Case Report Open Access
Interventional Radiological Techniques and Current Devices in the Management of Iliofemoral Deep Vein Thrombosis
| Ferguson D* and O’Sullivan GJ | |
| Department of Radiology, Galway University Hospitals, Newcastle Road, Galway, Republic of Ireland | |
| Corresponding Author : | Ferguson D Department of Radiology, Galway University Hospitals Newcastle Road, Galway, Republic of Ireland E-mail: David.Ferguson@mailn.hse.ie |
| Received May 04, 2013; Accepted June 26, 2013; Published July 01, 2013 | |
| Citation: Ferguson D, O’Sullivan GJ (2013) Interventional Radiological Techniques and Current Devices in the Management of Iliofemoral Deep Vein Thrombosis. OMICS J Radiology 2:133 doi: 10.4172/2167-7964.1000133 | |
| Copyright: © 2013 Ferguson D, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Visit for more related articles at Journal of Radiology
Abstract
Iliofemoral Deep Vein Thrombosis (IF-DVT) is different from other types of lower extremity DVT because it has a much higher morbidity and causes more long-term damage. Early treatment with removal of thrombus and restoration of vessel patency gives symptomatic relief and reduces these complications. In this article, we present three cases of iliofemoral DVT treated successfully with pharmaco-mechanical thrombolysis using a variety of devices currently available. We demonstrate pre and post procedural imaging findings and discuss the current interventional radiological techniques and devices available for the treatment of iliofemoral deep vein thrombosis.
| Introduction |
| The aetiology of DVT can be inferred back to Virchow’s triad of hypercoagulability, haemodynamic changes and endothelial damage. Specifically with respect to the ilio-femoral segments, this may commonly be secondary to trauma, radiation or venous catheter insertion (endothelial damage) or to stasis from external compression e.g. tumour/nodal, immobility or iliac vein compression syndrome (May-Thurner) (haemodynamic changes). |
| Morbidity from the presence of DVT within the iliofemoral venous system may result from physical obstruction by the thrombus itself or via embolisation. The degree of symptoms experienced is related to the venous segment affected, the more proximal being worse, and also to the development or non-development of a sufficient collateral venous return system [1]. Furthermore, an inflammatory response may ensue within the vessel causing valvular destruction and incompetency leading to the onset of the ‘post thrombotic syndrome’ (PTS) [2,3]. Early treatment with removal/lysis of intraluminal thrombus reduces these associated morbidities by restoration of vessel patency [4-6]. There is considerable evidence that the greater the percentage of thrombus removal, the lower the risk of development of DVT recurrence and PTS [7-9]. |
| Options available for the treatment of IF-DVT include surgical venous thrombectomy, Catheter Directed thrombolysis (CDT) in isolation or with mechanical thrombectomy, aspiration thrombectomy and Pharmaco-mechanical thrombolysis (PMT). This latter technique combines thrombolysis and mechanical thrombectomy in a single device to remove thrombus. Current research demonstrates equivalent outcomes for PMT with increased rapidity when compared with CDT. However, there is a need for a large randomised control trial to further assess; this is the aim of the ATTRACT trial currently nearing end recruitment in the USA [10]. Using a case series, we demonstrate the use of PMT in the treatment of IF-DVT. |
| Case 1 |
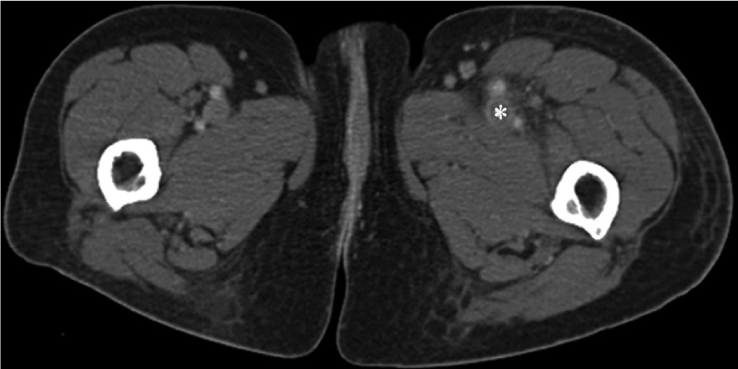
| A 69-year-old lady presented with an acutely swollen tender left lower limb with associated pitting oedema on a background of a left lower limb DVT 2 years previous. Ultrasound demonstrated absent flow with thrombus extending from the left external iliac vein into the left popliteal vein and posterior tibial vein. An ultrasound of the pelvis was also performed which was normal. The patient commenced anticoagulation therapy. The symptoms were slow to resolve and a CT venogram was performed at 2 weeks (Figure 1). This demonstrated thrombus that extended from the left common iliac vein to the distal left superficial vein with apparent patency of the left popliteal vein. The left common iliac vein was significantly narrowed in keeping with a chronic occlusion secondary to May-Thurner syndrome. There was marked thrombophlebitis and oedema of the soft tissues of the thigh. |
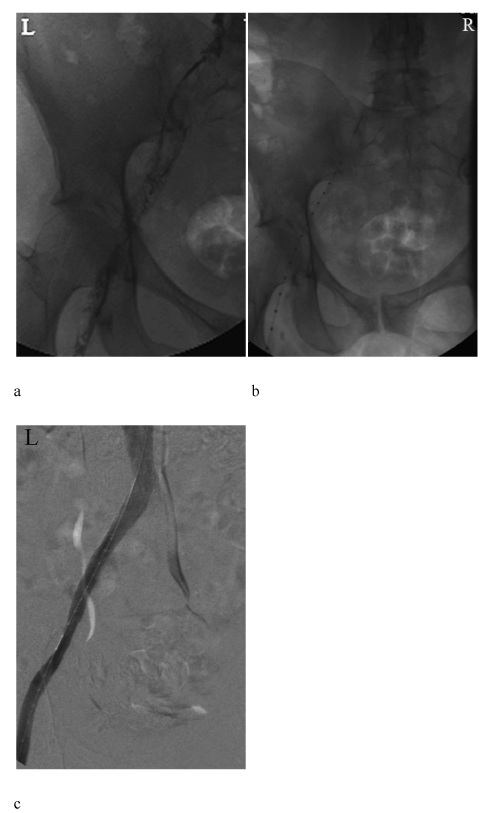
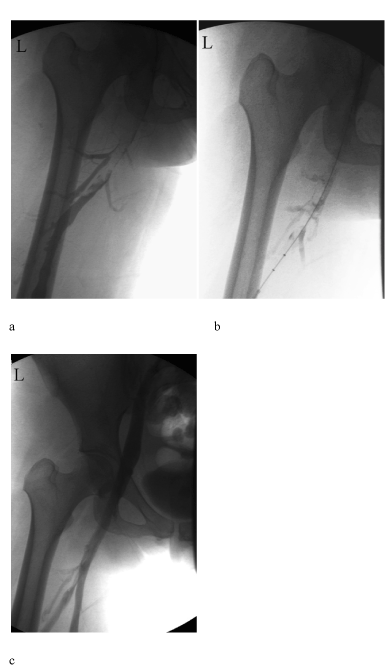
| Following informed consent, the patient was transferred to the interventional radiology suite. Under ultrasound, thrombosis of the popliteal vein was confirmed (Figure 2). The left popliteal vein was then punctured using a 4 Fr microdilator system. A 5 Fr catheter was introduced over a 0.035 hydrophilic wire and advanced into the left common iliac vein. A venogram was performed using gentle pulses (2-5cc) of contrast and confirmed occlusive thrombus extending from the left common iliac vein distally to and inclusive of the left popliteal vein (Figure 3a). |
| The EKOS Endowave Infusion Catheter System (EKOS Corporation, Bothell, WA) was placed and TPA was commenced at 1mg/hr. in combination with intravenous heparin (Figure 3b). The patient was transferred to the high dependency unit for monitoring. Following 19 hours of therapy, venography was repeated within the interventional suite. This demonstrated complete dissolution of thrombus (Society of Interventional Radiology Grade 3 [11,12]). As expected there was occlusion of the left common iliac vein with collateral flow through the left internal iliac vein and uterine plexus into the right common iliac vein and IVC. |
| The left common iliac vein occlusion was crossed using a chronic total occlusion (CTO) wire-Asahi Astato 30 (Asahi Intecc, Seto, Aichi, Japan). |
| Balloon dilatation from 3 mm up to 14 mm was followed by insertion of a 0.035 wire system, and then angioplasty was performed up to 14 mm. A 14 mm Cook Zilver stent was placed from the IVC to the left external iliac vein. This was post-dilated to 14 mm using a high pressure Zelos PTA balloon (Optimed, Karlsuhe, DE) with immediate rapid inline flow demonstrated from left popliteal vein through the left common iliac vein into the IVC (Figure 3c). |
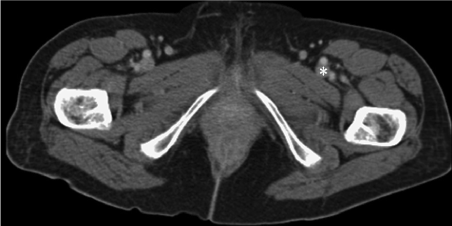
| The patient’s symptoms demonstrated immediate improvement. Ultrasound and CT venogram, performed post stenting at day 1 and at 6 weeks respectively, confirmed maintained patency without recurrent thrombus formation (Figure 4). |
| Case 2 |
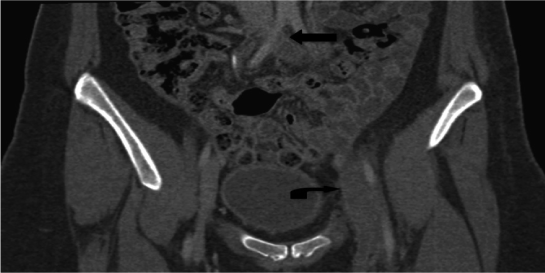
| A 30-year-old female presented with acute painful left thigh and calf swelling four weeks post partum on a background of a left below-knee DVT 4 years previously. On clinical examination, the patient was haemodynamically stable. The left thigh measured 3 cm greater than the right thigh. Ultrasound and CT venogram revealed the presence of thrombus extending from the mid-segment of the left common femoral vein up to the left common iliac vein at the level of its crossover with the right common iliac artery consistent with a May-Thurner syndrome (Figure 5). The left popliteal vein was patent. |
| Following informed consent and under ultrasound guidance, and in the prone position, access was gained into the left popliteal vein with a 6Fr vascular sheath was inserted. Ascending venogram was performed demonstrating an abrupt occlusion and thrombosis of the femoral vein extending up to the common femoral vein with presence of thrombus up to the left common iliac vein (Figure 6a). |
| PMT was then performed. 10mgs of tissue plasminogen activator was dispersed throughout the thrombus and left to percolate into the thrombus. A Possis-Angio-Jet DVX (Medrad, Warrendale PA, USA) thrombectomy was then inserted in pulse-spray mode from the left popliteal vein up to the left common iliac vein with a further 20 mgs TPA inserted (Figure 6b). Following a 25minute interval to allow thrombolysis, the device was used in thrombectomy mode with further dissolution and subsequent removal of thrombus. Post procedural venogram demonstrated a small amount of residual thrombus within the common iliac vein with left common iliac vein occlusion in keeping with iliac vein compression syndrome. Aspiration using an 8 F Terumo Destination Detachable Hub Sheath removed this remaining thrombus (Figure 6c). The common iliac vein occlusion was stented using a 16 mm Optimed Sinus Venus (Karlsuhe, DE) Zilver Vena stent (dilated to 14 mm) extending from the IVC to the left external iliac vein. Completion venography demonstrated rapid inline flow from the popliteal vein through to the IVC without residual thrombus. |
| The patient was returned to the ward with intermittent lower limb pneumatic compression for 24 hours and thigh high Class II compression stockings. Marked reduction in the diameter of the left thigh with resolution of pain was noted. |
| Doppler ultrasound and CT venography, performed 24 hours and 3 months post procedure respectively, demonstrated continued venous patency without the presence of thrombus (Figure 7). The patient remained asymptomatic at 5-month review. |
| Case 3 |
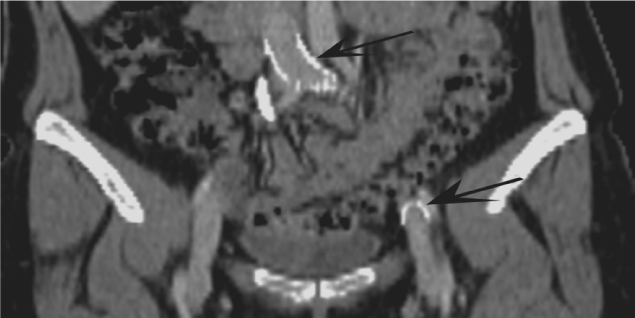
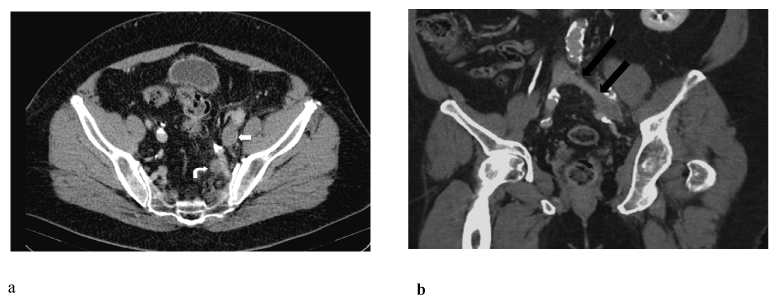
| A gentleman presented with lower back pain and swollen left thigh and calf. Ultrasound Doppler confirmed thrombus within the left common iliac and external iliac veins. CT venogram demonstrated a dilated distal left common iliac vein, external iliac vein and left internal iliac vein with enhancing rim indicative of acute thrombus (Figure 8a). The thrombus extended up the internal iliac vein into the lumbar vein which accounted for the back pain. The proximal left common iliac vein was contracted with little enhancement of the wall suggestive of a chronic thrombosed component (Figure 8b). The left popliteal vein was patent and compressible on ultrasound examination. |
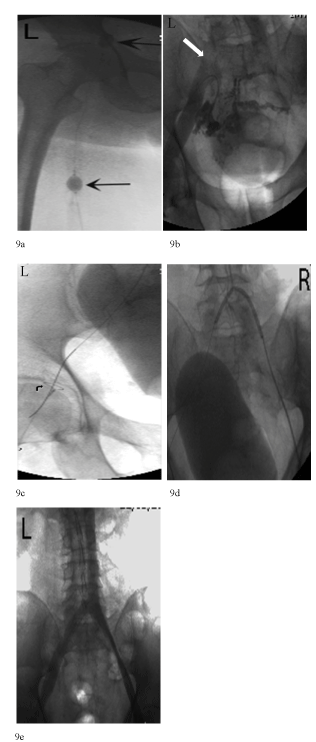
| Following informed consent, it was decided to proceed with pharmaco-mechanical thrombolysis. Under ultrasound guidance, access was gained through the left popliteal vein. A venogram confirmed the presence of thrombus as described on CT. Using a standard over-the-wire technique, a Trellis® system (Covidien, Santa Clara, CA), 80 cm shaft length, 30 cm infusion length, was inserted with the proximal end lying in the left common iliac vein, and the caudal end lying in the left common femoral vein at the level of the lesser trochanter (Figure 9a). A total of 30 mg (tPa) tissue plasminogen activator was then infused over approximately 25 minutes with the device in action. Following this, thrombus was aspirated using a 90 cm, 7 Fr Destination catheter. Venogram confirmed satisfactory thrombolysis/thrombectomy of the left common iliac, external iliac and common femoral segments. However, inline flow was reduced with stenosis and holdup within the proximal left common iliac vein, secondary to chronic thrombosis as demonstrated on CT (Figure 9b). Despite multiple attempts, retrograde access across the stenosed segment was unsuccessful from the left side. It was decided to attempt access using an antegrade approach. Access was gained through the right popliteal vein and using a chronic total occlusion (CTO) wire- Asahi Astato 30 (Asahi Intecc, Seto, Aichi, Japan), antegrade access was achieved across the left common iliac vein thrombosed segment and into the left common femoral vein. Using a snare, the guidewire was then brought through to the left popliteal vein giving ‘through and through’ access (Figures 9c and 9d). Over this, sequential balloon angioplasty of the stenosed segment was performed up to 6 mm with subsequent exchange of guidewire to a 0.035 Amplatz wire with the distal tip placed within the upper IVC. Further sequential angioplasty was performed up to 12 mm. A 14 mm diameter, 100 mm long Cook Zilver Vena stent was then inserted and dilated to 14mm. Dual sided venography confirmed satisfactory inline flow from both left and right popliteal veins into the IVC (Figure 9e). Persistent thrombus was demonstrated within the left internal iliac vein; this was not amenable to pharmaco-mechanical thrombolysis. Catheter-directed thrombolysis was performed for 24 hours via a 5 Fr infusion catheter placed within the distal left external iliac and proximal left common femoral venous segments. |
| Repeat venography at 24 hours confirmed satisfactory inline flow and dissipation of thrombus from within the left internal iliac vein also. |
| At 4.5-month review, the patient described significant and maintained symptomatic relief. CT venogram confirmed maintained patency of the left lower limb venous segments without evidence of recurrent thrombosis (Figure 10). |
| Discussion |
| With recent technological advances, radiology has evolved from offering a limited diagnosis of lower limb DVT using solely ultrasound/ conventional venography to providing a more substantial evaluation of aetiology using CT and MRI venography. Alongside this, we are now able to offer a variety of successful minimally invasive therapies. |
| Following the clinical diagnosis of DVT, in our institution, the patient primarily undergoes ultrasound Doppler imaging to assess specifically the patency of the popliteal vein, as this is our preferred access point. However, ultrasound is limited in the assessment of the proximal common iliac veins and distal IVC [13]. If there is thrombosis of the proximal ilio-femoral venous segments and in the absence of contraindications (e.g. contrast allergy, pregnancy), CT Pulmonary Angiogram (CTPA) and indirect CT venography are performed. This allows more detailed evaluation of thrombus within the distal inferior vena cava and common iliac venous segments and assessment of its chronicity as detailed (Table 1). Potential underlying thoracic/ abdominal/pelvic abnormality, that may be responsible for thrombus formation, will also be visualized on CT. The performance of CTPA also assesses for the presence of pulmonary embolus ± the presence of right ventricular dysfunction [14,15]. This allows appropriate preinterventional planning with respect to potential intra-procedural cardiopulmonary complications and specifically may aid the decision regarding the potential need for initial placement of an IVC filter [16]. |
| In conjunction with the pharmaco-mechanical thrombolysis therapy provided, the haematology services are also involved to further assess aetiology of thrombosis, where required, and also the management of pre and post procedural anticoagulation. |
| Current devices, used within our department, for the performance of Pharmaco-mechanical thrombolysis include the Trellis Peripheral Infusion System (Covidien, Santa Clara, CA), the Angiojet System (Medrad, Warrendale PA, USA) and more recently the EKOS EndoWave Infusion Catheter System (EKOS Corporation, Bothell, WA). |
| The EKOS Endowave Infusion Catheter System uses a 5.2Fr infusion catheter (106 cm in length) with incorporated microinfusion pores combined with an ultrasound core wire that promotes thrombus lysis. Segment lengths between 6 cm and 50 cm may be treated. Following its placement within the thrombus, continuous infusion of thrombolytic agent is commenced along with high frequency ultrasound waves into the thrombus segment. This results in enhanced delivery of the thrombolytic agent due to acoustic microstreaming formed from the ultrasound waves. There is also an increase of the thrombus surface area exposed to the thrombolytic agent due to the loosening of fibrin strands within the thrombus. Temperature changes associated with ultrasound energy dissipation, potential clot lysis and dissolution are monitored during the procedure. This allows automatic adjustment of ultrasound power to optimise treatment and also adjustment of saline infusion to aid heat dissipation to prevent damage to vessel walls. |
| Whilst ultrasound-enhanced thrombolysis is mostly used in the arterial system for acute ischaemic stroke, some studies have reviewed its use in the treatment of deep venous thrombosis. Motarjeme reported faster lysis times and successful achievement of venous patency of 83% in a case series of 12 patients [17]. Grommes et al. [18] also demonstrated successful complete lysis in 85% (>90% restored patency) [18]. Whilst complications of bleeding have been reported, these rates appear reduced when compared with catheter directed thrombolysis in isolation [19]. A more recent study failed to demonstrate significant overall clinical benefit in comparison with standard catheter directed thrombolysis in the treatment of IF-DVT [20]. Whilst only recently introduced in our institution, a significant benefit refers to the reduced time required within the interventional suite. Whilst overall time and monitoring is greater than the other methods to be discussed, the majority of this time occurs outside of the interventional department. This proves beneficial in increasing patient throughput within the confines of time-limited interventional sessions. Since its introduction within our department, we have noticed shorter times for infusion with equally successful results compared with CDT. In our practice it seems particularly suited to treating those patients with thrombus affecting both iliofemoral and below-knee venous segments. These patients benefit from its smaller catheter lumen size and reduced thrombolysis times compared to standard CDT. |
| The Angiojet system uses a 6 F catheter system of variable length of 50-140 cm in combination with a pump drive unit and pump set in the treatment of IF-DVT. Subsequent to placement within the appropriate segment, the thrombolytic agent is dispensed directly into the thrombus under high pressure. Following an interval period of 20-30 minutes, high velocity saline jets are commenced retrogradely to loosen the thrombus from the vessel wall. The high velocity of the saline jets creates a localised negative pressure area (as described by the Bernoulli effect) with resulting capture of the thrombus within the catheter tip. Further high-pressure jets within the catheter tip allow fragmentation and evacuation of the thrombus. |
| Currently, prospective observational studies are being undertaken with the use of the Angiojet rheolytic thrombectomy systems [21]. |
| Potential risks associated with this device include pulmonary emboli, more common in upper limb DVT therapy, systemic release of thrombolytic agent, haemolysis with resulting renal failure and fluid overload and bradyarrythmias [22,23]. |
| The Trellis Peripheral Infusion System consists of an 8 Fr multilumen catheter of 80-120 cm shaft length. It has an occluding proximal and distal balloon cuff with a dispersion wire contained between the cuffs. The dispersion wire performs mechanical oscillation stirring the thrombolytic agent through the thrombus to increase its surface area and therefore increase success of lysis. The balloon cuffs allow for the performance of isolated localised combined Pharmaco-mechanical thrombolysis (treatment zone of 15-30 cm) with minimisation of potential release of the lytic agent systemically. It is this feature that has allowed its use in those with contraindications secondary to risks of systemic haemorrhage. Furthermore, the lysed thrombus is aspirated with reduction of emboli risk. Other advantages include its use in both extremity and central venous thrombus obstruction and reduced incidence of haemolysis. Limitations refer to the 8F sheath, which may reduce its capability to treat below knee thrombi, and also to the maximum balloon expansion of <16 mm. The latter may limit its ability to acquire complete occlusion of the IVC. However, methods to overcome this have been described using a ‘pull-back’ technique [24]. |
| Within our institution, when choosing between the Trellis and Possis AngioJet devices, consideration is given to the lumen sizes of the venous segment for treatment and also to that of the access site. The narrower catheter luminal diameter of the Possis Angiojet system may prove beneficial in some cases. However, in our experience, these two devices are essentially interchangeable, particularly as aspiration is almost always performed thru an 8 F Hockey Stick Catheter or sheath that therefore requires a large bore sheath. Single sessional treatment of ilio-femoral DVT using either of these devices is successful in approximately 85% of patients. The remaining patients, where inline venous flow has not been adequately acquired, have subsequent adjunctive catheter directed thrombolysis for 24-36 hours prior to subsequent stenting. Therefore, in the majority of cases, there is a significant cost saving by avoiding intensive monitoring associated with CDT. However, the interventional component of the procedure is time-intensive (taking at least 2 hours) with resulting reduction of the patient case volume and throughput of the interventional suite. |
| Conclusion |
| Pharmaco-mechanical thrombolysis is effective in the treatment of Iliofemoral DVT. Multiple devices are available for its performance. The choice of device used may be determined by the following factors (1) patient’s clinical status including risk of systemic haemorrhage, (2) image findings including the length of thrombosed segment and patency of the popliteal vein and (3) the extent of thrombus and particularly below knee involvement (4) resource availability in terms of time, staff and cost both within the interventional radiology department and high dependency units. |
References |
|
Figures at a glance
 |
 |
 |
 |
 |
| Figure 1 | Figure 2 | Figure 3 | Figure 4 | Figure 5 |
 |
 |
 |
 |
 |
| Figure 6 | Figure 7 | Figure 8 | Figure 9 | Figure 10 |
Relevant Topics
- Abdominal Radiology
- AI in Radiology
- Breast Imaging
- Cardiovascular Radiology
- Chest Radiology
- Clinical Radiology
- CT Imaging
- Diagnostic Radiology
- Emergency Radiology
- Fluoroscopy Radiology
- General Radiology
- Genitourinary Radiology
- Interventional Radiology Techniques
- Mammography
- Minimal Invasive surgery
- Musculoskeletal Radiology
- Neuroradiology
- Neuroradiology Advances
- Oral and Maxillofacial Radiology
- Radiography
- Radiology Imaging
- Surgical Radiology
- Tele Radiology
- Therapeutic Radiology
Recommended Journals
Article Tools
Article Usage
- Total views: 14264
- [From(publication date):
September-2013 - Aug 16, 2025] - Breakdown by view type
- HTML page views : 9660
- PDF downloads : 4604
