Research Article Open Access
Is a Small Intestinal Biopsy Always Necessary to Diagnose Celiac Disease in Children?
Serge A. Sorser1, Tammy Tran1, Karen Hagglund3, Alexander Lyons2, Hernando Lyons3* and Kamran Kalim1
1Department of Gastroenterology, Providence Hospital, Southfield, MI, USA
2Department of Pediatric Gastroenterology, St. John Hospital and Medical Center, Detroit, MI, USA
3Department of Medical Education, St. John Hospital and Medical Center, Detroit, MI, USA
- Corresponding Author:
- Serge A. Sorser
MD, 16001 W. 9 Mile Rd., Southfield, MI 48075, USA
Tel: 248-560-1770
Fax: 248-443-2439
E-mail: ssorser@gmail.com
Received Date: February 10, 2015; Accepted Date: April 05, 2016; Published Date: April 11, 2016
Citation: Sorser SA, Tran T, Hagglund K, Lyons A, Lyons H, et al. (2016) Is a Small Intestinal Biopsy Always Necessary to Diagnose Celiac Disease in Children?. J Gastrointest Dig Syst 6:412. doi:10.4172/2161-069X.1000412
Copyright: © 2016 Sorser SA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Gastrointestinal & Digestive System
Abstract
Objectives: The purpose of this study was to assess the diagnostic accuracy of the tissue transglutaminase antibody (tTG-Ab) for celiac disease (CD) in children.
Methods: A retrospective chart review of children suspected to have CD from January 2007 to December 2011 was conducted. Patients were excluded if they had an Immunoglobulin A (IgA) deficiency, an autoimmune disorder or were following a gluten-free diet at the time of presentation. Gender, age at the time of small bowel biopsy, chief complaint, family history of celiac disease, serum IgA and tTG-Ab were recorded. Sensitivity, specificity, positive predictive values (PPV) and negative predictive values (NPV) of tTG-Ab compared to biopsy result were calculated, using three different cut-off values of tTG-Ab: >100 U/mL, >200 U/mL, and >300 U/mL.
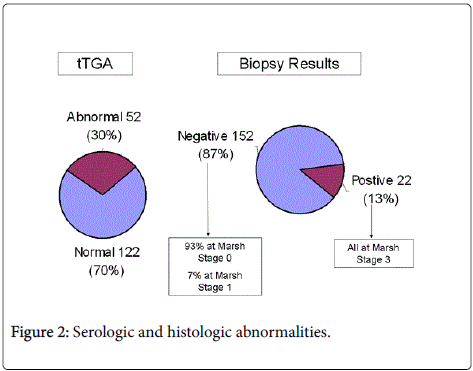
Results: 174 patients were included. 51% were male and the mean ± SD age was 9.8 ± 5.0 years. Chief complaints included abdominal pain (63.8%), diarrhea (14.9%), failure to thrive (14.4%), and vomiting (12.1%). 11.5% (20) of the patients had a family history of CD. 22 (13%) had a positive biopsy and 51(29%) had an abnormal tTG Ab level, with 13 patients >100 U/mL, 12 patients >200 U/mL, and 10 patients >300 U/mL. The specificity and PPV for the three groups were 97% and 77%, 99% and 92%, 100% and 100% respectively.
Conclusion: Low sensitivity precludes the use of tTG-Ab as a screening test, although tTG-Ab >300 U/mL has a very high specificity and PPV for celiac disease. In pediatric patients with clinical features suggestive of celiac disease, a tTG-Ab of >300 U/mL may be used to diagnose CD, avoiding duodenal biopsy.
Keywords
Celiac disease; Celiac sprue; Anti-tissue transglutaminase IgA; Malabsorption; Small intestinal biopsy; Chronic diarrhea
Introduction
Celiac Disease (CD) affects approximately 0.5 - 1% of the general population [1-5]. Damage to small intestinal villi results in malabsorption, leading to chronic diarrhea and failure to thrive. Nonspecific presenting symptoms include abdominal pain, osteoporosis, neurological symptoms and elevated transaminases [6-9]. The symptoms of CD usually resolve after gluten is removed from the diet [10,11]. The diagnosis typically rests on the appropriate clinical picture and ultimately, serologic, endoscopic and histologic findings, as well as response to a gluten free diet [12].
The gold standard for diagnosing CD is a small intestinal biopsy [13,14] which may show villous atrophy, intraepithelial lymphocytes or crypt hyperplasia. The major drawback of small intestinal biopsy is its invasive nature, requiring sedation for an endoscopic procedure and the associated risk of an endoscopy [15,16]. Furthermore, if mucosal changes are patchy, they may be missed on biopsy [17,18]. Serologic tests, on the other hand are easy to perform, relatively cheap and widely available [19]. Currently these tests are increasingly used in symptomatic patients as a tool for referral for a small intestinal biopsy. However, some drawbacks, including a high false positive rate in patients with other autoimmune conditions, are noted. Recently, the European Society for Pediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN) recommended that biopsy may be avoided if a pediatric patient has all of the following: 1) IgA anti tissue transglutaminase (tTG-Ab) >10X the upper limit of normal, 2) symptoms of celiac disease, 3) positive IgA anti-endomysial antibodies (EMA), 4) high risk human leukocyte antigens (HLA)-DQ2 or DQ8, and 5) a clinical response to a gluten-free diet [1]. This recommendation, along with other papers [20-22], suggests that conclusive serologic evaluation may negate the need for a biopsy.
In patients with symptoms suggestive of CD, the initial work up usually involves measuring IgA tTG-Ab and EMA. Currently, testing for IgA antibodies against tTG is considered to be the best initial serological test. ESPGHAN has recommended checking total IgA levels at the time of initial testing [20]. The sensitivity and specificity of the serological tests have increased in the recent years. The tTG-IgA Ab titer was shown to correlate well with severity of the biopsy results in both adult and pediatric patients [23,24]. Despite a high sensitivity and specificity, serological tests may have a low positive predictive values (PPV) and maybe difficult to interpret when one is positive and the other is negative [25].
In our retrospective review, we assessed the correlation between the tTG-IgA Ab positivity and the results of small intestinal biopsies. We also reviewed the correlation of serologic levels for tTG-IgA Ab at which the sensitivity, specificity, PPV and negative predictive value (NPV) are optimal and closely match the results of the small intestinal biopsy.
Materials and Methods
This was a retrospective chart review of all patients referred to the Pediatric Gastroenterology office at St John Hospital and Medical Center between January 2007 and December 2011 for suspected Celiac disease. The study was approved by the Institute Review Board (IRB). Patients between 2 and 18 years of age, who had both duodenal biopsy results and tTG-IgA Ab results, were included in the study. We excluded those patients on a gluten free diet, patients with IgA deficiency or patients with diabetes mellitus or other autoimmune disorders, as autoimmune disorders may give as high as a 50% false positive tTG-Ab [1]. Demographic data including age at biopsy, gender and ethnicity were collected. Clinical data including the presenting complaint, co-morbidities and family history of CD were recorded for each patient.
TTG-IgA Ab, and total IgA levels were measured. The assay used for tTG-IgA Ab testing was the human recombinant tissue transglutaminase antibody from Inova Biomechanics Research Laboratory. The cutoff for normal value of tTG-IgA was taken as less than 15 Units/mL. IgA deficiency was defined as IgA levels less than 7 mg/dL.
Duodenal biopsies were taken via esophagogastroduodenoscopy from the second portion of the duodenum and duodenal bulb. Histological diagnosis was made by the Department of Pathology at St. John Hospital. The pathologists were not informed of the results of the serological tests. Marsh classification was used to classify the histologic appearance of the duodenal mucosa [26,27]. Marsh I (increased intraepithelial lymphocytes) and Marsh II (crypt hyperplasia without villous atrophy) were not considered diagnostic of Celiac disease. Marsh III histology (crypt hyperplasia and increased intraepithelial lymphocytes with villous blunting) was considered diagnostic of Celiac disease.
Sensitivity, specificity, PPV and NPV were calculated for the tTGIgA Ab test using the duodenal biopsy as the gold standard. Frequency and descriptive statistics were calculated. Associations between categorical variables were measured using Chi-square. Differences between groups on continuous variables were measured using t-tests for independent groups. P values <0.05 were considered statistically significant. All analyses were performed using SPSS version 12.0.
Results
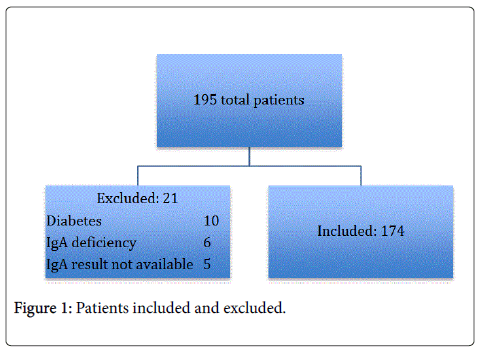
A total of 195 charts were reviewed. Twenty-one were excluded: 10 patients with diabetes mellitus, five patients with IgA deficiency and six patients in whom IgA levels were not available. A total of 174 patients were included in the final study. Mean age at the time of duodenal biopsy was 9.8 ± 5 years with 51% of the patients being male.
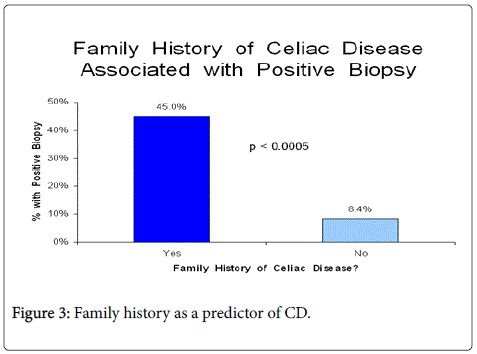
The most common presenting symptom was abdominal pain found in 64% of the patients followed by diarrhea in 15%. Of the 11.5% of patients with a family history of celiac disease, 45% had a positive biopsy, (p < 0.0005).
Overall 51 patients (29%) had an abnormal tTg-IgA Ab level of >15 Units/mL and 22 patients (13%) had biopsy proven CD. These findings are summarized in Figures 1, 2 and 3 and Table 1. The demographic data did not prove to be significant in regards to all correlations.
| Number of patients | 174 |
|---|---|
| Gender | 51% Male49% Female |
| Mean age (years) | 9.8 ± 5 |
| Biopsy diagnostic | 22 |
| Abnormal tTGA test | 51 (29%) |
| tTGAAb>100 | 13 |
| tTGAAb>200 | 12 |
| tTGAAb>300 | 10 |
Table 1: Demographic data.
Thirteen of these patients had tTG-IgA Ab level >100 units/mL, twelve patients had a level >200 Units/mL and ten patients had a level of >300 units/mL. The sensitivity, specificity, PPV and NPV at each of these cut-offs were calculated. With tTG-IgA Ab levels >100 units/mL, the specificity and NPV were high (97% and 94% respectively), but a low PPV of 77% and sensitivity of 59% were noted. At a tTG-IgA Ab level >200 units/mL, the specificity was 99%, NPV was 94%, while the sensitivity was 55% and PPV was 92%. At higher antibody levels of >300 units/mL, the PPV and specificity both increased to 100%, however this was associated with low sensitivity of 46%. These findings are summarized in Table 2.
| tTGA cut-off | Sensitivity | Specificity | PPV | NPV |
|---|---|---|---|---|
| >100 | 59% | 97% | 77% | 94% |
| >200 | 55% | 99% | 92 | 94% |
| >300 | 46% | 100% | 100% | 93% |
Table 2: Sensitivity, Specificity, PPV and NPV based on tTG cut-offs.
Discussion
Small intestinal biopsy is currently the gold standard for diagnosing CD. Endoscopy does not come without procedural risks as well as risks associated with anesthesia use. Less invasive modalities to accurately diagnose CD would not only be safer for the patients but less expensive. With improvement in sensitivity and specificity of serological tests, CD may be diagnosed without the need for small intestinal biopsy. The success of a serologic approach for diagnosis is dependent on targeting a patient population that has a high pretest probability of CD and the appropriate use of cutoff values is essential [2]. Previous studies in the adult and pediatric population have shown that tTG-IgA Ab >100 units/mL may be sufficient to diagnose CD [28-31]. However, this may compromise the positive predictive value of the test.
In our study, all of the patients with tTG-IgA antibody level >300 Units/mL had CD on small intestinal biopsy, with Marsh III classification noted histologically. Thus tTG-IgA Ab level of >300 Units/mL is strongly associated with Marsh III or higher histology on small intestinal biopsy. However, the sensitivity of the serologic tests is lacking as evidenced by the five patients who had normal tTG-IgA Ab level (<15 Units/mL) but small intestinal biopsy was diagnostic of CD (Marsh III histology). These patients were four to thirteen years of age.
Thus, while elevated serology correlated with Marsh III histology, a normal level of tTG-IgA Ab level does not rule out CD, particularly in the presence of symptoms suggestive of CD. Upper endoscopy with small intestinal biopsy should still be performed in patients with a high clinical suspicion for CD despite normal tTG-IgA Ab levels. Another important finding of our study is the low sensitivity and high specificity, PPV and NPV.
The limitations of this study are its retrospective study design and small sample size. However, it should be noted that for all of the patients who were enrolled, a complete history and physical was available and these patients have been followed in the clinic after their diagnosis was made.
Conclusion
Our study shows that a level of tTG-IgA Ab above 300 has a very high specificity for CD and that it can be used as definitive test to make the diagnosis in the pediatric population, particularly in patients with signs and symptoms suggestive of CD. The low sensitivity (46%) would preclude the use of this serologic test as a screening test for CD. Based on this retrospective review, a larger study should be conducted to further elaborate the diagnostic accuracy of tTG-IgA Ab test for diagnosing CD.
References
- Gujral N, Freeman HJ, Thomson AB (2012) Celiac disease: prevalence, diagnosis, pathogenesis and treatment.World J Gastroenterol 18: 6036-6059.
- Nelsen DA Jr (2002) Gluten-sensitive enteropathy (celiac disease): more common than you think.Am Fam Physician 66: 2259-2266.
- Hoffenberg EJ, MacKenzie T, Barriga KJ, Eisenbarth GS, Bao F, et al. (2003) A prospective study of the incidence of childhood celiac disease.J Pediatr 143: 308-314.
- Fasano A, Berti I, Gerarduzzi T, Not T, Colletti RB, et al. (2003) Prevalence of celiac disease in at-risk and not-at-risk groups in the United States: a large multicenter study.Arch Intern Med 163: 286-292.
- Rubio-Tapia A, Ludvigsson JF, Brantner TL, Murray JA, Everhart JE (2012) The prevalence of celiac disease in the United States.Am J Gastroenterol 107: 1538-1544.
- Mukherjee R, Egbuna I, Brar P, Hernandez L, McMahon DJ, et al. (2010) Celiac disease: similar presentations in the elderly and young adults.Dig Dis Sci 55: 3147-3153.
- Savilahti E, Kolho KL, Westerholm-Ormio M, Verkasalo M (2010) Clinics of coeliac disease in children in the 2000s.ActaPaediatr 99: 1026-1030.
- van der Windt DA, Jellema P, Mulder CJ, Kneepkens CM, van der Horst HE (2010) Diagnostic testing for celiac disease among patients with abdominal symptoms: a systematic review.JAMA 303: 1738-1746.
- McGowan KE, Castiglione DA, Butzner JD (2009) The changing face of childhood celiac disease in north america: impact of serological testing.Pediatrics 124: 1572-1578.
- Rubio-Tapia A, Hill ID, Kelly CP, Calderwood AH, Murray JA; American College of Gastroenterology (2013) ACG clinical guidelines: diagnosis and management of celiac disease.Am J Gastroenterol 108: 656-676.
- Nasr I, Leffler DA, Ciclitira PJ (2012) Management of celiac disease.GastrointestEndoscClin N Am 22: 695-704.
- Ludvigsson JF, Leffler DA, Bai JC, Biagi F, Fasano A, et al. (2013) The Oslo definitions for coeliac disease and related terms.Gut 62: 43-52.
- Oberhuber G (2000) Histopathology of celiac disease.Biomed Pharmacother 54: 368-372.
- Oberhuber G, Granditsch G, Vogelsang H (1999) The histopathology of coeliac disease: time for a standardized report scheme for pathologists.Eur J GastroenterolHepatol 11: 1185-1194.
- Bonamico M, Mariani P, Thanasi E, Ferri M, Nenna R, et al. (2004) Patchy villous atrophy of the duodenum in childhood celiac disease.J PediatrGastroenterolNutr 38: 204-207.
- Hart R, Classen M (1990) Complications of diagnostic gastrointestinal endoscopy.Endoscopy 22: 229-233.
- Singh R, Mei SL, Jayanna M (2013) Education and imaging: Gastrointestinal: Patchy distribution of coeliac disease diagnosed with narrow band imaging and optical magnification. J GastroenterolHepatol 28: 584.
- Ravelli A, Villanacci V, Monfredini C, Martinazzi S, Grassi V, et al. (2010) How patchy is patchy villous atrophy?: distribution pattern of histological lesions in the duodenum of children with celiac disease.Am J Gastroenterol 105: 2103-2110.
- Yagil Y, Goldenberg I, Arnon R, Ezra V, Ashkenazi I (2005) Serologic testing for celiac disease in young adults--a cost-effect analysis.Dig Dis Sci 50: 796-805.
- Husby S, Koletzo S, Korponay-Szabo IR (2012) European Society for Pediatric Gastroenterology, Hepatology and Nutrition Guidelines for the Diagnosis of Coeliac Disease. JPGN 54: 136-160.
- Mubarak A, Wolters VM, Gerritsen SA, Gmelig-Meyling FH, Ten Kate FJ, et al. (2011) A biopsy is not always necessary to diagnose celiac disease.J PediatrGastroenterolNutr 52: 554-557.
- Burgin-Wolff A, Mauro B, Faruk H (2013) Intestinal biopsy is not always required to diagnose celiac disease: a retrospective analysis of combined antibody testing. BMC Gastroenterology 13: 19-24.
- Baudon JJ, Johanet C, Absalon YB (2004) Diagnosing celiac disease: a comparison of human tissue transglutaminase antibodies with antigliadin and antiendomysium antibodies. Arch PediatrAdolesc Med 158: 584-588.
- Chand N, Mihas AA (2006) Celiac disease: current concepts in diagnosis and treatment.J ClinGastroenterol 40: 3-14.
- Dickey W, McMillan SA, Hughes DF (2001) Sensitivity of serum tissue transglutaminase antibodies for endomysial antibody positive and negative coeliac disease. Scand J Gastroenterol 36: 511-514.
- Clemente MG, Musu MP, Frau F, Lucia C, De Virgiliis S (2002) Antitissuetransglutaminase antibodies outside celiac disease.J PediatrGastroenterolNutr 34: 31-34.
- Marsh MN (1992) Gluten, major histocompatibility complex, and the small intestine. A molecular and immunobiologic approach to the spectrum of gluten sensitivity ('celiac sprue'). Gastroenterology 102: 330-354.
- Mills JR, Murray JA (2016) Contemporary celiac disease diagnosis: is a biopsy avoidable?CurrOpinGastroenterol 32: 80-85.
- Barker CC, Mitton C, Jevon G (2005) Can tissue transglutaminase antibody titers replace small-bowel biopsy to diagnose celiac disease in select pediatric populations? Pediatrics 115: 1341-1346.
- Wakim-Fleming J, Pagadala MR, Lemyre MS, Lopez R, Kumaravel A, et al. (2013) Diagnosis of celiac disease in adults based on serology test results, without small-bowel biopsy.ClinGastroenterolHepatol 11: 511-516.
- Mubarak A, Wolters VM, Gmelig-Meyling FH (2012) Tissue transglutaminase levels above 100 U/mL and celiac disease: a prospective study. World J Gastroenterol 18: 4399-4403.
Relevant Topics
- Constipation
- Digestive Enzymes
- Endoscopy
- Epigastric Pain
- Gall Bladder
- Gastric Cancer
- Gastrointestinal Bleeding
- Gastrointestinal Hormones
- Gastrointestinal Infections
- Gastrointestinal Inflammation
- Gastrointestinal Pathology
- Gastrointestinal Pharmacology
- Gastrointestinal Radiology
- Gastrointestinal Surgery
- Gastrointestinal Tuberculosis
- GIST Sarcoma
- Intestinal Blockage
- Pancreas
- Salivary Glands
- Stomach Bloating
- Stomach Cramps
- Stomach Disorders
- Stomach Ulcer
Recommended Journals
Article Tools
Article Usage
- Total views: 13183
- [From(publication date):
April-2016 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 12182
- PDF downloads : 1001