Research Article Open Access
Implantable Magnetic-Dielectric Composites for Prolonged Hyperthermia Treatment of Hepatic Lesions
Somesh Mohapatra1, KL Yadav2 and Alok Srivastava3*1Metallurgical and Materials Engineering Department, Indian Institute of Technology, Roorkee, India
2Department of Physics, Indian Institute of Technology, Roorkee, India
3Department of Physics, DAV (PG) College, Dehradun, India
- Corresponding Author:
- Alok Srivastava
Department of Physics, DAV (PG) College, Dehradun, India
Tel: +91-941205959
E-mail: alokshriv2@rediffmail.com, shriv.alok@gmail.com
Received Date: May 09, 2016; Accepted Date: May 23, 2016; Published Date: May 30, 2016
Citation: Mohapatra S, Yadav KL, Srivastava A (2016) Implantable Magnetic-Dielectric Composites for Prolonged Hyperthermia Treatment of Hepatic Lesions. J Biotechnol Biomater 6:230. doi:10.4172/2155-952X.1000230
Copyright: © 2016 Mohapatra S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Biotechnology & Biomaterials
Abstract
The inefficiency and the low success rates of surgical resection and side effects of chemotherapy used in the treatment of hepatocellular carcinoma have elicited substantial research interest in alternative methods for treatment. Here we report the use of magnetic-dielectric composite of Chromium doped Iron Oxide (Cr0.2Fe1.8O3) – Polyvinylidene Fluoride (PVDF) for the hyperthermia treatment of hepatic lesions. The magnetic oxide was prepared by sol-gel processing route and the formed phase was characterized by X-Ray Diffraction. The composite was formed in Acetic Acid media where the magnetic oxide was embedded in the PVDF matrix. Surface morphology of the formed composite was studied using Scanning Electron Microscopy and biocompatibility was ensured by MTTAssay studies. In silico studies were carried out using Finite Element Method simulation to depict the conditions at which hyperthermia treatment may occur in the hepatic tissue.
Keywords
Hepatocellular carcinoma; Hyperthermia; Magneticdielectric composites; Finite element calculations
Introduction
Hepatocellular Carcinoma is malignant tumor growth on or in the hepatic tissue, owing to in situ cellular degradation or metastases from other tumors in the body. In any case, this affects the whole of digestive tract and keeps other organs at risk pertaining to its role as blood purifier [1,2]. With hepatic resection as one of the major modes of treatment, the chances of success in the surgery and further survival without recurrence have been low. Even the use of chemotherapy and other drug based treatment modalities have not yielded satisfactory outcomes in the survival rates [3].
Alternate modes of treatment have been explored, hyperthermia using ferromagnetic materialsbeing one of them [4]. These methods have used the hysteretic heat loss as the major source of heat provision for the lesion and have worked on in vitro and in vivo models studying the effects on hepatic tissue [5].
All major studies have used the magnetic heat loss that occurs for a very short time period, while lesions require being at a reasonably high temperature for a longer period of time. This has not been a possibility due to the requirement of an alternative magnetic field near the affected region for a prolonged period, which is neither economical, nor ergonomically suitable.
This study deals with the usage of a dielectric materialin association with the magnetic oxide to act as a heat sink when the temperature goes beyond the safe limits for biological usage, and acts as a heat source when there is no power source. Using the model of liver and physical properties of the tissue, the heating procedure has been studied in silico. Cytotoxicity studies have been done on the material using MCF- 7 cancer cell line to ensure biocompatibility.
Materials and Methods
Synthesis and characterization
Ferric Nitrate Nonahydrate (Fe(NO3)3.9H2O, HiMedia) and Chromium Nitrate Nonahydrate (Cr(NO3)3.9H2O, Sigma-Aldrich) were used in stoichiometric proportion to prepare Cr0.2Fe1.8O3 (CFO) by sol-gel processing route. Further, Polyvinylidene Fluoride (PVDF, Sigma-Aldrich) was used to form the polymer matrix with embedded CFO.
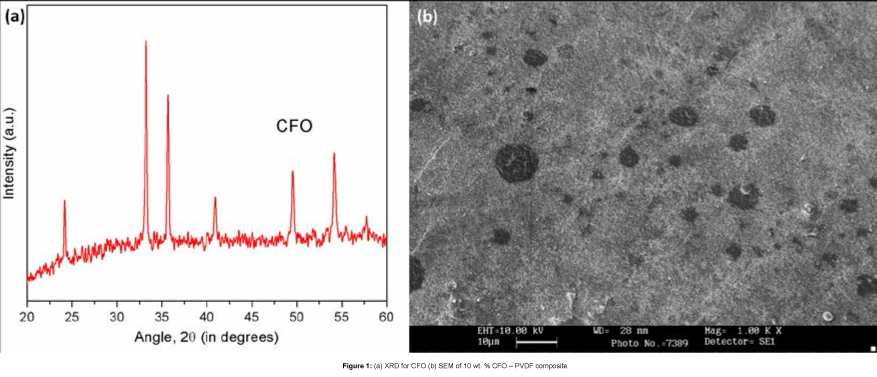
Structural confirmation of CFO was done using X-Ray Diffraction (Bruker D8 Advance) as shown in Figure 1(a), and SEM (LEO 435 VP) revealed the surface morphology of the composite as shown in Figure 1(b).
Cellular studies
In order to examine the biocompatibility of the nanocomposite, in vitrocytotoxicity assay was performed against MCF 7 cell lines which were chosen for the ease of the study on the static cancer cell line with proper depiction of the effect of the treatment modality. Cells were maintained in DMEM containing 10% FBS and 1% antibiotic in a humidified atmosphere of 5% CO2, at 37°C. The cytotoxic activity of the nanocomposite was evaluated against this cell line using 3-[4, 5- dimethythiazol-2-yl]-3, 5-diphenyltetrazolium bromide dye (MTT). For this assay, 5 x 103 cells were seeded in 200 μl of media in the wells of a 96-well microtiter plate and incubated in presence of nanocomposite for 24 h in a humidified atmosphere of 5% CO2, at 37°C. The cells in absence of nanocomposite served as control. Then 5 μl/well of MTT [10 mg/ml in phosphate buffer saline (PBS)] was added to each well and incubated for another 4 h under the same condition. After 4 h of incubation, the medium was removed and 200 μl/well of dimethyl sulfoxide (DMSO) was added to dissolve the formazan. The cell viability was determined by measuring the absorbance at 570 nm using a microplate reader (BMG Labtech, Germany). The cell viability was expressed as a percentage of the viability of the control culture. The cell viability (%) was calculated according to the following equation:
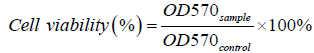 (1)
(1)
Where OD570 sample and OD570 control represent measurements from cells in the presence of nanocomposite and only cells, respectively. Results were expressed as mean ± SEM. Data were tested for normality and investigated for statistical significance using one-way analysis of variance (ANOVA), for p<0.05.
Modelling of the system
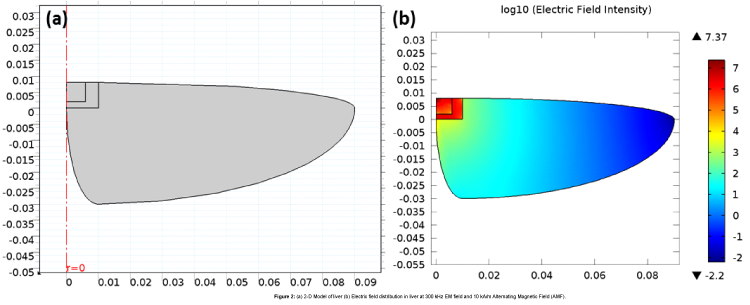
The liver tissue was modeled with appropriate physical parameters in terms of dimensions, thermal and electromagnetic properties, as shown in Table 1 [6]. A two dimensional axisymmetric model, Figure 2(a), was constructed and rendered in three dimensional structure for simulation.
| Materials | cp (J kg-1 K-1) | ρ (kg m-3) | λ (W m-1 K-1) | ε | µ | σ (S m-1) |
|---|---|---|---|---|---|---|
| Liver | 3540 | 1079 | 0.52 | 69.0 | 1 | 0.487 |
| Blood | 3617 | 1050 | 0.52 | 76.8 | 1 | 1.23 |
| PVDF | - | - | - | 8.4 | 1 | 5E-11 |
| Cr0.2Fe1.8O3 | - | - | - | 1140 | 780 | 3.952 |
Table 1: Physical properties of tissue.
The whole system was considered to be biological tissue, except the magnetic particle domain, and modeled under Fourier Heat Conduction equation with Pennes’ approximation, as in Equation 2 [7].
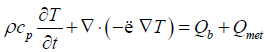 (2)
(2)
Where ρ stands for density, cp for heat capacity, T for Temperature (K), t for time, λ for the thermal conductivityand Qb is the heat due to blood perfusion modeled according to CEM 43 model [8].
Electromagnetic waves and electric field distribution in the tissue as wells as the magnetic composite was modeled using Maxwell’s equation, as in Equation 3.
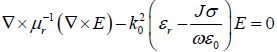 (3)
(3)
where μr is relative permeability, E is the electric field intensity, k0 is wave number of free space, εr is relative permittivity, J is current density, σ is electrical conductivity, ω is angular frequency of wave and ε0 is permittivity of free space.
Results
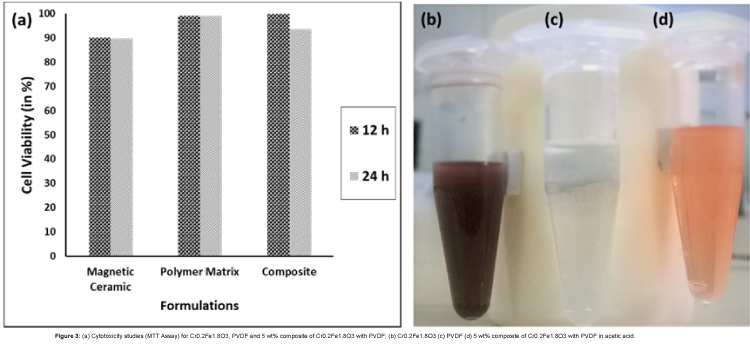
The cellular uptake studies based on the MTT Assay (Figure 3(a)) analysis show cell proliferation rather than cell apoptosis thereby agreeing to the fact that the cells are not in an environment that might be toxic owing to the presence of the material present.
The initial conditions were assumed to be normal body temperature at 300.15 K for all domains, and the electromagnetic frequency for excitation of magnetic particles was set at 300 kHz with 10 kA/m AMF, where saturation magnetization was found to be 37.3 emu/g for the Magnetic oxide.
The simulation was carried out for 1700 s, where the power was shut down at 600 s and the system was allowed to cool down on itself with the source of heat supply as the pre-heated dielectric PVDF.
Simulation results
The electric field distributionowing to the influence of the EM waves and the magnetic oxide has been shown in Figure 2(b).
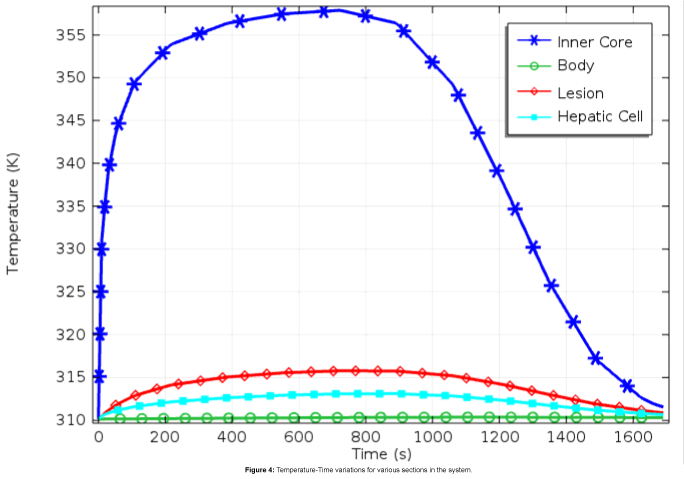
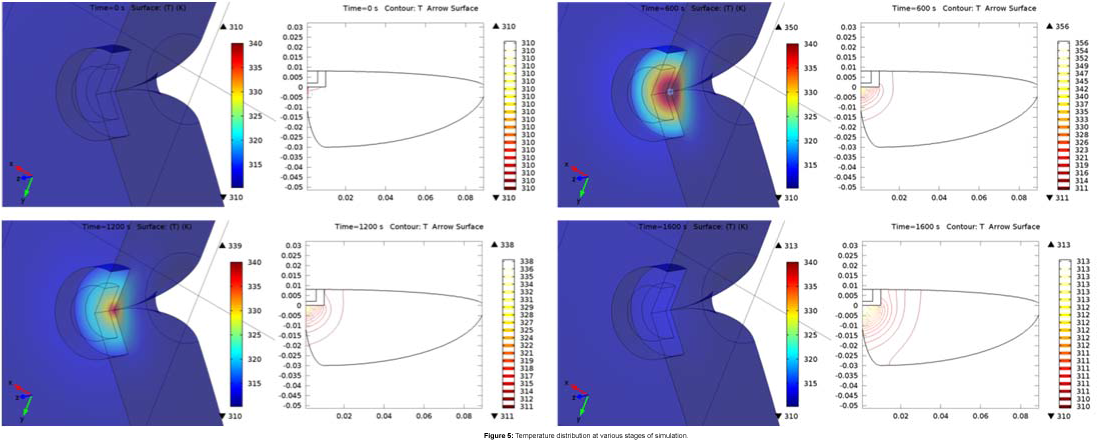
The temperature profile, as shown in Figures 4 and 5, depicts exemplary heat distribution in the lesion with heat in the hepatic tissue within safe biological limits.
The temperature remains at around 315.5 K, even after 300 s of switching off the power supply, and subsequently cools down to body temperature over another 1000 s. Thus, the presence of PVDF as the dielectric material helping in the regulation of heat in the tissue is established by the simulation results.
Discussion
The cytotoxic effect of the prepared formulations was studied by MTT assay (Figure 3(a)) on MCF-7 cell line. It is observed that even after 24 h, there was no significant cell death rather they continued to proliferate in a similar manner. This data further confirmed the biological compatibility of the formulations.
The magnetic-dielectric core reaches a maximum temperature of 355 K and the localized temperature in the lesion reaches a maximum of 316 K at around 700 s into simulation as can be seen from Figures 4 and 5. The surrounding hepatic tissue has an average temperature of 312 K, while the body temperature remains unchanged over the whole time.
Damage Tissue Analysis study over the region on which heat was applied stated the death of over 80% of the hepatic cells in the lesion, and a minimal damage of less than 5% in the region surrounding, thereby rendering the system viable for medical applications.
The prolonged application of the heat requires that the temperature be elevated and consistent for a longer period of time to be effective on the tissue [9,10]. The present methodology enables the heat to be dissipated even after there has been a withdrawal of the power source. This novelty makes it appropriate to have the heat presence persevere for a longer time thereby leading to a sustained therapy in the lesion affected regions.
Conclusion
Alternative treatment by hyperthermia was extensively discussed for the cure and management of hepatic lesions. Synthesis and characterization of highly ferromagnetic Cr0.2Fe1.8O3 embedded in a dielectric PVDF matrix was carried out. Cytotoxicity studies inclusive of MTT assay was carried out to confirm the cell viability in the case of the prepared formulations and to confirm their cytocompatability. Further, by 2-d axisymmetric modelling of the hepatic tissue the heat distribution due to the magnetic composites in the region was studied using Fourier heat conduction equation with Penne’s approximation and Maxwell’s electromagnetic equations. It was observed that the temperature remained adequate even after the removal of the power surface for sufficient amount of time to aid vasodilation and antiinflammation in the region. Thus, the use of dielectric as a temperature regulator in this case has been successful, and may be tried in different compositions and materials to achieve an optimized composite.
Acknowledgement
The authors would like to acknowledge Dr. Shabina Khanam, Chemical Engineering Department, Indian Institute of Technology, Roorkee for assisting in the usage of COMSOL. For financial support, authors would like to thank Sponsored Research and Industrial Consultancy, IIT Roorkee under grant SRICCONSL- 000-414 dt. 09.09.2015, and Council of Scientific Industrial Research (CSIR), New Delhi, India under the grant 03(1272)/13/EMR-II dated 12.04.2013. SS like to acknowledge Faculty Initiation Grant from IIT Roorkee.
References
- Cureley SA (1998) Liver Cancer. Springer Science and Business Media.
- Iwatsuki S, Dvorchik I, Madariaga JR, Marsh JW, Dodson F, et al. (1999) Hepatic resection for metastatic colorectal adenocarcinoma: a proposal of a prognostic scoring system. J Am CollSurg 189: 291-299.
- Fong Y, Fortner J, Sun RL, Brennan MF, Blumgart LH (1999) Clinical score for predicting recurrence after hepatic resection for metastatic colorectal cancer: analysis of 1001 consecutive cases. Ann Surg 230: 309-318.
- Moroz P, Jones SK, Winter J, Gray BN (2001) Targeting liver tumors with hyperthermia: ferromagnetic embolization in a rabbit liver tumor model. J SurgOncol 78: 22-29.
- Nagata Y, Hiraoka M, Nishimura Y, Masunaga S, Mitumori M, et al. (1997) Clinical results of radiofrequency hyperthermia for malignant liver tumors. Int J RadiatOncolBiolPhys 38: 359-365.
- Hasgall P, Neufeld E, Gosselin M, Klingenböck A, Kuster N (2011) ITIS Database for thermal and electromagnetic parameters of biological tissues.
- Pennes HH (1948) Analysis of tissue and arterial blood temperatures in the resting human forearm. J ApplPhysiol 1: 93-122.
- Sapareto SA, Dewey WC (1984) Thermal dose determination in cancer therapy. Int J RadiatOncolBiolPhys 10: 787-800.
- Jordan A, Scholz R, Wust P, Fähling H, Felix R (1999) Magnetic fluid hyperthermia (MFH): Cancer treatment with AC magnetic field induced excitation of biocompatible superparamagnetic nanoparticles. Journal of Magnetism and Magnetic Materials 201: 413-419.
- Chan DC, Kirpotin DB, Bunn PA (1993) Synthesis and evaluation of colloidal magnetic iron oxides for the site-specific radiofrequency-induced hyperthermia of cancer. Journal of Magnetism and Magnetic Materials 122: 374-378.
Relevant Topics
- Agricultural biotechnology
- Animal biotechnology
- Applied Biotechnology
- Biocatalysis
- Biofabrication
- Biomaterial implants
- Biomaterial-Based Drug Delivery Systems
- Bioprinting of Tissue Constructs
- Biotechnology applications
- Cardiovascular biomaterials
- CRISPR-Cas9 in Biotechnology
- Nano biotechnology
- Smart Biomaterials
- White/industrial biotechnology
Recommended Journals
Article Tools
Article Usage
- Total views: 12189
- [From(publication date):
June-2016 - Aug 28, 2025] - Breakdown by view type
- HTML page views : 11222
- PDF downloads : 967