Research Article Open Access
Comparative Study of the Protective Effect of Metformin and Sitagliptin against Doxorubicin-Induced Cardiotoxicity in Rats
Mahmoud M Kamel1*, Lubna Omar El-Farouk2, Afaf Sayed Osman3, Omayma Anwar Khorshid3 and Mohamed EL Shabrawy-Abdo31Department of Clinical Pathology, National Cancer Institute, Cairo University, Egypt
2Department of Pathology, Cairo University, Egypt
3Department of Medical Pharmacology, Cairo University, Egypt
- *Corresponding Author:
- Mahmoud M Kamel
Department of Clinical Pathology
National Cancer Institute
Cairo University, Egypt
Tel: +201018470990
E-mail: mm.kamel@yahoo.com
Received date: July 31, 2017; Accepted date: August 22, 2017; Published date: August 25, 2017
Citation: Kamel MM, El-Farouk LO, Osman AS, Khorshid OA, Shabrawy-Abdo ME (2017) Comparative Study of the Protective Effect of Metformin and Sitagliptin against Doxorubicin-Induced Cardiotoxicity in Rats. Clin Pharmacol Biopharm 6:174. doi: 10.4172/2167-065X.1000174
Copyright: © 2017 Kamel MM, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Clinical Pharmacology & Biopharmaceutics
Abstract
Background and aim: Cancer patients treated with doxorubicin are at high risk to develop cardiotoxicity and hyperglycemia. The present study was designed to compare the effect of the two anti-hyperglycemia drugs; metformin and Sitagliptin, in the prevention of doxorubicin-induced cardiotoxicity in rats.
Methods: Cardiotoxicity was induced in male Wistar rats by intraperitoneal injection of cumulative dose of doxorubicin (15 mg/kg over 3 weeks). Metformin or sitagliptin was administrated orally concomitant with intraperitoneal doxorubicin for 3 weeks. Mean body weight, systolic blood pressure, electrocardiographic changes, serum lactate dehydrogenase and creatine kinase-myocardium band, blood glucose and cardiac malondialdhyde level, cardiac histopathological examination and in vitro cardiac contractility in response to isoprenaline were be assessed.
Results: Doxorubicin induced marked cardiotoxicity evidenced by significant deterioration in body weight, systolic blood pressure and heart rate, elevation of ST segment, prolongation of QT interval, elevation in the serum level of creatine kinase-myocardiac band and lactate dehydrogenase, blood glucose and cardiac malondialdhyde level and reduced in vitro cardiac contractility in response to isoprenaline compared to control untreated rats. These changes were associated with histopathological evidence of cardiotoxicity. Administration of either metformin or sitagliptin with doxorubicin resulted in significant improvement in all tested parameters compared with doxorubicin treated rats. Metformin treated rats showed more significant improvement in systolic blood pressure, ST segment elevation, serum enzymes, cardiac malondialdhyde, histopathological finding and in vitro cardiac contractility than sitagliptin treated rats.
Conclusion: The present study showed that metformin ameliorated doxorubicin-induced cardiotoxicity better than sitagliptin.
Keywords
Doxorubicin; Metformin; Sitagliptin; Cardiotoxicity
Introduction
Doxorubicin (DOX) is one of the most potent anticancer agents, but its use is associated by development of cardiotoxicity that may lead to cardiomyopathy and congestive heart failure [1]. Heart is sensitive to Reactive Oxidative Species (ROS) induced damage because of its highly oxidative metabolism and fewer antioxidant defences compared to other organs [2]. Doxorubicin induces mitochondrial damage which causes continued production of free radicals and release of cytochrome c which induces apoptosis [3]. Doxorubicin-induced cardiotoxicity is usually dose dependent [4]. If the doxorubicin-induced cardiac complications prevented or at least reduced, higher doses could be utilized, thereby increasing cancer cure rates.
The prevalence of glucose intolerance is increased in patients with malignancy especially those treated by DOX [5]. Insulin resistance is correlated with increased risk for cancer. In addition, the rate of tumor recurrence, metastatic spread and fatal outcome is higher in cancer patients with hyperglycemia or type II diabetes [6].
Metformin is an oral biguanide antihyperglycemic drug which can enhance insulin sensitivity [7]. Metformin has been shown to have antioxidant properties and can decrease lipid peroxidation in various tissues [8]. Noteworthy, Metformin significantly improved left ventricular function and survival in vivo murine model of heart failure [9], improves outcomes in patients with advanced systolic heart failure [10] and may reduce the cardiovascular complications in diabetic patients [11].
Sitagliptin is a dipeptidyl peptidase-4 (DPP-4) inhibitor which improves hyperglycemia by inhibiting the inactivation of the incretin hormones, glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide. The GLP-1 receptor is expressed in islet cells, kidney, lung, brain, the gastrointestinal tract and in the heart [12]. The DPP-4 inhibitors caused decrease in the expression of myocardial fibrosis-related proteins and markers of cardiomyocyte stress in a rat model of uremic cardiomyopathy [13].
This work aimed to compare the potential protective effect of the antihyperglycemic drugs; metformin and sitagliptin against doxorubicin-induced cardiotoxicity in male Wistar rats.
Materials and Methods
Materials: Doxorubicin (Sigma Chemicals Company): Adriblastina ampoules (10 mg/5 ml) given by Intraperitoneal (i.p.) injection to rats. Metformin (Mina Pharm Company), Sitaglipitin (MERCK SHARP and DOHME Company) and Isoprenaline (Pharco Chemicals Company): all drugs are obtained as powder and dissolved in distilled water immediately before use. Reagent and chemicals: reagents used for measurement of Creatinine Kinase-M band Isoenzyme (CK-MB), Lactate Dehydrogenase (LDH) and blood glucose (Sigma Chemicals Company), kits for Measurement of Tissue Malondialdhyde (MDA) (Bio-diagnostic Company) and chemicals for modified Krebs- Henseleit buffer (Sigma Chemicals Company).
Animals: Experiments were performed on adult male Wistar rats 200-250 g. Animals were housed for at least 2 days before experiments under a 12 h light/dark cycle. Food and water were provided ad libitum. The study protocol was approved by the Institutional Reviewer Board of Faculty of Medicine, Cairo University and the animal experiments were carried out in accordance with the ethical guidelines of animal welfare.
Experimental design
The animals were classified into the following groups (24 rats each).
Group I (Control untreated group): Rats were injected with normal saline (2.5 ml/kg) intraperitoneally for 15 injections over 3 weeks.
Group II (MET group): Rats were treated daily with Metformin (MET) (150 mg/kg/day) orally for 3 weeks.
Group III (STG group): Rats were treated daily with Sitagliptin (STG) (10 mg/kg/day) orally for 3 weeks.
Group IV (DOX-induced cardiotoxicity group): Rats were injected intraperitoneally with a cumulative dose of 15 mg/kg of doxorubicin (each dose of 1.0 mg/kg for 15 injections) for 3 weeks (5 injections per week [13]).
Group V (MET+DOX group): Rats were injected with DOX as in group IV and treated daily with oral Metformin (150 mg/kg/day) over the same period (3 weeks).
Group VI (STG+DOX group): Rats were injected with DOX as in group IV and treated daily with oral sitagliptin (10 mg/kg/day) over the same period (3 weeks).
The doses of Metformin and Sitagliptin were calculated based on human therapeutic dose with reference to body surface area (BSA) rule [14].
Formula for dose translation based on BSA: Human dose (mg/ kg)=Animal dose (mg/kg) × Animal Km/Human Km (Km value for rat=6, Km value for human=37).
Measurement of body weight and mortality percent: Body weight was recorded daily and mortality percent was calculated.
Assessment of systolic blood pressure: Systolic Blood Pressure (SBP) was measured for conscious rats by a non-invasive blood pressure monitor (tail-cuff plethysmograph) (Ugo, Basile-Italy) according to the determination criterion described by Ref. [15]. SBP was measured on day one and day 21 one hour after drug administration. Five consecutive measures were recorded. The mean value of the lowest three measurements of systolic blood pressure was measured.
Biochemical analysis: Blood samples were collected on day one and on day 21 of the experiment from rat tails for measuring serum CKMB and LDH and blood glucose.
On day 21, rats of each group were classified into four subgroups (six rats each) for assessment of electrocardiography, measuring MDA, in vitro study and histopathological examination.
Assessment of electrocardiography (ECG): Six rats from each group were anaesthetized with urethane in a dose of 1 g/kg i.p. injection [16]. Electrocardiographic needle electrodes were inserted subcutaneously and ECG tracing (lead II) were recorded by AD instruments Power Lab 4/30-model ML 866, serial 430-0684.
Tissue sampling for lipid peroxidation: Six rats from each group were sacrificed by decapitation under ether anesthesia and hearts were rapidly isolated. Isolated hearts were placed into Petri dishes containing ice-cold isolation medium consisting of 125 mM KCl, 15 mM Tris, pH 7·4. The heart great vessels and valves were trimmed away and the ventricles and atria were cut open and rinsed free of blood. 20% homogenates were prepared with a Potter-Elvehjem homogenizer set at a standard velocity (500 r.p.m.) for determination of tissue Malondialdhyde (MDA) content. Malondialdhyde reacts with thiobarbituric acid producing Thiobarbituric Acid Reactive Substance (TBARS), a pink chromogen, which is measured spectrophotometrically at 532 nm [17].
Measurement of cardiac contractility: Six rats from each group were sacrificed by decapitation under ether anesthesia and hearts were rapidly isolated. The amplitude of ventricular contractions was recorded in response to variable concentrations of isoprenaline (3, 6 and 12 μg) using Power Lab Data Acquisition and Analysis Systems for Monitoring and recording signals from the isolated rat heart connected to the Langendorff’s coronary perfusion set [18].
Modified Krebs-Henseleit buffer solution at temperature 37°C was used. The heart was continuously bubbled with a mixture of 95% O2 and 5% CO2 taking care that no air bubbles entered to the aorta. Coronary perfusion is maintained at a constant pressure of 90 mmHg in a non-recirculating way. Modified Krebs–Henseleit buffer of the following composition (mM): NaCl 118, KCl 4.7, CaCl2 2.52, MgSO4 1.64, NaHCO3 24.88, KH2PO4 1.18, glucose 5.55, sodium pyruvate 2.0 [19].
Histopathological examination: Isolated hearts from six rats of each group were fixed in 10% formalin, embedded in paraffin wax, sectioned (3–5 μm), and stained with Hematoxylin and Eosin. The sections were examined under light microscope and then photomicrographs were taken for assessment of histopathological changes.
Statistical analysis
Data were coded and entered using the statistical package SPSS version 22. Data were summarized using mean ± standard deviation. Comparisons between groups were done using Analysis of Variance (ANOVA) with multiple comparisons post hoc Tukey test [20]. Pvalues less than 0.05 were considered as statistically significant.
Results
Groups treated with either metformin (group II) or sitagliptin (group III) didn't show any significant change regarding any of the tested parameters compared with control untreated group (group I). There was no mortality among rats of group I, II or III.
Body weight and mortality
Doxorubicin in group IV caused significant decrease in mean body weight on day 21 as compared to control untreated group (group I) (P<0.05) (Table 1). It is to be noted that percentage of mortality in group IV was 30%.
| Group | Mean Body weight (g) ± SD | |
|---|---|---|
| Day one | Day 21th | |
| Group I (Control untreated) | 231.67 ± 17.57 | 242.22 ± 15.55 |
| Group II (MET group) | 228.89 ± 6.76 | 238.33 ± 6.18 |
| Group III (STG group) | 223.33 ± 19.40 | 232.78 ± 18.09 |
| Group IV (DOX-induced cardiotoxicity group) | 228.33 ± 18.86 | 170.00 ± 7.48 a |
| Group V (MET+DOX group) | 230.56 ± 12.11 | 200.56 ± 7.25 ab |
| Group VI (STG+DOX group) | 229.44 ± 18.30 | 191.39 ± 10.96 ab |
Table 1: The mean body weight of rats in different studied groups. Data were represented in (mean ± SD) (n=24) MET: metformin, STG: sitagliptin and DOX: doxorubicin, a: Statistically significant difference compared to control untreated group (P<0.05), b: Statistically significant difference compared DOX-induced cardiotoxicity group (P<0.05).
Co-administration of either metformin in group V or sitagliptin in group VI showed significant increase in the mean body weight on day 21 compared with doxorubicin group (group IV) (P<0.05) (Table 1). There was no recorded mortality among rats in group V and VI.
Systolic Blood Pressure (SBP)
Rats received cumulative dose of doxorubicin (group IV) showed significant decline in the level of SBP on day 21 of the experiment as compared to control untreated group (group I) (P<0.05) (Table 2).
| Systolic blood pressure (mmHg) | Group | |
|---|---|---|
| Day 21th | Day one | |
| 123.48 ± 8.75 | 130 ± 11.38 | Group I (Control untreated) |
| 121.47 ± 8.11 | 130.56 ± 9.98 | Group II (MET group) |
| 125.51 ± 9.40 | 134.44 ± 10.24 | Group III (STG group) |
| 75.81 ± 6.65a | 130.56 ± 14.34 | Group IV (DOX-induced cardiotoxicity group) |
| 97.85 ± 12.56a,b | 135 ± 16.18 | Group V (MET+DOX) |
| 84.25 ± 4.21a-c | 135 ± 11.50 | Group VI (STG+DOX) |
Table 2: The mean values of rats systolic blood pressure in different studied groups. Data were represented in (mean ± SD) (n=24), MET: Metformin, STG: Sitagliptin and DOX: Doxorubicin. a: Statistically significant difference compared to control untreated group (P<0.05), b: Statistically significant difference compared to doxorubicin group (P<0.05), c: Statistically significant difference compared to doxorubicin + metformin group (P<0.05).
Meanwhile, co-administration of either metformin (group V) or sitagliptin (group VI) with doxorubicin produced significant increase in the SBP on day 21 compared with doxorubicin group (group IV). Systolic blood pressure was significantly improved in rats of group V compared with rats of group VI (P<0.05) (Table 2).
Serum level of cardiac enzymes
Administration of doxorubicin in group IV caused significant elevation in serum level of CK-MB and LDH on day 21 compared to control untreated group (group I) (P<0.05). Co-administration of either metformin (group V) or sitagliptin (group VI) with doxorubicin produced significant reduction in the elevated serum level of CK-MB and LDH on day 21 compared to doxorubicin group (group IV) (P<0.05). Serum level of CK-MB and LDH were significantly reduced in rats of group V compared with rats of group VI (P<0.05) (Table 3).
| lactate dehydrogenase enzyme (U/L) | CK-MB (ng/ml) | Group | ||
|---|---|---|---|---|
| Day 21th | Day one | Day 21th | Day one | |
| 458.89 ± 26.76 | 450.22 ± 24.45 | 3.78 ± 0.24 | 3.74 ± 0.27 | Group I (Control untreated) |
| 441.22 ± 18.64 | 442.89 ± 18.60 | 3.77 ± 0.30 | 3.97 ± 0.50 | Group II (MET group) |
| 442.11 ± 8.62 | 442.11 ± 10.54 | 3.97 ± 0.50 | 3.73 ± 0.29 | Group III (STG group) |
| 1145.56 ± 8.53a | 449.00 ± 26.57 | 97.28 ± 6.03a | 3.74 ± 0.26 | Group IV (DOX-induced cardiotoxicity group) |
| 789.44 ± 19.62a,b | 443.44 ± 11.86 | 51.67 ± 8.55a,b | 3.76 ± 0.32 | Group V (MET+DOX) |
| 892.67 ± 27.44a-c | 445.67 ± 26.79 | 68.56 ± 6.14a-c | 3.79 ± 0.23 | Group VI (STG+DOX) |
Table 3: The mean value of rats’ serum cardiac enzymes level in different studied groups.
Data were represented in (mean ± SD) (n=24), MET: Metformin, STG: Sitagliptin and DOX: Doxorubicin. a: Statistically significant difference compared to control untreated group (P<0.05), b: Statistically significant difference compared to doxorubicin group (P<0.05), c: Statistically significant difference compared to doxorubicin + metformin group (P<0.05).
Blood glucose level
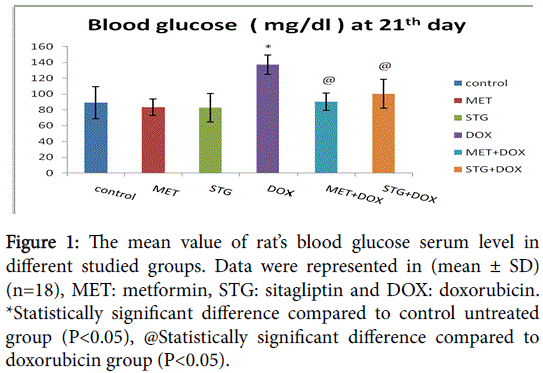
Administration of doxorubicin (group IV) caused significant elevation in blood glucose level on day 21 as compared to control untreated group (group I) (P<0.05). Co-administration of either metformin (group V) or sitagliptin (group VI) with doxorubicin produced significant reduction the elevated blood glucose level on day 21 compared to group IV (P<0.05) to became near to the blood glucose level in control untreated group (group I) (Figure 1 and 2).
Figure 1: The mean value of rat’s blood glucose serum level in different studied groups. Data were represented in (mean ± SD) (n=18), MET: metformin, STG: sitagliptin and DOX: doxorubicin. *Statistically significant difference compared to control untreated group (P<0.05), @Statistically significant difference compared to doxorubicin group (P<0.05).
Data were represented in (mean ± SD) (n=6), MET: metformin, STG: sitagliptin and DOX: doxorubicin. a: Statistically significant difference compared to control untreated group (P<0.05), b: Statistically significant difference compared to DOX-induced cardiotoxicity group (P<0.05), c: Statistically significant difference compared to doxorubicin+ metformin group (P<0.05).
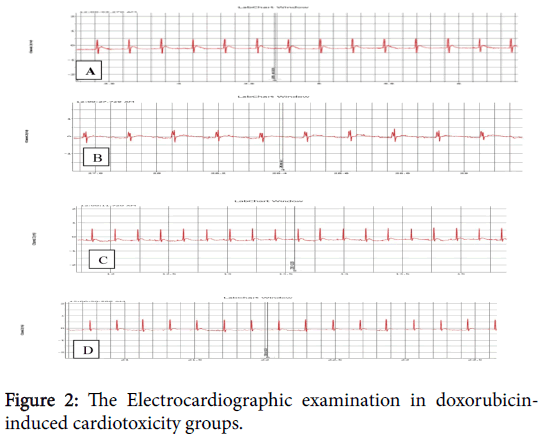
Electrocardiographic evaluations
Malondialdehyde (MDA) in cardiac tissue
Doxorubicin in group IV caused significant increase in the mean level of MDA in the cardiac tissue on day 21 as compared to control untreated group (group I) (P<0.05). Co-administration of either metformin (group V) or sitagliptin (group VI) with doxorubicin produced significant reduction in the elevated level of MDA in the cardiac tissue on day 21 compared to group IV (P<0.05). Reduction in cardiac MDA was more significant in group treated by metformin with doxorubicin (group V) than in group treated by sitagliptin with doxorubicin (group VI) (P<0.05) (Table 5).
| Electrocardiographic parameters | Group | ||
|---|---|---|---|
| QT (ms) | ST (mv) | HR (beat/min) | |
| 29.62 ± 0.74 | 0.056 ± 0.012 | 280.50 ± 4.41 | Group I (Control untreated) |
| 29.50 ± 0.76 | 0.056 ± 0.012 | 282.88 ± 5.79 | Group II (MET group) |
| 29.62 ± 0.74 | 0.056 ± 0.012 | 283.38 ± 4.41 | Group III (STG group) |
| 46.25 ± 2.38 a | 0.181 ± 0.008 a | 209.75 ± 3.77a | Group IV (DOXinduced cardiotoxicity group) |
| 36.99 ± 1.09 ab | 0.078 ± 0.004 ab | 250.25 ± 3.33a,b | Group V (MET+DOX) |
| 35.84 ± 0.86 ab | 0.109 ± 0.013 abc | 242.25 ± 9.39a,b | Group VI (STG+DOX) |
Table 4: The mean values of rats’ Electrocardiographic parameter in different studied groups.
| Mean cardiac MDA level (nmol/g) at 21th day | Group |
|---|---|
| 44.63 ± 4.30 | Group I (Control untreated) |
| 40.85 ± 2.46 | Group II (MET group) |
| 44.20 ± 3.54 | Group III (STG group) |
| 269.89 ± 16.72a | Group IV (DOX-induced cardiotoxicity group) |
| 159.98 ± 8.39ab | Group V (MET+DOX) |
| 200.29 ± 8.88a-c | Group VI (STG+DOX) |
Table 5: The mean values of malondialdehyde (MDA) in rats’ cardiac tissue in different studied groups.
Data were represented in (mean ± SD) (n=6), MET: Metformin, STG: Sitagliptin and DOX: Doxorubicin. a: Statistically significant difference compared to control untreated group (P<0.05), b: Statistically significant difference compared to doxorubicin group (P<0.05), c: Statistically significant difference compared to doxorubicin + metformin group (P<0.05).
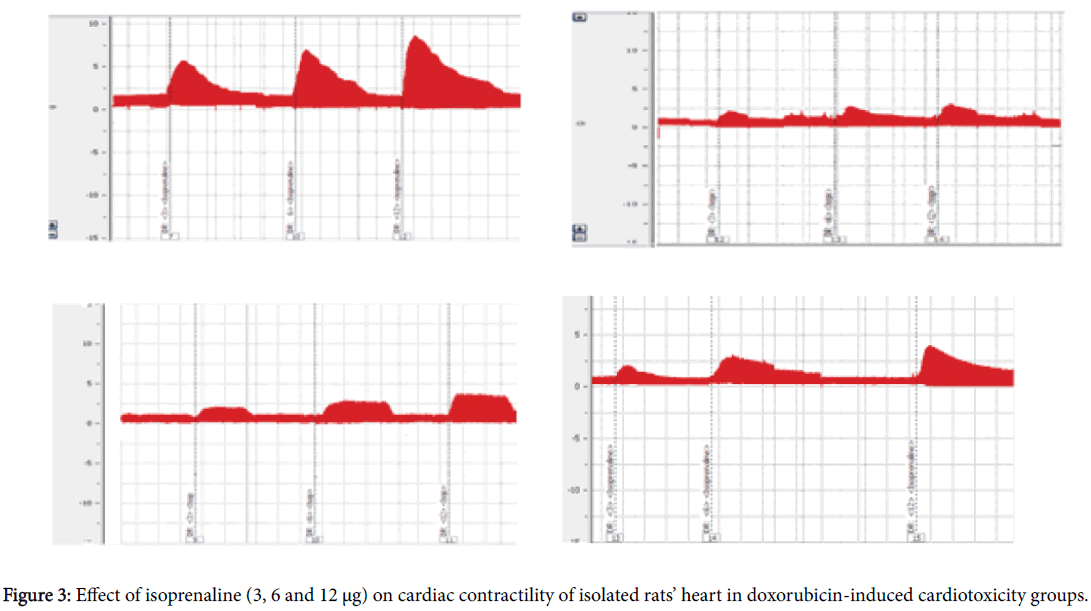
In vitro cardiac contractility
A cumulative dose of doxorubicin (group IV) caused significant regress in the mean percent changes of isolated heart contractility in response to as compared to negative control group (P<0.05). Coadministration of either metformin (group V) or sitagliptin (group VI) with doxorubicin produced significant increase in the mean percent change of isolated heart contractility induced by isoprenaline as compared to group IV (P<0.05). Rats treated with metformin (group V) showed significant increase in the mean percent changes of isolated heart contractility induced by isoprenaline compared with rats treated with sitagliptin (group VI) (P<0.05) (Table 6 and Figure 3).
| Inotropic response to isoprenaline concentration | Group | ||
|---|---|---|---|
| 12 μg | 6 μg | 3 μg | |
| 431.23 ± 1.53 | 325.00 ± 1.10 | 193.83 ± 2.71 | Group I (Control untreated) |
| 430.00 ± 2.90 | 325.50 ± 3.27 | 192.00 ± 5.22 | Group II (MET group) |
| 427.50 ± 2.07 | 322.33 ± 3.50 | 190.17 ± 2.79 | Group III (STG group) |
| 169.23 ± 2.21a | 100.00 ± 1.41a | 76.90 ± 1.19a | Group IV (DOX-induced cardiotoxicity group) |
| 270.17 ± 3.19a,b | 190.00 ± 2.76a,b | 140.00 ± 1.67a,b | Group V (MET+DOX) |
| 233.29 ± 1.60a-c | 177.78 ± 2.00a-c | 133.33 ± 0.82a-c | Group VI (STG+DOX) |
Table 6: The mean Percent changes of cardiac contractility in the isolated rats` hearts in response to isoprenaline in different studied groups.
MET: Metformin, STG: Sitagliptin and DOX: Doxorubicin, A: Group I (Control untreated group), B: Group IV (Doxorubicininduced cardiotoxicity group) showing bradycardia, prolonged QT interval and low voltage, C: Group V (MET+DOX group), D: Group VI (STG+DOX group).
Data were represented in (mean ± SD) (n=6), MET: Metformin, STG: Sitagliptin and DOX: Doxorubicin. a=Statistically significant difference compared to control group (P<0.05), b=Statistically significant difference compared to doxorubicin group (P<0.05). c=Statistically significant difference compared to doxorubicin +metformin group (P<0.05).
(MET: Metformin, STG: Sitagliptin and DOX: Doxorubicin). A: Group I (Control untreated group), B: Group IV (DOX-induced cardiotoxicity group), C: Group V (MET+DOX), D: Group VI (STG +DOX).
Histopathological examination
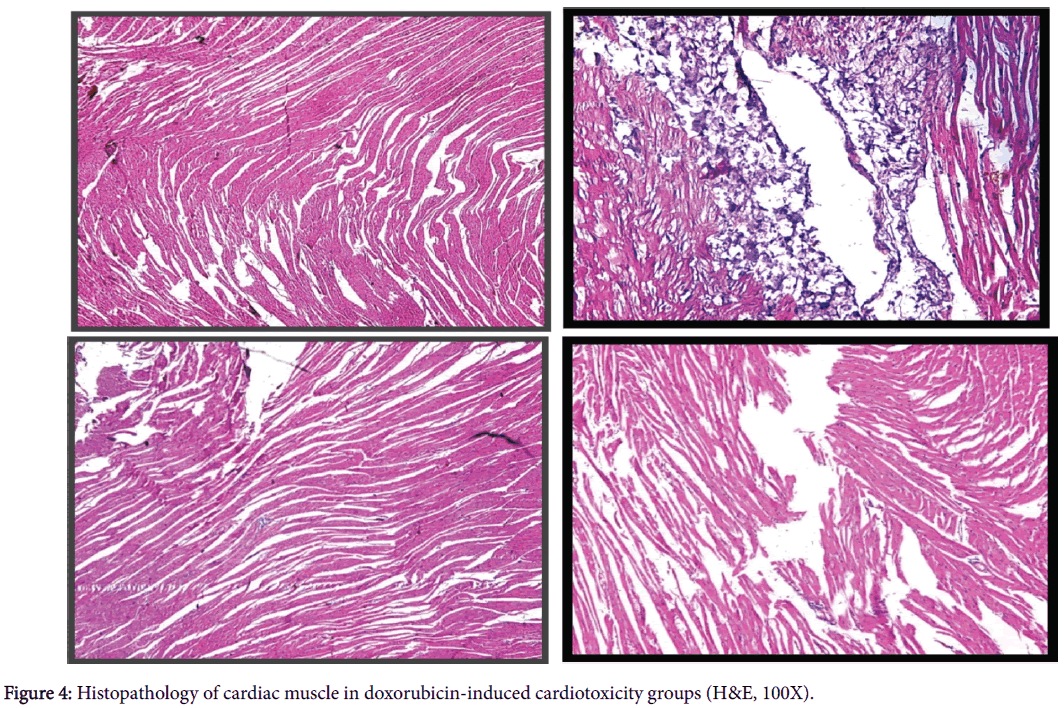
The heart from Groups I, II and III showed normal histologic appearance, that is showing normal arrangement of cardiac muscle fibres, regularity of cell and nuclear membrane, normal nuclear pattern of cells, absent vacuolation of cells, normal blood vessels pattern, no inflammatory infiltration around blood vessels, as well as normal appearances of endocardium and pericardium
Histopathological examination of isolated rats` hearts in doxorubicin induced cardiotoxicity group showed marked interstitial edema, inflammatory cellular infiltration around blood vessels, perinuclear vacuolation, highly oesenophilic cytoplasm, interfibrillar hemorrhage, disarrangement and degeneration of the myocardium.
Co-administration of metformin with doxorubicin produced improvement in the histopathological findings which appear as reversion to normal cellular pattern, little cellular infiltration and absent interfibrillar hemorrhage. Co-administration of sitagliptin with doxorubicin produced improvement in the histopathological findings which appear as mild interstitial edema, disarrangement of muscle fibres, cellular infiltration and absent interfibrillar hemorrhage (Figure 4).
(MET: Metformin, STG: Sitagliptin and DOX: Doxorubicin), A: Group I: Control untreated rats, B: Group Π (DOX-induced cardiotoxicity group) shows small cluster of myocardial fibers with small and large cytoplasmic vacuoles (indicated by left arrow) and inflammatory infiltration (indicated by bent-up arrow), C: Group Π (MET+DOX group) shows normal myocardium, reversion to normal pattern, no vacuolated fibers and no hemorrhage, D: Group IV (STG +DOX group) shows disarrangement of fibers, no vacuolated fibers and no hemorrhage.
Discussion
The aim of the present work was to compare the ability of metformin and sitagliptin to protect the heart against the cardiotoxicity induced by cumulative dose of doxorubicin in male rats which may help in cardio protection of the patients treated with doxorubicin.
In the present study, a cumulative dose of doxorubicin induced significant deterioration in body weight, blood pressure and heart rate. Significant elevation of ST segment and prolongation of QT interval, significant elevation in the serum level of CK-MB, LDH, blood glucose and cardiac MDA, histopathological signs of cardiotoxicity and reduced myocardial contractility in response to isoprenaline compared to control untreated group.
The results of the present study are in agreement with Xu [21] who showed weight loss, [22] who demonstrated significant reduction in systolic blood pressure [23] who detected ST segment elevation and QT interval prolongation in doxorubicin treated rats. Demonstrated increase cardiac [24] MDA and Shao [25] detected cardiac histopathological changes in rats treated with doxorubicin.
Arunachalam et al. [26] showed that doxorubicin can alter glucose metabolism through inhibition of insulin secretion or increase the insulin resistance. Elevation of cardiac enzymes in rats following cumulative dose of doxorubicin was detected by Ref.’s [27,28].
Different cellular mechanisms may explain doxorubicin-induced cardiac toxicity as increase intracellular iron accumulation causing increased oxidative stress [29], preventing the repair of damaged DNA strands [30], changes in vascular endothelium-derived vasoactive mediators (endothelin-1 and cardiac nitric oxide) [31] and alteration of cardiac-specific gene expression including structural, metabolic and enzyme activities [32].
Co-administration of either metformin with doxorubicin (group V) or sitagliptin with doxorubicin (group VI) in the present study resulted in significant improvement of the body weight, blood pressure and heart rate, significant attenuation of doxorubicin-induced ST segment elevation and QT interval prolongation, significant reduction in the elevated serum level of CK-MB, LDH, blood glucose and the elevated cardiac MDA level, significant improvement of cardiac contractility in response to isoprenaline and significant improvement of histopathological signs of cardiotoxicity compared to doxorubicin treated group (group IV).
The present study prove that metformin produced more significant improvement than that produced by sitagliptin regarding systolic blood pressure, ST segment elevation, serum level of CK-MB, LDH, cardiac MDA level and histopathological finding and isolated heart contractility in response to isoprenaline.
The findings of the present study are in agreement with Aleisa et al. [33] who reported that pre-treatment with metformin could improve doxorubicin-induced increase in MDA in mice and Kelle et al. [34] who detected the protective effect of metformin could reduce cardiac MDA and the serum level of CK-MB and LDH in doxorubicin treated rats.
Metformin can ameliorate doxorubicin-induced oxidative stress through; increase cardiac superoxide dismutase (SOD) activity [35], increase in the glutathione level in heart tissue [36], enhancement of endothelium-dependent relaxation factor protection [37], modulation of ferritin heavy chain (FHC), that is responsible for sequestration of any excess intracellular iron [38] and prevent mitochondrial damage induced by doxorubicin [39].
Metformin through its activation of 5′-Adenosine Monophosphateactivated Protein Kinase (AMPK) reduces the generation of ROS [40] and increases endothelial nitric oxide synthase phosphorylation which prevents the opening of the mitochondrial permeability pores [41]. Also, AMPK can up regulate the activity of peroxisome proliferatoractivated receptor gamma coactivator-1α (PGC-1α), a tissue-specific transcriptional co-activator which plays a key role in regulating energy metabolism and mitochondrial bioenergetics responses [42].
Metformin increases the circulating levels of adiponectin and increase the expression of adiponectin receptors (adipoR1 and adipoR2) in skeletal muscle and adipose tissue [43]. Adiponectin is an adipokine synthesized in the adipose tissue, exerts vasodilator, antiapoptotic, anti-inflammatory and anti-oxidative activities in both cardiac and vascular cells [44] and attenuate the apoptosis induced by doxorubicin [45].
Prevention of the activation of caspase-3 may attribute in the metformin induced cardio protection [39]. The caspase-3 protein is a member of protease (caspase) family that plays a central role in the execution phase of cell apoptosis [46].
Cardio protective effect of sitagliptin may be due to reduction in inflammatory markers by down-regulation of COX-2 expression [47] and iNOS expression [48], decreasing free radicals [49] and nitro oxidative stress parameters (MDA and NOx) by increase SOD activity [50].
Stromal cell-derived factor 1α (SDF1α) is degraded by DPP4, so, Sitagliptin by inhibition of DPP4 can increase the stromal cell-derived factor 1α (SDF1α) concentration that promote vascular repair and neoangiogenesis. Also, DPP4 inhibition exerts ant atherosclerotic effects and reduces inflammation via inhibition of toll-like receptor 4- mediated up regulation of IL1β, IL6, and other proinflammatory cytokines [51].
Furthermore, sitagliptin may decrease the expression of fibrosis markers as transforming growth factor b (TGF-b1), tissue inhibitor of matrix metalloproteinases (TIMP-1) and collagen (Col1a1 & Col3a1) [52] and vascular calcification marker, osteopontin [53] that is known to be associated with vascular calcification and cardiovascular morbidity in humans [54].
The observed beneficial effects of metformin and sitagliptin could generate increasing interest in their potential use as adjunctive therapy to patients under treatment with doxorubicin. Clinical trials are needed to confirm the protective effect of metformin and sitagliptin on cardiotoxicity induced by doxorubicin. Also, safety profile for the concomitant use of either metformin or sitagliptin with doxorubicin for longer periods and in patients with malignant tumours must be studied.
Footnotes
This research did not receive any specific grants from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of Interest
The authors declare no conflict of interest.
References
- Minotti G, Menna P, Salvatorelli E, Cairo G, Gianni L (2004) Anthracyclines: molecular advances and pharmacologie developments in antitumor activity and cardio toxicity. Pharmacol Rev 56: 185-229.
- Herman EH, Zhang J, Chadwick DP, Ferrans V (2000) Comparison of the protective effects of amifostine and dexrazoxane against the toxicity of doxorubicin in spontaneously hypertensive rats. Cancer Chemother Pharmacol 45: 329-334.
- Danz ED, Skramsted J, Henry N, Bennett JA, Keller RS (2009) Resveratrol prevents doxorubicin cardiotoxicity through mitochondrial stabilization and the Sirt1 pathway. Free Radic Biol Med 46: 1589-1597.
- Horenstein MS, Vander Heide RS, L’Ecuyer TJ (2000) Molecular basis of anthracycline-induced cardiotoxicity and its prevention. Mol Genet Metab 71: 436-444.
- Ioannidis JP, Hesketh PJ, Lau J (2000) Contribution of dexamethasone to control of chemotherapy-induced nausea and vomiting: a meta-analysis of randomized evidence. J Clin Oncol 18: 3409-3422.
- Suba Z, Ujpál M (2006) Correlations of insulin resistance and neoplasms. Magy Onkol 50: 121-135.
- El Messaoudi S, Rongen GA, De-Boer RA, Riksen NP (2011) The cardioprotective effects of metformin. Curr Opin Lipidol 22: 445-453.
- Bhamra GS, Hausenloy DJ, Davidson SM (2008) Metformin protects the ischemic heart by the Akt-mediated inhibition of mitochondrial permeability transition pore opening. Basic Res Cardiol 103: 274-284.
- Gundewar S, Calvert JW, Jha S (2009) Activation of AMP-activated protein kinase by metformin improves left ventricular function and survival in heart failure. Circ Res 104: 403-411.
- Shah DD, Fonarow GC, Horwich TB (2010) Metformin therapy and outcomes in patients with advanced systolic heart failure and diabetes. J Card Fail 16: 200-206.
- Kooy A, De Jager J, Lehert P, Bets D, Wulffelé MG, et al. (2009) Long-term effects of metformin on metabolism and microvascular and macrovascular disease in patients with type 2 diabetes mellitus. Arch Intern Med 169: 616-625.
- Nikolaidis LA, Mankad S, Sokos GG, Miske G, Shah A, et al. (2004) Effects of glucagon-like peptide-1 in patients with acute myocardial infarction and left ventricular dysfunction after successful reperfusion. Circulation 109: 962-965.
- Chaykovska L, von Websky K, Rahnenfu¨hrer J, Alter M, Heiden S. et al. (2011) Effects of DPP-4 inhibitors on the heart in a rat model of uremic cardiomyopathy . PLoS ONE 6: e27861.
- Reagan-Shaw S, Nihal M, Ahmed N (2008) Dose translation from animal to human studies revisited. FASEB J 22: 3659-3661.
- Irvine RJ, White J, Chan R (1997) The influence of restraint on blood pressure in the rat. J Pharmacol Toxicol Methods 38: 157-162.
- Buelke-Sam J, Holson JF, Bazare JJ, Young JF (1978) Comparative stability of physiological parameters during sustained anesthesia in rats. Lab Anim Sci 28: 157-162.
- Kobashigawa LC, Xu YC, Padbury JF, Tseng YT, Yano N (2014) Metformin protects cardiomyocyte from doxorubicin induced cytotoxicity through an AMP-activated protein kinase dependent signaling pathway: an in vitro study. PloS ONE 9: 104888.
- De Nigris F, Rienzo M, Schiano C, Fiorito C, Casamassimi A (2008) Prominent cardioprotective effects of third generation beta blocker nebivolol against anthracycline-induced cardiotoxicity using the model of isolated perfused rat heart. Eur J Cancer 44: 334-340.
- Kozlovski VI, Lomnicka M, Chlopicki S (2006) Nebivolol and carvedilol induce NO-dependent coronary vasodilatation that is unlikely to be mediated by extracellular ATP in the isolated guinea pig heart. Pharmacol Rep 58: 103-110.
- Chan YH (2003) Biostatistics 102: quantitative data-parametric & non-parametric tests. Singapore Med J 44: 391-396.
- Xu Y, Liu Z, Sun J, Pan Q, Sun F, et al. (2011) Schisandrin B prevents doxorubicin-induced chronic cardiotoxicity and enhances its anticancer activity in vivo. PloS ONE 6: 28335.
- Tatlidede E, Şehirli Ö, Velioğlu-Öğünç A, Çetinel Ş, Yeğen BÇ, et al. (2009) Resveratrol treatment protects against doxorubicin-induced cardiotoxicity by alleviating oxidative damage. Free Radic Res 43: 195-205.
- Khatib NA, Medi S, Patel J (2011) Evaluation of concomitant treatment of simvastatin and zingiber officinale in doxorubicin induce cardiotoxicity in wistar rats. IJRAP 2: 660-664.
- Zhou S, Palmeira CM, Wallace KB (2001) Doxorubicin-induced persistent oxidative stress to cardiac myocytes. Toxicol Lett 121: 151-157.
- Shao J, Nangaku M, Inagi R, Kato H, Miyata T, et al. (2007) Receptor-independent intracellular radical scavenging activity of an angiotensin II receptor blocker. J hyper 25: 1643-1649.
- Arunachalam S, Pichiah PT, Achiraman S (2013) Doxorubicin treatment inhibits PPARγ and may induce lipotoxicity by mimicking a type 2 diabetes-like condition in rodent models. FEBS Lett 587: 105-110.
- Rashikh A, Najmi AK, Akhtar M, Mahmood D, Pillai KK, et al. (2011) Protective effects of aliskiren in doxorubicin-induced acute cardiomyopathy in rats. Hum Exp Toxicol 30: 102-109.
- Hadi N, Yousif NG, Al-Amran FG, Huntei NK, Mohammad BI, et al. (2012) Vitamin E and telmisartan attenuates doxorubicin induced cardiac injury in rat through down regulation of inflammatory response. BMC Cardiovasc Disord 12: 1.
- Ducroq J, Moha ou Maati H, Guilbot S, Dilly S, Laemmel E, et al. (2010) Dexrazoxane protects the heart from acute doxorubicin‐induced QT prolongation: a key role for IKs. Br J Pharmacol 159: 93-101.
- Machado NG, Baldeiras I, Pereira GC, Pereira SP, Oliveira PJ (2010) Sub-chronic administration of doxorubicin to Wistar rats results in oxidative stress and unaltered apoptotic signaling in the lung. Chem Biol Interact 188: 478-486.
- Alberto D, Roberto D, Daniela C, Maria T, Mauro P (2008) Biochemical Markers for Prediction of Chemotherapy-Induced Cardiotoxicity. Am J Clin Pathol 130: 688-695.
- Zordoky BN, Anwar-Mohamed A, Aboutabl ME, El-Kadi AO (2010) Acute doxorubicin cardiotoxicity alters cardiac cytochrome P450 expression and arachidonic acid metabolism in rats. Toxicol Appl Pharmacol 242: 38-46.
- Aleisa AM, Al-Rejaie SS, Bakheet SA (2007) Effect of metformin on clastogenic and biochemical changes induced by adriamycin in Swiss albino mice. Mutat Res 634 (1-2): 93-100.
- Kelleni MT, Entesar FA, Aly MA (2015) Effect of Metformin and Sitagliptin on Doxorubicin-Induced Cardiotoxicity in Rats: Impact of Impact of Oxidative Stress, Inflammation, and Apoptosis. J Toxicol 20: 15.
- Jayakumar P, Pugalendi KV, Sankaran M (2014) Attenuation of hyperglycemia-mediated oxidative stress by indole-3-carbinol and its metabolite 3, 3′-diindolylmethane in C57BL/6J mice. J Physiol Biochem 70: 525-534.
- Ashour AE, Sayed MM, Abd-Allah AR, Korashy HM, Maayah ZH, et al. (2012) Metformin rescues the myocardium from doxorubicin-induced energy starvation and mitochondrial damage in rats. Oxidat med cel long 43: 1-13.
- Xie W, Zhang SD, Ou XP, Yang TL (2009) Protective effects of metformin on low-density lipoprotein-induced endothelial dysfunction in rats. J Sout Med Univers 29: 890-893.
- Asensio-López MC, Sánchez-Más J, Pascual-Figal DA, Abenza S, Pérez-Martínez MT, et al. (2013) Involvement of ferritin heavy chain in the preventive effect of metformin against doxorubicin-induced cardiotoxicity. Free Radic Biol Med 57: 188-200.
- Asensio-Lopez MC, Sanchez-Mas J, Pascual-Figal DA, De-Torre C, Valdes M, et al. (2012)Ferritin heavy chain as main mediator of preventive effect of metformin against mitochondrial damage induced by doxorubicin in cardiomyocytes. Free Radic Biol Med 67: 19-29.
- Kewalramani G, Puthenveetil P, Wang F, Kim MS, Deppe S, et al. (2009) AMP-activated protein kinase confers protection against TNF-α-induced cardiac cell death. Cardiovasc Res 84: 42-53.
- Noeman SA, Hamooda HA, Baalash AA (2011) Biochemical study of oxidative stress markers in the liver, kidney and heart of high fat diet induced obesity in rats. Diabetol Metab Syndr 3: 17.
- Ashabi G, Khodagholi F, Khalaj L, Goudarzvand M, Nasiri M (2014) Activation of AMP-activated protein kinase by metformin protects against global cerebral ischemia in male rats: Interference of AMPK/PGC-1α pathway. Metab Brain Dis 29: 47-58.
- Asensio-López MC, Lax A, Pascual-Figal DA, Valdés M, Sánchez-Más J (2011) Metformin protects against doxorubicin-induced cardiotoxicity: involvement of the adiponectin cardiac system. Free Radic Biol Med 51:1861-1871.
- Li P, Shibata R, Unno K, Shimano M, Furukawa M, et al. (2010) Evidence for the importance of adiponectin in the cardioprotective effects of pioglitazone. Hypertension 55: 69-75.
- Zhu W, Cheng KK, Vanhoutte PM, Lam KS, Xu A (2008) Vascular effects of adiponectin: molecular mechanisms and potential therapeutic intervention. Clin Sci 114: 361-374.
- Agosto M, Azrin M, Singh K, Jaffe AS, Liang BT (2011) Serum caspase-3 p17 fragment is elevated in patients with ST-segment elevation myocardial infarction: a novel observation. J Am Coll Cardiol 57: 220-221.
- Liu L, Liu J, Tian XY, Wong WT, Lau CW, et al. (2014) Uncoupling protein-2 mediates DPP-4 inhibitor-induced restoration of endothelial function in hypertension through reducing oxidative stress. Antioxid Redox Signal 21: 1571-1581.
- Nader MA, El-Awady MS, Shalaby AA, El-Agamy DS (2012) Sitagliptin exerts anti-inflammatory and anti-allergic effects in ovalbumin-induced murine model of allergic airway disease. Naunyn Schmiedebergs Arch Pharmacol 385: 909-919.
- Chang G, Zhang P, Ye L, Lu K, Wang Y, et al. (2013) Protective effects of sitagliptin on myocardial injury and cardiac function in an ischemia/reperfusion rat model. Eur J Pharmacol 718: 105-113.
- Nader MA (2014) Sitagliptin ameliorates lipid profile changes and endothelium dysfunction induced by atherogenic diet in rabbits. Naunyn-Schmiedeberg's archives of pharmacology 387: 433-444.
- Ussher JR, Drucker DJ (2012) Cardiovascular biology of the incretin system. Endocr Rev 33: 187-215.
- Susana R, Amaia Z, Dıez J (2012) GLP-1 and Cardioprotection: From Bench to Bedside. Cardiovasc Res 94: 1-8.
- Yan X, Sano M, Lu L, Wang W, Zhang Q, et al. (2010) Plasma concentrations of osteopontin, but not thrombin-cleaved osteopontin, are associated with the presence and severity of nephropathy and coronary artery disease in patients with type 2 diabetes mellitus. Card Diab 9: 70-78.
- Tokudome T, Mizushige K, Noma T, Manabe K, Murakami K, et al. (2000) Prevention of doxorubicin (adriamycin)-induced cardiomyopathy by simultaneous administration of angiotensin-converting enzyme inhibitor assessed by acoustic densitometry . J Cardiovasc Pharmacol 36: 3618.
Relevant Topics
- Applied Biopharmaceutics
- Biomarker Discovery
- Biopharmaceuticals Manufacturing and Industry
- Biopharmaceuticals Process Validation
- Biopharmaceutics and Drug Disposition
- Clinical Drug Trials
- Clinical Pharmacists
- Clinical Pharmacology
- Clinical Research Studies
- Clinical Trials Databases
- DMPK (Drug Metabolism and Pharmacokinetics)
- Medical Trails/ Drug Medical Trails
- Methods in Clinical Pharmacology
- Pharmacoeconomics
- Pharmacogenomics
- Pharmacokinetic-Pharmacodynamic (PK-PD) Modeling
- Precision Medicine
- Preclinical safety evaluation of biopharmaceuticals
- Psychopharmacology
Recommended Journals
Article Tools
Article Usage
- Total views: 4662
- [From(publication date):
September-2017 - Aug 17, 2025] - Breakdown by view type
- HTML page views : 3633
- PDF downloads : 1029