Research Article Open Access
Kiln Condition Investigation Based on Microscopically Study, Ono Method and Bouge Calculation in Fars Cement Factory, South West Iran
Zahra Ahmadi*Faculty of Natural sciences, Department of Earth sciences, University of Tabriz, Tabriz, Iran
- *Corresponding Author:
- Zahra Ahmadi
Faculty of Natural sciences, Department of Earth sciences
University of Tabriz, Tabriz
Iran
Tel: +984133341300;
E-mail: metamorphic2016@gmail.com
Received date: March 07, 2017; Accepted date: May 18, 2017; Published date: May 22, 2017
Citation: Ahmadi Z (2017) Kiln Condition Investigation Based on Microscopically Study, Ono Method and Bouge Calculation in Fars Cement Factory, South West Iran. J Earth Sci Clim Change 8: 401. doi: 10.4172/2157-7617.1000401
Copyright: © 2017 Ahmadi Z, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Earth Science & Climatic Change
Abstract
<p>Cement is one of the most traditional construction materials, as well as, the most widely used cement in the building industry today. The rising costs of energy required for the manufacture of cement and the needed to understand the mechanism of concrete deterioration as well as the necessity of optimizing the use of Portland cement in high quality concrete, emphasizes the fact that understanding of this important building material is essential for correct industry practice. Clinker <a href="https://www.omicsonline.org/scholarly/virtual-microscopy-journals-articles-ppts-list.php" title="https://www.omicsonline.org/scholarly/virtual-microscopy-journals-articles-ppts-list.php" target="_blank">microscopy</a> is evidently one of the key factors which determine cement quality. In this research cement quality control of Fars Cement factory (SW Iran) were studied by Clinkers Polished sections and <a href="https://www.omicsonline.org/scholarly/chemical-toxicology-journals-articles-ppts-list.php" title="https://www.omicsonline.org/scholarly/chemical-toxicology-journals-articles-ppts-list.php" target="_blank">Chemical</a> analysis of clinker and Portland cements that were done by Fars cement factory’s XRF Lab. Thus, Bouge chemical indices were calculated. Also, Lime saturation Factor, silica and alumina ratios were used to compare the Fars cements and clinker by universal standards and the Ono method was used to investigate the kiln conditions. The Results show the presence of Alite, Belite, Aluminate and quartz phases in polished sections, the type of fuel was from gas fields and the increasing rate of temperature, cooling rate were slow in clinker and burning time of clinker was in long time. Also, Fars clinker and Portland cements have rather conform to clinker and cement universal standards.</p>
Keywords
Quality Control; Portland cement; Clinker; Mineralogy and Fars Cement Factory
Introduction
Cement microscopy is a very valuable technique used for examining clinker, cement, raw material, raw feed and fuel type. Every stage of the cement manufacturing process can be improved using a microscope [1]. Most cement microscopy is done using a petrographic microscope as a technique for kiln control with clinker samples being examined continuously. Usually all the specimen is a polished section of cement clinker examined using reflected light, although it may be powder mount or a thin section examined using transmitted light [1]. With only a basic assemblage of equipment, microscopically analysis can be easily performed in many cases within a few minutes. Study of cement microscopy quickly reveals several details of crystal size, morphology, abundance and distribution, leading almost intuitively to interpretations relating these data to certain features of the raw material and burning conditions [2]. For example, nests of tightly packed belite crystals form in silica rich areas of the clinker and suggest the possibility of 10 to 15 microns may indicate an undesirably rapid rate of temperature rise in the clinker as it passes through the kiln, large clusters of free lime suggest coarse limestone particles and coarse Alite may indicate a slow heating rate in kiln [2]. Portland cement clinker is a dark grey nodular material made by heating ground limestone and clay at a bout temperature of 1400 to 1500 centigrade degree [3]. The nodules are ground up to a fine powder to produce cement with a small amount of gypsum added to control the setting properties. The nodules can be anything from 1millimeter to 25 millimeter or more in diameter. Portland cement is a material made by heating mixture of limestone and clay in a kiln at about 1450 centigrade degree then grinding to a fine powder with a small addition of gypsum [3].
Portland cement clinker contains four principal minerals [4]:
Alite (approximately tricalcium silicate or C3A, typically about 65 percentage of the total) Belite (approximately dicalcium silicate or C2S, typically about 15 percentage of the total). Aluminate (very Approximately tricalcium aluminate or C3A, typically about 7 percentage of the total). Ferrite (very approximately tetra calcium Alumina ferrite or C4AF, typically about 8 percentage of the total). Alite is a solid solution series of trigonal, monoclinic and triclinic. Alite may include up to approximately 4 percent impurity [4]. Its Crystals are idiomorphous, vitreous, compact and a quant, usually six sided in cross section depending on the angle of cut. Normal crystal size range from 25 to 65 microns. Alite crystallized between 1200 to 1450 centigrade degree from the melt [1]. Its birefringence ranging from 0.002 to 0.010 as a function of ionic substitution. Crystal zonation is common in alite crystals. Smaller alite crystals form in the lime rich regions while larger crystals form in the silica rich regions of a nodule by conversion of belite [1].
Belite is a solid solution series of trigonal, orthorhombic and monoclinic. Belite usually occurs as rounded crystals frequently made up of lamellae or marked by striations indicating twinning or exsolution from solid solution both effects resulting from polymorphic transformations. Belite crystals may occur in clusters which if large indicate an unacceptable level of heterogeneity in the kiln raw feed [5]. If a belite cluster is centered on a pore, a site formerly occupied by a silica particle is indicated. In a slowly cooled clinker small belite crystals separate from the melt. Belite size is 1 to 4 microns below 1300 centigrade degree but recrystallizes to 20 to 40 microns with treatment at elevated temperaturek (approximately 1500 centigrade degree) [1]. Tricalcium Aluminate and the ferrite or interstitial phases crystallize from the melt with a degree of separation which increases with decreasing rate of cooling [1]. The crucial cooling is that occurring in the kiln itself, which is above about 1300 centigrade degree. If clinker is air quenched from the burning temperature then these individual phases may not be resolved by optical microscopy [1]. Normally tricalcium aluminate is cubic but in the presence of alkali not combined with sulfate and alkali containing solid solutions may form. The alkali is retained by the tricalcium aluminate during cooling and part or all the resulting solid solution will be orthorhombic.
This phase grows as elongated, lath-like crystals and exhibits birefringence [1]. The ferrite phase is distinguished by its color in thin section and its high reflectivity in polished section [1]. The black color of clinker is believed to result from elements such as magnesium, titanium and silicon in solid solution in the ferrite phase and oxidation as it cools in the kiln-cooler system [6]. Figures 1-3 shows the mineralogical phases in the Fars cement factory clinkers.
The aim of this study was to evaluate the clinker and cement quality control that produced in the Fars cement factory, SW Iran, by microscopically study. The main phases were recognized by this method and then Fars clinker and two different cement types compare with the universal standards.
Experimental Methods
Methods of sample preparation of the microscopically investigation of clinker and cement are essentially the classic geologic techniques of optical mineralogy and petrography. Sample preparation for clinker mineralogy studies were done by the following method:
• Vacuum impregnates the clinker fragments or whole clinkers with epoxy resin.
• Cure in an oven or slide warmer at approximately 42 centigrade degree.
• Cut with the small table top saw or grind the base of the encapsulation to expose the particles.
• Grind the expose surface with the grinder or polisher using NO. 320 adhesives-backed silicon- carbide papers for approximately two to three minutes.
• Grind on NO. 600, adhesive – backed, silicon – carbide paper for approximately two to three minutes.
• Gently coarse polish the mount on Texmet TM (a low - nap chemo textile polishing cloth) with 6 micron’s diameters for approximately four minutes.
• Find polished on TexmetTM or equivalent with 0.3 micron’s alumina for roughly four minutes.
• Clean the polished section with a forceful isopropyl alcohol spray and dry with forced warm air.
In this research, to study the mineralogy of cement clinkers that produced in the Fars cement factory (SW Iran), five polished sections of this cement factory clinkers with fine grading were prepared based on the above method and were investigated by reflected polarizing microscope type Olympus BH2-UMA in the economic geology laboratory, the University of Tabriz. Also for geochemical studies of clinker and different cement type’s production in this cement factory, chemical analysis by XRF method were done to determine the major oxides, in the Fars cement factory chemistry laboratory and results were drawn in the CaO-Al2O3-SiO2 phase diagram [7]. To calculate the clinker, cement type 2 and cement Portland pozzolanic phase composition based on the values of chemical analysis, Bogue calculation method was used [8] and Fars cement factory clinkers, cement type 2 and cement Portland pozzolanic were compared with clinker and cement universal standards. Tables 1-5 show the Comparison of Fars clinker and Portland cements with universal standards also Chemical Analysis result of Cement type 2 and Cement Portland pozzolanic that were be done by XRF method in the Chemistry Laboratory of Fars cement factory show in Tables 6 and 7.
| Burning Condition | Poor | Average | Good | Excellent |
|---|---|---|---|---|
| Heating Rate | Quick | Slow | - | - |
| AS (Micron) | 15-20 | 40-60 | 30-40 | 20-30 |
| Maximum Temperature | Low | - | - | High |
| AB (Micron) | 0.005-0.002 | 0.006-0.005 | 0.007-0.006 | 0.010-0.008 |
| Burning Time | Short | - | - | Long |
| BS (Micron) | 5-10 | 15-20 | 20-25 | 25-40 |
| Cooling Rate | Slow | - | - | Quick |
| BC | Amber | Yellow | Faint Yellow | Clear |
Table 1: Ono’s method of classification [1].
| Oxide | Cement 1-325 | Cement Portland pozzolanic | Cement type2 | Clinker | Cement 1-425 |
|---|---|---|---|---|---|
| SiO2 | 21.27 | 21.90 | 21.26 | 24.75 | 21.36 |
| Al2O3 | 5.07 | 5.11 | 4.96 | 4.87 | 4.99 |
| Fe2O3 | 5.69 | 4.11 | 5.63 | 3.94 | 5.29 |
| CaO | 60.75 | 64.20 | 60.77 | 58.32 | 60.85 |
| MgO | 2.64 | 2.95 | 2.61 | 2.77 | 2.80 |
| Na2O | 0.34 | - | 0.12 | 0.06 | 0.36 |
| K2O | 0.85 | - | 0.24 | 0.17 | 0.91 |
| SO3 | 2.06 | - | 2.10 | 2.08 | 1.94 |
| LOI | 0.68 | 0.63 | 0.62 | 1.67 | 0.88 |
Table 2: Major oxides in Fars clinker and different cement types (Percent).
| LSF | SR | AR | C3S | C2S | C3A | C4AF | composition |
|---|---|---|---|---|---|---|---|
| 91.6 | 2.3 | 1.2 | 54.6 | 21.5 | 6.5 | 12.4 | Fars clinkers |
| 104 - 90 | 1.6 – 1.4 | 3.7 – 1.4 | 85 - 52 | 27 – 0.2 | 16 - 7 | 16 - 4 | Clinker universal standard |
Table 3: Compare Fars cement factory clinker versus universal clinker standard [13].
| LSF | SR | AR | C3S | C2S | C3A | C4AF | composition |
|---|---|---|---|---|---|---|---|
| 75.1 | 2.8 | 1.2 | 10.9 | 62.7 | 6.2 | 11.9 | Fars Portland pozzolanic cement |
| 90-98 | 2-4 | 1-4 | 45-65 | 7-30 | 2-8 | 7-10 | Cement universal standard |
Table 4: Compare Fars Portland pozzolanic cement versus universal cement standard [14].
| LSF | SR | AR | C3S | C2S | C3A | C4AF | Composition |
|---|---|---|---|---|---|---|---|
| 88.00 | 2.0 | 0.8 | 44.3 | 27.4 | 3.6 | 17.1 | Cement type 2 |
| 90-98 | 2-4 | 1-4 | 45-65 | 7-30 | 2-8 | 7-10 | Cement universal standard |
Table 5: Compare Fars cement type 2 versus universal cement standard [14].
| Days | SiO0 | Al2O3 | Fe2O3 | CaO | MgO | SO3 | Na2O | K2O | L.O.I | Total |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 21.23 | 4.98 | 6.60 | 60.51 | 2.62 | 2.15 | 0.00 | 0.00 | 0.55 | 98.64 |
| 2 | 21.28 | 5.08 | 5.58 | 60.74 | 2.48 | 2.16 | 0.33 | 0.56 | 0.78 | 98.99 |
| 3 | 21.43 | 4.64 | 5.12 | 60.94 | 2.33 | 2.03 | 0.00 | 0.00 | 0.73 | 97.22 |
| 4 | 21.41 | 4.31 | 5.74 | 60.57 | 2.43 | 2.23 | 0.00 | 0.00 | 0.78 | 97.47 |
| 5 | 21.30 | 4.58 | 5.83 | 60.58 | 2.48 | 2.14 | 0.33 | 0.58 | 0.76 | 98.58 |
| 6 | 21.38 | 5.10 | 5.68 | 60.74 | 2.38 | 2.11 | 0.00 | 0.00 | 0.57 | 97.96 |
| 8 | 21.37 | 4.98 | 5.82 | 6.61 | 2.38 | 2.11 | 0.00 | 0.00 | 0.57 | 97.84 |
| 9 | 21.28 | 5.08 | 5.74 | 61.07 | 2.48 | 1.87 | 0.33 | 0.55 | 0.59 | 98.99 |
| 10 | 21.19 | 5.08 | 5.65 | 60.34 | 2.62 | 2.12 | 0.00 | 0.00 | 0.58 | 97.58 |
| 11 | 21.09 | 5.12 | 5.70 | 61.27 | 2.52 | 1.98 | 0.00 | 0.00 | 0.45 | 98.06 |
| 12 | 21.18 | 5.01 | 5.63 | 60.97 | 2.66 | 2.09 | 0.33 | 0.57 | 0.48 | 98.92 |
| 13 | 21.30 | 5.07 | 5.60 | 60.60 | 2.68 | 1.98 | 0.34 | 0.67 | 0.63 | 98.87 |
| 15 | 21.31 | 5.03 | 5.57 | 60.54 | 2.62 | 2.14 | 0.00 | 0.00 | 0.78 | 97.99 |
| 16 | 21.40 | 4.95 | 5.62 | 60.74 | 2.65 | 2.04 | 0.00 | 0.00 | 0.87 | 98.27 |
| 17 | 21.38 | 4.64 | 5.58 | 60.94 | 2.76 | 1.98 | 0.34 | 0.56 | 0.67 | 98.85 |
| 18 | 21.20 | 4.91 | 5.67 | 60.67 | 2.95 | 2.12 | 0.00 | 0.00 | 0.73 | 98.25 |
| 19 | 21.12 | 5.03 | 5.53 | 60.81 | 2.67 | 2.29 | 0.00 | 0.00 | 0.67 | 98.12 |
| 20 | 21.16 | 4.95 | 5.50 | 60.73 | 2.79 | 2.32 | 0.35 | 0.58 | 0.78 | 99.16 |
| 22 | 21.18 | 5.05 | 5.56 | 60.67 | 2.95 | 2.09 | 0.00 | 0.00 | 0.62 | 98.12 |
| 23 | 21.08 | 5.16 | 5.53 | 60.94 | 2.81 | 2.16 | 0.00 | 0.00 | 0.49 | 98.17 |
| 24 | 21.26 | 5.21 | 5.59 | 61.07 | 2.67 | 2.06 | 0.34 | 0.87 | 0.49 | 99.56 |
| 25 | 21.36 | 5.18 | 5.57 | 60.51 | 2.67 | 2.12 | 0.00 | 0.00 | 0.55 | 97.96 |
| 26 | 21.21 | 5.11 | 5.61 | 61.14 | 2.57 | 2.01 | 0.00 | 0.00 | 0.49 | 98.14 |
| 27 | 21.23 | 5.01 | 5.12 | 60.82 | 2.68 | 2.15 | 0.34 | 0.87 | 0.43 | 98.65 |
| Ave | 21.26 | 4.96 | 5.63 | 60.77 | 2.61 | 2.10 | 0.12 | 0.24 | 0.62 | 98.34 |
| Max. | 21.43 | 5.21 | 6.60 | 61.27 | 2.95 | 2.32 | 0.35 | 0.87 | 0.87 | 99.56 |
| Min | 21.02 | 4.31 | 5.12 | 60.34 | 2.33 | 1.87 | 0.00 | 0.00 | 0.43 | 97.22 |
Table 6: Chemical analysis result of cement type 2 by XRF method in the Chemistry Laboratory of Fars cement factory (Percent).
| Days | SiO2 | Al2O3 | Fe2O3 | CaO | MgO | SO3 | Na2O | K2O | L.O.I | Total |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 24.88 | 4.88 | 3.95 | 58.87 | 2.62 | 2.08 | 0.00 | 0.00 | 1.38 | 98.66 |
| 2 | 24.52 | 5.02 | 4.06 | 59.24 | 2.76 | 2.02 | 0.00 | 0.00 | 1.47 | 99.09 |
| 3 | 24.36 | 4.93 | 4.12 | 58.88 | 2.67 | 2.02 | 0.00 | 0.00 | 1.54 | 98.52 |
| 4 | 24.20 | 4.95 | 3.97 | 59.01 | 2.67 | 2.07 | 0.32 | 0.83 | 1.61 | 99.63 |
| 5 | 24.42 | 5.08 | 4.13 | 58.79 | 2.68 | 1.90 | 0.00 | 0.00 | 1.57 | 98.57 |
| 6 | 24.30 | 5.15 | 4.23 | 58.87 | 2.76 | 1.89 | 0.00 | 0.00 | 1.54 | 98.74 |
| 8 | 24.42 | 4.96 | 4.24 | 58.67 | 2.81 | 1.82 | 0.33 | 0.84 | 1.57 | 99.66 |
| 9 | 24.92 | 4.92 | 3.98 | 58.28 | 2.76 | 2.15 | 0.00 | 0.00 | 1.70 | 98.71 |
| 10 | 24.86 | 4.82 | 3.66 | 57.89 | 2.68 | 2.07 | 0.00 | 0.00 | 1.84 | 97.82 |
| 11 | 25.05 | 4.87 | 3.79 | 57.68 | 2.81 | 2.28 | 0.33 | 0.84 | 1.98 | 99.63 |
| 12 | 25.23 | 4.78 | 3.77 | 57.49 | 2.66 | 2.16 | 0.00 | 0.00 | 1.88 | 97.97 |
| 13 | 25.24 | 4.75 | 3.99 | 57.14 | 2.81 | 2.23 | 0.00 | 0.00 | 1.95 | 98.41 |
| 15 | 25.31 | 4.67 | 3.99 | 57.48 | 2.74 | 2.18 | 0.00 | 0.00 | 1.76 | 98.13 |
| 16 | 25.20 | 4.78 | 4.04 | 57.67 | 2.90 | 2.22 | 0.00 | 0.00 | 1.50 | 98.31 |
| 17 | 25.06 | 4.79 | 3.95 | 57.65 | 2.85 | 2.00 | 0.00 | 0.00 | 1.57 | 97.87 |
| 18 | 24.82 | 4.57 | 3.79 | 58.21 | 2.95 | 2.31 | 0.32 | 0.82 | 1.63 | 99.42 |
| 19 | 24.55 | 4.89 | 4.07 | 58.61 | 2.90 | 1.95 | 0.00 | 0.00 | 1.54 | 98.51 |
| 20 | 24.70 | 4.82 | 3.94 | 58.55 | 2.84 | 2.25 | 0.00 | 0.00 | 1.69 | 98.79 |
| 21 | 24.46 | 5.03 | 3.63 | 58.57 | 2.85 | 2.03 | 0.00 | 0.00 | 1.73 | 98.30 |
| 22 | 24.58 | 4.98 | 3.66 | 58.52 | 2.88 | 1.94 | 0.00 | 0.00 | 1.64 | 98.20 |
| 23 | 24.58 | 4.92 | 3.68 | 58.47 | 2.81 | 2.19 | 0.00 | 0.00 | 1.73 | 98.38 |
| 24 | 24.70 | 4.83 | 3.94 | 58.44 | 2.65 | 2.18 | 0.00 | 0.00 | 1.69 | 98.43 |
| 25 | 24.82 | 4.79 | 4.01 | 58.21 | 2.81 | 2.21 | 0.33 | 0.83 | 1.85 | 99.86 |
| 26 | 24.58 | 4.72 | 4.14 | 58.56 | 2.66 | 1.98 | 0.00 | 0.00 | 1.80 | 98.44 |
| Ave. | 24.75 | 4.87 | 3.94 | 58.32 | 2.77 | 2.08 | 0.06 | 0.17 | 1.67 | 98.66 |
| Max | 25.54 | 5.15 | 4.24 | 59.24 | 2.95 | 2.31 | 0.33 | 0.84 | 1.98 | 99.86 |
| Min | 24.2 | 4.57 | 3.63 | 57.14 | 2.62 | 1.82 | 0.00 | 0.00 | 1.38 | 97.82 |
Table 7: Chemical analysis result of cement Portland pozzolanic by XRF method. Chemistry laboratory of Fars cement Factory (Percent).
Study area
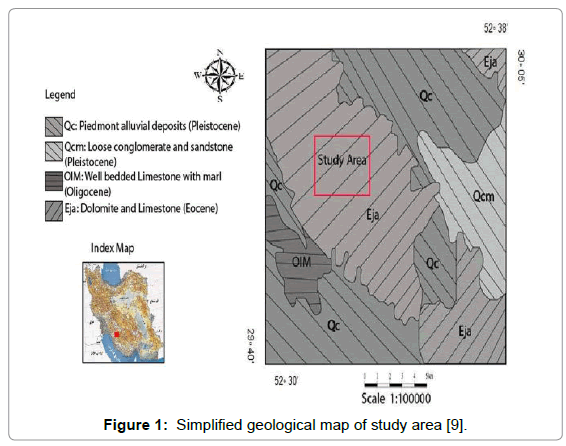
Fars cement factory is located among longitude 29◦40′ 35₺ to 30◦05′ 48₺ E and latitude 52◦ 30′ 10₺ to 52◦38′ 26₺ N in south west Iran. This cement factory is located at the beginning of Shiraz-Boshehr road and was established in 1328. It has two cement production lines with daily production capacity of 2500 tons cement per day. Lithological units in the study area including thick beds of white to cream dolomitic limestone and inter bedded white marly limestone and marl, dark grey ferrogenous dolomitic limestone in upper portions (Jahrom Formation) also well bedded to thick bedded cream, brown and grey limestone and marly limestone inter bedded with marl (Asmari Formation). Figure 1 shows the simplified geological map of study area in the Shiraz Toposheet [9].
The Ono Method
Yoshio Ono’s major contribution to cement production technology has been largely using transmitted, polarized-light microscopy as one of the methods of quality control. Natio and Ono [10] reported the relationship between three size fractions of raw feed (greater than 30 microns, between 20 to 30 microns and between 10 to 20 microns) and their burnabilities, concluding that coarse quartz increased the difficulty of burning and a few percent alkalies and magnesia as mineralizes greatly improved sintering. Microscopically it was shown that with the same raw mix burned for 20 minutes, type 2 belite with a single set of parallel lamellae characterized a clinker burned at 1380 centigrade degree, and type one belite with several sets of multidirectional lamellae formed at a temperature of 1430 centigrade degree. This method of cement kiln evaluation is based on observation of clinker phases. The principal kiln conditions and microscopically parameters evaluated by Ono’s technique including Rate of heating (according to alite size, AS), Burning time at elevated temperature (according to belite size, BS). Maximum temperature (according to alite birefringence, AB). Rate of cooling (according to belite color, BC). Table 1 shows the summary of Ono’s method.
Bouge Calculation
The Bouge calculation is used to calculate the approximate proportions of the four main minerals in Portland cement clinker, the standard bogue calculation refers to cement clinker rather than cement but it can be adjusted for use with cement [1]. Although the result is only approximate, the calculation is an extremely useful and widely used calculation in the cement industry. In practice, the bogue calculation can determine four main minerals and clinker compositional parameters such as below equations which the oxides represent the weight percentages of the oxides in the clinker [8].
Chemical parameters based on the oxide composition are very useful in describing clinker characteristics. The fallowing parameters are widely used which chemical formula represents weight percentages; Lime saturation factor (LSF) is a ratio of CaO to the other three main oxides. Applied to clinker it is calculated as equation [1]. Often this is referred to as a percentage and therefore multiplied by 100. The LSF controls the ratio of alite to belite in the clinker [11]. A clinker with a higher LSF has a higher proportion of alite to belite than will a clinker with a low LSF [11]. Typical LSF values in modern clinkers are 0.92 to 0.98 or 92 to 98 percent. Values above 1 or 100 percent indicate that free lime is likely to be present in the clinker. This is because, in principle, at LSF 1 or 100 percent all the free lime should have combined with belite to form alite. If the LSF is higher than 1 or 100 percent the surplus free lime has nothing with which to combine and will remain as free lime [11]. In practice, the mixing of raw materials is never perfect and there are always regions within the clinker where the LSF is locally a little above or a little below, the target for the clinker. This means that there is almost always some residual free lime, even where the LSF is considerably below 1. It also means that to covert virtually all the belite to alite, an LSF slightly above 1 is needed. The silica ratio or silica modulus is defined as equation [2]. A high silica ratio means that more calcium silicates are present in the clinker and less aluminate and ferrite. Aluminate ratio or AR is defined as equation [3]. This determines the potential relative proportions of aluminate and ferrite. SR is typically between 2 and 3. Aluminate ratio or AR is defined as equation [3]. This determines the potential relative proportions of aluminate and ferrite phases in the clinker [11]. An increase in clinker AR means there will proportionally more Aluminate and less ferrite in the clinker. In ordinary Portland cement clinker the AR is usually between 1 and 4. Figure 2 shows the clinker and Portland cements position in the phase diagram also Table 2 shows the major oxides in Fars clinker and different cement types.
Clinker Grandibility
The grandability of clinker depends on its chemistry and on the conditions, it experiences in burning [12]. Hard burning and high melt Contents resulting from a low silica ratio increase initial grandability since they result in a clinker with low porosity. However, after most of the aggregates of crystals have been Broken, the fracture properties of the individual phase dominate the rate of increase in fineness. Hardness is less important than brittleness, since Alite cracks more readily than Belite in a micro hardness measurement, it is considered that clinker with a high Lime saturation factor are ground more readily than those with a low lime saturation factor. The specific power consumption for grinding clinker increases when the amount of fine material increases. The production of fine grained or dusty clinker is avoided by ensuring that strong nodules of clinker are formed by agglomeration in the kiln. The requirement is met when 90-95 per cent of the void space is filled with liquid melt, which means that Between 15 and 25 per cent by mass of the charge must be melted [12]. Two properties of the melt are decisive in determining the Rate of agglomeration of the clinker and the strength of the resulting nodules, namely its surface tension and its viscosity. It has been observed that increasing content of SO3 in the feed Results in a dustier clinker, and this has been ascribed in melt surface tension associated with increased K2SO4 levels. The Grandability of clinker is related to the packing density of the individual clinker mineral grains suspended in the clinker melt. Due to their elongated habit, Alite crystals pack less densely than the belite crystals, resulting in a clinker which is easier to grind. The grandability of a clinker sample sintered to a density of 3000 kg/m3 is determined by its fracture energy and the size of the micro cracks present [12]. The number and size of the latter can be related to the cooling regime experienced by the clinker. Generally high content of Alite and low content of Belite and interstitial phases is easier to grind than the clinker rich in belite and interstitial phases. Alite crystal size, micro cracks within alite crystals and belite cluster also affect Grandability [13,14].
Discussion
Study of polished section of Portland cement clinker quickly reveals several details of crystallize, morphology, abundance and distribution, leading almost intuitively to interpretations relating these data to certain features of the raw material and burning conditions. Based on microscopically examination of clinker polished section, four main phases including Alite, Belite, Quartz and Aluminate were recognized in Fars cement factory clinkers. Belite phase was the most abundant phase in this cement factory fine grained clinker.
Based on chemical analysis results, CaO is the major oxide in clinker. Chemical composition of clinker and four cement types in CaO-Al2O-SiO2 phase diagram were in the range of C3S (Alite) and C2S (Belite) and Belite phase frequency in clinker polished sections confirm the geochemical results.
Based on Bouge calculations the amount of lime saturation factor in Fars cement factory clinkers is less than 1 (about 0.91) that represents the belite phase rich in clinkers and this calculation confirm the microscopically examinations in clinker polished sections.
According to Ono method, increasing rate of temperature, based on Alite size that was about 30-40 microns in Fars cement factory clinkers and cooling rate, based on belite color that was yellow in this clinker were slow in clinker and burning time of clinker, based on Belite size that was about 25 to 35 microns in this cement factory clinkers was performance in long time and type of fuel was from gas fields.
Conclusion
Based on Bogue calculations also clinker and cement universal standards, Fars cement factory clinkers were partly in accordance with clinker universal standard but cements Portland pozzolanic and cement type 2 produced in Fars cement factory showed more belite phase and less Alite phase than cement universal standards because of less Lime saturation factor in this cement composition.
Grandibility of Portland cement clinker is affected by the chemical composition and mineralogical properties. In the Fars cement factory clinkers, due to presence of high Belite and aluminate phases, lower Alite content and small alite crystal size show the worse Grandability.
Acknowledgements
The Author is grateful to Mr. Yaser Kamgar- mining Chief of Fars cement factory- and Mrs. Fatemeh Ahmadi Master Student in the BU-Ali Sina university- Hamadan and Dr Asghar Asghari Moghadam, Head of Earth science faculty, the University of Tabriz, for suggesting that a study be done on kiln conditions and cement quality control in the Fars cement factory.
References
- Bye GC (1999) Portland cement, (2ndedn), ASCE Press 54-60.
- Donald HC(1999) Microscopical examination and interpretation of Portland cement and clinker, (2ndedn), publishing by Portland cement Association, USA.
- Locher FW (2006) Cements principles of production and use, part3, 2: 20-70.
- Ghosh SN (2002) Advance in cement technology; chemistry, manufacture and testing, (2ndedn), published by Tech Books International, New Delhi, India.
- Cement association of Japan (2006) Portland cement clinker and their relationship obtained through microscope investigation. Reviews 1: 28-32.
- Campbell DH, Weiss SJ (1987) Some effects of kiln atmosphere and shape on clinker characteristics, Proceedings of the 9th International Conference on Cement Microscopy, International Cement Microscopy Association, Reno, Nevada. 2: 168-204.
- Taylor FW (1997) Cementchemistry, (2ndedn), Thomas Telford, London.
- Bouge RH (1974) Calculation of phase composition in the chemistry of Portland cement, Reinhold publishing 2: 184-203.
- Asadian A (2001) Geological Survey of Iran, Shiraz Center, 1: 100000 Geological Maps.
- Natio R, Ono Y (1953) Studies on the influence of the quartz sand in raw mixes upon cement burning, Journal of Research, Onoda Cement co. 5: 104-126.
- Bye GC (1983) Portland cement: Composition, production and properties(1st edn), Pergamon press Oxford.
- Maki I, Tanioka T, Ohno Y, Fukude K (1993) Clinker grandability and textures of alite and belite, cement concrete research 32: 1078-1094.
- Vanoss HG,Padorani AC (2003) Cement and the environment, Part1, Journal of Industrial Ecology 6: 89.
- Van OSS HG (2005) Background facts and issues concerning cement and cement data. Open file report, Department of Interior U.S. Geological Survey, USA.
Relevant Topics
- Atmosphere
- Atmospheric Chemistry
- Atmospheric inversions
- Biosphere
- Chemical Oceanography
- Climate Modeling
- Crystallography
- Disaster Science
- Earth Science
- Ecology
- Environmental Degradation
- Gemology
- Geochemistry
- Geochronology
- Geomicrobiology
- Geomorphology
- Geosciences
- Geostatistics
- Glaciology
- Microplastic Pollution
- Mineralogy
- Soil Erosion and Land Degradation
Recommended Journals
Article Tools
Article Usage
- Total views: 5668
- [From(publication date):
May-2017 - Aug 17, 2025] - Breakdown by view type
- HTML page views : 4613
- PDF downloads : 1055