Research Article Open Access
Investigation on Biological Properties of Dental Implant by Ce-TZP/Al2O3/ HA Bio-nano-composites
M. Yousefpour1,2,3, N. Askari2,3*, H. Abdollah-Pour3, A. Amanzadeh3 and N. Riahi21Materials Science and Engineering Department, Semnan University, Semnan, Iran
2Materials Group, Engineering Department, Imam Khomeini International University, Qazvin, Iran
3National Cell Bank of Iran (NCBI), Pasteur Institute of Iran, Tehran, Iran
- Corresponding Author:
- N. Askari
Materials Group, Engineering Department
Imam Khomeini International University, Qazvin, Iran
Tel: +98 09127877567
Fax: +98 0281-3780073
E-mail: nayereh_askari_20@yahoo.com
Received date: December 28, 2010; Accepted date: April 10, 2011; Published date: May 05, 2011
Citation: Yousefpour M, Askari N, Abdollah-Pour H, Amanzadeh A, Riahi N (2011) Investigation on Biological Properties of Dental Implant by Ce-TZP/Al2O3/HA Bio-nano-composites. J Biotechnol Biomaterial 1:105. doi:10.4172/2155-952X.1000105
Copyright: © 2011 Yousefpour M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Biotechnology & Biomaterials
Abstract
An attempt is made to convert the calcium carbonate skeleton of widely available garden snail shell (Helix aspersa) to hydroxyapatite based bioceramics. The snail shell was found to decompose within 850°C to all the carbonate phases. The calcined snail shells were then treated with acids followed by different chemicals in ammoniacal media maintaining proper stoichemetry to produce fine Hydroxyapatite (HAP) as filter cake with Ca/P molar ratio of 1.67. The dried HAP powder was extremely pure with specific surface area of 15m2/g. The different characterization techniques were adopted both for calcined snail shell and HAP synthesized like X-ray Diffraction (XRD), Thermal Analysis (DTA/TGA) Figure 1, Fourier Transform Infrared Spectroscopy (FTIR) and Scanning Electron Microscopy (SEM). The surface area and the particle size, of the HAP powder prepared by chemical precipitation route, were also determined by BET and Malvern particle size analyzer respectively. The synthesized powder was soaked in stimulated body fluid (SBF) medium for various periods of time in order to evaluate its bioactivity. The changes of the pH of SBF medium were measured. High bioactivity of prepared HAP powder due to the formation of apatite on its surface was observed.
Keywords
Biocompatibility; Nanocomposite; Cellular response; Osteoblast
Introduction
Hydroxyapatite (HA, Ca10 (PO4)6(OH)2), Zirconia (ZrO2) and Alumina (Al2O3) are known as important implant materials [1-3]. HA has often been considered as a candidate for use in load -bearing applications due to bonds and promotes the new bone formation necessary for implant osseointegration to bone directly. However, its poor mechanical properties have limited its application to load-bearing parts [4-11].
Alumina and zirconia have excellent biocompatibility. The main advantages of Al2O3 is its high hardness and wear resistance, while ZrO2 exhibits higher strength and fracture toughness and the composite consisting of the two has higher ductility and fracture toughness than each constituent [12]. Some interest has been paid to composites comprising of hydroxyapatite and another phase of high mechanical properties and bio-inertness, such as alumina and zirconia [13]. Cerium oxide or Ceria is used to stabiles the tetragonal polycrystalline structure of zirconia at room temperature, the product called Ceria-partially stabilized zirconia (Ce-TZP) [15]. Ce-TZP/Al2O3is a composite in which Ce-TZP particles are added to Al2O3 matrix as the second phase to modify the fracture behavior of composite because, it has high fracture toughness [14]. In additional, Ce-TZP/Al2O3 nano-composites could be superior to Y-TZP in clinical use [16,17]. The biocompatibility of zirconia-alumina (ZA) nano-composites in load-bearing applications such as dental/ orthopedic implants was significantly enhanced by the addition of bioactive HA [4].
In this work, the effect of HA is studied on biological properties of Ce-TZP/Al2O3. In order to, bio-nano-composites were fabricated by mechanical milling of pre-synthesized nano-scaled constituents, and cold pressing and sintering Phase analysis; strength and biological properties of produced composites were evaluated by different characterization methods.
Experimental
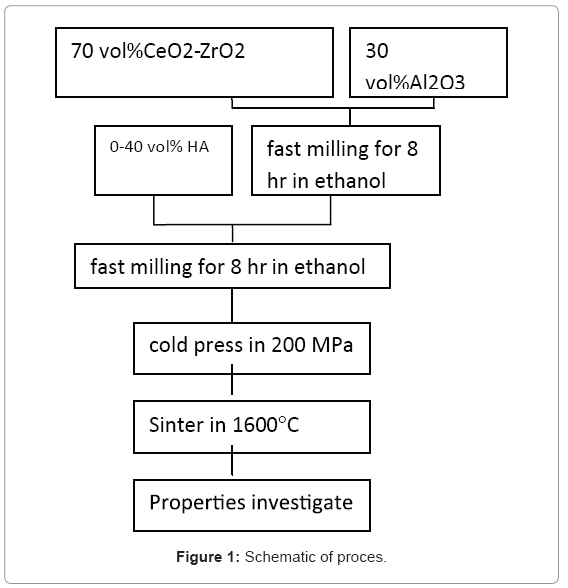
For this study, we used of Al2O3, HA and ZrO2 nanopowders synthesized by sol-gel, chemical precipitation and hydrothermal methods (with particle sizes 112,9,12 nm respectively). The process Schematic is given in Figure 1.
As is observed ZA nano -composite powders were prepared by fast milling machine (sharifi co. iran) of 70 vol% 10Ce-TZP and 30 vol% Al2O3 powder in ethanol for 8 hr. Obtained powder was mixed with HA (up to 40vol%) in ethanol by fast milling machine again for 8 hr. Mixed slurry was dehydrated, crushed and sieved, and uniaxial cold pressed at a pressure of 200 MPa by universal test machine (Zwick roll-Z100). Cold pressed samples were sintered at 1600°C for 2 hr by electrical furnace (Vecstar). The density of the specimens was measured by the Archimedes method [ASTM B311-08]. Crystalline phase analysis was performed by XRD (Bruker model D8). The bending strength was measured by means of three- point bend test with universal test machine (Zwick roll-Z100) [ASTM C1674-08].
In order to investigation the proliferation behavior on the ZA-HA composites, the samples used for cell tests were prepared from disc specimens with 10mm diameter that after polishing both sides with diamond slurries down to 1 mm, the specimens were washed and sterilized at 121°C for 20 minutes. The samples were placed in a 24 well tissue culture plate and seeded with 25µl of the 3x104 cells/cm2 MG63 (National Cell Bank of Iran-no NCBI C.) cell density suspension. Cells were allowed to adhere for 2 hours then flooded with 1 ml of Dulbecco's modified Eagle's medium (DMEM, Gibco-USA), 2mM L-glutamine (Gibco), 100 IU/ml of penicillin and 100µg/ml of streptomycin (Gibco) supplemented with 10% fetal bovine serum (FBS, Gibco-USA) and placed back into the incubator. The fabricated specimens, including pure HA as a positive control, were placed into a 24-well plate, which was followed by plating on all the discs.
The cells were cultured for 7 days in a humidified incubator under an atmosphere of 5% CO2 at 37°C. After detaching the cells from the specimens with a trypsin-EDTA solution (0.25% trypsin, 0.05% EDTAGibco) and staining with 0.4% trypan blue, the living cells were counted using a hemocytometer (Paul Marienfeld GmbH & Co-Germany). Each set of tests was performed in triplicate.
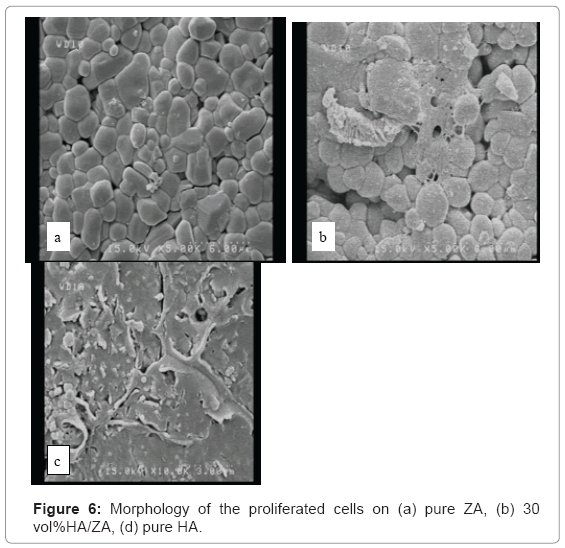
For observation the morphology of the proliferated cells with FESEM (Hitachi s-4160) cells were cultured for 5 days as above then fixation with glutaraldehyde(2.5%),dehydration with graded ethanol (50%,60%,70%,80%,90%and 100%).
The results were expressed as means with standard errors. The data were then analyzed by one-way ANOVA, followed by the post hoc paired Student's two-tailed t-test. The significance levels were notified significant (*, if p< 0.05) and highly significant (**, if p<0.01) in the tables.
Results
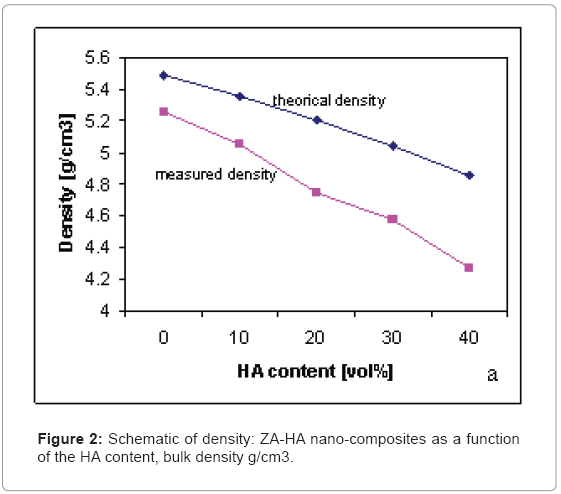
Figure 2 shows the densities of the ZA nano-composites containing various amounts of HA. Compared to the theoretical density, which was calculated by the rule of mixture, the density of the specimens with a low HA content almost matched with the theoretical value. However, as the amounts of HA were increased, the discrepancy between the measured and theoretical densities increased.
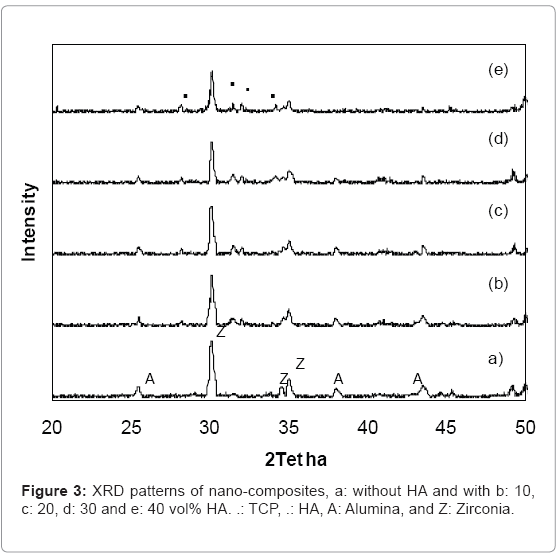
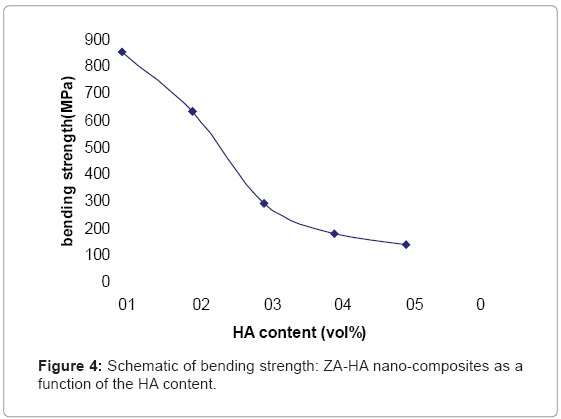
Figure 3, illustrates the XRD patterns of different composite materials. When no HA was added, only peaks corresponding to ZrO2 and Al2O3 are present, as shown in Figure (3-a). With addition of 10, 20, 30 and 40 vol% HA (Figure (3-b-c-d-e), respectively), as well as the peaks corresponding to HA, TCP peaks, were also detected, that TCP component was originated from the reaction between HA and ZrO2. The bending strengths of the nanocomposites are shown in Figure 4, as the HA content was increased up to 40vol% the strength decreased of 850 to 137 MPa.
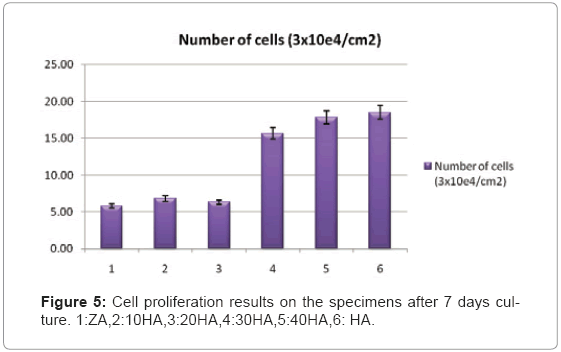
Biocompatibility tests are in progress. Figure 5 illustrates Cell proliferation results on the specimens after 7 days culture. The number of cells was significantly increased by the addition of 10% HA to the ZA (ANOVA, p<0.05). The number of cells increased steadily with further addition of HA (ANOVA, p±0.001). When 40% HA was added to the ZA, the proliferation rate on that specimen became comparable to that on the pure HA means of wells±S.D.
Figuer 6 show morphology of the proliferated cells on the specimens. As was observed specimens with30vol%HA show most of the cell proliferation.
| Mean | |||
|---|---|---|---|
| Comparison | Difference | q | P value |
| Column A vs Column F | -12.667 | 20.121*** | P<0.001 |
| Column A vs Column E | -12.000 | 19.062*** | P<0.001 |
| Column A vs Column D | -9.833 | 15.620*** | P<0.001 |
| Column A vs Column C | -1.167 | 1.853 ns | P>0.05 |
| Column A vs Column B | -1.000 | --- ns | P>0.05 |
| Column B vs Column F | -11.667 | 18.533*** | P<0.001 |
| Column B vs Column E | -11.000 | 17.474*** | P<0.001 |
| Column B vs Column D | -8.833 | 14.032*** | P<0.001 |
| Column B vs Column C | -0.1667 | --- ns | P>0.05 |
| Column C vs Column F | -11.500 | 18.268*** | P<0.001 |
| Column C vs Column E | -10.833 | 17.209*** | P<0.001 |
| Column C vs Column D | -8.667 | 13.767*** | P<0.001 |
| Column D vs Column F | -2.833 | 4.501** | P<0.01 |
| Column D vs Column E | -2.167 | 3.442* | P<0.05 |
| Column E vs Column F | -0.6667 | 1.059 ns | P<0.05 |
Discussion
As a bio-inert implant material, ZA nano-composite is considered to have sufficient strength. However, its biocompatibility such as satisfactory osseointegration and faster bone regeneration has to be improved by the addition of calcium phosphates such as HA, and TCP, [1,4]. TCP component was originated from the reaction between HA and ZrO2. Both HA and TCP are formed of calcium and phosphate component, which are essential to satisfactory osseointegration and faster bone regeneration to be achieved [4,7]. Just as shown in the XRD patterns ,intensities of the HA and TCP peaks increased steadily with increasing HA content.
Conclusion
Hydroxyapatite (HA)-zirconia-alumina (ZA) bio-nano-composites were produced by mechanical blending of separately synthesized nano-scaled powder. Composite materials were characterized by phase analysis and cellular response test. From mechanical and biological properties evaluation of the 10Ce-TZP/Al2O3/HA bio nano composites, specimens with 30 vol% of HA, was found to be the optimal composition for biological applications.
Acknowledgement
Authors of paper, gratefully thank Semnan University, Qazvin Imam Khomeini International University and Pasteur Institute of Iran for the research support.
References
- Rapacz-Kmita A, Slosarczyk A, Paszkiewicz Z, Paszkiewicz C (2004) Phase stability of hydroxyapatite-zirconia(HAP-ZrO2) composites for bone replacement. J Molecular Structure 704: 333-340.
- Benzaid R, Chevalier J, Saâdaoui M, Fantozzi G, Nawa M, et al. (2008) Fracture toughness,strength and slow crack growth in a ceria stabilized zirconiaalumina nanocomposite for medical application. Biomaterials 29: 3636-3641.
- Sung YM, Kim DH (2003) Crystallization characteristics of yttria-stabilized zirconia/ hydroxyapatite composite nano powder. J Crystal Growth 254: 411-417.
- Kong YM, Bae CJ, Lee SH, Kim HW, Kim HE (2005) Improvement in biocompatibility of ZrO2- Al2O3nanocomposite by addition of HA. Biomaterial 26: 509- 517.
- Miao X, Chen Y, Guo H, Khor KA (2004) Spark plasma sintered hydroxyapatiteyttria stabilized zirconia composites. J Ceramics International 30: 1793-1796.
- Yoshida K, Hashimoto K, Toda Y, Udagawa S, Kanazawa T (2006) Fabrication of structure-controlled hydroxyapatite/zirconia composite. J Eur Cer So 26: 515-518.
- Rapacz-Kmita A, Slósarczyk A, Paszkiewicz Z (2006) Mechanical properties of HAP-ZrO2 composites. J Eur Cer So 26: 1481-1488.
- Khalil KA, Kim SW, Kim HY (2007) Consolidation and mechanical properties of nanostructured hydroxyapatite-( ZrO2+3mol%Y2O3)bioceramics by highfrequency induction heat sintering. J Material science and ingineering A 456: 368-372.
- Chiu CY, Hsu HC, Tuan WH (2007) Effect of zirconia addition on the microstructural evolution of porous hydroxyapatite. J Ceramics International 33: 715-718.
- Evis Z, Doremus RH (2007) Effect of YF3 on hot-pressed hydroxyapatite and monoclinic zirconia composites. Materials Chemistry and Physics 105: 76-79.
- Rapacz-Kmita A, Slósarczyk A, Paszkiewicz Z (2005) Hap- ZrO2 composite coatings prepared by plasma spraying for biomedical applications. J Ceramics International 31: 567-571.
- Santos1 C, Souza RC, Daguano JKMF, Elias CN, Rogero SO (2007) Development of ZrO2- Al2O3bioceramic composites. Congresso brasileiro de ceramica.
- Li W, Gao L (2003) Fabrication of HAP- ZrO2(3Y) nanocomposite by SPS. Biomaterials 24: 937-940.
- Tsukuma K, Shimada M (1985) Strength, fracture toughness and Vickers hardness of CeO2-stabilized tetragonal ZrO2 polycrystals(Ce-TZP). J Materials Science 20: 1178-1184.
- Yang G, Li JC, Wang GC, Min SL, Chen TC, et al. (2006) Investigation on strengthening and toughening mechanisms of Ce-TZP/Al2O3 nano-composites. J Metallurgical and Materials A 37: 1969-1975.
- Fischer J, Stawarczyk B (2007) Compatibility of machined Ce-TZP/Al2O3 nanocomposite and a veneering ceramic. J Dental materials 23: 1500-1505.
- Bhaduri S, Bhaduri SB (1997) Enhanced low temperature toughness of Al2O3- ZrO2 nano/nano composites. J Nanostructure Materials 8: 755-763.
Relevant Topics
- Agricultural biotechnology
- Animal biotechnology
- Applied Biotechnology
- Biocatalysis
- Biofabrication
- Biomaterial implants
- Biomaterial-Based Drug Delivery Systems
- Bioprinting of Tissue Constructs
- Biotechnology applications
- Cardiovascular biomaterials
- CRISPR-Cas9 in Biotechnology
- Nano biotechnology
- Smart Biomaterials
- White/industrial biotechnology
Recommended Journals
Article Tools
Article Usage
- Total views: 15268
- [From(publication date):
May-2011 - Dec 21, 2025] - Breakdown by view type
- HTML page views : 10515
- PDF downloads : 4753