Make the best use of Scientific Research and information from our 700+ peer reviewed, Open Access Journals that operates with the help of 50,000+ Editorial Board Members and esteemed reviewers and 1000+ Scientific associations in Medical, Clinical, Pharmaceutical, Engineering, Technology and Management Fields.
Meet Inspiring Speakers and Experts at our 3000+ Global Conferenceseries Events with over 600+ Conferences, 1200+ Symposiums and 1200+ Workshops on Medical, Pharma, Engineering, Science, Technology and Business
Review Article Open Access
Imaging In Neuro-Ophthalmology
| Ravindra Kamble*, Raghavendra S, Rohit Shetty, Sanjeev Mudakavi and Diwakar N | |
| Consultant Neuroradiologist, Vikram Hospital, Bangalore, Karnataka | |
| Corresponding Author : | Ravindra Kamble Consultant Neuroradiologist Vikram Hospital, Radiology Millers Road Bangalore, Karnataka-560052, India Tel: +91 9845781307 E-mail: drravindra31@rediffmail.com |
| Received: June 03, 2015 Accepted: July 09, 2015 Published: July 13, 2015 | |
| Citation: Ravindra K, Raghavendra S, Rohit S, Diwakar N, Sanjeev M (2015) Imaging In Neuro-Ophthalmology. OMICS J Radiol 4:193. doi:10.4172/2167-7964.1000193 | |
| Copyright: © 2015 Kamble R, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Visit for more related articles at Journal of Radiology
Abstract
Neuro-ophthalmological pathologies require dedicated imaging studies for proper localisation and appropriate diagnosis. Technical advancements in MRI and CT have facilitated early diagnosis and treatment of various neuroophthalmological disorders. This review article has tried to focus on specific imaging techniques and imaging features of these neuro-ophthalmological disorders.
|
Abstract
Neuro-ophthalmological pathologies require dedicated imaging studies for proper localisation and appropriate diagnosis. Technical advancements in MRI and CT have facilitated early diagnosis and treatment of various neuro-ophthalmological disorders. This review article has tried to focus on specific imaging techniques and imaging features of these neuro-ophthalmological disorders.
Keywords:
Optic nerve; Cranial nerve; Cavernous sinus; MRI
Introduction
Ophthalmologists are the first to examine the patient and recommend specific imaging modality, need of contrast and additional imaging. They should have adequate knowledge of specific imaging sequences required to localise the lesion and should request for the same. Lack of these facts can lead to inadequate and incomplete study causing errors in diagnosis and subsequent delay in treatment. The various indications for neuro-ophthalmological imaging includes [1]
Imaging Modalities and Techniques Various imaging modalities can be used for imaging in neuro-ophthalmological diseases like X-rays, ultrasound, Doppler, CT scan, Magnetic resonance imaging and catheter angiography. Role of X-rays for orbit is very limited except for visualising foreign bodies and bony abnormalities. Ultrasound and Doppler studies are used for abnormalities of the globe.
Computed tomography (CT)
CT scan plays important role in identifying haemorrhage, calcification, fractures, bony lesions and foreign bodies. It is useful in finding orbital extension of diseases from paranasal sinuses into cavernous sinus or orbital apex. 3D multiplanar reconstructions, maximum intensity projections, CT angiography add value to CT scan. Thin cuts of 0.6 mm with coronal/Sagittal /oblique reconstruction is the protocol followed in our institution.
Magnetic resonance imaging (MRI)
MRI due to its high spatial and temporal resolution, good tissue contrast and multiplanar reconstruction capabilities is preferred choice in neuro-ophthalomology. MRI with contrast is more useful in characterising the diseases and to locate subtle findings. MR angiography is useful to find vascular abnormalities causing secondary ocular symptoms. Advanced techniques like tensor imaging (for optic nerve) and CISS 3D can be used in some pathologies. Our Protocol includes-T1W and T2W axial and Coronal-3 mm slice thickness with 1mm interslice gap, Fat suppressed T1 Coronal and Sagittal pre and post contrast and fat sat T2 coronal apart from routine sequences of brain and spine as indicated.
Catheter angiography
The most common indication is suspicion of carotid cavernous fistula which causes proptosis and chemosis. The various neuro-ophthalmological disorders can be described according to signs and symptoms as per the indications.
Diseases Causing Visual Problems
Various disorders can cause unilateral or bilateral visual problems like blurring or field defects. Afferent pupillary defects can be seen in pathologies affecting optic nerves and chiasm. Detailed anatomy of the visual pathway is needed for proper localization (Table 1).
Diseases which can cause unilateral visual problem are located in retinal region, optic nerve head and optic nerve. However bilateral visual problems are caused due to pathologies affecting bilateral optic nerves, optic chiasm, retrochiasmal tract and cortex. Pathologies Affecting Optic Nerve
Lesions affecting optic nerves can be divided as
Inflammatory lesions affecting optic nerves
Optic neuritis presents with sudden onset blurring of vision or visual loss which can be unilateral or bilateral and on examination shows relative afferent pupillary defect (RAPD). Investigation of choice is MRI with contrast. MR imaging in acute optic neuritis should include dedicated imaging sequences for orbit, brain and spinal cord along with contrast study.
On imaging, optic nerve appears Hyperintense on T2 and isointense on T1 and may be swollen. The contrast enhancement in acute optic neuritis is very sensitive (94%) and it can affect variable length and region. Longer segment and foraminal region involvement suggests poor initial vision but it does not predict recovery [2]. Inflammatory pathologies which affect optic nerve are predominantly demyelinating diseases like multiple sclerosis (MS) and neuromyelitis optica (NMO). NMO was initially considered to be spectrum of multiple sclerosis but it is well known now as a different entity. The involvement of brain and spinal cord in both these entities is different. In NMO, lesions in brain are atypical (not like MS) and may involve medulla and hypothalamus, whereas in MS involvement of brain is typical (periventricular involvement). Cord involvement in NMO is seen for more than 3 segments and central part of cord is involved. In MS, cord is seen involved for one segment and in posterolateral aspect. Both these diseases can present with unilateral optic neuritis however can be bilateral. In 10% cases of NMO, simultaneous spinal cord and optic nerve can be involved [3]. The risk of developing MS by 15 years after onset of acute neuritis is 25% in patients who do not have brain lesions in initial scan and 72% in patients who have 1 or more lesions at initial scan [4] (Figure 1). Ischemic optic neuropathy (ION)
Ischemic optic neuropathy can be divided into anterior (AION) and posterior (PION). AION can be further divided into arteritic and non arteritic [5].
Arteritic AION is seen in elderly individuals more than 60 years with systemic signs of myalgia, temporal pain and transient visual loss or diplopia and is commonly associated with giant cell arteritis [6]. Non arteritic AION is seen in individuals more than 50 years with systemic vascular disease like hypertension and diabetes [7]. Differentiation between them can be done fundoscopically by cup to disc ratio in other eye [6]. Role of MRI is primarily to rule out demyelinating diseases and mass lesions. Non arteritic AION does not show any abnormality on T1W or contrast imaging [8]. However posterior ischemic optic neuropathy may show contrast enhancement on MRI [9]. Sometimes acute ischemic optic neuropathy can show diffusion restriction on MRI like acute infarct [10]. Our experience in non arteritic AION shows atherosclerotic changes in ophthalmic segment of internal carotid artery on MR angiogram with hyperintense signal changes in optic nerve on T2W without swelling and contrast enhancement (Figure 2). Toxic / nutritional optic neuropathy
Common causes of toxic optic neuropathy includes methanol, ethanol, tobacco and drugs like ethambutol, amiodaron, Chloramphenicol, cisplatin etc [11-14]. These toxins affect pappilomacular bundles and cause central and centrocaecal scotoma. Nutritional deficiencies of vitamin B1, B6, B12 cause similar effects like toxic optic neuropathy. We found subtle loss of CSF space surrounding both optic nerves without contrast enhancement on MRI (Figure 3). Differentiating features of various types of optic neuritis is described in Table 2.
Infiltrative lesions of optic nerve
Systemic malignancies like leukemia, lymphoma and multiple myeloma can involve optic nerve as sole manifestation or as a part of systemic disease. It can infiltrate optic nerve causing swelling and enhancement on imaging [15-17]. The optic nerve sheath can be infiltrated causing compression of central retinal artery and vein leading to visual symptoms [18] (Figures 4A and 4B).
Tumors of optic nerve
It can be divided into tumors of optic nerve and optic nerve sheath. Glioma is commonest tumor that affects optic nerve however uncommon tumors include ganglioglioma, medulloepitheliomas and haemangioblastoma. The most common optic nerve sheath tumor is meningioma. Optic nerve tumors commonly present with progressive vision loss with proptosis.
Optic nerve gliomas are unilateral and frequently seen in females and commonly associated with neurofibromatosis type 1. On imaging they are seen as T2 hyperintense enlarged nerve with kinking and enhancement. The symptoms are usually due to compression of central retinal vein. Optic nerve meningiomas are usually unilateral seen commonly in middle age but can be seen in children and young adults and can be bilateral. On MR imaging, optic nerve is enlarged demonstrating uniform peripheral contrast enhancement with non-enhancing central optic nerve (tram track sign) and can be associated with pneumosinus dilatans [19,20] (Figure 4C). Compressive pathologies of optic nerve
Optic nerve can be compressed by the mass lesions anywhere along its course. The common lesions that cause optic nerve compression are meningiomas, pituitary adenomas and paranasal sinus lesions. The vision loss is usually gradual. Sometimes vision loss can be rapid in cases of aneurismal compression or in pituitary apoplexy [21] (Figure 5). Some congenital rare causes of progressive visual field defects are described below.
Optic nerve head drusen: Optic disc drusen is uncommon cause of progressive visual field defect seen as small calcified lesion at the optic nerve head on CT scan. It is bilateral in 13% with incidence of 2.4%. They occur due to progressive calcification of mucoproteins and mucopolysaccharides globules [22] (Figure 6A). Coloboma: Coloboma are developed due to failure of closure of fetal fissure posteriorly with absent choroid and retina at that region. It is seen inferiorly and on nasal side as focal bulges and may present with field defects [23] (Figure 6B). Staphyloma: Staphyloma are the bulges of sclera on posterior aspect of eyeball with longer and wider globe seen commonly in high myopics and can be the cause of field defects [24] (Figure 6C). Lesions of optic chiasm
Optic chiasm can be affected by infectious/inflammatory or neoplastic diseases of basal cisterns .Rarely can be affected by drugs. The presenting symptoms are usually bitemporal hemianopia in compressive etiology and junctional scotoma if the lesion is compressing optic nerve and chiasm junction.
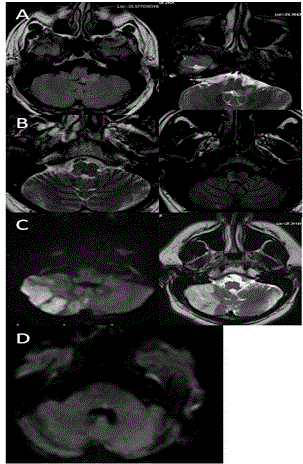
Opto-chiasmatic arachnoiditis: Opto-chiasmatic arachnoiditis can occur due to trauma, infection like tuberculosis, tumors and subarachnoid haemorrhage [25-27]. However most common cause in developing countries is tuberculosis. It is seen commonly in young females and frequently associated with paradoxical response to antitubercular treatment. Clinically it is very important to identify this entity as it causes severe visual loss and thus has to be treated promptly. On imaging there is frequent enhancing exudates surrounding the optic chiasm with extension along the post chiasmal tract (Figure 7). Toxic opto-chiasmatis: Various substances like ethambutol, isoniazid, methanol and tobacco are toxic to optic nerves and optic chiasm. Toxicity of the anti-tubercular drugs typically present after 2 to 8 months of treatment with dyschromatopsia (especially for blue and yellow colour) as earliest sign of toxicity. It causes central scotomas, but bitemporal and peripheral field defects can also be seen [28-30]. On Imaging, the optic chiasm is swollen with hyperintense signal changes (Figure 8). Compressive pathologies of optic chiasm: Optic chiasm can be compressed by large lesions in basal cistern like craniopharyngioma, pituitary adenoma, arachnoid cyst, epidermoid, aneurysms, meningioma etc. The most common clinical feature is bitemporal field defects. Sometimes it can cause junctional scotoma if compression is at the optic nerve/chiasm junction (Figure 9). Tumors of optic chiasm: The most common tumor of optic chiasm is glioma however few rare tumors like primary chiasmatic germinoma and glioblastoma are also described [31-33]. Different pattern of affection of chiasmal gliomas is described between neurofibromatosis (NF) and non-neurofibromatosis group. Gliomas in NF group, affect optic nerve more commonly (66%) as compared to optic chiasm (62%). On imaging, the chiasm is thickened with preserved contour. In non NF group, chiasmal glioma is most commonly seen with mass like cystic component, variable enhancement, distortion of the contour and extension beyond optic pathway [33] (Figure 10). Cortical lesions causing field defects
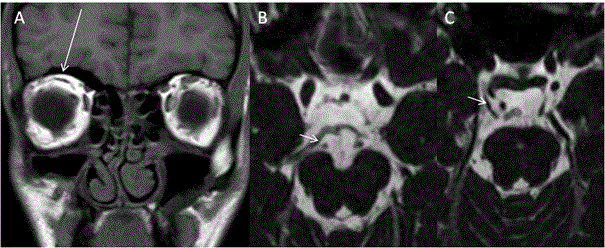
Field defects can be variable depending on the site of insult to optic radiation due in brain parenchyma. Various lesions causing field defects are hypoxic injury, arterial/venous infarcts, tumors, granulomas, traumatic contusions etc. Thus depending on site of lesions the field defects can be homonymous hemianopia or quandratanopia (Figure 11).
Bilateral visual problem due to raised intracranial pressure
Patients presenting with bilateral gradual visual loss or blurring of vision often shows features of papilloedema on fundoscopy, suggesting raised intracranial pressure. Raised intracranial pressure causes enlargement of the optic nerve sheath as optic nerve sheath communicates with the brain subarachnoid space.
Raised intracranial pressure can be idiopathic or secondary to venous thrombosis/stenosis, dural arteriovenous malformations, tumors, obstructive hydrocephalus, meningitis and trauma. MRI findings which suggest raised intracranial pressure are increased optic nerve sheath diameter, indentation on optic nerve head, vertical tortuosity and kinking of optic nerve, empty sella and widening of foramen ovale. There is direct correlation with raised intracranial pressure and increase in optic nerve sheath diameter. The cut off value of 5.82 mm of optic nerve sheath diameter is considered as best predictor of raised intracranial pressure [34] (Figure 12). Diseases causing strabismus and lid abnormalities
Squint and lid abnormalities like ptosis occur due to diseases affecting cranial nerves or muscles. Depending on the pathology, patient may present with only ptosis (as seen in isolated 3rd nerve palsy) or squint due to involvement of one or more extra ocular muscles. Various congenital and acquired pathologies can cause symptoms of ocular movement disorders.
Abnormalities of 3rd, 4th and 6th cranial nerves
Congenital fibrosis of extra-ocular muscles (CFEOM) is a non-progressive congenital restrictive movement disorder of eye, either due to abnormality in innervation of extraocular muscles by 3rd and/or 4th cranial nerves (all or part of nerve) or abnormality in the muscles innervated by these nerves. Depending on clinical features and genetics, it is classified into 3 types. Whether CFEOM is a primary muscle disorder or due to neurogenic cause is debatable. There are few case descriptions of muscle fibrosis on pathology [35,36]. There are other reports favouring neurogenic cause, as there is frequent association of CFEOM with Marcus–Gunn Jaw winking phenomenon [37]. In CFEOM, the third nerve is either normal or may be hypoplastic (bilaterally or unilaterally) on MR Imaging. The normal diameter of third nerve on CISS 3D was found to be 2.01+/-0.36 mm and abnormal nerve was 1.14+/-0.61 mm suggesting hypoplasia. 3rd nerve hypoplasia can also be seen in congenital third nerve palsy. [38]. Additionally, there are atrophic extraocular muscles with hyperintense signal changes (Figure 13).
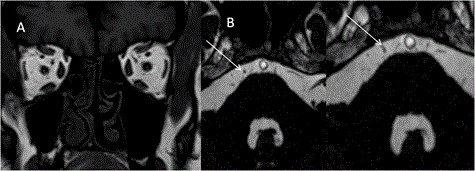
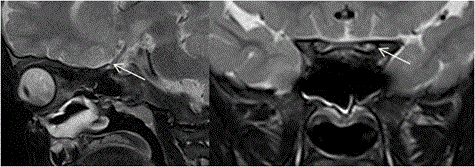
In some cases where there is CFEOM with synergistic divergence, it was found that 3rd nerve is hypoplastic bilaterally with absent 6th nerve on the side of synergistic divergence [39] (Figure 14). In Mobius syndrome, 6th and 7th nerves are hypoplastic and patients present clinically with facial paresis and abduction deficits. In Duane retraction syndrome, 6th nerve is hypoplastic or absent with innervation of lateral rectus by 3rd nerve (miswiring). In CFEOM 3rd, 6th and 4th nerves are known to be hypoplastic with hypoplasia of corresponding extraocular muscles [40]. 3rd, 4th and 6th cranial nerves can be compressed along their course by tumors, vascular lesions like aneurysms and leptomeningeal diseases (Figure 15). Tumors like schwannoma of 3rd, 4th and 6th cranial nerves are rare and on imaging appears similar to other types of schwannomas. They appears isointense to brain and frequently shows homogenous bright contrast enhancement but can be heterogenous and appears along the course of the involved nerve [41-43] (Figure 16). Tumours and Granulomatous lesions like cysticercal cyst, tuberculomas etc. can involve cranial nerve nuclei, causing ocular movement abnormality (Figure 17). Table 3 shows motility disorder with etiology and localisation of cranial nerve palsies. Abnormalities of extraocular muscles (EOM)
Brown syndrome: Brown syndrome is rare abnormality affecting superior oblique tendon and trochlea leading to limited elevation of the affected eye. It can be congenital or acquired. Acquired cases are either due to trauma or surgery, which causes scarring/avulsion of trochlea. When congenital, the superior oblique muscle belly is also atrophic as may be demonstrated by high resolution MRI [44] (Figures 17 and 18).
Isolated myositis of EOM: Isolated myositis of extraocular muscle is usually a subgroup of idiopathic orbital inflammatory syndrome. It commonly affects the recti mucles (inferior rectus is least affected). The involvement of EOM in inflammatory diseases is due to its high blood flow and high mitochondria which makes these muscles prone to increased circulating inflammatory cells. On MRI, the affected muscle belly is bulky and swollen with hyperintensity on T1 and T2 weighted images as compared to normal muscle and also shows bright contrast enhancement [45,46] (Figure 19). Cysticercal cyst affecting EOM: Isolated EOM can be infested with cysticercal cyst. There is fusiform enlargement of the muscle with T2 hyperintense cystic lesion with eccentric scolex within, well demonstrated on MRI [47] (Figure 20). Isolated metastasis to extraocular muscles: Isolated metastasis to EOM muscles is extremely rare. It can be initial presentation of metastases. The most common primary can be from breast and lungs but reports from thyroid and gastric malignancies are also reported. It mostly affects muscle belly with medial rectus being most commonly involved muscle. In 17% cases it can be bilateral [48-50] (Figure 21). Diseases causing proptosis and external ophthalmoplegia
Proptosis is usually due to enlargement of extraocular muscles, presence of tumor or space occupying lesion in orbit and vascular engorgement. Similarly these patients may present with ophthalmoplegia due to involvement of multiple muscles in the orbital apex or due to involvement of multiple nerves at the apex or cavernous sinus. Patient may present with redness of eyes, chemosis and excessive watering. These patients need dedicated studies concentrating at orbital apex and cavernous sinus.
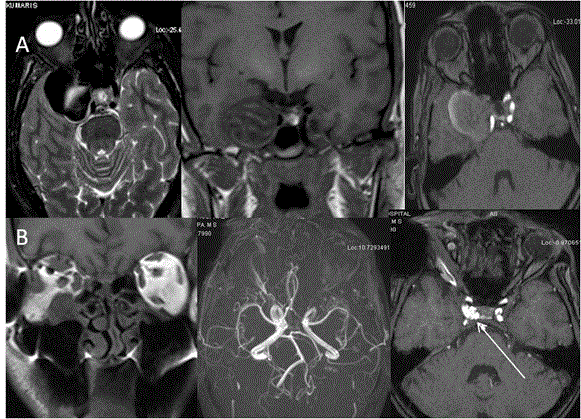
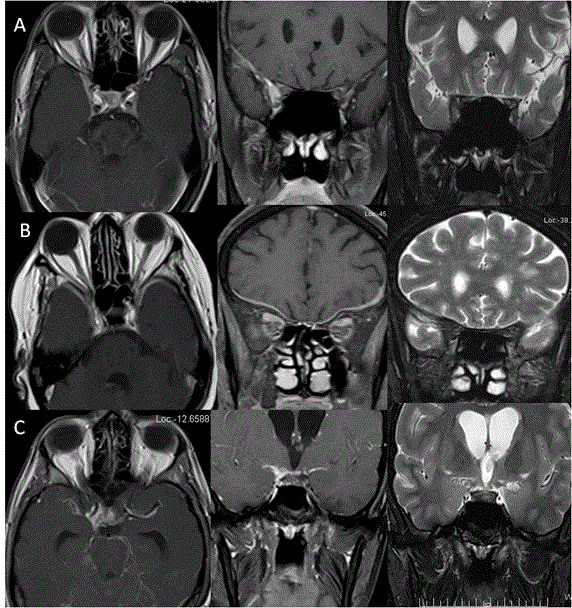
Orbital psuedotumor: It is an inflammatory non-granulomatous disease process affecting globe, orbit with extraorbital extension. It can manifest as myositis, dacryoadenitis, periscleritis, trochleitis, perineuritis and tumor-like masses. It can present either as mass in orbital apex (focal or diffuse form) or as myositis. It can extend into cavernous sinus, Meckel’s cave, middle cranial fossa, petrous apex, pterygopalatine fossa, infratemporal fossa etc. Imaging features depends on the type of affection. It appears as T2 hypointense and T1 isointense to gray matter with bright contrast enhancement. In cases of extraorbital extensions, it can show bright dural enhancement. This disease usually responds well to steroid treatment [51] (Figure 22). Grave’s ophthalmoplegia: Thyroid eye disease manifests with enlarged muscles causing ophthalmoplegia. It usually affects bilateral muscles and sometimes causes optic neuropathy due to crowding of enlarged muscles at the apex. On CT, the muscles appear bulky and enlarged with sparing of tendons and bowing of medial orbital wall [52]. On MRI the muscles appears enlarged with hyperintensity in acute phase. In chronic burnt out phase the muscles undergo fibrosis and does not show stretching on Dynamic MRI [53] (Figure 23). Tolosa hunt syndrome: Tolosa hunt syndrome is important cause of painful ophthalmoplegia showing good response to steroid therapy. The disease is seen affecting cavernous sinus and or orbital apex. On MRI, the affected cavernous sinus is enlarged and shows lateral convexity. The lesions are isointense to muscle and hypointense to fat on T1W and isointense to fat on T2W images. On contrast administration, it shows bright enhancement. On MRI, the important differential diagnosis includes pseudotumor, sarcoidosis, meningioma and lymphoma as all these lesions shows T2 hypointensity [54] (Figure 24A). Wegeners granulomatosis: It is necrotising granulomatous disease affecting paranasal sinuses, nasal cavity and orbit. Intracranial involvement shows meningeal thickening, which can be seen extending to cavernous sinus. It also shows T2 hypointensity on MR imaging. There can be remote T2 hyperintense signal changes in brainstem and cerebral white matter [55] (Figure 24 B). Tuberculous meningitis: Tuberculosis can involve cavernous sinus and basal meninges, which appear hypointense on T2 and can be nodular in contour. Bright enhancement is seen on post contrast studies. This picture is identical to sarcoidosis [56] (Figure 24C). Sarcoidosis: Sarcoidosis shows involvement of dura mater which extends to cavernous sinus. The lesions are dark on T2 with bright contrast enhancement. Other features includes intraprenchymal nodular lesions with edema, thickening of cranial nerves, periventricular lesions and hypothalamic involvement [57]. Fungal infection: Invasive fungal infections are commonly due to mucormycosis and aspergillosis, and are mainly seen in immunocompromised patients. These occur by extension from surrounding paranasal sinus infection with or without bony destruction. They can lead to cavernous sinus thrombosis with wall thickening and narrowing of the cavernous ICA. On MRI, fungal infections are hypointense on T2 and T1 due to ferromagnetic and calcium substance and can show mass-like lesions or dural enhancement [58,59] (Figure 5B). Hypertrophic pachymeningitis: It is rare disease which causes diffuse dural thickening and can involve cavernous sinus. It is one of the cause of cavernous sinus thrombosis and parenchymal edema [59,60]. Cavernous sinus thrombosis: Thrombosis of cavernous sinus is seen due to infection extending from nasal cavity, paranasal sinuses or face. On MRI, there are filling defects in cavernous sinus due to clot. Indirect signs include enlargement of cavernous sinus with dural enhancement lateral to cavernous sinus extending to tentorium, enlargement of superior ophthalmic vein and exophthalmos [59,61] ( Figure 25). Cavernous sinus aneurysm: Cavernous ICA aneurysms cause symptoms due to mass effect, inflammation and rupture into cavernous sinus leading to carotid cavernous fistula. 5% of these are giant aneurysms. On imaging, these appear as flow voids in the large aneurysm on SE sequences and mixed intensity if the aneurysm is partially thrombosed [59,62,63] (Figure 26A). Carotid cavernous fistula (CCF): CCF is abnormal communication of carotid artery and cavernous sinus. It can be direct or indirect through dural branches of ICA. Most common cause of direct CCF is trauma. On imaging the cavernous sinus is enlarged in size with multiple flow voids and enlarged superior ophthalmic vein. On MR angiography, flow related enhancement can be seen in the cavernous sinus. Associated findings like orbital fat stranding, enlarged veins and muscles with proptosis can be noted in affected eye. 3D FISP sequence shows sensitivity of 83% and specificity of 100% to accurately diagnose shunt flow [59,64] (Figure 26B). Tumors affecting cavernous sinus: Schwannomas are frequently seen affecting cavernous sinus, of which 5th nerve schwannoma is most common. Small lesions show bright homogenous contrast enhancement and large tumors are heterogenous. They follow the course of the nerve. These lesions are T1 isointense and T2 hyperintense. Plexiform variety shows fusiform enlargement of the nerve with heterogenous contrast enhancement. Cavernous sinus haemangiomas are bright on T1 and T2 with progressive intense contrast enhancement and attached to outer wall of cavernous sinus. Meningiomas arise usually from lateral wall of cavernous sinus and is hypo to isotense to gray matter on all sequences and on contrast they show intense contrast enhancement with dural tail sign and narrowing of the ICA. Other tumors that invade cavernous sinus are pituitary adenoma, metastases, nasopharyngeal carcinoma, sphenoid sinus carcinoma, chordoma, chondrosarcoma, lymphoma, leukemia and perineural spread of the nasopharyngeal tumors [59] (Figures 27 and 5C). Nystagmus
Nystagmus is oscillating rhythmic movement disorder of the eyes. It can be congenital or acquired. There are different types of acquired nystagmus which can be localised precisely on imaging of brain.(Table 4) So one must be aware of type of nystagmus for proper localisation of the lesion [65] (Figure 28).
Trauma
Trauma can cause vision loss either due to injury to globe and optic nerve, or can cause ocular movement disorder due to cranial nerve injury. Muscle entrapment in fracture fragment can also cause restricted eye movements following trauma.
Traumatic optic neuropathy: Trauma to optic nerve can be direct or indirect. Direct trauma to optic nerve can cause avulsion or transection of optic nerve. There can be optic nerve sheath haemorrhage or intra-orbital hematoma. Indirect trauma to optic nerve can be due to fracture of optic strut with fracture fragment causing compression of the optic nerve. Damage to the optic nerve can also be due to shearing/traction stresses at the optic canal as it is fixed to the dural fold [66] (Figure 29). Rarely trauma can cause isolated or combined 3rd, 4th or 6th cranial nerve palsy usually due to stretching. 3rd cranial nerve palsy is usually associated with severe head injury, 4th nerve palsy associated with intermediate head injury and 6th nerve palsy associated with minor head injury [67-68] (Figure 30). References
|
Tables and Figures at a glance
| Table 1 | Table 2 | Table 3 | Table 4 |
Figures at a glance
 |
 |
 |
 |
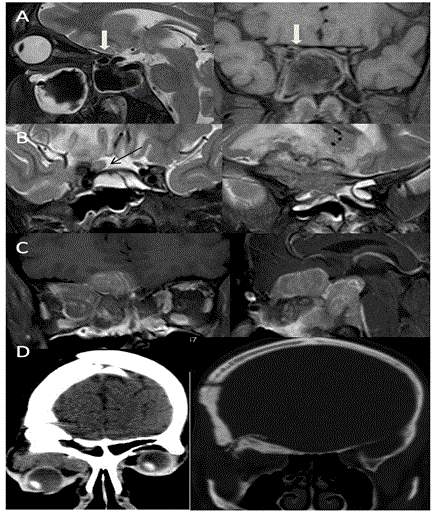 |
| Figure 1 | Figure 2 | Figure 3 | Figure 4 | Figure 5 |
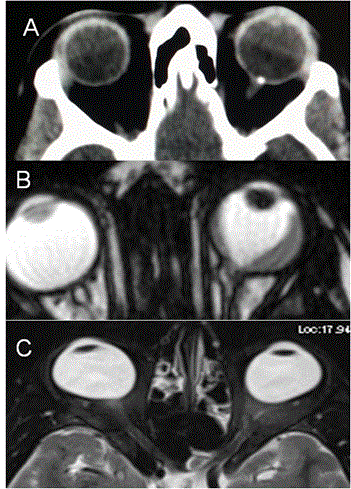 |
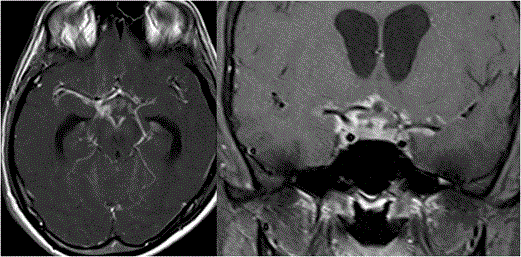 |
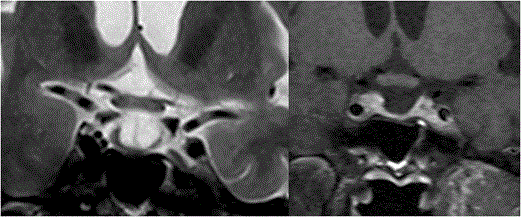 |
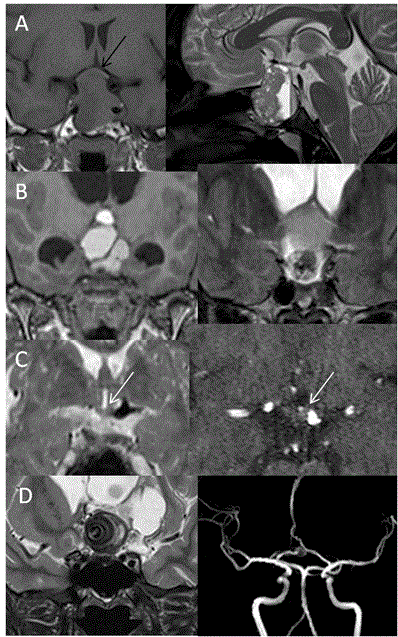 |
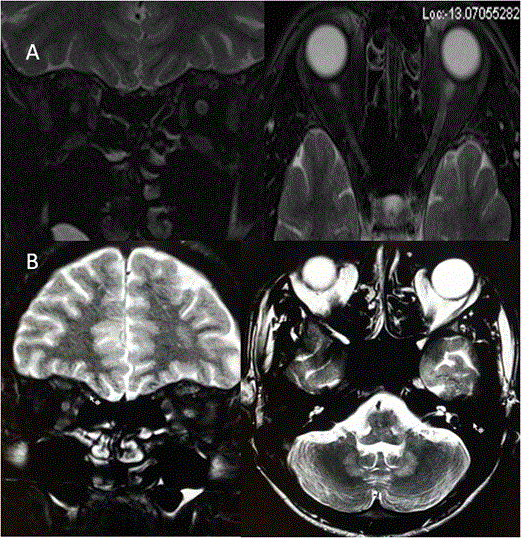 |
| Figure 6 | Figure 7 | Figure 8 | Figure 9 | Figure 10 |
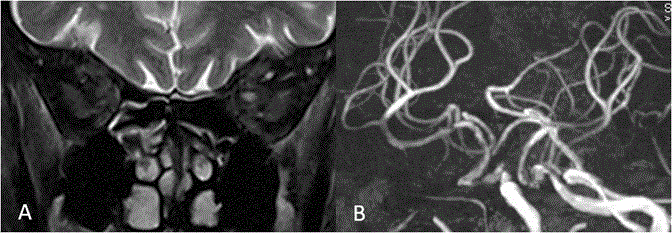 |
 |
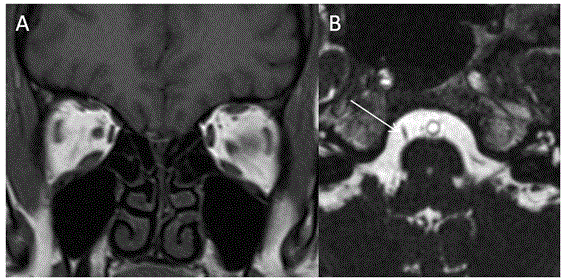 |
 |
 |
| Figure 11 | Figure 12 | Figure 13 | Figure 14 | Figure 15 |
 |
 |
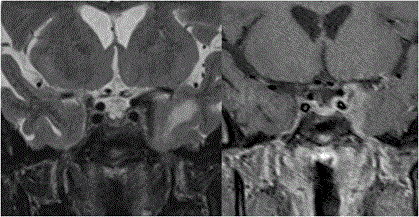 |
 |
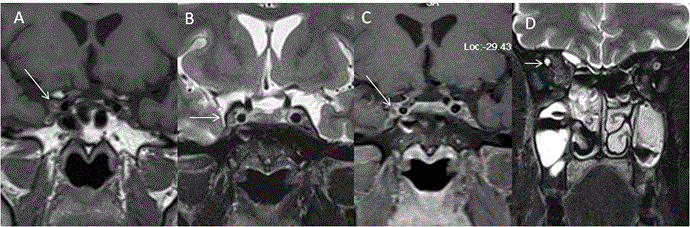 |
| Figure 16 | Figure 17 | Figure 18 | Figure 19 | Figure 20 |
 |
 |
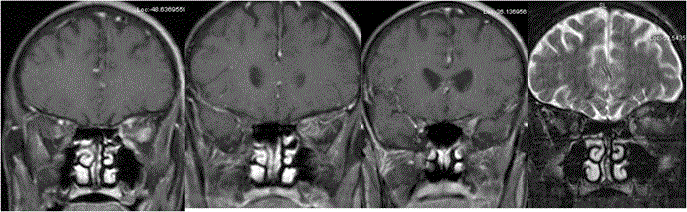 |
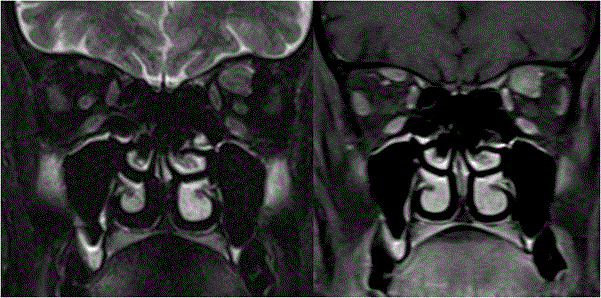 |
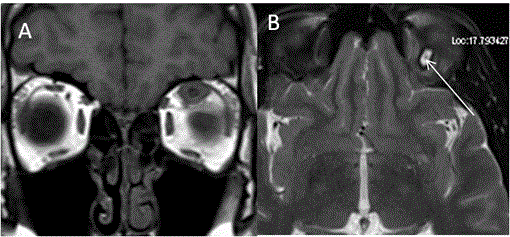 |
| Figure 21 | Figure 22 | Figure 23 | Figure 24 | Figure 25 |
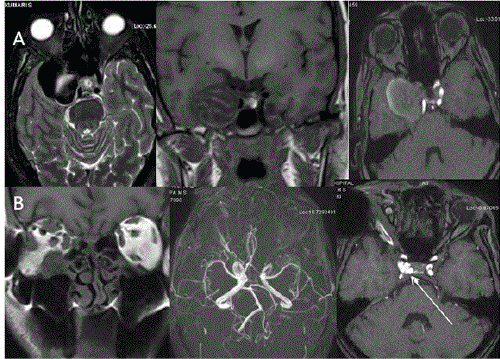 |
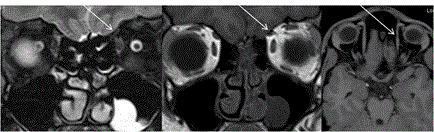 |
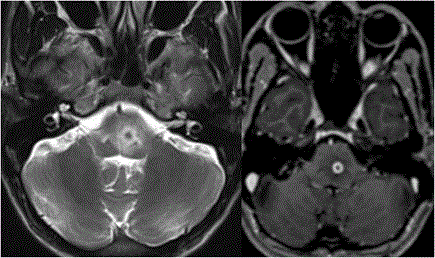 |
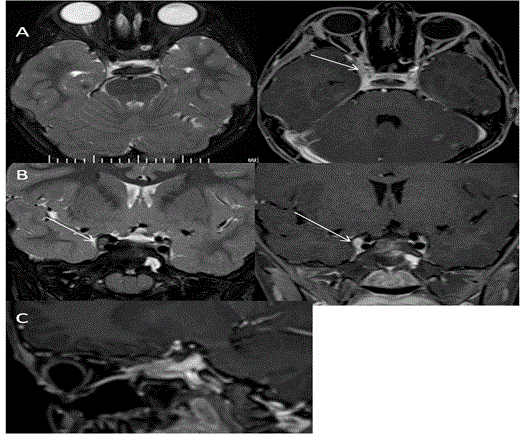 |
 |
| Figure 26 | Figure 27 | Figure 28 | Figure 29 | Figure 30 |
Post your comment
Relevant Topics
- Abdominal Radiology
- AI in Radiology
- Breast Imaging
- Cardiovascular Radiology
- Chest Radiology
- Clinical Radiology
- CT Imaging
- Diagnostic Radiology
- Emergency Radiology
- Fluoroscopy Radiology
- General Radiology
- Genitourinary Radiology
- Interventional Radiology Techniques
- Mammography
- Minimal Invasive surgery
- Musculoskeletal Radiology
- Neuroradiology
- Neuroradiology Advances
- Oral and Maxillofacial Radiology
- Radiography
- Radiology Imaging
- Surgical Radiology
- Tele Radiology
- Therapeutic Radiology
Recommended Journals
Article Tools
Article Usage
- Total views: 17150
- [From(publication date):
August-2015 - Aug 28, 2025] - Breakdown by view type
- HTML page views : 12239
- PDF downloads : 4911
