Carbohydrate Deficient Transferrin: How reliable is it as a Biomarker for Chronic Alcohol Consumption?
Received: 01-Jan-1970 / Accepted Date: 01-Jan-1970 / Published Date: 30-Jun-2018 DOI: 10.4172/1522-4821.1000392
Abstract
Alcohol use disorders are a major cause of a number of health, economic and social challenges for individuals, their families and health care systems worldwide. The inadequate and inaccurate assessment of long-term drinking demeanors is a significant and substantial hindrance to its diagnosis and management. Biomarkers for chronic alcohol consumption are now well established as reliable diagnostic aids but their sensitivity and specificity still need to improve. Therefore, there is a definitive need for the development of more sensitive and specific markers of alcohol abuse and addiction. Biological markers of alcoholism are divided into two cohorts: conventional and circumstantial indices. Lineal markers are detected in some biological fluids including blood and urine. The other matrices encompassing hair, saliva and sweat are not yet internationally accepted and approved, despite some studies seems to be promising for some. Among the conventional biomarkers which ate tested for alcohol misuse and abuse are ethanol, ethyl glucuronide and ethyl sulfate. The conventional biomarkers, directly detect the alcohol consumption, with variable degrees of sensitivity and reliability. The circumstantial markers including MCV, γ-GT, transaminase enzymes SGOT (Serum Glutamate Oxaloacetate Transaminase) & SGPT (Serum Glutamate Pyruvate Transaminase) and carbohydrate-deficient transferrin. These biological markers are affected by heavy alcohol consumption for long periods. Objective: Our prime objective of this article is to review the available literature on CDT (Carbohydrate Deficient Transferrin) as a biomarker for chronic alcohol consumption and its role in diagnosing and monitoring alcohol use disorders. We also aim to enrich and add to the scientific debate and knowledge about the manifest reliability of this biomarker.
Keywords: Alcohol use disorders, Biological markers, Laboratory findings, Alcoholism, Transferrin isoforms, CDT
Introduction
Alcohol use is popular and wide spread around the globe. Alcoholism causes a myriad of medical and psychiatric syndromes and complications. For example, alcohol use is a major cause of road traffic accidents. The impact of alcohol abuse is reflected in the biological, psychological, and social domains of our lives. Therefore, there is pressing need for reliable, safe and specific markers for early detection of potential alcohol abuse and follow up of recovering patients. Laboratory testing of alcohol consumption can be of an added value in identifying alcohol ingestion. Yet, the conventional biomarkers, including transaminase enzymes SGOT (Serum Glutamate Oxaloacetate Transaminase) and SGPT (Serum Glutamate Pyruvate Transaminase), Gamma-Glutamyl Transferase (GGT) and (MCV), have mutable and finite sensitivity and specificity.
Discovered in 1976, Carbohydrate-Deficient Transferrin (CDT) was introduced to detect heavy and long-term alcohol abuse. CDT refers to the less sialylated forms of human transferrin: Asialoand disialo-transferrin (Kent E Vrana et al.-March 25, 2011). It is presumed that alcohol intake of ≥ 50-80 g/day for a period of at least two weeks augments the increase of CDT concentrations. Despite the fact that mechanism of CDT increase still remains poorly understood, a large number of studies suggest and manifest that CDT is a good biomarker for the diagnosis of heavy alcohol consumption, with higher sensitivity and specificity than any of the other traditional markers. CDT has become a focal point for alcohol abuse research and clinical studies, besides forensic and judicial applications (Peterson, K.-2005, Steven Kipnis-2006).
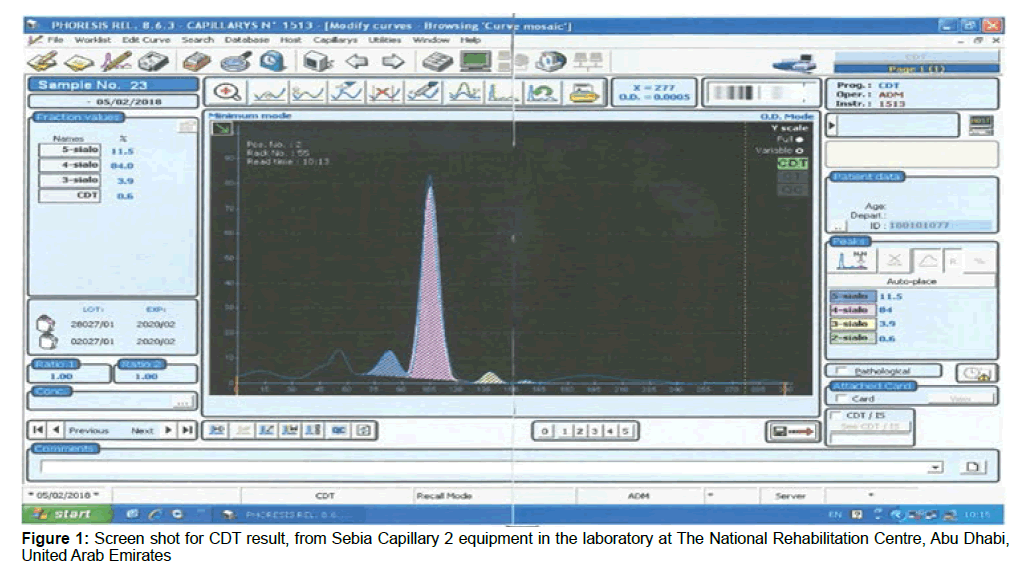
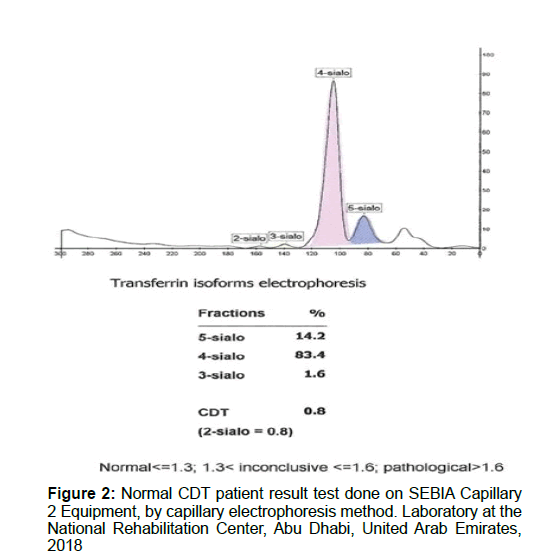
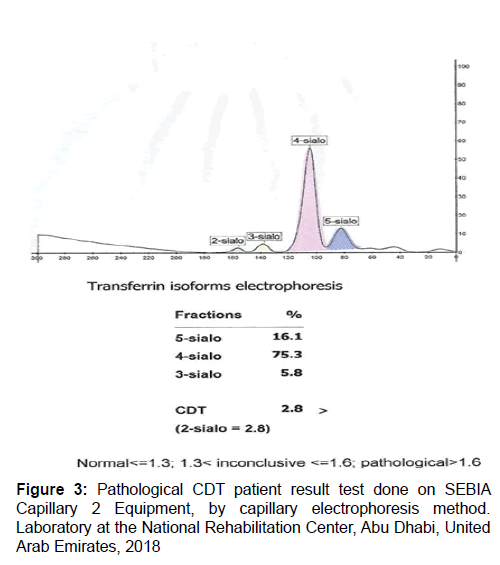
The novel advances in proteomic technologies have immensely boosted the potential for alcohol abuse biomarker discovery. Quantification of Carbohydrate Deficient Transferrin (CDT) by capillary electrophoresis is used for screening patient serum samples to detect chronic alcohol abuse. Serum transferrin isoforms are separated into five major fractions according to their sialylation level. The human transferrin consists of several isoforms. Each isoform has a different mobility according to its sialic acid content (Isoelectric point between 5.2 and 5.9). The tetrasialo transferrin is the prevalent isoform and represents about 70 to 80% of total transferrin content. Other isoforms that can be revealed are 6-sialo Tf, 5-sialo Tf, 3-sialo Tf, 2-sialo Tf and occasionally 0-sialo Tf. When analysing serum from an alcohol abuser one can find an increase in 2-sialo-Tf and 0-sialo Tf will progressively manifest (Figures 1-4 and Table 1).
| Transferrin Isomer | Pl (Isoelectric Point) |
|---|---|
| 0-sialo Transferrin | pI=5.9 |
| Hexasialo Transferrin | ++++ |
| Pentasialo Transferrin | pI=5.2 |
| Tetrasialo Transferrin | pI=5.4 |
| Trisialo Transferrin | pI=5.6 |
| 2-sialo Transferrin | pI=5.7 |
| Monosialo Transferrin | pI=5.8 |
Table 1. Isoelectric point for human transferrin isomers
Methodology
Literature search and was conducted on the databases of PubMed, Medline and Embase for all articles published using the key words CDT test & chronic alcohol use. A total, of 907 articles, were found, search date 22/02/2018. For the brief description of the different methodologies generally used for detection and quantification of CDT, several laboratory manuals and standard operation procedures were reviewed.
Carbohydrate Deficient Transferrin
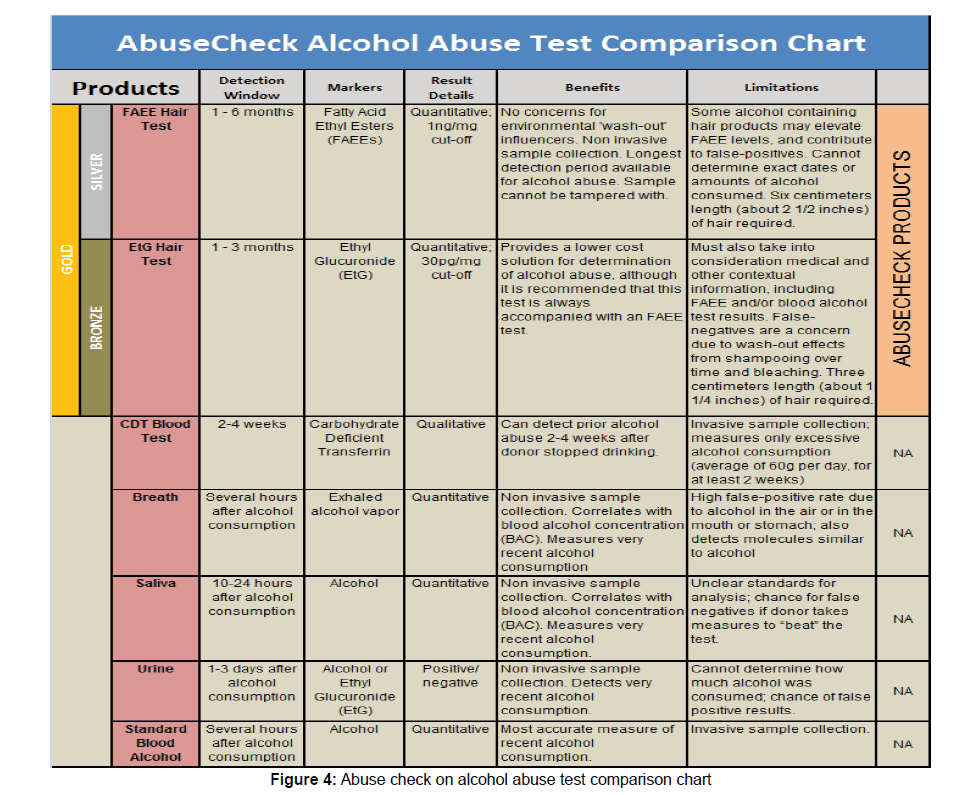
Transferrin is a carbohydrate-protein that relates and carries Fe ions through the blood. In healthy normal individuals, transferrin has specific sialic acid arrangement through its molecule. The sialic acid component of CDT might range from four to six per molecule. The unique structure of transferrin is disrupted as a result of alcohol consumption due to the impediment of sialic acids bonding to Transferrin, which leads to the presence of inadequate amount of sialic acid in blood and consequently carbohydrate deficient transferrin. Transferrin result is reported as % of total transferrin. A result that exceeds 5%, suggests heavy drinking. The FDA ratified this test in 2001. Prolonged alcohol consumption for two weeks or more will increase the rate of formation of CDT in blood, resulting in a higher percentage value raised CDT blood levels return to normal within fourteen to thirty days after the first day of ceasing intense alcohol intake (SAMSHA).
The levels of CDT in females are always higher than in males, regardless of the any aspect (Allen 2000). Misleading false positive results for CDT testing are seen in many cases and diseases, including, Obstructive liver disease, Post hepatic obstruction, Hepatitis, Liver cirrhosis, Liver Carcinoma, Cardiac insufficiency, Mononucleosis, Renal transplant, Hyperthyroidism, Myotonic dystrophy, Diabetes mellitus, Pancreatitis, Cystic Fibrosis, Low Ferritin, primary biliary cirrhosis, combined kidney and pancreatic transplant, CDG (Carbohydrate Deficient Glycoprotein), congenital glycoprotein metabolism flaw and hereditary variants of transferrin. False negative CDT test result is sometimes seen in females, where further studies are needed to explain the relation of gender to the sensitivity of the test.
The induction or inhibition of sialyl transferase and plasma sialidase may be implicated in the rise of CDT level. Transposition of protein transport during post-translational modification could be a primary mechanism in the decadence of protein metabolism connected with chronic alcohol abuse. Hormonal variations affect sensitivity of CDT. Iron homeostasis and stable equilibrium is interrupted in women due to hormonal fluctuation and imbalance in situations of gestation, use of contraceptives, menstruation, menopause, etc. CDT levels are significantly affected by iron homeostasis. Many CDT assay methods appeared to be auspicious, but it is not clearly conspicuous which technique is the most precise and accurate. Furthermore, false-positive results of CDT have been notified in non-alcohol related hepatic failure and in rare conditions. Subsequently, clinical interpretation of CDT result necessitates rigorous assessment in patients with alcohol-related or non-alcohol-related health problems. CDT levels below 1.3% are regarded as normal, but CDT levels above 1.6% are considered to be abnormal and indicative of chronic alcohol abuse. CDT values above 1.3% and beneath 1.6% are inconclusive results (Figures 1-4).
Concentration of Alcohol in Object Blood (BAC)
For neotric and fresh alcohol consumption, it is advisable to determine the blood or breath alcohol concentrations as the most precise and accurate measures with the ease of using simple equipment. Another advantage of BAC is the possibility of correlating the test result with the findings and diagnosis of the treating physician.
The extent of ailment is influenced by degree of indulgence. The metabolism and clearance of alcohol from the body is easy and full. In the majority of people the range is 8-10 hours. As a result of this rapid removal, BAC is regarded as an ineffectual test outside this timeframe, where the window of alcohol metabolism in some people ranges from four to six hours (Table 2).
| 20-99 mg% | Loss of muscular coordination |
| 100-199 mg% | Neurological impairment, ataxia, prolonged reaction time, mental impairment, incoordination |
| 200-299 mg% | Nausea, vomiting, ataxia |
| 300-399 mg% | Hypothermia, dysarthria, amnesia, stupor |
| 400- > mg% | Coma |
Table 2. The assessment of alcohol use utilizing biomarkers
Discussion
Biomarkers revealing chronic alcohol consumption continue to be a substantial tool for inpatient management and effective outpatient handling. Conventional laboratory investigations, including SGOT, SGPT, GGT, and MCV, are still used as the standard markers to in defining alcoholism treatment course. CDT, on the other-hand, considered to be a relatively new marker that added an extra value to better interpretation of heavy alcohol consumption with higher sensitivity and specificity. CDT has been extensively studied to help correlate effectiveness in conjunction with other commonly used tests.
Conclusion
CDT has bounded sensitivity with regards to being used for confirmation of ponderous alcohol use. Caution should be exercised when ordering and interpreting % CDT results, particularly in women, patients with cirrhosis and those with a high BMI. In deduction, % CDT has limited sensitivity as a thematic biomarker to recognize and characterize subjects consuming mischievous amounts of alcohol. We believe in the very near future, High Resolution Mass Spectrometry, might soon become the method of choice when available for its high degree of accuracy and sensitivity. HPLC and capillary zone electrophoresis are currently widely used for their ease of use and accuracy despite the HPLC methodology being a little bit lengthy.
Definitely, % CDT result would be of higher value in defining heavy alcohol consumption for longer terms when combined with the other conventional markers GGT, SGOT, SGPT and MCV for more informative interpretation and hence a clinical decision.
References
- Chen, J., Katherine, M.C., Macaskill, P., Whitfield, J.B. & Irwig, L. (2003). Combining carbohydrate-deficient transferrin and gamma-glutamyltransferase to increase diagnostic accuracy for problem drinking. Alcohol Alcohol, 38,(6): 574-582. Abuelgasim Elrasheed, Hamad Al Ghafri, Ahmed Yousif, Helander, A., Peter, O., & Zheng1, Y. (2012). Monitoring of the alcohol biomarkers PEth, CDT and EtG/EtS in an outpatient treatment setting. Alcohol Alcohol, 47(5): 552-557. Aertgeerts, B., Buntinx, F., Ansoms, S., & Fevery, J. (2001). Screening properties of questionnaires and laboratory tests for the detection of alcohol abuse or dependence in a general practice population. Br J Gen Pract, 51(464): 206-217. Anton, R.F., Lieber, C., Tabakoff, B., & CDTect Study Group. (2002). Carbohydrate-deficient transferrin and g-glutamyltransferase for the detection and monitoring of alcohol use: Results from a multisite study. Alcohol Clin Exp Res, 26(8): 1215-1222. Arndt, T. (2001). Carbohydrate-deficient transferrin as a marker of chronic alcohol abuse: a critical review of preanalysis, analysis, and interpretation. Clin Chem, 47(1): 13-27. Balldin, J., Berggren, U., Berglund, K., Blennow, K., Fahlke, C., Zetterberg, H. (2010). Gamma-glutamyltransferase in alcohol use disorders: Modification of decision limits in relation to treatment goals? Scand J Clin Lab Invest, 70(2): 71-74. Bergstrom, J.P., & Helander, A. (2008). Clinical characteristics of carbohydrate deficient transferrin (% disialotransferrin) measured by HPLC: Sensitivity, specificity, gender effects, and relationship with other alcohol biomarkers. Alcohol Alcohol, 43(4): 436-441. Bergstrom J.P., Helander, A. (2008). Influence of alcohol use, ethnicity, age, gender, BMI and smoking on the serum transferrin glycoform pattern: Implications for use of carbohydrate-deficient transferrin (CDT) as alcohol biomarker. Clin Chim Acta, 388(1-2): 59-67. Bianchi, V., Ivaldi, A., Raspagni, A., Arfini, C., & Vidali, M. (2010). Use of carbohydrate-deficient-transferrin (CDT) and a combination of GGT and CDT (GGT-CDT) to assess heavy alcohol consumption in traffic medicine. Alcohol Alcohol, 45(3): 247-251. Bortolotti, F., De Paoli, G., & Tagliaro, F. (2006). Carbohydrate-deficient transferrin (CDT) as a marker of alcohol abuse: A critical review of the literature 2001-2005. J Chromatogr B Analyt Technol Biomed Life Sci, 841(1-2): 96-109. Chrostek, L., Cylwik, B., Korcz, W., Krawiec, A., Koput, A., Supronowicz, Z., et al. (2007). Serum free sialic acid as a marker of alcohol abuse. Alcohol Clin Exp Res, 31(6): 996-1001. Conigrave, K.M., Davies, P., Haber, P., & Whitfield, J.B. (2003). Traditional markers of excessive alcohol use.Addiction, 98(Suppl 2): 31-43. Dawes, M.A., Frank, S., & Rost, K. (1993). Clinician assessment of psychiatric comorbidity and alcoholism severity in adult alcoholic inpatients. Am J Drug Alcohol Abuse, 19(3): 377-386. Findley, J.K., Park, L.T., Siefert, C.J., Chiou, G.J., Lancaster, R.T., Demoya, M., et al. (2010). Two routine blood tests-mean corpuscular volume and aspartate aminotransferase-as predictors of delirium tremens in trauma patients. J Trauma, 69(1): 199-201. Fagan, K.J., Irvine, K.M., McWhinney, B.C., Fletcher, L.M., Horsfall, L.U., Johnson, L., et al. (2014). Diagnostic sensitivity of carbohydrate deficient transferrin in heavy drinkers. BMC Gastroenterology, 14: 97. Gautier, A., Balkau, B., Lange, C., Tichet, J., Bonnet, F., & DESIR Study Group. (2010). Risk factors for incident type 2 diabetes in individuals with a BMI of <27 kg/m2: The role of gammaglutamyltransferase. Data from an Epidemiological Study on the Insulin Resistance Syndrome (DESIR). Diabetologia, 53(2): 247-253. Goldberg, D.M. (1980). Structural, functional, and clinical aspects of gamma-glutamyltransferase. CRC Crit Rev Clin Lab Sci, 12(1): 158. Harasymiw, J., & Bean, P. (2001). The combined use of the early detection of alcohol consumption (EDAC) test and carbohydrate-deficient transferrin to identify heavy drinking behavior in males. Alcohol Alcohol, 36(4): 349-353. Hartmann, S., Aradottir, S., Graf, M., Wiesbeck, G., Lesch, O., Ramskogler, K., et al. Phosphatidylethanol as a sensitive and specific biomarker: comparison with gamma-glutamyl transpeptidase, mean corpuscular volume and carbohydrate-deficient transferrin. Addict Biol, 12(1): 81-84. Das, S.K., & Vasudevan, D.M. (2004). Should we use carbohydrate deficient transferrin as a marker for alcohol abusers? Indian J Clin Biochem, 19(2): 36-44. Tavakoli, H.R., Hull, M., & Michael Okasinski, L. (2011). Review of current clinical biomarkers for the detection of alcohol dependence. Innov Clin Neurosci, 8(3): 26-33. Jeppsson, J.O., Arndt, T., Schellenberg, F., Wielders, J.P., Anton, R.F., Whitfield, J.B., et al. (2007). Toward standardization of carbohydrate-deficient transferrin (CDT) measurements: I. Analyte definition and proposal of a candidate reference method. Clin Chem Lab Med, 45(4): 558-562. Junghanns, K., Graf, I., Pfluger, J., Wetterling, G., Ziems, C., Ehrenthal, D., et al. (2009). Urinary ethyl glucuronide (EtG) and ethyl sulphate (EtS) assessment: Valuable tools to improve verification of abstention in alcohol-dependent patients during in-patient treatment and at follow-ups. Addiction, 104(6): 921-926. Koivisto, H., Hietala, J., Anttila, P., Parkkila, S., & Niemela, O. (2006). Long-term ethanol consumption and macrocytosis: Diagnostic and pathogenic implications. J Lab Clin Med, 147(4): 191-196. Kravos, M., & Malesic, I. (2010). Glutamate dehydrogenase as a marker of alcohol dependence. Alcohol Alcohol, 45(1): 39-44. Loomba, R., Bettencourt, R., & Barrett-Connor, E. (2009). Synergistic association between alcohol intake and body mass index with serum alanine and aspartate aminotransferase levels in older adults: The rancho bernardo study. Aliment Pharmacol Ther, 30(11-12): 1137-1149. Lovinger, D.M., & Crabbe, J.C. (2005). Laboratory models of alcoholism: treatment target identification and insight into mechanisms. Nat Neurosci, 8(11): 1471-1480. Marcos MartÃn, M., Pastor Encinas, I., & Laso Guzman, F. (2005). Biological markers for alcoholism. Revista Clinica Espanola, 205(9): 443-445. Martensson, O., Harlin, A., Brandt, R., Seppa, K., & Sillanaukee, P. (1997). Transferrin isoform distribution: Gender and alcohol consumption. Alcohol Clin Exp Res, 21(9): 1710-1715. Morini, L., Marchei, E., Vagnarelli, F., Garcia Algar, O., Groppi, A., Mastrobattista, L., et al. (2010). Ethyl glucuronide and ethyl sulfate in meconium and hairpotential biomarkers of intrauterine exposure to ethanol. Forensic Sci Int, 196(1-3): 74-77. Morini, L., Politi, L., & Polettini, A. (2009). Ethyl glucuronide in hair. A sensitive and specific marker of chronic heavy drinking. Addiction, 104(6): 915-920. http://www.nida.nih.gov/about/welcome/MessageBathSalts211.html. Peterson, K. (2005). Biomarkers for alcohol use and abuse-a summary. Alcohol Res Health, 28(1): 30-37. Pragst, F., Rothe, M., Moench, B., Hastedt, M., Herre, S., & Simmert, D. (2010). Combined use of fatty acid ethyl esters and ethyl glucuronide in hair for diagnosis of alcohol abuse: Interpretation and advantages. Forensic Sci Int, 196(1-3): 101-110. Reif, A., Fallgatter, A.J., & Schmidtke, A. (2005). Carbohydrate-deficient transferrin parallels disease severity in anorexia nervosa. Psychiatry Res, 137(1-2): 143-146. Rinck, D., Frieling, H., Freitag, A., Hillemacher, T., Bayerlein, K., Kornhuber, J., et al. Combinations of carbohydratedeficient transferrin, mean corpuscular erythrocyte volume, gamma-glutamyltransferase, homocysteine and folate increase the significance of biological markers in alcohol dependent patients. Drug Alcohol Depend, 89(1): 60-65. Substance Abuse and Mental Health Services Administration (SAMHSA) https://www.samhsa.gov/ Schroder, H., De la Torre, R., Estruch, R., Corella, D., Martinez-Gonzalez, M.A., Salas-Salvado, J., et al. (2009). Alcohol consumption is associated with high concentrations of urinary hydroxytyrosol. Am J Clin Nutr, 90(5): 1329-1335. Skipper, G.E., Wurst, F., Weinmann, W., & Liepman, M. (2009). Ethanol-based hand sanitizing gel vapor causes positive alcohol marker, ethylglucuronide, and positive breathalyzer. J Addict Med, 3(2): 1-5. Song, B., Zhu, J., Wu, J., Zhang, C., Wang, B., Pan, B., et al. (2014). Determination of carbohydrate-deficient transferrin in a Han Chinese population. BMC Biochemistry, 15:5. Stibler, H., & Kjellin, K.G. (1976). Isoelectric focusing and electrophoresis of the CSF proteins in tremor of different origins. J Neurol Sci, 30(2-3): 269-285.https://www.oasas.ny.gov/admed/documents/workbook4.pdf Turgut, O., Tandogan, I., & Gurlek, A. (2009). Association of gammaglutamyltransferase with cardiovascular risk: A prognostic outlook. Arch Med Res, 40(4): 318-320. Turgut, O., Yilmaz, A., Yalta, K., Karadas, F., & Birhan Yilmaz, M. (2006). gamma-Glutamyltransferase is a promising biomarker for cardiovascular risk. Med Hypotheses, 67(5): 1060-1064. Walter, H., Hertling, I., Benda, N., Konig, B., Ramskogler, K., Riegler, A., et al. (2001). Sensitivity and specificity of carbohydrate-deficient transferrin in drinking experiments and different patients. Alcohol Alcohol, 25(3): 189-94. World Health Organization. (2004). Consequences of alcohol use. WHO Global Status Report. p: 35-88.
Select your language of interest to view the total content in your interested language
Share This Article
Open Access Journals
Article Tools
Article Usage
- Total views: 12967
- [From(publication date): 0-2018 - Dec 19, 2025]
- Breakdown by view type
- HTML page views: 11816
- PDF downloads: 1151