Effective Management of Severe Diverticular Bleeding in Japanese Patients
Received: 03-Mar-2016 / Accepted Date: 08-Mar-2016 / Published Date: 11-Mar-2016
Abstract
Aim: Diverticular bleeding is a common cause of lower gastrointestinal bleeding. Aggressive treatment because of spontaneous hemostasis is typically required only in severe cases. To determine the clinical characteristics of Japanese patients with diverticular bleeding and evaluate the treatment methods used with the aim of developing an effective management approach.
Method: We conducted a retrospective cohort study of 626 Japanese patients (769 endoscopic procedures) who underwent diagnostic lower gastrointestinal endoscopy at Fujita Health University Hospital between November 2011 and April 2014.
Results: Diverticular bleeding was the most frequent cause of lower gastrointestinal bleeding, occurring in 155 patients. Concurrent illness, such as hypertension, was common and 92 patients were receiving anticoagulant or antiplatelet therapy on admission. However, no significant clinical differences were observed between patients with severe and mild diverticular bleeding. Computed tomography (CT) was useful for a treatment policy decision of the one from diverticular bleeding as well as other lower gastrointestinal bleeding. Clipping via colonoscopy could stop bleeding effectively for patients with an identified bleeding point. Barium impaction had utility in patients with persistent bleeding. Angiographic hemostasis or colectomy (intestinal resection) was only performed in uncontrolled patients in whom clipping and barium impaction was not possible.
Conclusion: No significant clinical differences were observed between patients with severe and mild diverticular bleeding. CT angiography was useful for a treatment policy decision of the one from diverticular bleeding. Endoscopic clipping and barium impaction had efficacy in severe cases of diverticular bleeding, at first.
Keywords: Angiographic hemostasis; Colectomy; CT angiography; Diverticular bleeding; Endoscopic clipping; High-dose barium impaction
5162Introduction
Lower gastrointestinal bleeding is defined as bleeding from the anal side of the ligament of Treitz [1-3]. Gastrointestinal bleeding can be broadly classified into small intestinal and large intestinal bleeding. Colonic diverticular bleeding is considered a major cause of large intestinal bleeding in addition to benign anorectal diseases, such as ischemic colitis, colorectal tumors, infectious enteritis, hemorrhoidal bleeding, and hemorrhagic rectal ulcers [2,3].
Treatment options for lower gastrointestinal bleeding depend on the underlying etiology. Ischemic colitis reportedly resolves with conservative treatment and requires aggressive treatment only in cases of vaso-occlusive disease [4,5]. Benign anorectal diseases, such as hemorrhoids or hemorrhagic rectal ulcers, are treated transanally and/or by pharmacological therapy [6] and bleeding sites can usually be easily identified. Arteriovenous malformations can often be treated by endoscopic clipping or argon plasma coagulation [7]. Furthermore, bleeding caused by colorectal tumors, inflammatory bowel disease, or infectious colitis should be controlled by treating the respective underlying causes. Diverticular bleeding is known to spontaneously resolve in the majority of cases [8-10] and therefore requires aggressive treatment only in severe cases. At our hospital, the treatment options for severe cases of diverticular bleeding include endoscopic clipping [11-14], high-dose barium impaction [15-17], angiographic hemostasis [18,19], and intestinal resection. However, currently, there is a lack of consensus regarding the most appropriate treatment methods for particular clinical presentations. Furthermore, the severity of diverticular bleeding cannot be accurately determined in all cases. Patients may be at increased risk due to the time required to decide on treatment plans where there is a lack of definitive treatment strategy. Furthermore, endoscopists in our hospital have varied levels of skills and competencies.
In the present study, we retrospectively analyzed the characteristics of patients with lower gastrointestinal bleeding in our hospital, including clinical characteristics, concurrent illness, use of anticoagulant or antiplatelet therapy on presentation, and recurrence of bleeding. We further evaluated the treatment outcomes of patients with diverticular bleeding for effective hemostasis.
Patients and Methods
Patients
High-dose barium impaction for hemostatic treatment of diverticular bleeding was introduced at Fujita Health University Hospital in November 2011. We conducted a retrospective cohort study in this hospital between November 2011 and April 2014. The study was conducted in accordance with the ethical standards of the responsible committee on human experimentation and with the Helsinki Declaration of 1975, as revised in 1983. Only data from patients who provided written informed consent was used in this study. We evaluated disease etiology, concurrent illness, and use of anticoagulant or antiplatelet therapy in 626 patients (769 endoscopic procedures) who underwent diagnostic lower gastrointestinal endoscopy in our hospital for lower gastrointestinal bleeding. Patients with positive fecal occult blood tests and those undergoing follow-up endoscopy for inflammatory bowel disease were excluded.
Treatments
Colonoscopy was performed by 19 endoscopists in our department. Colonoscopy in cases of diverticular bleeding without pretreatment with oral intestinal lavage liquid (pretreatment) was performed by 12 experienced endoscopists who had a cumulative total experience of >400 colonoscopies. The clinical characteristics of patients with and without lower gastrointestinal diverticular disease were compared. Patient characteristics, bleeding sites, concurrent illness, use of anticoagulant and antiplatelet therapy, recurrent or persistent bleeding, and treatment modalities performed in cases of diverticular bleeding were analyzed. We defined recurrence as bleeding occurring for >2 weeks following previous instance of hemostasis. Persistent bleeding was defined as bleeding re-occurring within 2 weeks. In addition, the efficacy and rates of recurrent bleeding for endoscopic clipping, highdose barium impaction, angiographic hemostasis, and surgical treatment were evaluated. In endoscopic clipping, hemostasis is achieved by attaching a metal clip to the hemorrhage site using an endoscope [11-14]. In high-dose barium impaction, hemostasis is achieved by performing enema to fill diverticula with highly concentrated barium [15-17]. Angiographic hemostasis involves ablation of the vein of the responsible lesion under angiographic guidance [18,19]. Surgical treatment (colectomy) for diverticular bleeding involves partial resection of the large intestine that includes the hemorrhage site.
Statistical analysis
The chi-square and Fisher’s exact tests were used for statistical analyses. Statistical significance was set at a P-value of <0.05. An effective approach for the management of patients with diverticular bleeding in our hospital was devised following data analysis.
Results
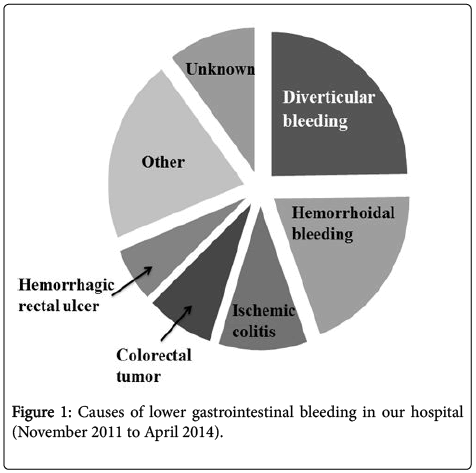
A total of 626 patients suffered lower gastrointestinal bleeding during the study period. Etiologies, in order of frequency, included diverticular bleeding (including strongly suspected cases), hemorrhoidal bleeding, ischemic colitis, colorectal tumor, and hemorrhagic rectal ulcer (Figure 1). Less frequent causes of lower gastrointestinal bleeding included arteriovenous malformations, infectious colitis, collagen disease, and initial inflammatory bowel disease. Lower gastrointestinal bleeding of unknown cause was recorded in cases with two or more suspicious bleeding lesions.
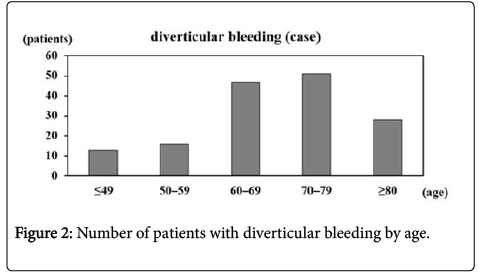
We compared patients with and without lower gastrointestinal diverticular bleeding. The median age of the 155 patients with diverticular bleeding was 69 years, with the frequency peaking between the ages of 60 and 79 years (Figure 2 and Table 1a).
| Diverticular bleeding | Non-diverticular lower gastrointestinal bleeding | p value | |
|---|---|---|---|
| Number of patients | 155 | 471 | |
| Age (median) | 28-92 (69) | 3-98 (67) | |
| Concurrent illness | 119 (76.8%) | 317 (63.7%) | 0.0261 |
| Anticoagulant and/or Antiplatelet | 42 (27.1%) | 104 (22.1%) | 0.2002 |
Table 1a: Patient characteristics; Comparison of retrospectively collected clinical characteristics of patients with and without lower gastrointestinal diverticular bleeding in our hospital between November 2011 and April 2014. Bold values indicate significant differences (P < 0.05).
The median age of patients with non-diverticular lower gastrointestinal bleeding was 67 years, indicating a trend toward patients with diverticular bleeding being older than those with other etiologies (Table 1a). Common concurrent illness included cardiovascular diseases (including hypertension), diabetes, cerebrovascular diseases, and malignant neoplasms. Concurrent illness were more common in patients with diverticular bleeding than in those with non-diverticular lower gastrointestinal bleeding (Tables 1a and 1b). Cardiovascular diseases, particularly hypertension, were found to be significantly more prevalent in patients with diverticular bleeding than in those with non-diverticular lower gastrointestinal bleeding. Conversely, malignant neoplasms were significantly more prevalent in patients with non-diverticular lower gastrointestinal bleeding than in those with diverticular bleeding (Table 1b). However, no significant difference in the prevalence of malignant neoplasms was observed when patients with malignant tumors and radiation colitis were excluded (data not shown). Chronic renal failure was significantly more prevalent in patients with diverticular bleeding than in those with non-diverticular lower gastrointestinal bleeding (Table 1b). However, this difference could not be accurately evaluated because of the small number of cases. Use of anticoagulant and particularly antiplatelet therapy was more prevalent in patients with diverticular bleeding than in those with non-diverticular lower gastrointestinal bleeding (Table 1a and 1c).
| Concurrent illness | Diverticular bleeding | Non-diverticular lower gastrointestinal bleeding | p value |
|---|---|---|---|
| Cardiovascular disease | 103 (66.5%) | 224 (47.6%) | <0.0001 |
| Hypertension | 84 (54.2%) | 172 (36.5%) | 0.0001 |
| Arrhythmia | 26 (16.8%) | 53 (11.3%) | 0.0726 |
| Ischemic heart disease |
24 (15.5%) | 63 (13.4%) | 0.5401 |
| Other cardiovascular disease |
22 (14.2%) | 46 (9.8%) | 0.1244 |
| Diabetes mellitus | 32 (20.6%) | 75 (15.9%) | 0.1756 |
| Cerebrovascular disease | 18 (11.6%) | 52 (11.0%) | 0.8445 |
| Chronic renal failure | 12 (7.7%) | 15 (3.2%) | 0.0154 |
| Malignant neoplasm | 22 (14.2%) | 119 (25.3%) | 0.0042 |
| Other disease | 4 (2.6%) | 11 (2.3%) | 0.8625 |
| Total | 119 (76.8%) | 317 (63.7%) | 0.0261 |
Table 1b: Concurrent illness; Comparison of retrospectively collected clinical characteristics of patients with and without lower gastrointestinal diverticular bleeding in our hospital between November 2011 and April 2014. Bold values indicate significant differences (P < 0.05).
| Drug | Diverticular bleeding | Non-diverticular lower gastrointestinal bleeding | p value |
|---|---|---|---|
| Anticoagulant | 12 (7.7%) | 20 (4.3%) | 0.0865 |
| Warfarin | 11 | 13 | |
| Rivaroxaban | 1 | 3 | |
| Heparin | 0 | 2 | |
| Fondaparinux | 0 | 1 | |
| Dabigatran | 0 | 1 | |
| Antiplatelet | 33 (21.3%) | 67 (14.2%) | 0.0373 |
| Aspirin | 19 | 45 | |
| Clopidogrel | 7 | 7 | |
| Ticlopidine | 1 | 3 | |
| Cilostazol | 0 | 2 | |
| Icosapentate | 3 | 1 | |
| Aspirin + Clopidogrel | 1 | 8 | |
| Aspirin + Cilostazol | 2 | 0 | |
| Clopidogrel + Cilostazol | 0 | 1 | |
| Anticoagulant + Antiplatelet | 3 (1.9%) | 17 (3.6%) | 0.4445 |
| Warfarin + Aspirin | 3 | 15 | |
| Rivaroxaban + Aspirin | 0 | 1 | |
| Ticlopidine + Aspirin | 0 | 2 | |
| Total | 42 (27.1%) | 104 (22.1%) | 0.2002 |
Table 1c: Prescribed anticoagulant or antiplatelet agents; Comparison of retrospectively collected clinical characteristics of patients with and without lower gastrointestinal diverticular bleeding in our hospital between November 2011 and April 2014. Bold values indicate significant differences (P < 0.05).
We next conducted a detailed analysis of patients with diverticular bleeding (Table 2). Patients with diverticular bleeding tended to be older and were more likely to be male; 69.0% were ≥65 years and 60.6% were male (Figure 2 and Table 2a). There was no difference in the rate of pretreatment use (Table 2a). Of the 155 included patients, 29% (n = 45) had diverticula affecting the right colon (cecum and ascending colon), 15.5% (n = 24) had diverticula affecting the left colon (descending and sigmoid colon), and 55% (n = 86) had diverticula affecting both sides of the colon. Diverticular bleeding occurred in the right colon in 25.2% (n = 39) patients, in the left colon in 16.1% (n = 25) patients, and could not be determined in 58.7% (n = 91) patients (Table 2a). Diverticular bleeding sites were less frequently determined in patients who had received pre-treatment. Diverticular bleeding occurred only once in 70.3% (n = 109) patients (Table 2a).
| Diverticular bleeding | 155 patients |
|---|---|
| Age (young/elderly)* | 48/107 |
| Sex (male/female) | 94/61 |
| Pretreatment (yes/no) | 75/80 |
| Site of diverticula (right/both/left) | 45/86/24 |
| Bleeding site (right/left/unclear) | 39/25/91 |
| Bleeding times (once/ more than twice) | 109/46 |
| Hemostatic treatment (yes/no) | 25/130 |
Table 2a: Patient characteristics; Clinical characteristics of patients with diverticular bleeding in our hospital (November 2011 to April 2014).
Approximately 84% patients with diverticular bleeding improved following fasting, bed rest, and intravenous fluid administration. Interventions to restore hemostasis following diverticular bleeding were performed in 25 patients (Table 2a) and included high-dose barium impaction (n=15), endoscopic clipping (n=12), angiographic hemostasis (n=1), and colectomy (n=1; Table 2b). Five patients experienced bleeding recurrence and required repeat interventions to restore hemostasis (Table 2b). Clinical characteristics, including age, sex, diverticulum site, concurrent illness, and use of anticoagulant or antiplatelet therapy, were compared between patients with and without recurrent bleeding after interventions to restore hemostasis during the follow-up period. No significant differences in any clinical characteristic were observed (data not shown). Therefore, patients requiring repeated interventions to restore hemostasis were assessed as reference when determining the appropriate methods for achieving hemostasis in patients with severe diverticular bleeding.
| Hemostatic treatment | Case | Recurrent bleeding after hemostatic treatment |
|---|---|---|
| High-dose barium impaction | 15/155 (9.7%) | 3/15 (20.0%) |
| Endoscopic clipping | 12/155 (7.7%) | 3/12 (25.0%) |
| Intervention | 1/155 (0.6%) | 0/1 (0.0%) |
| Colectomy | 1/155 (0.6%) | 1/1 (100.0%) |
| Total | 25/155 (16.1%) | 5/25 (20.0%) |
Table 2b: Hemostatic treatment (multiple interventions performed in some patients); Clinical characteristics of patients with diverticular bleeding in our hospital (November 2011 to April 2014).
In patients with stable vital signs without renal failure, computed tomography (CT) angiography was performed when possible to identify the hemorrhage site. Endoscopy was then used to diagnose lower gastrointestinal bleeding. Endoscopic clipping was initially attempted in cases where the bleeding source was determined. In the present study, endoscopic clipping was performed in 12 patients with complete cessation of bleeding achieved after a single intervention in 9 (75.0%) patients (Tables 2b). High-dose barium impaction was performed for patients in whom the diverticular bleeding point could not be detected, who exhibited recurrent or persistent bleeding without becoming shocked, and who definitely did not have intestinal perforation. Fifteen patients received barium impaction and 12 patients (80.0%) had complete cessation of bleeding following a single intervention (Tables 2b). All patients receiving at least one treatment with barium impaction were discharged early.
In the present study, angiographic embolization was performed to achieve hemostasis in cases where repeated attempts at endoscopic clipping failed to resolve prolonged active bleeding.
One patient who underwent colectomy was admitted to our hospital on seven occasions between May 2011 and February 2012 for diverticular bleeding. This patient received repeated interventions with high-dose barium impaction. On each occasion, hemostasis was achieved and the patient was successfully discharged. However, the patient suffered frequent bleeding recurrences over a prolonged period of time. Surgery was performed in February 2012 in accordance with the patient’s preference (laparoscopic resection of the left hemicolon).
Overall, difficult-to-treat patients with diverticular bleeding were found to require a combination of a number of interventions to achieve adequate hemostasis.
Discussion
The elderly (age ≥ 65 years) population of Japan was estimated to be 31,860,000 as of September 15, 2013, according to data from the Statistics Bureau of Japan, accounting for 25% of the general population. This figure and the corresponding proportion of the general population represent the highest totals ever recorded (Statistics Bureau of Japan. <http://www.stat.go.jp/data/topics/topi721.htm> (Accessed Sep, 2015)). In our hospital, the number of elderly patients presenting with lower gastrointestinal bleeding is increasing annually (data not shown). Moreover, the prevalence of diverticular disease has recently increased in elderly Japanese patients, albeit not affecting all elderly patients. Previously, in Japan, a greater number of patients presented with right-sided diverticula than with left-sided diverticula. However, there has been a recent increase in the number of patients presenting with left or both-sided diverticula [8-10,20]. Our data was consistent with the reported nation-wide Japanese prevalence (Table 2a).
In the present study, we analyzed data from 626 Japanese patients who underwent lower gastrointestinal endoscopy in our hospital for lower gastrointestinal bleeding, with the exclusion of patients with positive fecal occult blood tests and those undergoing follow-up endoscopy for inflammatory bowel disease. Diverticular bleeding was the most common cause of lower gastrointestinal bleeding in the present study, accounting for approximately one quarter of lower gastrointestinal bleeding presentations in our hospital (Figure 1). Diverticular bleeding was relatively common in elderly patients (Figure 2). Furthermore, a high proportion of patients had lifestyle-associated comorbid diseases, including cardiovascular diseases, diabetes, and cerebrovascular diseases. Of particular note, over half of all patients had hypertension. Moreover, a significantly high number of patients with lower gastrointestinal bleeding were currently receiving anticoagulant or antiplatelet therapy. The results indicate diverticular bleeding is associated with increased age, presence of cardiovascular diseases, and use of anticoagulant or antiplatelet therapy (Tables 1b and 1c).
Approximately 84% patients with diverticular bleeding in our hospital demonstrated clinical improvements with conservative treatment including fasting, bed rest, and intravenous fluid administration (Table 2a). However, bleeding recurrence occurred in approximately one-third of patients and a number of patients were particularly difficult to management regardless of age (Tables 2b). In other words, although most patients with diverticular bleeding improve with conservative treatment, a small proportion develop recurrent and/or persistent bleeding that is difficult to treat, potentially resulting in serious outcomes. There were reports re-bleeding case of diverticular bleeding were able to predict by risk factors about hypertension, elderly, et al. [21,22]. However, in the present study, no significant differences were observed between severe and mild cases of diverticular bleeding. Appropriate selection of the treatment modality, including endoscopic clipping, high-dose barium impaction, angiographic hemostasis, and intestinal resection, is critical in difficult cases.
Endoscopic clipping may be attempted when a hemorrhage point has been clearly determined by endoscopy. Bleeding diverticula may also be identified by fitting a hood to the tip of the colonoscope and turning over each diverticulum one by one [13]. The effect of hemostasis had been reported to be excellent [14]. However, this method is not performed in our hospital due to restrictions associated with time and personnel. A definite hemorrhage source was identified in a small number of patients in our hospital (n=12).
High-dose barium impaction is considered a relatively less strenuous procedure that may be used to achieve hemostasis and an earlier discharge. However, it is contraindicated in cases of suspected colorectal perforation. Physicians must also be cautious regarding patients with intestinal stricture due to multiple episodes of diverticulitis and patients with colorectal cancer due to the risk of colon ileus development. Further, achieving hemostasis with the use of high-dose barium impaction is often difficult in patients with large amounts of bloody stool, which causes sudden shock. Administered barium was found to have disappeared within 1 month in the majority of patients included in this study. Therefore, residual barium was not found to be an issue associated with this surgical procedure.
Angiographic ablation has a strong hemostatic effect. However, it may be difficult to identify the responsible lesion in patients with mild bleeding. In patients with decreased renal function, acute renal failure may be precipitated by use of a contrast medium. Accordingly, angiographic ablation is indicated only in the following patients: those with a normal renal function; those in whom the hemorrhage source has been identified by colonoscopy, CT angiography, or scintigraphy; and those in whom bleeding is uncontrolled by conservative therapy, endoscopic clipping, or high-dose barium impaction [19].
Surgical treatment (colectomy) is indicated for patients in whom hemostasis cannot be achieved through the use of endoscopic clipping, high-dose barium impaction, or angiographic hemostasis or those in whom severe bleeding prevents rapid improvement in general conditions because other hemostatic interventions cannot be applied. A number of studies have evaluated the utility of laparoscopy in reducing the surgical invasiveness of colectomy [23,24]. However, colectomy remains the most invasive method for the treatment of severe cases of lower gastrointestinal bleeding. As described previously, diverticular bleeding commonly occurs in elderly patients with numerous high-risk factors, indicating that colectomy should be considered as the last treatment option.
In the present study, we decided that an appropriate management strategy was necessary for the effective treatment of all patients with diverticular bleeding due to the considerable number of difficult cases and the lack of clinical characteristics allowing distinction between severe and mild cases of diverticular bleeding. Based on the results of the present study and the above discussion, we developed a treatment approach that comprised the following steps. First, in cases of suspected diverticular bleeding, the following steps are taken until a decision is reached regarding the requirement for colonoscopy. When patients exhibit bloody stools (an occurrence not limited to patients with diverticular bleeding), vital signs are assessed to confirm shock. Identified cases of shock are immediately treated with initiation of fasting and emergency interventions. Furthermore, substantial hemorrhage is likely to lead to cases of shock. Therefore, CT angiography is attempted upon improvement in the clinical signs of shock with angiographic embolization or intestinal resection performed once bleeding has reduced. Colonoscopy is performed only in patients with stable vital signs. In cases where the bleeding source cannot be determined, emergency interventions are continued. CT angiography is performed to locate hemorrhage sites in patients without renal failure once vital signs no longer indicate a state of shock. Moreover, CT angiography also has utility in assessing the presence of diverticula, diverticulitis, abscess, perforation, malignant colitis, and large tumors. Where bleeding sources are located, fasting is initiated, the bowel is prepared, and colonoscopy is considered. However, emergency colonoscopy without bowel cleansing performed by experienced endoscopists may be considered in cases where bleeding sources are not identified because emergency colonoscopy for diverticular bleeding carries a high risk of perforation and shock.
Endoscopic clipping is initially attempted in cases where the bleeding source is identified by colonoscopy. Angiographic embolization and intestinal resection are considered in cases where bleeding remains uncontrolled despite the use of endoscopic clipping. A conservative approach, involving fasting and close observation, is followed in cases where the hemorrhage source cannot be determined but the amount of bleeding is insufficient to cause shock. In such cases, high-dose barium impaction is considered upon prolonged or recurrent episodes of lower gastrointestinal bleeding. High-dose barium impaction may be used to achieve hemostasis, allowing early discharge. High-dose barium impaction may be repeated in cases of recurrent bleeding. Furthermore, angiographic embolization or intestinal resection may be considered in cases where hemostasis is not achieved, depending on the patient background and identification of hemorrhage sources. Although these methods often result in reliable hemostasis, their use should be restricted to high-risk cases due to the large burden placed on patients.
We introduced this approach for the treatment of diverticular bleeding at the end of April 2014. All patients with diverticular bleeding have been successfully treated according to this treatment approach to date (September 2015). We plan to analyze the validity of this treatment strategy and make necessary revisions to further improving the safety and efficacy of this approach for the management of diverticular bleeding.
Acknowledgments
This study was supported by grants from Fujita Health University and the staff of the Gastroenterology Department of Fujita Heath University. We thank Ulatus and Enago for their assistance in manuscript translation and editing, respectively.
Conflict of Interest Disclosure
The authors declare no disclosure statement.
References
- Zuckerman DA, Bocchini TP, Birnbaum EH (1993) Massive hemorrhage in the lower gastrointestinal tract in adults: diagnostic imaging and intervention. Am J Roentgenol 161: 703-711.
- Ellis DJ, Reinus JF (1995) Lower intestinal hemorrhage. Crit Care Clin 11: 369-389.
- Vernava AM, Moore BA, Longo WE, Johnson FE (1997) Lower gastrointestinal bleeding. Dis Colon Rectum 40: 846-858.
- Gandhi SK, Hanson MM, Vernava AM, Kaminski DL, Longo WE (1996) Ischemic colitis. Dis Colon Rectum 39: 88-100.
- Greenwald DA, Brandt LJ (1998) Colonic ischemia. J ClinGastroenterol 27: 122-128.
- Kamm MA (1998) Diagnostic, pharmacological, surgical and behavioral developments in benign anorectal disease. Eur J SurgSuppl 582: 119-123.
- Yano T, Yamamoto H, Sunada K, Miyata T, Iwamoto M, et al. (2008) Endoscopic classification of vascular lesions of the small intestine (with videos). GastrointestEndosc 67: 169-172.
- Miura S, Kodaira S, Aoki H, Hosoda Y (1996) Bilateral type diverticular disease of the colon. Int J Colorectal Dis 11: 71-75.
- Nakaji S, Danjo K, Munakata A, Sugawara K, MacAuley D, et al. (2002) Comparison of etiology of right-sided diverticula in Japan with that of left-sided diverticula in the West. Int J Colorectal Dis 17: 365-373.
- Takano M, Yamada K, Sato K (2005) An analysis of the development of colonic diverticulosis in the Japanese. Dis Colon Rectum 48: 2111-2116.
- Rino Y, Imada T, Iwasaki H, Tanabe H, Toyoda H, et al. (1999) Hemostasis of colonic diverticular bleeding with hemoclips under endoscopic control: report of a case. Hepatogastroenterology 46: 1733-1735.
- Pilichos C, Bobotis E (2008) Role of endoscopy in the management of acute diverticular bleeding. World J Gastroenterol 14: 1981-1983.
- Shibata S, Shigeno T, Fujimori K, Kanai K, Yoshizawa K (2014) Colonic diverticular hemorrhage: the hood method for detecting responsible diverticula and endoscopic band ligation for hemostasis. Endoscopy 46: 66-69.
- Jensen DM, Machicado GA, Jutabha R, Kovacs TO (2000) Urgent colonoscopy for the diagnosis and treatment of severe diverticular hemorrhage. N Engl J Med 342: 78-82.
- Matsuhashi N, Akahane M, Nakajima A (2003) Barium impaction therapy for refractory colonic diverticular bleeding. AJR Am J Roentgenol 180: 490-492.
- Fujimoto A, Sato S, Kurakata H, Nakano S, Igarashi Y (2011) Effectiveness of high-dose barium enema filling for colonic diverticular bleeding. Colorectal Dis 13: 896-898.
- Niikura R, Nagata N, Yamano K, Shimbo T, Uemura N (2013) High-dose barium impaction therapy is useful for the initial hemostasis and for preventing the recurrence of colonic diverticular bleeding unresponsive to endoscopic clipping. Case Rep Gastrointest Med 2013: 365954.
- Goldberger LE, Bookstein JJ (1977) Transcatheter embolization for treatment of diverticular hemorrhage. Radiology 122: 613-617.
- Feld RS, Zink S, Posteraro A (2010) Empiric embolization of a diverticular bleed with CT angiographic mapping: enlarging the therapeutic window of transcatheter arterial intervention. J VascIntervRadiol 21: 593-595.
- Tursi A (2015) The role of colonoscopy in managing diverticular disease of the colon. J Gastrointestin Liver Dis 24: 85-93.
- Nishikawa H, Maruo T, Tsumura T, Sekikawa A, Kanesaka T, et al. (2013) Risk factors associated with recurrent hemorrhage after the initial improvement of colonic diverticular bleeding. ActaGastroenterolBelg 76: 20-24.
- Watanabe S, Kodera S, Shimura H (2014) Investigation of risk factors predicting recurrence of colonic diverticular hemorrhage and development of a recurrence risk score. ActaGastroenterolBelg 77: 408-412.
- Senagore AJ, Luchtefeld MA, Mackeigan JM, Mazier WP (1993) Open colectomy versus laparoscopic colectomy: are there differences? Am Surg 59: 549-553.
- Gonzalez R, Smith CD, Mattar SG, Venkatesh KR, Mason E, et al. (2004) Laparoscopic vs open resection for the treatment of diverticular disease. SurgEndosc 18: 276-280.
Citation: Nakagawa Y, Kamano T, Ohmiya N, Tahara T, Shibata T, et al. (2016) Effective Management of Severe Diverticular Bleeding in Japanese Patients. J Colitis Diverticulitis 1: 106.
Copyright: ©2016 Nakagawa Y, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Usage
- Total views: 11944
- [From(publication date): 3-2016 - Aug 19, 2025]
- Breakdown by view type
- HTML page views: 11043
- PDF downloads: 901