Research Article Open Access
3D-CT Mammary Lymphography Can Help Selective Axillary Dissection of Breast Lymph Flow Differed from the Arm
| Koji Yamashita* and Kazuo Shimizu | |
| Department of Breast Surgery, Nippon Medical School, Japan | |
| Corresponding Author : | Koji Yamashita Department of Breast Surgery, Nippon Medical School 1-5, Sendagi-1, Bunkyo-ku, Tokyo, 113-8602, Japan Tel: +81-3-5814-6219 Fax: +81-3-5685-0985 E-mail: yamasita@nms.ac.jp |
| Received December 19, 2013; Accepted January 25, 2014; Published January 27, 2014 | |
| Citation: Yamashita K, Shimizu K (2014) 3D-CT Mammary Lymphography Can Help Selective Axillary Dissection of Breast Lymph Flow Differed from the Arm. OMICS J Radiol 3:158. doi:10.4172/2167-7964.1000158 | |
| Copyright: © 2014 Yamashita K, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Visit for more related articles at Journal of Radiology
Abstract
Background: Axillary Nodes (AN) dissection causes many complications such as lymphedema. Three Dimensional Computed Tomographic (3D-CT) Lymphography (LG) can show the precise individual lymphatic flow from the breast tumor to Sentinel Node (SN), and through AN to venous angle, which means breast lymphatic channel. We applied 3DCT LG to distinguish them from the arm channel to avoid any arm complications.
Methods: 3D-CT LG used contrast medium injected subcutaneously above the tumor, near the areola and the inner upper arm. CT images were taken 1, 3 and 5 minutes after injection. SN biopsy and AN dissection were performed by dye-method using endoscopy.
Results: We performed SN biopsy with 3D-CT LG on 160 patients. 3D-CT LG clearly showed the precise lymphatic flow from the tumor to SN one minute after injection and five axillary node groups into venous angle 5 minutes after injection. Detection rate of SN, the second, the third, and the fifth group was 100%, 88%, 80%, and 30% respectively. The arm lymph flow was distinguished by the connecting ducts between the breast and the arm channel, observed 39% and 71% in the fourth and the fifth group. Five patients (3.1%) had the common SN from the breast and the arm channel, and suffered arm lymphedema only after SN biopsy. We dissected AN from the breast by endoscopic surgery on the SN-positive patients.
Conclusions: 3D-CT LG can helps us easily to distinguish between the breast and the arm channel by finding the connecting ducts.
| Keywords |
| Arm lymph flow; Axillary dissection; Sentinel lymph node biopsy; Lymphography; 3D-CT; Breast cancer |
| Background |
| In early breast cancer, the presence of metastasis in Axillary Lymph Nodes (AN) is an important factor in prognosis and further treatment. However, AN dissection causes many complications such as contracture of shoulder joint, lymph edema, and paralysis of upper extremities [1]. Sentinel Node (SN) biopsy provides us valuable information about no need to dissect AN for node-negative patients [2,3]. However, on nodepositive patients, the conventional AN dissection has been performed. |
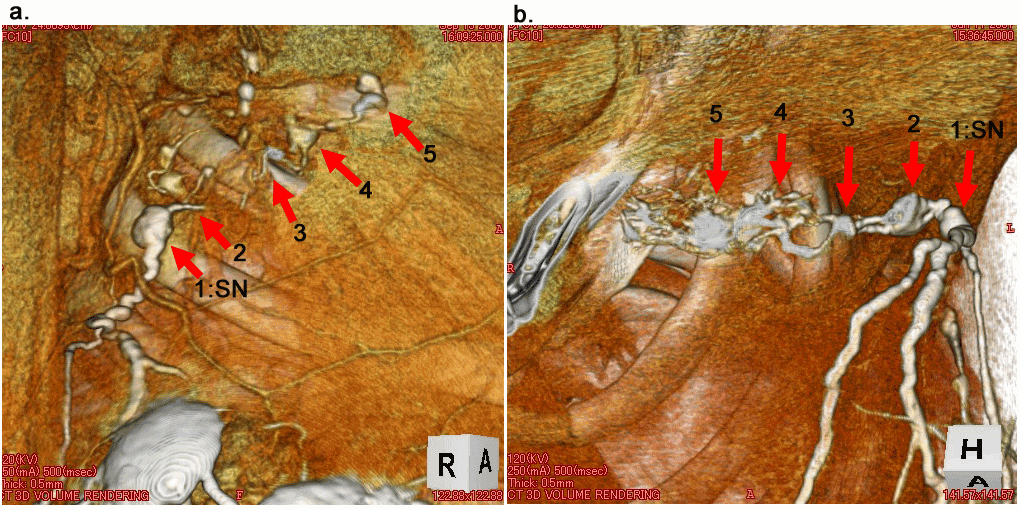
| SN is defined as the first lymph node drained of lymph flow from the tumor [4,5]. The most commonly used methods to identify the SN are dye-staining [6,7] and radioisotope incorporation [8,9]. Multidetectorrow three-Dimensional Computed Tomography (3D-CT) mammary Lymphography (LG) can be used to mark the precise location of the SN on the skin before the operation [10-12]. The detailed relations between lymph nodes and lymph flow can be clarified using 3D-CT LG [13]. And the late phase of 3D-CT LG of the breast can show the axillary lymphatic architecture from SN into venous angle [14]. These nodes are classified into 5 groups. |
| We applied 3D-CT LG technique to the lymphography of the arm. The lymph flow from the arm runs along the axillary vein. In the axilla, the connections can be observed between the lymph channel from the breast and from the arm. We will use this information to avoid any arm complications. |
| Materials and Methods |
| Patients |
| From July 2002 to March 2009, 600 patients with breast disease were treated surgically by us in the Department of Breast Surgery at the Nippon Medical School of Musashikosugi Hospital and Main Hospital. We performed Video-Assisted Breast Surgery (VABS) for the patients with a limited small breast cancer at surgery [15,16]. We also performed SN biopsy for the patients without clinical axillary node metastasis. A SN biopsy was performed for 200 patients, 160 of which involved SN biopsy using VABS and 3D-CT LG. 3D-CT LG was performed on 160 patients. The arm LG was performed on 30 patients. |
| 3D-CT lymphography |
| Interstitial 3D-CT LG was performed using a 16-channel multidetector-row helical 3D-CT scanner (Toshiba Aquilion 16; Toshiba Medical Systems Corporation, Tochigi, Japan). The patients were placed in the supine position with arms positioned in the lateral abduction direction suitable for the operating position. After local anesthesia by subcutaneous injection of 1% lidocaine (0.5 ml), 2 ml of iopamidol (Iopamiron 300; Nihon Shering, Osaka, Japan) were injected intracutaneously into the periareolar skin and the skin above the tumor. A CT image with a 3-mm slice thickness was taken 1 and 5 minutes after injection. SN was identified on transaxial CT images, and their location was marked on the skin surface with an oil-painting pen using a laser pointer of CT the day before the surgery. Next, 3D-CT images were reconstructed from transaxial enhanced CT images, which clearly showed the lymph ducts and SN. 3D-CT LG was also performed for the arm LG. 2 ml of iopamidol were injected in the upper inner arm along the medial intramuscular groove of the ipsilateral arm after local anesthesia. A CT image was taken 5 minutes after injection on the same condition. |
| Surgical methods |
| Previously, VABS has been described in detail [15]. The following operative procedures were performed: skin incision in the axilla and/ or periareola, skin flap formation via the tunnel method [17], pectoral muscle fascia dissection, vertical section of the mammary gland, SN biopsy by the dye-staining method guided by preoperative 3D-CT LG marking, and axillary node dissection (levels 1 and 2) on the SNpositive patients. We performed only partial mastectomy of VABS in this study. Radiotherapy and chemotherapy were performed after surgery on the indicated patients for adjuvant therapy. |
| The SN biopsy was performed by the dye-staining method using a part of the VABS technique at the beginning of the operation before gland resection. In the periareolar region and over the tumor, 2 ml of 1% indocyanine green were injected subcutaneously. A 1-cm-long skin incision was made along wrinkles in the axilla at the position marked by 3D-CT LG. A Visiport optical trocar (Tyco Healthcare Japan, Tokyo, Japan) was inserted into the incision after 20 minutes. The endoscopic view was observed through the Visiport with a 10-mm-diameter straight-angled rigid endoscope (Olympus Optical, Tokyo, Japan), and the stained nodes were found by following the dye in the lymph ducts. |
| The lymph nodes were sampled, and metastasis was determined from fast frozen sections. To remove them, we left the external tube of the Visiport and grasped the nodes by the endodissector (Johnson & Johnson Company, New Brunswick, NJ, USA), then cut and shielded the surrounding lymph duct and vessels using the harmonic scalpel (Johnson & Johnson Company). Axillary node dissection was performed at levels 1 and 2 with bipolar scissors through the same incision, which was lengthened to 2.5 cm. The inferior pectoral nerve, long thoracic nerve, second and third intercostobrachial nerves, thoracodorsal nerve, artery, and vein were observed and preserved. The lateral pectoral artery was preserved for the lateral tissue flap. After surgery, SN and axillary nodes were pathologically examined by standard hematoxylin and eosin staining. Breast reconstruction was simultaneously performed either by mobilization of the remnant mammary gland and by filling the Lateral thoracic fat Tissue Flap (LTF) or by filling an absorbent synthetic fiber [17]. |
| Informed consent for the procedure was obtained from all the patients before surgery. This study was approved by Institutional Review Board and Ethics Committee and obtained the written Informed Consent. |
| Results |
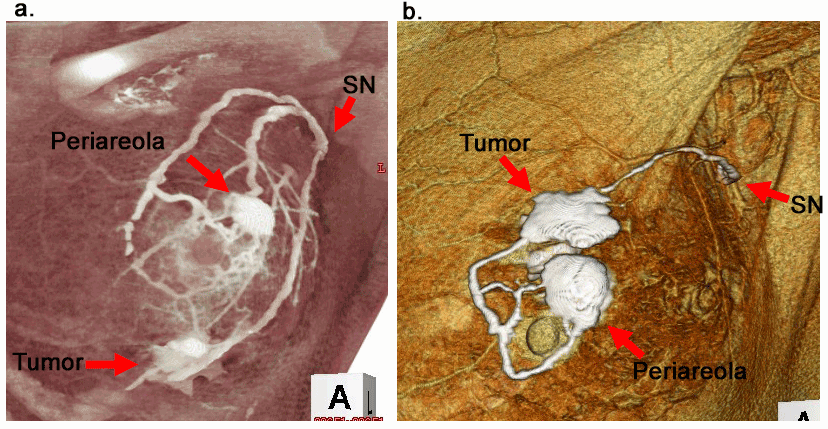
| We have performed 3D-CT LG on 160 patients, and the arm lymphography on 30 patients. The patient characteristics are shown in Table 1. Down-staged cases managed by Preoperative Systemic Chemotherapy (PST) are included in this study. These patients did not have any clinical metastasis of axillary nodes after PST. 3D-CT LG clearly showed the precise lymphatic flow from the tumor to SN (Figure 1). 3D-CT LG showed periareolar circular lymph ducts and complicated radial breast subcutaneous lymph ducts flow. They were connected to make a network. It can show SN at only one minute after injection. By following up to 5 minutes, we can follow the lymph ducts beyond SN into the second to the fifth node groups toward the venous the forth, and the fifth group was 100%, 88%, 80%, 36%, and 30% respectively. |
| The complications of 3D-CT have not been experienced except for the slight injection pain of the anesthesia. There was not allergic reaction and any other complications. |
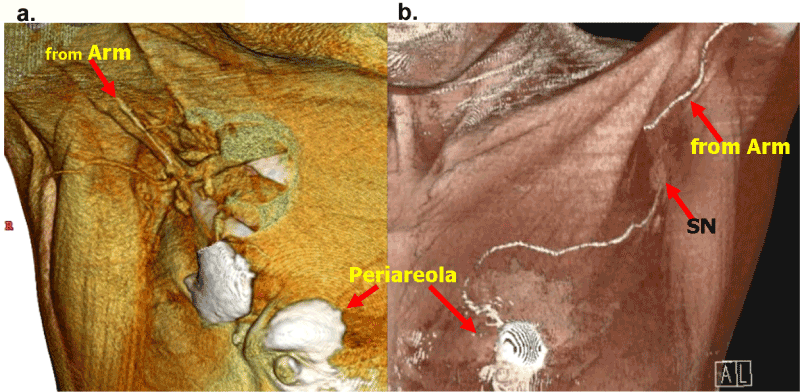
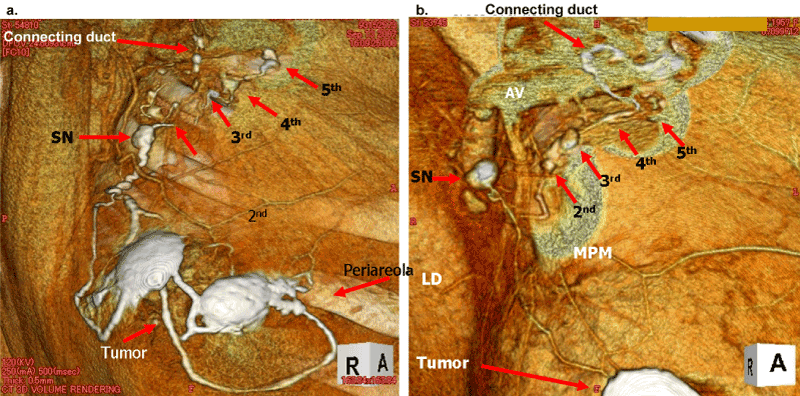
| The lymph flow originated from the arm can be visualized by injecting contrast enhancing medium iopamidol into the inner upper arm along the medial intramuscular groove on 3D-CT LG (Figure 3). The lymph ducts from the arm run along the arm vein and onto the axillary vein. Then they were joined to the cross-over connecting ducts over the axillary vein, branched from the axillary lymphatic system originated from the breast (Figure 4). |
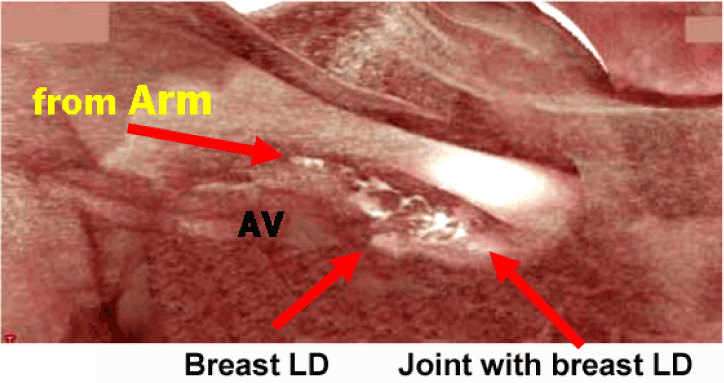
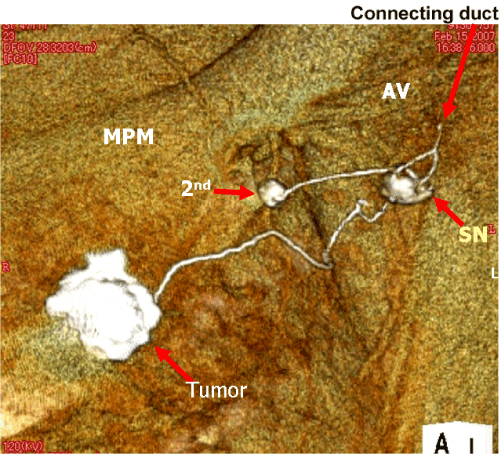
| We reviewed the previously performed 160 cases of 3D-CT LG to find those connecting lymph ducts (Figure 5). The location of them is shown in Table 2. The arm lymph flow was distinguished by the connecting ducts, observed at a rate of 39% and 71% beyond the fourth and the fifth groups of axillary nodes respectively. Most ducts were branched over the fourth group. However, five patients had a single common SN from the arm and the breast (Figure 6). Although these five patients (3.1%) underwent only SN biopsy, they suffered from mild lymphedema of the arm without axillary dissection. They could be relieved by hand massage and bandage. And there was no local recurrence and no distant metastasis on these patients. |
| We have performed the endoscopic SN biopsy oriented by 3DCT LG on 160 patients, and the endoscopic axillary dissection on 220 patients. We could distinguish and sample sentinel nodes and axillary grouped nodes drained from the breast, guided by 3D-CT LG and dyestaining using endoscopic surgery. |
| Discussion |
| We previously reported that 3D-CT LG could show the precious lymphatic architecture of the breast and the axilla, and its usefulness in SN biopsy [13,14,18]. The procedure takes only about 1 minute after injection of iopamidol subcutaneously over the tumor and near the nipple for representing SN precisely (Figure 1). CT Examination of iopamidol flow 1, 3, and 5 minutes after injection revealed that the flow extended over the SN into the next nodes, then to the next nodes repetitively, and finally into the venous angle in half of the patients examined (Figure 2). Therefore, we can easily ascertain the tracts through which cancer cells will spread during metastasis. As a representative example, Figure 2 shows a clear view of a string of five bead-like grouped lymph nodes beyond the SN into the center of the body. We classified these into five node groups in relation with the location of the pectoralis minor muscle. |
| We verified the identification of SNs detected by RI uptake and 3DCT LG in the fusion study of SPECT and 3D-CT LG, which is referred to the section of SPECT-CT of the previous manuscript [19]. During surgery, we conventionally use dye and RI to detect SN. However, we can approach from the marking on the skin made by 3D-CT LG directly to SN. We can detect the other lymph nodes without RI-uptake or dye-stain by the relative position on the lymph node map of the axilla, which is also made by 3D-CT LG in the anatomical architecture. |
| In this study, we injected contrast enhancing material into the upper inner arm along the medial intramuscular groove in 3D-CT LG to show the lymph flow from the arm to the axilla. This arm lymph flow runs along the upper arm vein and the axillary vein into the axilla and joins with the flow from the breast at many different sites, which are coincided with the cross-bridge over the axillary vein. We defined the cross-bridge duct on the axillary vein as a connecting lymph duct between the breast and the arm lymphatic channels in the axilla. |
| Therefore, we reviewed all patient data of 3D-CT LG for about the sites of connecting duct from the breast lymphatic channel over axillary vein. The two figures in Figure 5 show the connecting ducts, which means the lymph flow of the breast lymphatic channel joins to the arm channel. |
| We can use this 3D-CT LG to know about the key point to avoid harming the lymphatic pathway from the arm. In order to avoid arm lymphedema, it is necessary to be restricted to dissect axillary lymph nodes and fat tissue prior to the connecting ducts and caudal to the axillary vein. The selective axillary dissection needs such information. |
| We performed the selective axillary dissection on eleven patients with arm LG. There was no increasing of postoperative seroma in the axilla and no lymphedema at six years after surgery on average. The pathological findings showed the node metastasis was found only in SN and the second node group, and the number was less than four, which means pathological n1 diseases. And there was no local recurrence and no distant metastasis. I think this selective axillary dissection is better management on the patients without massive axillary metastasis. |
| The connecting lymph ducts were located almost after 4th and 5th node groups. However, some patients had early junctions. Figure 6 shows the example that SN was directly connected to the arm lymph flow. This means the common SN from both the breast and the arm. We experienced five patients had the same lymph duct connection with SN. Actually, although these patients underwent only SN biopsy, they suffered from arm lymphedema even without axillary dissection. The arm lymphedema suffered five years after SN biopsy was reported its rate as 5%, compared with the rate after axillary dissection as 16% [20]. High body weight, infection, and injury were significant risk factors for developing lymphedema. However, radiation and type of breast surgery (breast conserving surgery v mastectomy) were not associated with the prevalence of lymphedema. Therefore, we cannot predict the incidence of lymphedema after SN biopsy by the other than high body weight. The common SN shown by 3D-CT LG will give us important information about the risk of lymphedema. If we know the risk, we can prevent it by the aggressive rehabilitation of arm after SN biopsy like after axillary dissection. |
| Dual identification technique of breast and arm lymph flow has been reported recently by using different materials. Britton TB, et al reported to inject two types of isotopes 99mTc and 111In into near areola and hand respectively [21]. And two colors of fluorescence were injected to visualize their pathway through the skin in mice [22]. SN biopsy and axillary dissection were attempted by arm reverse mapping by two colors of dyes [23,24]. The lymphoscintigraphy using isotopes has been still poor spatial resolution (>1 cm), but also showed common SN in some patients. The fluorescence study can show the precise lymph flow in mice, but in human study the thick subcutaneous fat tissue disturbs the permeability of fluorescence to be visualized through the skin. Two colors of dye-staining mapping can show the existence of lymph flow from the arm in the axilla, but it may be through collateral connecting ducts. The main pathway from the arm may exist out of the lymph dissected area. 3D-CT LG can show the main pathway and the connecting ducts from the breast lymph channel. |
| A new approach of lymphangiography has been reported by using MRI in mice [25,26]. It can show the precise lymphatic system in the anatomical architecture. It will be helpful for us to perform the selective axillary dissection and to preserve the arm lymph channel, like 3D-CT LG. However, it needs long time to exam, so it cannot follow the rapid lymph flow as one minute and three minutes after injection like CT. |
| We have performed endoscopic surgery for breast diseases, named as Video-Assisted Breast Surgery (VABS) [27,28]. It is also useful for SN biopsy. The incisional wound is only 1 cm long and inconspicuous in the axilla. VABS SN biopsy needs only narrow blunt dissection of about 1 cm in diameter with a distance from the skin to SN. It makes lower infestation than the open SN biopsy. The 3D-CT LG procedure can help VABS in finding the accurate SN as well as the second and the third node groups [29]. It will also be able to distinguish the connecting lymph ducts on VABS axillary dissection for preserving the arm lymphatic channel. |
| By 3D-CT LG, we can recognize the accurate and more precise lymph flow in the surrounding anatomical architecture. It helps us easily to distinguish the breast channel and the arm channel by finding the connecting ducts, and to pursuit lymph flow and to remove SN and axillary nodes from the breast selectively using endoscopic surgery, for preventing any arm complications like lymphedema. |
References |
|
Tables and Figures at a glance
| Table 1 | Table 2 |
Figures at a glance
 |
 |
 |
| Figure 1 | Figure 2 | Figure 3 |
 |
 |
 |
| Figure 4 | Figure 5 | Figure 6 |
Relevant Topics
- Abdominal Radiology
- AI in Radiology
- Breast Imaging
- Cardiovascular Radiology
- Chest Radiology
- Clinical Radiology
- CT Imaging
- Diagnostic Radiology
- Emergency Radiology
- Fluoroscopy Radiology
- General Radiology
- Genitourinary Radiology
- Interventional Radiology Techniques
- Mammography
- Minimal Invasive surgery
- Musculoskeletal Radiology
- Neuroradiology
- Neuroradiology Advances
- Oral and Maxillofacial Radiology
- Radiography
- Radiology Imaging
- Surgical Radiology
- Tele Radiology
- Therapeutic Radiology
Recommended Journals
Article Tools
Article Usage
- Total views: 14171
- [From(publication date):
April-2014 - Aug 23, 2025] - Breakdown by view type
- HTML page views : 9584
- PDF downloads : 4587
