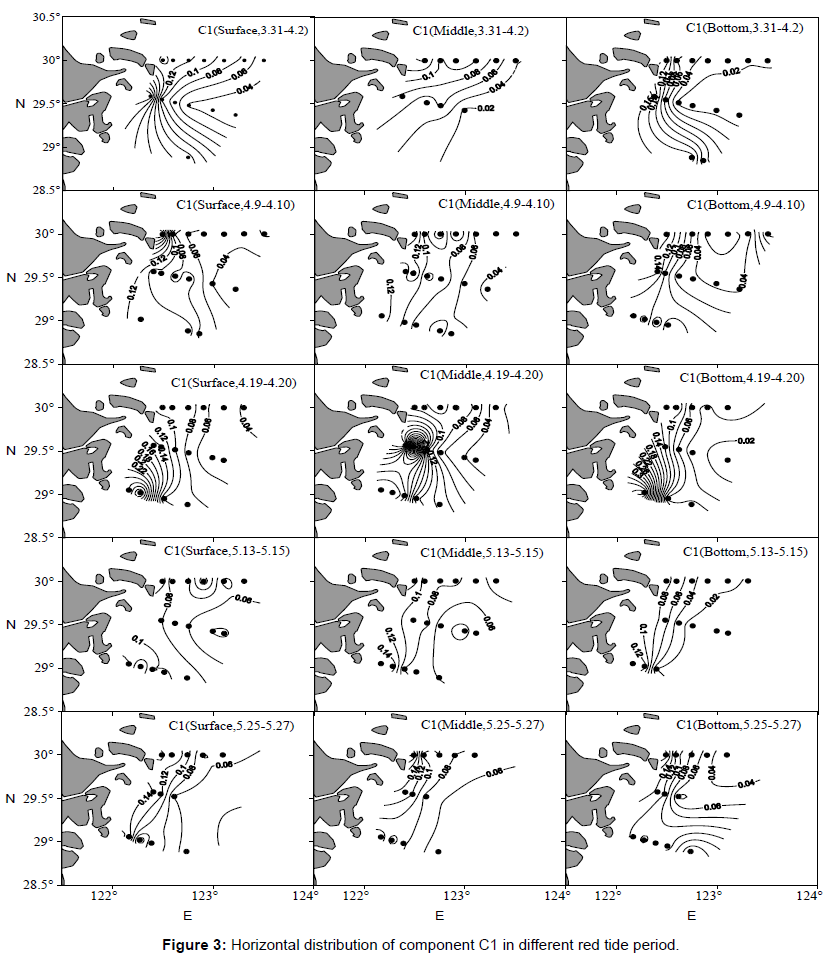
Owing to the high relationship between the three components and the fluorescence intensity of component C1 was the highest, we chose component C1 as characteristic component. Figure 3 displayed the horizontal distribution variation of component C1 with the red tide succession. On the whole, fluorescence intensity of DOM inshore was higher than that in the open sea. Diatom bloom erupted far offshore during March 31 to April 2 and the highest fluorescence intensity appeared in offshore. Then diatom bloom began to dispersion in April 9 to 10, the highest fluorescence intensity transfer toward north area. Following it appeared in southern area of surveyed waters in April 19 to 20, the distribution patterns kept until May 13 to15, which accord with relatively high dinoflagellate density in this area. During May 25 to 27, dinoflagellate bloom broke full-scale, fluorescence intensity were relatively high in the southern coastal area, conform to the result that dinoflagellates finally outbreak in the near shore, which demonstrated that phytoplankton activity had certain contribution to fluorescent components.
Spectroscopic indication of the sources of DOM in the East China Sea
To analysis the influence of terrestrial input on DOM in different red tide period, Table 4 showed the relationships between the three components and salinity. Although tyrosine-like C1 component had significant correlation with salinity, the correlation coefficient can reach -0.6749 in April 9 and April 10. Combining with the process of algal bloom showed that the C1 component was affected by algal succession in other periods. Tryptophan-like C2 component and humic-like C3 component had different correlation with salinity in different periods of red tide. C2 and C3 had better correlation with salinity during the diatom erupted until it dispersed, which showed that terrestrial input was a major contributor to them. The result was consisting with the conclusion that terrestrial input contributed a lot of tryptophan-like matter to the East China Sea. However, correlation had been reduced during the outbreak of dinoflagellate bloom, which indicated that the biological effects bad been increased.
| Component | Mar.31 to April 2 | April 9 to 10 | April 19 to 20 | May 13 to 15 | May 25 to 27 |
|---|---|---|---|---|---|
| C1 | R=-0.4267 P<0.01 N=77 |
R=-0.6749 P<0.01 N=50 |
R=-0.4489 P<0.01 N=45 |
R=-0.3672 P<0.01 N=72 |
R=-0.4853 P<0.01 N=57 |
| C2 | R=-0.7253 P<0.01 N=77 |
R=-0.7226 P<0.01 N=50 |
R=-0. 6021 P<0.01 N=45 |
R=-0.5612 P<0.01 N=72 |
R=-0.5863 P<0.01 N=57 |
| C3 | R=-0.6908 P<0.01 N=77 |
R=-0.6682 P<0.01 N=50 |
R=-0.6942 P<0.01 N=45 |
R=-0.6424 P<0.01 N=72 |
R=-0.5583 P<0.01 N=57 |
Table 4: Positions of the fluorescence maxima of the three components.
| Time | RC1/DOC | RC2/DOC | RC3/DOC | N |
|---|---|---|---|---|
| 3.31-4.2 | 0.1893 | 0.309* | 0.3069* | 77 |
| 4.9-4.10 | 0.121 | 0.2089 | 0.1068 | 50 |
| 4.19-4.20 | -0.3045* | -0.1982 | 0.0635 | 45 |
| 5.13-5.15 | 0.1422 | 0.301** | 0.4483** | 72 |
| 5.25-5.27 | 0.1488 | 0.3209* | 0.3385* | 57 |
*significant level P<0.05 **significant level P<0.01
Table 5: Relationship between fluorescence intensity of three components and DOC.
FI values can infer the relative contributions from autochthonous and allochthonous organic matter in natural waters. It suggested that FI values of 1.4 or less indicated humic DOM of terrestrial origin and values of 1.9 or higher correspond to microbially-derived material [24]. In this study, the FI in the East China Sea is in the range of 0.40~2.11, average 1.08. Most of the samples are less than 1.4, only one of them is larger than 1.9, indicating that terrestrial origin is the mainly sources of DOM in this area, which is consistent with above the relationship between DOM fluorescence components and salinity.
BIX Values between 0.8 and 1.0 correspond to freshly produced DOM of biological or microbial origin, whereas values below 0.6 are considered to contain little autochthonous OM [24]. In this area, BIX values were in the range from 0.39~1.48, average 0.9, about 81% samples were larger than 0.8, only 7 samples were less than 0.6. Red tide process is an important organic synthetic process; freshly produced DOM undoubtedly became an important distributor to the CDOM in the investigated water area.
The humification index (HIX) is used to investigate the degree of humification of CDOM [22,23]. Huguet et al. [33] measured HIX for samples from the Gironde Estuary (France). The high values between 10 and 16 were of terrestrial origin, but the low values (<4) were related to autochthonous organic matter. Yang et al. [34] got HIX values 0.53 and 2.11 sampled from the submarine hydrothermal vents off NE Taiwana indicating a biological or aquatic bacterial source. We calculated HIX values ranged from 0.45~7.53, averaged 1.37. Only several near shore or bottom samples have higher HX values (>2), especially near the Changjiang Estuary, the HIX value was 7.53. Most of samples had relatively lower HIX values (<2), indicating the DOM had the low humification degree and was the freshly produced by biological activity.
The fluorescence intensity means variation of the three components on the typical sections in the red tide succession process
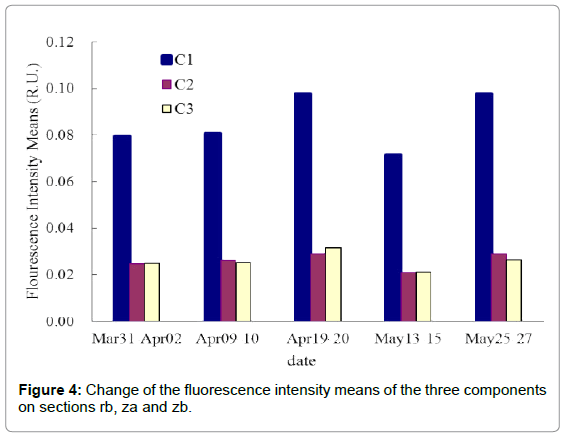
Figure 4 showed the change of the fluorescence intensity means of the three components on sectionsrb, za and zb. Combining with Table 3, we knew that fluorescence intensities of the three components reached maximum in April 19 and April 20. Moreover, fluorescence intensities of tyrosine-like C1 component changed distinctly with red tide succession, while C2 component and C3 component varied not obviously.
Tryptophan fluorescence peak can only be tested if a protein molecule containing both tyrosine and tryptophan. So tyrosine-like fluorescence peak only be detected from tryptophan monomer or protein molecules only containing tyrosine without tryptophan [35,36]. Determann [37] effectively explored the fluorescence properties of protein from phytoplankton and bacteria through laboratory culture experiment and identified distinct tryptophan-like fluorescence peak from algae cells and bacteria, while tyrosine-like fluorescence peak only existed in phytoplankton and fluorescence intensities of tyrosine only a third of tryptophan-like. So the tyrosine-like fluorescence can be used to judge the composition and source of protein in water. Figure 4 showed that fluorescent intensities of tyrosine-like component C1 is three times more than that of tryptophan-like component C2, speculating that phytoplankton is the main source of protein in water.
Basing on the above characteristics of component C1 and its correlation with salinity, we conjectured that phytoplankton activity is the significant cause of the variation of C1 fluorescence intensities, diatom decomposition released plenty of tyrosine-like matter into water causing C1 fluorescence intensities rising in late March and early April; 8μm unknown algae took advantage of organic matter making C1fluorescence intensities reduce since April 20; then the outbreak of dinoflagellates enabled it rise up until the middle of May.
Relationship between fluorescence intensity of three components and DOC
Many scholars attempted to establish the correlation between fluorescence and dissolved organic carbon (DOC) and tried to use fluorescence to invert the content of DOM [38]. Ferrari [39] found that humic-like was positively correlated with DOC (R>0.5) in the Mediterranean and the Atlantic coast, and Ji et al. [38] discovered that protein-like substance had good linear relation with DOC and humic-like substance had bad liner relation with DOC during red tide occurrence in Jiaozhou bay, Qingdao in China. In this study only part of the five curises showed somecorrelations between the three components and DOC listed in Table 4, which suggested the source complexity of the CDOM in the investigated water area.
Conclusions
Dissolved fluorescent organic matter was measured using EEMs combined with PARAFAC. The fluorescence intensity of tyrosine-like C1 was relatively high and changed along with the succession of red tides. Phytoplankton activity was the important factor effecting on the fluorescence intensity change of tyrosine-like C1 components. The fluorescence intensities of component C2 and C3 were low relatively and changed not very noticeable. All of the relationships between the three fluorescence components and salinity, low FI, higher BIX and low HIX indicated that terrestrial input and biological activity were the major sources of the CDOM components in different red tide periods. Our studies showed that the application of EEMs-PARAFAC modeling can characterize the qualities and sources of the dissolved organic matter in the coastal red tide-frequent-occurrence area.
References
- Mopper K, Schultz CA (1993) Fluorescence as a possible tool for studying the nature of water column distribution of DOC components. Marine Chemistry 41: 229-238.
- Green SA, Blough, NV (1994) Optical absorption and fluorescence properties of chromophoric dissolved organic matter in natural waters. Limnology & Oceanography 39: 1903–1916.
- Vodacek A, Blough NV, DeGrandpre MD, Peltzer ET, Nelson RK. Seasonal variation of CDOM and DOC in the Middle Atlantic Bight: terrestrial inputs and photooxidation. Limnology &Oceanography 42: 674-686.
- Rochelle-Newall EJ, Fisher TR (2002) Chromophoric dissolved organic matter and dissolved organic carbon in Chesapeake Bay. Marine Chemistry 77: 23-41.
- Huguet A, Vacher L, Relexans S, Saubusse S, Froidefond JM, et al. (2009) Properties of fluorescent dissolved organic matter in the Gironde Estuary. Organic Geochemistry 40: 706-719.
- Yang LY, Hong HS, Guo WD, Chen-Tung Arthur Chen, Pei-I Pan, et al. (2012) Absorption and fluorescence of dissolved organic matter in submarine hydrothermal vents off NE Taiwan. Marine Chemistry 128-129: 64-71.
- Hall GJ, Clow KE, Kenny JE (2005) Estuarial fingerprinting thorough multidimensional fluorescence and multivariate analysis. Environ Sci Technol 39: 7560-7567.
- Coble P (1996) Characterization of marine and terrestrial DOM in seawater using excitation–emission matrix spectroscopy. Marine Chemistry 51: 325-346.
- Hudson N, Baker A, Reynolds D (2007) Fluorescence analysis of dissolved organic matter in natural, waste and polluted waters – a review. River Research &Applications 23: 631-649.
- Gao L, Fan DD, Li DJ, Cai JG (2010) Fluorescence characteristics of chromophoric dissolved organic matter in shallow water along the Zhejiang coasts, southeast China. Mar Environ Res 69: 187-197.
- Blough NV, Zafiriou OC, Bonillam J (1993) Optical absorption spectra of waters from the Orinoco River outflow: terrestrial input of coloured organic matter to the Caribbean. Journal Geophysical Research 98: 2271–2278.
- ZhuoPeng-Ji, Zhao Wei-hong (2009) Fluorescence Characterization of dissolved organic matter in the East China Sea after diatom red tide dispersion. Guang Pu Xue Yu Guang Pu Fen Xi 29: 1349-1353.
- Zhao Weihong, ZhuoPengj (2011) Dissolved organic matter and its role in red tide succession in the East China Sea in spring, Chinese Journal of Oceanology and Limnology 29: 795-799.
- Zhou Ming-Jiang, Zhu Ming-Yuan, Zhang Jing (2001) Status of harmful algal blooms and related research activities in China. Chinese Bulletin of life sciences 13: 54-59.
- Su Jilan (2001) Harmful algal bloom and its research in China. China Academic Journal electronic publishing house 5: 339-342.
- Zhang CS, Wang XL, Shi XY, Tang HJ, Han XR, et al. (2007) Seasonal variation and spatial distribution of nutrients and their relationships with harmful algal blooms in coastal area of the east china sea. Environmental Sciences 28: 2416-2424.
- Zhang Y, van Dijk M A, Liu M, Zhu G, Qin B et al. (2009) The contribution of phytoplankton degradation to chromophoric dissolved organic matter (CDOM) in eutrophic shallow lak es: Field and experimental evidence. Water Research 43: 4685-4697.
- Li M, Xu K, Watanabe M, Chen Z (2007) Long-term variations in dissolved silicate, nitrogen, and phosphorus flux from the Yangtze River into the East China Sea and impacts on estuarine ecosystem. Estuarine, Coastal and Shelf Science 71: 3-12.
- Zhou MJ, Shen ZL, Yu RC (2008) Responses of a coastal phytoplankton community to increased nutrient input from the Changjiang (Yangtze) River. Continental Shelf Research 28: 1483-1489.
- Lawaetz AJ, Stedomon CA (2009) Fluorescence intensity calibration using the Raman scatter peak of water. Applied Spectroscopy 63: 936-940.
- Stedmon CA, Markager S, Bro R (2003) Tracing dissolved organic matter in aquatic environments using a new approach to fluorescence spectroscopy. Marine Chemistry 82: 239-254.
- Zsolnay á, Baigar E, Jimenez M, Steinweg B, Saccomandi F (1999) Differentiating with fluorescence spectroscopy the sources of dissolved organic matter in soils subjected to drying. Chemosphere 38: 45–50.
- OhnoT (2002) Fluorescence inner-filtering correction for determining the humification index of dissolved organic matter. Environ Sci Technol 36: 742-746.
- Birdwell JE, Engel AS (2010) Characterization of dissolved organic matter in cave and spring waters using UV-vis absorbance and fluorescence spectroscopy. Organic Geochemisty 41: 270-280.
- Matthews BJH, Jones NK, Theodorou, Tudhope AW, et al. (1996) Excitation-emission-matrix fluorescence spectroscopy applied to humic acid bands in coral reefs. Marine Chemistry 55: 317-332.
- Parlanti P, Worz K, Geoffroy L, M. Lamotte (2000) Dissolved organic matter fluorescence spectroscopy as a tool of estimate biological activity in a coastal zone submitted to anthropogenic inputs. Organic Geochemistry 31: 1765-1781.
- Baghoth SA, Sharma Sk, Ary GL (2011) Tracking natural organic matter (NOM) in a drinking water treatment plant using fluorescence excitation emission matrices and PARADAC. Water Research 45: 797-809.
- Yamashita Y, Rudolf J, Nafamitsu M, Eiichiro Tanoue (2008) Assessing the dynamics of dissolved organic matter (DOM) in coastal environments by excitation emission matrix fluorescence and parallel factor analysis (EEM-PARAFAC). Limnol Oceanogr 53: 1900-1908.
- Stedmon CA, Markager S (2005) Resolving the variability in dissolved organic matter fluorescence in a temperate estuary and its catchment using PARAFAC analysis. Limnology & Oceanography 50: 686-697.
- Stedmon CA, Markager S (2005) Tracing the production and degradation of autochthonous fractions of dissolved organic matter using fluorescence analysis. Limnology & Oceanography 50: 1415-1426.
- Murphy KR, Stedmon CA, Waite TD, Ruiz GM (2008) Distinguishing between terrestrial and autochonous organic matter sources in marine environments using fluorescence spectroscopy. Marine Chemistry 108: 40-58.
- Chen RF, Bada JL (1992) The fluorescence of dissolved organic matter in seawater. Marine Chemistry 37: 191-221.
- Huguet A, Vacher L, Relexans S, Saubusse S, Froidefond JM, et al. (2009) Properties of fluorescent dissolved organic matter in the Gironde Estuary. Organic Geochemistry 40: 706-719.
- Yamashita Y, Tanoue E (2003) Chemical characterization of protein-like fluorophores in DOM in relation to aromatic amino acids. Marine Chemistry 82: 255-271.
- Mayer LM, Schik LL, Loder TC (1999) Dissolved protein fluorescence in two Maine estuaries. Marine Chemistry 64: 171-179.
- Vodeack A, Hoge FE, Swift RN (1995) The in situ and airborne fluorescence measurements to determine UV absorption coefficients and DOC concentrations in surface waters. Limnology &Oceanography 40: 411-415.
- Determann S, Lobbes J, Reuter R, Jürgen Rullkötter (1998) Ultraviolet fluorescence excitation and emission spectroscopy of marine algae and bacteria. Marine Chemistry 62: 137-156.
- Ji NY, Zhao WH, Wang JT, Cui X, Miao H (2006) Fluorescence characteristics of dissolved organic matter during algal bloom in Jiaozhou Bay. Environmental Science 27: 257-262.
- Ferrari GM (2000) The relationship between chromophoric dissolved organic matter and dissolved organic carbon in the European Atlantic coastal area and in the West Mediterranean Sea (Gulf of lions), Marine Chemistry 70: 339-357.
Relevant Topics
- Algal Blooms
- Blue Carbon Sequestration
- Brackish Water
- Catfish
- Coral Bleaching
- Coral Reefs
- Deep Sea Fish
- Deep Sea Mining
- Ichthyoplankton
- Mangrove Ecosystem
- Marine Engineering
- Marine Fisheries
- Marine Mammal Research
- Marine Microbiome Analysis
- Marine Pollution
- Marine Reptiles
- Marine Science
- Ocean Currents
- Photoendosymbiosis
- Reef Biology
- Sea Food
- Sea Grass
- Sea Transportation
- Seaweed
Recommended Journals
Article Tools
Article Usage
- Total views: 16021
- [From(publication date):
February-2014 - Dec 19, 2025] - Breakdown by view type
- HTML page views : 11173
- PDF downloads : 4848