Research Article Open Access
Antimicrobial Effect of Coated Leather Based on Silver Nanoparticles and Nanocomposites: Synthesis, Characterisation and Microbiological Evaluation
Isabel Maestre-López M, Federico Payà-Nohales J, Natalia Cuesta-Garrote, Francisca Arán-Ais, Miguel Ángel Martínez-Sánchez, César Orgilés-Barceló and Marcelo Bertazzo*INESCOP, Footwear Technological Institute, 03600 Elda (Alicante), Spain
- Corresponding Author:
- Marcelo Bertazzo
INESCOP, Footwear Technological Institute
03600 Elda (Alicante), Spain
Tel: +34 965 39 52 13
Fax: +34 965 38 10 45
E-mail: mbertazzo@inescop.es
Received date: December 29, 2014; Accepted date: January 30, 2015; Published date: February 05, 2015
Citation: Isabel Maestre-López M, Federico Payà-Nohales J, Cuesta-Garrote N, Arán-Ais F, Martínez-Sánchez MÁ, et al. (2015) Antimicrobial Effect of Coated Leather Based on Silver Nanoparticles and Nanocomposites: Synthesis, Characterisation and Microbiological Evaluation. J Biotechnol Biomater 5:171. doi:10.4172/2155-952X.1000171
Copyright: © 2015 Isabel Maestre-López M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Biotechnology & Biomaterials
Abstract
This paper describes the assessment of the antimicrobial effect of leathers employed for footwear production, which were obtained by different tanning techniques and treated with silver nanoparticles emulsions synthesised from different reducing agents (sodium borohydride, gelatine/glucose and Aloe vera extract), as well as the assessment of the silica nancomposites obtained from said nanoparticles. After the characterisation of silver nanoparticles and nanocomposites, the emulsions were applied onto different leather samples. Scanning Electron Microscopy was used to verify that silver nanoparticles were deposited on the collagen fibres surface and inside the collagen matrix of leather. The antibacterial effect of different silver nanoparticles and silver nanocomposites was assessed on gram positive and gram negative bacterial strains in agar diffusion assays and turbidimetric tests. The results obtained proved a powerful antibacterial effect on all bacterial strains tested of both silver nanoparticles and silica nanocomposites. This allows us to consider the incorporation of these nanocomposites into leather as an alternative to other commercially available products in order to obtain leather with antimicrobial properties that confer better functionality and added value.
Keywords
Antimicrobial effect; Silver nanoparticles; Nano composites; Treated leather
Introduction
Footwear is a garment that is subjected to highly demanding conditions of use. Being in continuous contact with the foot for long periods of time in a poorly ventilated environment, the humidity and temperature conditions favour the proliferation of microorganisms. Such microorganisms are involved in the generation of foot odour, as a result of the degradation of footwear components and sweat produced by the wearer’s [1] foot bacterial flora. Furthermore, the presence of certain microorganisms can lead to some foot conditions, as is the case of athlete’s foot [2], or make pre-existing conditions worse, as for instance the infections in diabetic patients [3,4]. Apart from the preventive measures recommended by specialists, such as foot care and hygiene, there are currently other ways to prevent and/or reduce these problems, among which is the use of synthetic or natural antimicrobial agents or biocides that prevent or reduce microbial growth in the shoe-foot environment [5,6]. To this end, it is extremely important to be able to determine accurately the efficacy of the antimicrobial activity of such products. Among the wide range of products that can be used for this purpose, it would be interesting to use broad spectrum, long-lasting products that do not pose biocompatibility or toxicity problems, since they are to be in direct contact with the skin. This is the case of silver, copper or other metals [7].
Indeed, although silver has been used from time immemorial, in recent years it has been employed more extensively for numerous biomedical [8-10] and industrial (electrical appliances, garments [11,12] applications as it shows excellent antimicrobial properties against a wide range of bacteria and fungi [13]. Silver ions achieve significant reductions in microbial growth at very low concentrations (oligodynamic effect) and even though the mechanisms that cause this effect have not been completely elucidated yet, some authors have attributed the cytotoxicity of silver to several possible mechanisms, including the generation of large amounts of free radicals inducing high oxidative stress, the disruption of cell membrane integrity, or protein or DNA binding and damage to genetic material [14,15].
Although the biocidal properties of silver compounds are well described, today these properties have been enhanced through nanotechnology by means of silver nanoparticles which has allowed the size of metal nanoparticles to be modularly and accurately reproduced in order to improve the stability of the colloidal emulsions as well as a controlled release of the silver nanoparticles, obtaining a long-term antimicrobial activity and reducing unwished potential effects, among others [16]. This has made it possible to limit dosage to avoid cytotoxic effects, to control the product’s useful life, to optimise the release profile for specific tissue-targeted or intracellular-targeted delivery, or to minimise the release of silver ions into the environment [17].
In order to obtain footwear components with new functionalities, more specifically with the capacity to inhibit microorganism growth (antimicrobial effect) colloidal silver nanoparticle (AgNPs) emulsions were synthesised using different reducing agents: sodium borohydride [18], gelatine/glucose [19] and glycolic Aloe vera extract [20]. The first one was used as a control for the other two, which are involved in “green” synthesis processes that are more innocuous and environmentallyfriendly [17]. Furthermore, silver nanoparticles (AgNPs) have a strong tendency to agglomeration and poor dispersability in the polymeric matrices. So in this work AgNPs have been protected with a porous coating, such as a silica matrix, by forming silica nanocomposites (Ag@SiO2) capable of preventing the aggregation of the nanoparticles but allowing the antimicrobial mechanism, thus reducing the possible incompatibility with materials matrices. The silica structure acts as a convenient carrier for incorporating the fine silver particles into plastics, textiles, and coatings. A further advantage is that the immobilization of silver nanoparticles within the silica structure limits the potential for release and disposal of the nanoparticles themselves. This property may be highly desirable because of the possible ability of nanoparticles to go through biological membranes and other barriers.
The synthesised silver nanoparticles and nanocomposites and their antimicrobial properties were characterised by means of UV-Vis spectroscopy, and Scanning and Transmission Electron Microscopy (SEM and TEM). Furthermore, leathers obtained by different tanning methods and impregnated with silver nanocomposites were assessed in terms of their antimicrobial properties. The influence of the antimicrobial treatments on the physico-mechanical properties was also evaluated, as well as the distribution of silver-nanoparticles onto leather fibres. Additionally, the influence of the leather tanning method employed (whether conventional chrome tannage or alternative tannages based on oxazolidine, titanium or aluminium) on the antimicrobial properties of the treated leathers was also analysed.
Materials and Methods
Test bacteria and media
Two gram positive (Staphylococcus aureus, CECT 239 and Bacillus subtilis, 168 ATCC 23857) and two gram negative (Escherichia coli, SG13009, QIAGEN, and Klebsiella pneumoniae, CECT 141) bacterial strains were used to evaluate the activity of AgNPs and Ag@SiO2 nanocomposites, as well as treated leathers.
The antimicrobial activity was measured in nutrient broth (NB) [19] by means of turbidimetric assays and the inhibition halos assay was carried out in nutrient agar (NA) [19] medium.
Leather samples
Different tanned custom-made bovine leathers were used including metal-tanned leathers, 3% of tanning product (chrome, aluminium and titanium sulphate) respectively, and non-metal-tanned ones (3% oxazolidine) (supplied by Curtidos Requena SL, Caudete, Albacete, Spain). Each of the leathers was split to obtain three different thicknesses (0.8, 1.2 and 1.6 mm), which correspond to leather thicknesses usually employed in the footwear industry as insock, lining and upper materials.
Synthesis of silver nanoparticles (AgNPs)
Colloidal silver emulsions were synthesised from silver nitrate using three different reducing agents: sodium borohydride (Merck, Darmstadt, Germany), gelatine/glucose and an Aloe vera extract [20]. Gelatine and Aloe vera extract were used as natural reducing agents which are more environmentally-friendly than the traditional sodium borohydride.
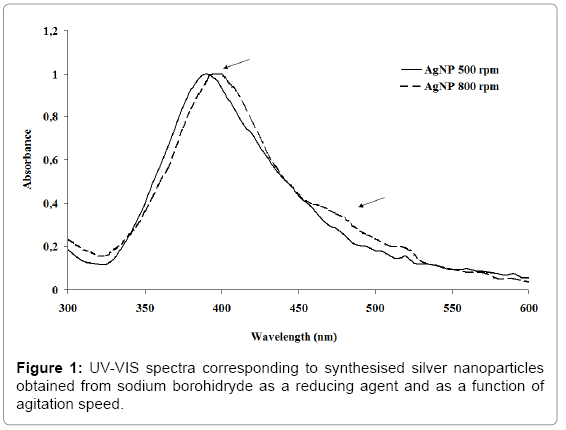
Firstly, silver nanoparticles (AgB), which were to be used as a control, were synthesised by the traditional co-precipitation Creighton- Blatchford method [18] using sodium borohydride as a reducing agent of silver nitrate (ASC reagent Sigma-Aldrich, Barcelona, Spain) and sodium citrate as a stabiliser (ASC reagent Sigma-Aldrich, Barcelona, Spain) equation 1). Silver nanoparticles (AgB) were prepared by reduction of 10-3 M AgNO3 with excess ice-cold NaBH4 solution. A 10-3 M solution of sodium citrate was used as stabiliser. The solutions of these salts were mixed rapidly with vigorous agitation to aid monodispersity for 10 min. The obtained solution was yellow and showed a single visible extinction band near 400 nm (Figure l), characteristic of silver particles substantially smaller than the wavelength of light. Silver nanoparticles were stored at 10ºC in absence of light.
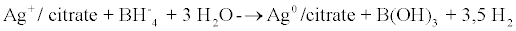 (1)
(1)
Secondly, AgG nanoparticles were synthesised by the Ahmad modified method [21], using a gelatine/glucose mixture (Sigma-Aldrich, Barcelona, Spain) as a reducing/stabilising agent of silver nitrate (equation 2). In this case, 5 g of gelatine type B were dissolved in 100 mL deonised water until a transparent solution is obtained. Then 5 g of glucose solution were added. After 10 min, 1 g of AgNO3 was added and the temperature was raised up to 80ºC and maintained for 3 h under magnetic agitation until a light brown is obtained. Silver nanoparticles were stored at 10ºC in absence of light
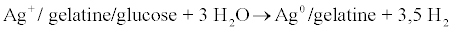 (2)
(2)
Finally, AgAV nanoparticles were synthesised by the Zhang method [20] using a glycolic Aloe vera extract (Guinama, Valencia, Spain) as a reducing/stabilising agent (equation 3). The evolution of the reaction was then followed by UV-Vis spectroscopy. 5 mL of a glycolic Aloe Vera extract and 5 mL of 10-2 M AgNO3 solution were added to 200 mL of deionised water under magnetic agitation. The mixture was maintained at 80ºC for 3 h. Finally, silver nanoparticles were stored at 10ºC in absence of light.
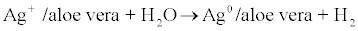 (3)
(3)
In order to determine the optimum reaction time as well as to confirm the obtaining of silver particles at nanosize, the reactions were followed by UV-Vis spectroscopy at different times. In addition, the influence of the agitation rate (500 and 800 rpm) on the nucleation process during the synthesis of the silver nanoparticles was analysed in all cases mentioned above.
Synthesis of Ag@SiO2 nanocomposites
In order to improve colloidal emulsions stability avoiding silver nanoparticle aggregation, Ag@SiO2 nanocomposites were synthesised. First of all, hollow silica nanoparticles were prepared by the Stöber [22] method. Then, Ag@SiO2 nanocomposites were obtained from each previously described silver colloidal emulsion by adding them during the hydrolysis process of the tetraethyl orthosilicate (TEOS) (98% purity, Sigma-Aldrich, Barcelona, Spain) in alkaline conditions (pH = 14). The condensation reaction generally started immediately allowing silver nanoparticles to be embebed into silica matrix. Finally, Ag@SiO2 were centrifugated and resuspended in deonised water and then stored at 4ºC.
Characterisation of AgNPs and Ag nanocomposites
A UV-Vis NanoDrop ND-1000 spectrophotometer (NanoDrop products, Wilmington, USA) operating in the range from 200 to 700 nm wavelength, a JEOL JEM-2010 transmission electron microscope (TEM) (JEOL Ltd, Tokyo, Japan) operating at 200 kV and provided with an X-Ray EDX OXFORD INCA Energy TEM 100 detector (Oxford Instruments Analytical, High Wycombe, UK) for microanalysis, and a JEOL JSM-840 scanning electron microscope (SEM) (JEOL Ltd, Tokyo, Japan), were used for morphological and size nanoparticle characterisation.
Application of AgNPs on different tanned leathers
For tear and tensile strength and spotting tests, synthesised AgNPs and Ag@SiO2 nanocomposites were applied on the grain side of 200×400 mm leather test-pieces by spreading 400 μL of the corresponding solution twice (200 μL each, allowing it to dry between applications), using the side of a glass microscope slide.
For antimicrobial assays and SEM imaging, leather test-pieces consisting of 9 mm diameter circles were treated with the corresponding silver emulsions (AgNPs and Ag@SiO2 nanocomposites) by applying 50 μL of the emulsion on the grain side, and 50 μL on the flesh side using a micropipette, and allowing them to dry at room temperature between and after each application, for at least 24 h.
SEM analysis of AgNP-treated leathers
In order to test AgNPs and Ag@SiO2 deposition and penetration inside the leather collagen structure, SEM analysis was performed on both the grain and flesh sides, and also within the leather matrix, of cryofractured leather samples. All leather samples were fixed to aluminium stubs using a double-sided carbon conductive adhesive tape (Nisshin EM Co, Tokyo, Japan) and gold-coated in a SCD 004 sputter coater (Balzers, Liechtenstein). Samples were visualised in a JEOL JSM- 840 microscope (JEOL Ltd, Tokyo, Japan).
Physical and mechanical evaluation of AgNP-treated leathers
All the physical and mechanical tests on AgNP-treated leathers were based on international standards. The tear strength test was performed according to EN ISO 3377-2:2003. The tensile strength test was performed according to EN ISO 3376:2003. The determination of thickness was performed according to EN ISO 2589:2003.
The test for colour fastness was performed in accordance with EN ISO 15700:1998, modified for a better adaptation to the characteristics of the samples under study. This International Standard specifies a method for assessing the effect of water spotting on all kinds of leather. The method is suitable for assessing changes in the physical appearance and colour of leather. Instead of water, AgNPs and Ag@SiO2- emulsions were used. Drops of each solution were deposited on test-pieces of the corresponding leather sample. After 30 min, the excess solution was spread with the help of a glass slide. Samples were allowed to dry overnight and the colour change was evaluated by visual analysis.
Assessment of antibacterial activity
The antibacterial activity of leather samples treated with the nanosilver colloidal solutions and Ag@SiO2 nanocomposites was tested by means of liquid (turbidity evaluation) and solid medium (agar diffusion) antibacterial tests against the four above-mentioned test bacteria. To this end, 9 mm diameter leather test-pieces were used.
For turbidity evaluation, 100 μL of the corresponding bacterial culture, obtained from an overnight culture grown until OD546= 0.1 [23], were inoculated into a test tube containing 1.9 mL of liquid nutrient broth. Treated leather samples were added to the corresponding tube, and the cultures were then incubated overnight at 37ºC and 160 rpm. A sample free tube was inoculated as a negative control. After the incubation period, the turbidity of the samples was compared to that of the controls, assigning a “+” symbol to the samples with a positive growth and “-“ to those with absence of bacterial growth, meaning the presence of an antibacterial effect.
In order to assess the antimicrobial activity by means of the agar diffusion method, overnight cultures, with OD546 = 1.3 were added to nutrient broth liquid medium (100 μL bacterial culture/100 mL nutrient agar) and poured into sterile Petri dishes. Leather samples were placed on the surface of the solidified agar plates, grain side facing up, and then incubated at 37ºC overnight. After incubation, the inhibition zones were measured, and expressed in mm, the “0” value corresponding to samples showing no inhibition.
Results and Discussion
Synthesis and characterisation of silver nanoparticles (AgB, AgG, AgAV) and nanocomposites (Ag@SiO2)
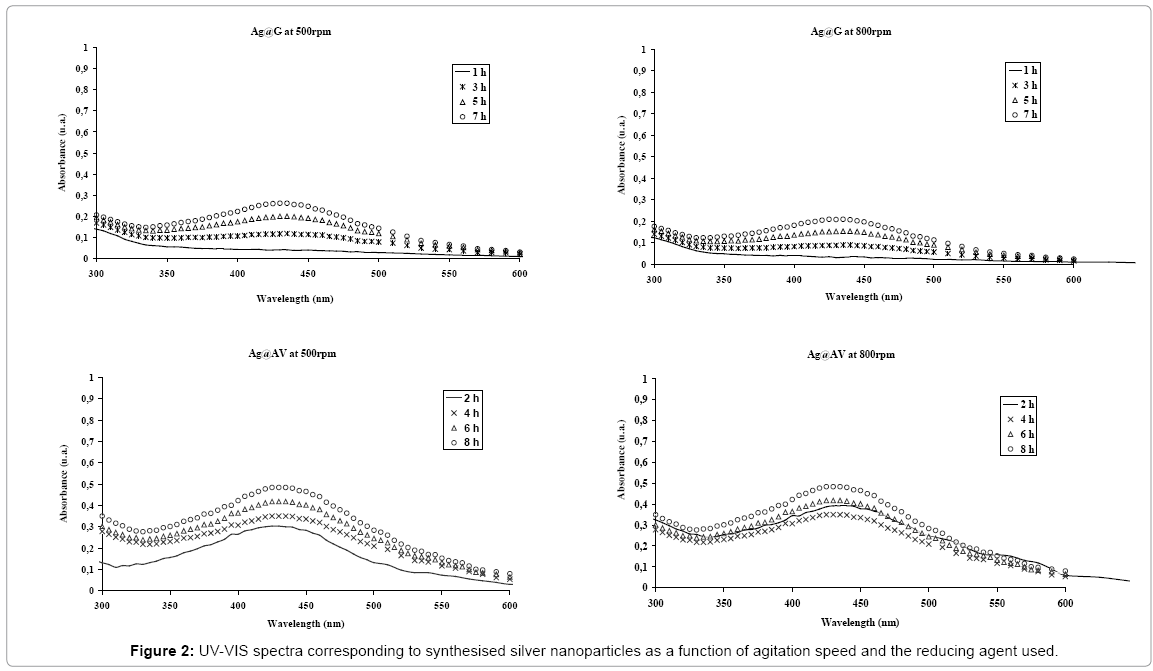
The formation of colloidal silver nanoparticles using different reducing agents was evaluated by UV-Vis spectroscopy [20,24-26]. Figures 1 and 2 show the UV-Vis spectra of the different silver nanoparticles synthesised at two different agitation rates and as a function of time.
In general, all spectra showed a well-defined absorption band around 400 nm attributed to the phenomenon of plasmon resonance characteristic of the silver particles at nano scale. The wavelength of the absorption band provides information on the average particle size, whereas its full-width at half-maximum (fwhm) provides an estimation of particle size distribution.
Figure 1 corresponds to silver nanoparticles obtained using sodium borohydride as a reducing agent at 500 and 800 rpm. The spectrum of the AgNPs synthesised at 500 rpm showed a maximum of absorbance centred in 391 nm. The increase in the agitation speed during the reduction reaction produced a slight displacement towards higher wavelengths of the absorbance maximum, which could be attributed to a slight increase in silver nanoparticle diameter. In addition, a second particle distribution at higher wavelengths around 450-525 nm appeared, possibly due to the aggregation of silver nanoparticles at higher agitation rates. In both cases the average particle size was around 5-10 nm.
In addition, when gelatine and Aloe vera extract were used as green reducing agents, a displacement of the plasmon resonance band towards higher wavelengths, around 435 nm, was observed (Figure 2) corresponding to an average particle size of 10-20 nm [19,25]. In this case, there was no influence of the two different agitation rates studied on the average silver particle size, which might be due to a stabilising effect of both green reducing agents characterised by the immobilisation of the silver nanoparticles embedded in a polymeric matrix that avoids their aggregation and/or precipitation.
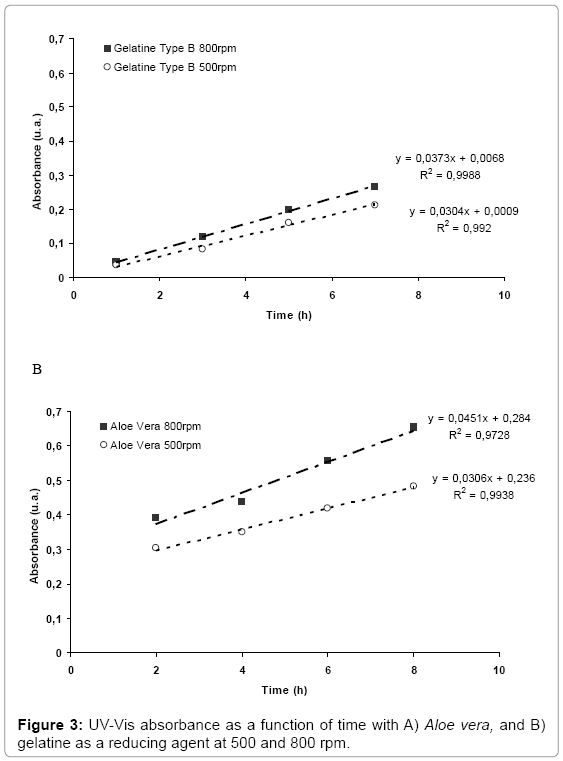
Furthermore, according to Figure 2, an increase in the absorbance of the plasmon resonance bands as a function of time was observed due to slower kinetics of the reduction reactions when green reducing agents were used instead of sodium borohydride. Figure 3 represents the intensity of the plasmon resonance bands as a function of time at two different agitation rates corresponding to AgG and AgAV synthesis reactions. In both cases, an increase in the reaction kinetics was achieved when the agitation rate increased. This effect might be attributed to a higher number of particle collisions in the reaction media improving silver nanoparticle nucleation process.
The average size of the synthesised nanoparticles estimated by UVVis spectroscopy was confirmed by means of Transmission Electron Microscopy. Figure 4 shows TEM images of the different silver nanoparticles which also revealed that the silver nanoparticles were predominantly spherical.
Different synthesised silver nanoparticles were used to obtain silver silica nanocomposites with a mean particle size of around 300 nm, according to SEM images (Figure 5). In addition, TEM images of the silver silica nanocomposites (Figure 6) show that silver nanoparticles were located at silica nanosphere surface.
Evaluation of AgNP-treated leathers
SEM analysis
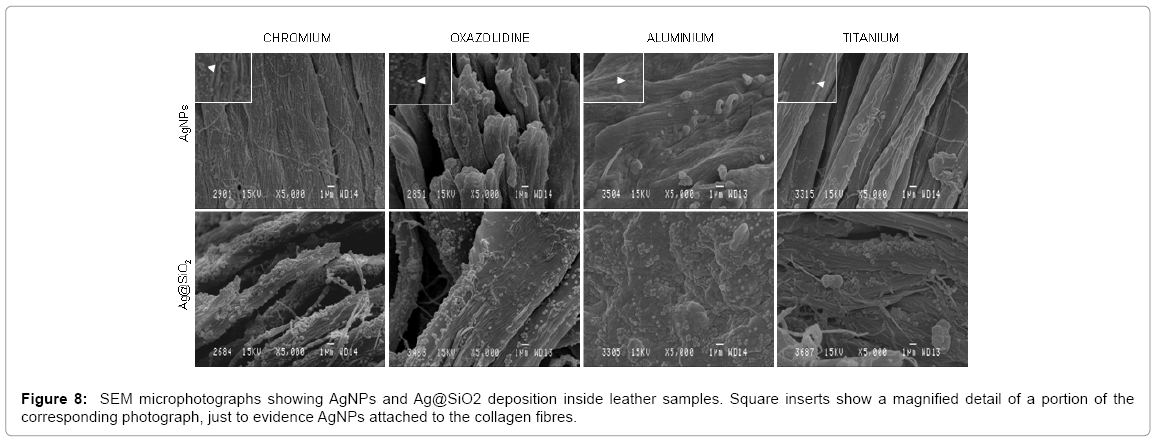
The Scanning Electron Microscopy analyses carried out on the different treated leathers revealed the presence of colloidal emulsions on their surface and inside the leather matrix. Figure 7 shows leather samples treated with AgNPs, Ag@SiO2 and a control sample (distilled water). As can be seen in Figure 7, the control sample (distilled water) showed a smooth and homogenous texture of collagen fibres, unlike the leather samples impregnated with AgNPs and Ag@SiO2, which confer a coarse aspect on collagen fibres. The collagen fibres of AgNP-treated leathers had a dotted appearance, while the Ag@SiO2-impregnated leathers showed silica nanospheres along collagen fibres.
Nanosilver colloidal solutions and Ag@SiO2 nanocomposites deposition was observed both on the external layers of the samples (grain and flesh sides) and in their internal structure, adhered to collagen fibres (Figure 8). As shown in Figures 7 and 8, the presence of AgNPs and Ag@SiO2 deposited on collagen fibres proves the penetration capacity of these composites. Particle distribution, especially that of Ag@SiO2 nanocomposites, seemed to be quite uniform, regardless of the tanning agent used.
Tear and tensile strength: Tensile and tear strength tests (Tables 1 and 2) were performed to determine whether the application of silver emulsions and nanocomposites could alter the physical properties of leathers, which could have an effect on their quality and compliance with some standard requirements for certain types of footwear.
| Sample | Water | AgNPS | AgNPs@SiO | |||
|---|---|---|---|---|---|---|
| Tensile Strength (N/mm2) | Elongation (%) | Tensile Strength (N/mm2) | Elongation (%) | Tensile Strength (N/mm2) | Elongation (%) | |
| Chromium | 26,6 | 90 | 24,5 | 90 | 24,1 | 89 |
| Oxazolidine | 31,4 | 110 | 33,7 | 106 | 30,0 | 98 |
| Aluminium | 19,0 | 80 | 19,8 | 76 | 21,2 | 85 |
| Titanium | 30,1 | 79 | 31,4 | 86 | 31,9 | 87 |
Table 1: Tensile strength and elongation at break results under different conditions.
| Sample | Water | AgNPS | AgNPs@SiO | |||
|---|---|---|---|---|---|---|
| Thickness (mm) | Strength (N) | Thickness (mm) | Strength (N) | Thickness (mm) | Strength (N) | |
| Chromium | 2,1 | 298 | 2,1 | 301 | 2,0 | 310 |
| Oxazolidine | 2,1 | 400 | 2,1 | 408 | 2,0 | 439 |
| Aluminium | 1,9 | 256 | 1,8 | 280 | 1,8 | 316 |
| Titanium | 2,0 | 275 | 2,0 | 303 | 2,0 | 322 |
Table 2: Tear strength results under different conditions.
Table 1 lists the tensile strength and elongation at break values, and Table 2 shows the tear strength values, corresponding to differently tanned leathers, with or without synthesised silver nanoparticle or nanocomposite treatment. The results obtained indicated that there were no significant differences with regard to the mechanical properties of leathers, between the different treatments compared to the control (non-treated) samples (data not provided).
Spotting: Figure 8 shows the images of the test-pieces that underwent spotting tests. The results of the visual inspection of the testpieces are listed in Table 3. The results obtained showed that the tested colloidal emulsions had a negative effect on the surface appearance of some of the leathers, as is the case of aluminium-tanned leather since all the test-pieces were spotted to some extent, thus proving that aluminium tanning or the finishing of this type of leather were not the most suitable ones to avoid spotting. The test-pieces of oxazolidinetanned leather evidenced spotting in the case of the test-pieces that had been impregnated with the different colloidal emulsions, but not with silver nanocomposite emulsions. This would represent an additional advantage of Ag@SiO2 nanocomposites. Chrome-tanned and titanium tanned leathers did not show spotting, regardless of whether silver nanoparticles had been added in free form or as silica nanocomposites.
| SiO2 | AgNPs@SiO2 | AgNPS | Water | Negative Control | |||||
| Av | Gi | Bh | Av | Gi | Bh | ||||
| Chromium | no | no | no | no | no | no | no | no | no |
| Oxazolidine | no | no | no | no | yes | yes | yes | yes | no |
| Aluminium | yes | yes | yes | yes | yes | yes | yes | yes | no |
| Titanium | no | no | no | no | no | no | no | no | no |
Table 3: Visual assessment results of the spotting test under different conditions.
Antimicrobial assays
Table 4 shows the results of the turbidimetric tests performed to determine the antimicrobial effect of the different leathers impregnated with silver emulsions. All leather samples and emulsions used showed a marked inhibitory effect evidenced by the absence of turbidity in test tubes, except for chrome-tanned leathers treated with silver nanocomposites.
| AgNPs@SiO2 | AgNPs | SiO2 | |||||
| Av | Gl | Bh | Av | Gl | Bh | ||
| CHROMIUM | |||||||
| B. subtilis | + | - | + | - | - | - | + |
| E. coli | - | - | + | - | - | - | + |
| K. pneumoniae | + | - | + | - | - | - | + |
| S. aureus | + | - | + | - | - | - | + |
| OXAZOLIDINE | |||||||
| B. subtilis | - | - | - | - | - | - | + |
| E. coli | - | - | - | - | - | - | + |
| K. pneumoniae | - | - | - | - | - | - | + |
| S. aureus | - | - | - | - | - | - | + |
| ALUMINIUM | |||||||
| B. subtilis | - | - | - | - | - | - | + |
| E. coli | - | - | - | - | - | - | + |
| K. pneumoniae | - | - | - | - | - | - | + |
| S. aureus | - | - | - | - | - | - | + |
| TITANIUM | |||||||
| B. subtilis | - | - | - | - | - | - | + |
| E. coli | - | - | - | - | - | - | + |
| K. pneumoniae | - | - | - | - | - | - | + |
| S. aureus | - | - | - | - | - | - | + |
Table 4: Bacterial growth (+) or absence (-) of bacterial growth in the four strains under study under different turbidimetric testing conditions.
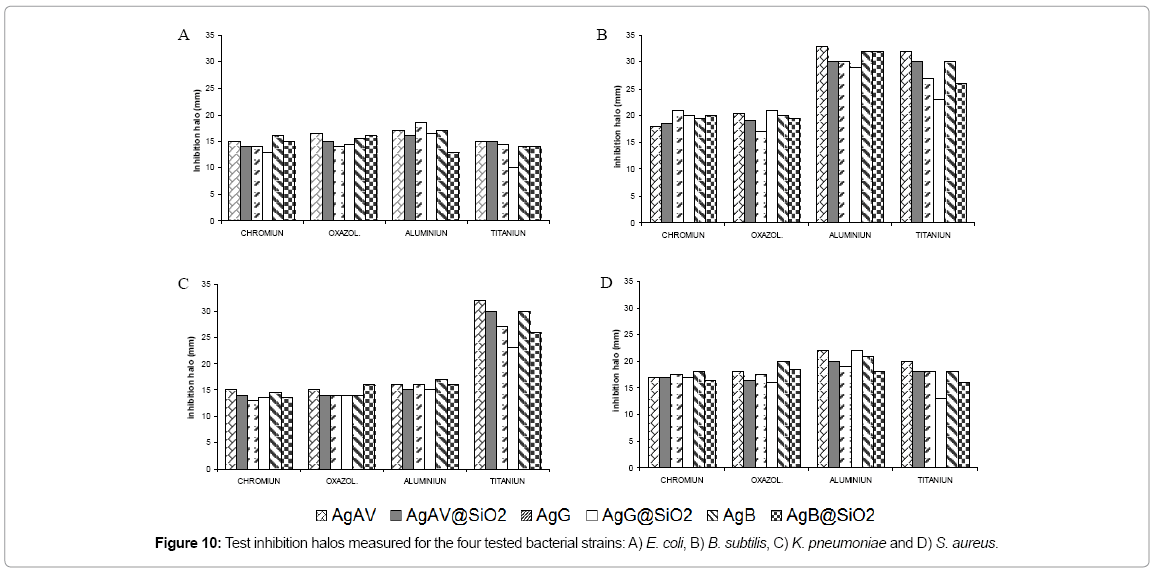
As shown in Figure 9 (agar diffusion tests), the different leathers showed significant differences in terms of the size of inhibition halos caused by the effect of the different treatments applied, except for Bacillus subtilis, which showed bigger halos. This proves that, regardless of the tannage applied, silver present in AgNPs and Ag@SiO2 accounts for the inhibition of bacterial growth. Furthermore, aluminium-tanned and titanium-tanned leathers showed a significant increase in their inhibition halos against Bacillus subitilis (Figure 10). This effect was also observed in the sample impregnated with distilled water (control) for Bacillus subtilis (data not supplied), where the inhibition halo could be attributed to the tanning agent, and halos showed bigger diameters when nanoparticles had been applied than those of samples tanned with other tanning agents. This could be partly due to an additive effect corresponding to a moderate antibacterial activity of aluminium or titanium against Bacillus subtilis that would add to that of silver nanoparticles [27-29].
Conclusion
This paper describes the assessment of the antibacterial effect of leathers obtained by different tanning methods that had been impregnated with silver nanoparticle emulsions or silver silica nanocomposites with the aim of verifying the potential use of these methodologies in footwear production. This would make it possible to prevent or minimise the presence of microorganisms that cause foot odour or are involved in the development or worsening of various foot diseases. For this purpose, silver nanoparticles were synthesised using three reducing agents: sodium borohydride, gelatine/glucose and glycolic Aloe vera extract. The type of reducing agent used determined the kinetics of the reduction reaction. The nanoparticles obtained with gelatine/glucose and Aloe vera extract showed slow formation kinetics and bigger particle size, since in these cases silver nanoparticles were embedded in a polymeric matrix that produced a stabilising effect, thus preventing their aggregation. The increase in the agitation rate during the reaction improved the kinetics without altering nanoparticle size.
The results obtained suggest that the use of silica nanocomposites obtained from said silver nanoparticles constitutes an effective method to inhibit bacterial growth.
Although the application of the different silver nanoparticles emulsions and nanocomposites seemed not to affect the physical properties of leathers, such as their tensile and tear strength, the use of nanosilver emulsions produced a more marked spotting effect in more types of leathers than Ag@SiO2 nanocomposites, especially in the case of aluminium-tanned leather. The use of Ag@SiO2 nanocomposites is regarded as more suitable for footwear manufacture, in that they offer long-term delivery. In order to avoid the above-mentioned spotting problems, the incorporation of these composites during the leather finishing process and not a posteriori should be considered.
Acknowledgements
The authors thank IVACE and the European Regional Fund for their financial support to the project IMDEEA/2012/46.
References
- Ara K, Hama M, Akiba S, Koike K, Okisaka K, et al. (2006) Foot odor due to microbial metabolism and its control.Can J Microbiol 52: 357-364.
- Boboschko I, Jockenhofer S, Sinkgraven R, Rzany B (2005) [Hyperhidrosis as risk factor for tineapedis].Hautarzt 56: 151-155.
- Karchmer AW, Gibbons GW (1994) Foot infections in diabetes: evaluation and management.CurrClin Top Infect Dis 14: 1-22.
- Lipsky BA, Berendt AR, Deery HG, Embil JM, Joseph WS, et al. (2004) Diagnosis and treatment of diabetic foot infections.Clin Infect Dis 39: 885-910.
- Sanchez MM, Perez MA, Bertazzo M, Aran F, Orgile C (2013) Antimicrobial effect of microencapsulated essential oil mixtures in footwear, In Worldwide Research Efforts in the Fighting Against Microbial Pathogens: From Basic Research to Technological Developments, Ed. BrownWalker Press, Boca Raton 58-62.
- Cuesta N, Sanchez MM, Aran F, Orgile C (2010) Natural antimicrobial agents against the microbiota associated with insoles. Science and Technology Against Microbial Pathogens. Research, Development and Evaluation. Ed. World Scientific: 109-113.
- Usman MS, Zowalaty ME, Shameli K, Zainuddin N, Salama M, et al. (2013) Synthesis, characterization, and antimicrobial properties of copper nanoparticles.Int J Nanomedicine 8: 4467-4479.
- Panyam J, Labhasetwar V (2003) Biodegradable nanoparticles for drug and gene delivery to cells and tissue.Adv Drug Deliv Rev 55: 329-347.
- Park JH, Gu L, von Maltzahn G, Ruoslahti E, Bhatia SN, et al. (2009) Biodegradable luminescent porous silicon nanoparticles for in vivo applications.Nat Mater 8: 331-336.
- Uludag Y, Tothi IE (2010) Development of a sensitive detection method of cancer biomarkers in human serum (75%) using a quartz crystal microbalance sensor and nanoparticles amplification system.Talanta 82: 277-282.
- Zuhuang J (2003) Bactericidal nanosilver cloth and its making process and use. Patent number CN 1387799.
- Lee HJ, Yeo SY, Jeong SH (2003) Antibacterial effect of nanosized silver colloidal solution on textile fabrics. J Mat Sci 38: 2199-2204.
- Zhao G, Stevens SE Jr (1998) Multiple parameters for the comprehensive evaluation of the susceptibility of Escherichia coli to the silver ion.Biometals 11: 27-32.
- Bressan E, Ferroni L, Gardin C, Rigo C, Stocchero M, et al. (2013) Silver nanoparticles and mitochondrial interaction.Int J Dent 2013: 312747.
- Phrabhu S, Poulose EK (2012) Silver nanoparticles: mechanism of antimicrobial action, synthesis, medical applications, and toxicity effects. International Nano Letters 2: 32.
- Sambhy V, Sen A (2008) Novel process of incorporating silver biocides into polymers. Chem. Today 26: 316-318.
- Egger S, Lehmann RP, Height MJ, Loessner MJ, Schuppler M (2009) Antimicrobial properties of a novel silver-silica nanocomposite material.Appl Environ Microbiol 75: 2973-2976.
- Creighton JA, Blatchford CG, Albrecht MG (1979) Plasma resonance enhancement of Raman sacttering by pyridinde adsorbed on silver or gold particles of size comparable to the excitation wavelength. Journal of the Chemical Society, Faraday Transactions 2: Molecular and Chemical Physics 75: 790-798.
- Darroudi M, Ahmad MB, Abdullah AH, Ibrahim NA (2011) Green synthesis and characterization of gelatin-based and sugar-reduced silver nanoparticles.Int J Nanomedicine 6: 569-574.
- Zhang Y, Yag D, Kong Y, Wang X, Pandoli O, et al. (2010) Synergetic antimicrobial effects of silver nanoparticles@Aloevera prepared via a green method. Nano Biomedicine and Engineering 2: 252-257.
- International Standard ISO 16187:2013: Footwear and footwear components — Test method to assess antibacterial activity.
- Yazdimamaghani M, Vashaee D, Assefa S, Shabrangharehdasht M, Rad AT, et al. (2014) Green synthesis of a new gelatin-based antimicrobial scaffold for tissue engineering.Mater SciEng C Mater BiolAppl 39: 235-244.
- Stober W, Fink A (1968) Controlled growth of monodisperse silica spheres in the micron size range. Journal of Colloid and Interface Science 26: 62-69.
- Ramirez LS (2012) Evaluation of antibacterial activity of essentials oils and ethanolic extracts using agar diffusion and well dilution methods. ScientiaetTechnicaAño XVII 50: 152-157. ISSN 0122-1701.
- Kobayashi Y, Katakami H, Mine E, Nagao D, Konno M, et al. (2005) Silica coating of silver nanoparticles using a modified Stober method.J Colloid Interface Sci 283: 392-396.
- Ahmad MB, Lim JJ, Shameli K, Ibrahim NA, Tay MY (2011) Synthesis of silver nanoparticles in chitosan, gelatin and chitosan/gelatin bionanocomposites by a chemical reducing agent and their characterization. Molecules 16: 7237-7248.
- Matsunaga T, Tomoda R, Nakajima T, Wake H (1985) Photochemical sterilization of microbial cells by semiconductor powders. FEMS MicrobiolLett 29: 211-214.
- Ghoranneviss M, Shahidi S (2013) Effect of various metallic salts on antibacterial activity and physical properties of cotton fabrics. Journal of Industrial Textiles 42: 193-203.
- Kullman RMH, Reinhardt RM (1978) Aluminum Salt Catalysts in Durable-Press Finishing Treatments. Text Res J 48: 320.
Relevant Topics
- Agricultural biotechnology
- Animal biotechnology
- Applied Biotechnology
- Biocatalysis
- Biofabrication
- Biomaterial implants
- Biomaterial-Based Drug Delivery Systems
- Bioprinting of Tissue Constructs
- Biotechnology applications
- Cardiovascular biomaterials
- CRISPR-Cas9 in Biotechnology
- Nano biotechnology
- Smart Biomaterials
- White/industrial biotechnology
Recommended Journals
Article Tools
Article Usage
- Total views: 17602
- [From(publication date):
April-2015 - Sep 02, 2025] - Breakdown by view type
- HTML page views : 12627
- PDF downloads : 4975