Research Article Open Access
Development of HPTLC Method for Determination of Amlodipine Besylate and Olmesaratan Medoxomil Using Human Plasma by Liquid Liquid Extraction
Ambadas. R Rote* and Sadhana.K .KandeDepartment of Pharmaceutical Chemistry, M. G. V.’s Pharmacy College, Panchavati, Mumbai - Agra Road, Nashik- 422003, Maharashtra, India
- *Corresponding Author:
- Ambadas.R.Rote
Department of Pharmaceutical Chemistry
M. G. V.’s Pharmacy College, Panchavati
Mumbai - Agra Road, Nashik- 422003, Maharashtra, India
Tel: +91 9579574199
Fax: +91 2532511931
E-mail: roteambadas@gmail.com
Received date: November 13, 2011; Accepted date: December 21, 2011; Published date: December 23, 2011
Citation: Rote AR, Kande SK (2011) Development of HPTLC Method for Determination of Amlodipine Besylate and Olmesaratan Medoxomil Using Human Plasma by Liquid Liquid Extraction. J Anal Bioanal Tech 2:128. doi: 10.4172/2155-9872.1000128
Copyright: © 2011 Rote AR, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Analytical & Bioanalytical Techniques
Abstract
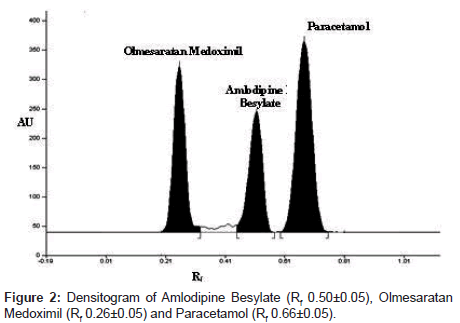
A simple, sensitive, rapid and economic high performance thin layer chromatographic method has been developed for determination of amlodipine besylate and olmesaratan medoxomil in human plasma by liquid liquid extraction using paracetamol as an internal standard. The plasma sample was extracted using mixture of methanol: acetonitrile (3.0: 0.1). A concentration range from 500-3000 ng/spot of amlodipine besylate and olmesaratan medoxomil was used for calibration curve respectively. The percent recovery of amlodipine besylate and olmesaratan medoxomil was found to be 90.41 and 90.64 % respectively. The mobile phase consists of chloroform: methanol (9:1, v/v). Densitometric analysis was carried out at wavelength 254 nm. The Rf values for amlodipine besylate, olmesaratan medoxomil and paracetamol was 0.50 ± 0.05, 0.26 ± 0.05 and 0.66 ± 0.05 respectively. The stability of amlodipine besylate and olmesaratan medoxomil in plasma was confirmed during three freeze-thaw cycles at ‘-20°C’, on bench during 24 h and post preparative during 48 h. The proposed method was validated statistically and by performing recovery study for determination of amlodipine besylate and olmesaratan medoxomil in human plasma by liquid liquid extraction.
Keywords
HPTLC; Amlodipine besylate; Olmesaratan medoxomil; Human plasma; Liquid liquid extraction
Introduction
Amlodipine besylate (3-ethyl 5-methyl 2-[(2-aminoethoxy) methyl]-4-(2-chlorophenyl)-6-methyl-1,4-dihydropyridine-3,5-dicarboxylate, as shown in Figure 1) is antihypertensive agent, vasodilator agent and calcium channel blocker [1,2]. Olmesaratan medoxomil is (5-methyl-2-oxo-2H-1,3-dioxol-4-yl)methyl 4-(2-hydroxypropan-2-yl)- 2-propyl-1-({4-[2-(2H-1,2,3,4-tetrazol-5-yl)phenyl]phenyl}methyl)- 1H-imidazole-5 carboxylate [3,4]. A dosage form of amlodipine besylate with olmesaratan medoximil in combination is more effective than either drug alone in the management of hypertension. For the determination of amlodipine besylate in human plasma, methods based on UV-spectrophotometric method, [5], stability indicating RP-HPLC [6] has been reported. Similarly, a number of methods for the determination of omesaratan medoximil in human plasma have been developed based on liquid chromatography coupled to tandem mass spectrometry [7], RP-LC and HPTLC [8], spectrophotometric estimation [9], RPHPLC with diode array detection [10] has been repotred. No HPTLC method is reported for the simultaneous estimation of amlodipine besylate and olmesaratan medoxomil in human plasma. This paper describes the development and validation of method to simultaneously quantify amlodipine besylate and olmesaratan medoxomil in human plasma using HPTLC by liquid liquid extraction.
Materials and Methods
Instrumentation
HPTLC Camag with precoated silica gel plate 60F254 (20cm×10 cm) 250μm thicknesses (E. Merck, Darmstadt, Germany) was used as stationary phase. Sample application was done by using Camag 100μl syringe and Camag Linomat V applicator. The sample was sprayed in the form of narrow bands of 8 mm length at a constant rate 2μl/s. Linear ascending development was carried out in 20 cm×10cm twin trough glass chamber (Camag, Muttenz, Switzerland). The densitometric scanning was performed by using Camag TLC scanner III supported by win CATS software (V1.4.2.8121 Camag). Evaluation of densitogram was done by using ratio of peak areas of drugs with internal standard.
Chemicals
Amlodipine besylate and olmesaratan medoximil (Ajanta Pharmaceuticals, Mumbai, Maharashtra, India) and paracetamol (Kirti Pharmachem Ltd., Sinner, Maharashtra, India) were received having 99.82%, 98.75% and 100.2 % purity respectively. They were used as such by without checking their purity. The HPLC grade methanol and Analytical Reagent grade Chloroform and Methanol was purchased from s d Fine Chem. Ltd., Mumbai, India. Human plasma used for research work was supplied by Arpan Blood Bank, Nashik, Maharashtra, India.
Preparation of stock solution and working standard solution
Stock solutions 1.0 mg/ml each of amlodipine besylate, olmesaratan medoxomil and paracetamol were prepared in methanol.
Preparation of plasma sample
In a 15ml centrifuge tube 0.5, 1, 1.5, 2, 2.5, 3 μl of working stock solution of amlodipine besylate and olmesaratan medoxomil was added to drug-free plasma to provide calibration standards of 500, 1000, 1500, 2000,2500, 3000 ng/ml and 1000 ng/ml of paracetamol (internal standard)was kept constant. The quality control (QC) samples were prepared in plasma concentration range 1000, 2000, 3000 ng. Protein precipitation and extraction was carried out by using mixture of methanol and acetonitrile (3.0:0.1ml) on by vigorous vortex using remi mixer for 1min and centrifuged at 5,000 rpm at 10min. The organic phase was recovered and evaporated to dryness on a hot plate. The residual mass was reconstituted with 1 ml methanol. The analysis was carried on HPTLC.
Chromatographic condition
Mobile phase was selected as mixture of chloroform and methanol in the ratio of (9:1, v/v) for the development of plates. Time for chamber saturation was optimized to 20 min. The length of chromatographic development was 70 mm. The densitometric scanning was performed at 254 nm.
Method validation
The method was validated for sensitivity, selectivity, precision, accuracy, linearity, recovery and stability. The validation of the method was based on FDA guidelines and on standard bioanalytical method validation recommendation. The selectivity of method was investigated by analyzing six blank plasma samples. Each blank sample was tested for interference using proposed extraction procedure. Five replicate of three QC sample low, mid and high were used for the determination of precision and accuracy. Intra-day and inter-day precision were carried out. Precision and accuracies showed 15% relative standard deviation (RSD) from nominal values, at LLOQ these were both 20%. The recovery of amlodipine besylate and olmesaratan medoxomil was calculated by comparison of the peak areas of low, mid, and high QC sample (1000, 2000, 3000ng/ml) prepared in plasma (extracted) with unextracted amlodipine besylate and olmesaratan medoxomil respectively.
Stability experiments were undertaken to detect degradation of amlodipine besylate and olmesaratan medoxomil under certain condition. Freeze–thaw stability was determined at two QC concentrations (low, high) after freezing (−20°C) and thawing for three cycles and compared with nominal value. Bench-top stability was assessed for low and high QC samples by comparing with nominal value which stored at room temperature for 12 h. The effect of storage within the auto-sampler was assessed by comparing QC samples injected immediately after preparation with those left in auto-sampler for 48 h.
Result and Discussion
Extraction procedure optimization
One of the most difficult task during the method development was to achieve a high and reproducible recovery from the solvent which is used for extraction of the drug and also difficult to select such single extracting solvent from which both the drugs are extracted. Different solvents were tried for the extraction of amlodipine besylate and olmesaratan medoxomil from human plasma. First 5 ml each of chloroform and ethyl acetate was tried for the precipitation of plasma but the recovery was very less. It gave 50- 70% of recovery because of less precipitation of protein from plasma. At the last methanol was tried and 60–80% of recovery was obtained. It was found that the addition of acetonitrile (0.1 ml) increases the precipitation of protein and also the recovery which is reproducible and high as compare to other solvents. So methanol: acetonitrile (3.0: 0.1) was kept as final solvent for extraction of amlodipine besylate and olmesaratan medoxomil.
Optimization of chromatographic condition
Initially plane solvents like methanol, ethyl acetate, chloroform, toluene, acetone, formic acid were tried. Then chloroform and acetone in the ratio of (7:3, v/v) was tried but no proper resolution obtained. Then I have increased by 1ml showing good resolution but amlodipine besylate show greater Rf value. Lastly by increasing the concentration of chloroform from 7 to 9 ml and addition methanol instead of acetone shows good resolution with symmetrical peaks of mlodipine esylate, lmesaratan edoximil and paracetamol were obtained. Finally mobile phase consisted of chloroform: methanol (9: 1v/v) was used. The Rf values for amlodipine besylate, olmesaratan medoximil and paracetamol was found to be 0.50 ± 0.05, 0.26 ± 0.05, and 0.66 ± 0.05 respectively. Well defined spots were obtained by prewashing the plate using methanol followed by activating at 120°C for 20min. Chamber was saturated with mobile phase for 30 min at room temperature, which gave reproducible Rf values for amlodipine besylate, olmesaratan medoximil and paracetamol respectively (Figure 2).
Calibration curve
The six point calibration curve was constructed by plotting the peak response ratio of amlodipine besylate to paracetamol and olmesaratan medoximil to paracetamol in plasma. Correlation coefficients are 0.9969 and 0.9949 for amlodipine besylate and olmesaratan medoximil respectively. Linearity were found over the range 0.5-3.0 μl/ml for amlodipine besylate and olmesaratan medoximil respectively. The lower limit of quantification was defined as lowest concentration in the calibration curve. The amlodipine besylate and olmesaratan medoximil can be determined at LLOQ 0.5μl/ml.
Recovery
Absolute recovery was calculated by comparing peak areas obtained from freshly prepared sample extracted with unextracted standard solutions of the same concentration. Recovery data was determined in triplicates at three concentrations (low, mid, high) as recommended by the FDA guidelines [11]. The recovery of amlodipine besylate and olmesaratan medoximil for HPTLC at the three concentrations 1000, 2000, 3000 ng/spot were found to be 85.22, 96.61 and 89.40% and 70.45, 84.86 and 96.43% respectively (Table 1).
| Sr.no | Concentration | Recovery | %RSD | |||
| AML | OLM | AML | OLM | AML | OLM | |
| 1 | 1000 | 1000 | 85.22 % | 70.45 % | 1.89 % | 1.65% |
| 2 | 2000 | 2000 | 96.61 % | 84.86% | 2.02% | 2.26% |
| 3 | 3000 | 3000 | 89.40% | 96.43% | 2.56% | 3.25% |
Table 1: Result of recovery of amlodipine besylate and olmesaratan Medoxomil in human plasma.
Precision and accuracy
Precision of the method was determined by repeatability (intraday) and intermediate precision (inter-day) and accuracy for set of quality control (QC) sample (low, mid, high) in replicate (n = 5).The inter-day and intra-day precision and accuracy for the amlodipine besylate and olmesaratan medoximil evaluated by assaying the QC samples (low, mid, high) (n = 5) in (%RSD). In this assay the intra-run precision was found to be in the range of 2.39-6.03% and 2.44-17.28% and the inter-run precision was 5.46-8.49% and 1.06-6.78%. The accuracy was within 4.59-10.63%. The above values were within the acceptable range, it shows that the methods are accurate and precise. The low percent relative standard deviation (%RSD) and percent relative error (%RE) were within the acceptable limit. The results of inter-day, intra-day precision and accuracy for the amlodipine besylate and olmesaratan medoximil are shown in Table 2.
| Precision | Concentration(ng) | %RSD | %RE | |||
| AML | OLM | AML | OLM | AML | OLM | |
| INTRA | 1000 | 1000 | 12.76 | 18.46 | 0.94 | 0.04 |
| 2000 | 2000 | 14.37 | 19.70 | 7.52 | 1.9 | |
| 3000 | 3000 | 9.64 | 14.39 | 5.84 | 0.56 | |
| INTER | 1000 | 1000 | 11.69 | 16.17 | 17.90 | 0.07 |
| 2000 | 2000 | 18.62 | 18.13 | 19.63 | 5.23 | |
| 3000 | 3000 | 14.86 | 13.74 | 17.07 | 5.98 | |
Table 2: Results of precision of amlodipine besylate and olmesaratan medoxomil in human plasma.
Sensitivity and selectivity
Selectivity or specificity should be assessed to show that the intended analytes are measured and that their quantitation is not affected by the presence of the biological matrix. For HPTLC method by LLE there is no interference of the biological matrix in the quantitation of amlodipine besylate and olmesaratan medoximil. There were no changes in retention factor of amlodipine besylate and olmesaratan medoximil, the method is selective. Sensitivity of the method is defined as the lowest concentration that can be measured with an acceptable limit of accuracy and precision which is lower than 20%. The accuracy and precision at lower limit of quantitation (LLOQ) analyzed by using five replicate (n = 5) of the sample at the LLOQ concentration. The accuracy is determined by %RE at this LLOQ concentration. The lower limit of quantitation which could be detected were found to be 500 ng/ spot with %RE = 15.06, 2.92 % and %RSD= 2.11, 11.28 % for amlodipine besylate and olmesaratan medoximil is within acceptable limit.
Analysis speed
In case of HPTLC 18 spot can be applied on one plate so less time consuming.
Stability
In bench top stability the low and high QC sample were thawed and left at room temperature for 12 h. Comparison of the results for QC sample (low and high) with freshly prepared stock solution showed that there was no significant difference between response of freshly prepared solution and sample of amlodipine besylate and olmesaratan medoximil after 12 h. Freeze–thaw stability was determined after two freezes–thaw cycles for three replicate of low and high QC sample. The samples were stored at −20°C temperature for 24 h. Then thaw at room temperature. No significant difference between freeze–thaw sample and freshly prepared sample was observed. The result of stability experiments shows that no significant degradation occurred at ambient temperature for 48 h for post-preparative stability. Results of stability for HPTLC method are shown in Table 3.
| Parameters | Concentration | S.D | %RSD | |||
| AML | OLM | AML | OLM | AML | OLM | |
| Freez thaw stability Low |
1000 | 1000 | 0.007 | 0.021 | 0.70 | 2.19 |
| High | 3000 | 3000 | 0.016 | 0.014 | 1.65 | 1.46 |
| Bench top stability Low |
1000 | 1000 | 0.027 | 0.013 | 2.78 | 1.36 |
| High | 3000 | 3000 | 0.012 | 0.063 | 1.25 | 6.35 |
| post-preparative stability Low |
1000 | 1000 | 0.0079 | 0.002 | 0.79 | 0.22 |
| High | 3000 | 3000 | 0.019 | 0.007 | 1.93 | 0.72 |
Table 3: Stability study of amlodipine besylate and olmesaratan Medoxomil in human plasma.
Conclusion
The proposed HPTLC method for the estimation of amlodipine besylate and olmesaratan medoximil in human plasma by LLE is selective and sensitive. The method is economical and faster than earlier published methods. In future these method can be used for bioequivalence study.
Acknowledgements
The authors are thankful to the Management and Principal Dr. Rajendra S. Bhambar, M. G. V.’s Pharmacy College, Nashik for providing necessary facilities for the research work. The authors are also thankful to Ajanta Pharmaceuticals, Mumbai, Maharashtra, India for providing amlodipine besylate and olmesaratan medoximil and paracetamol by Kirti Parma Sinner, Maharashtra, India as a gift sample for the research work and Arpan Blood Bank, Nashik for providing human plasma.
References
- Indian Pharmacopeia (2007) Government of Indian. The Controller of Publications Delhi 2: 714-715.
- British Pharmacopeia (2007) Government of Indian. The Controller of Publications, Delhi 1: 132-133.
- Olmesartan Medoximil
- Olmesartan Medoximil
- Kasture AV, Ramteke M (2006) Simultaneous UV-spectrophotometric method for the estimation of atenolol and amlodipine besylate in combined dosage form. Indian J Pharm Sci 68: 394-396
- Naidu KR, Kale UN, Shingare MS (2005) Stability indicating RP-HPLC method for simultaneous determination of amlodipine and benazepril hydrochloride from their combination drug product. J Pharm Biomed Anal 39: 147-155.
- Liu D, Hu P, Matsushima N, Li X, Li L, et al. (2007) Quantitative determination of olmesartan in human plasma and urine by liquid chromatography coupled to tandem mass spectrometry. J Chromatogr B 856: 190-197.
- Bari PD, Rote AR (2009) RP-LC and HPTLC Methods for the Determination of Olmesartan Medoxomil and Hydrochlorothiazide in Combined Tablet Dosage Forms. Chromatographia 69: 1469-1472.
- Rote AR, Bari PD (2010) Spectrophotometric Estimation of Olmesartan Medoxomil and Hydrochlorothiazide in Tablet. Indian J Pharm Sci 72: 111-113.
- Ritesh N. Sharma, Shyam S Pancholi (2010) RP-HPLC-DAD method for determination of olmesartan medoxomil in bulk and tablets exposed to forced conditions. Acta pharmaceutica Zagreb Croatia 60: 13-24
- US Department of Health and Human Services (2001) FDA Guidance for Industry: bioanalytical Method validation, US Department of Health and Human Services, US FDA.
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 15209
- [From(publication date):
December-2011 - Dec 19, 2025] - Breakdown by view type
- HTML page views : 10422
- PDF downloads : 4787