Liposomes for Drug Delivery
Received: 25-Sep-2017 / Accepted Date: 30-Oct-2017 / Published Date: 06-Nov-2017 DOI: 10.4172/2155-952X.1000276
Abstract
Liposomes are a novel drug delivery system (NDDS), they are vesicular structures consisting of bilalyers which form spontaneously when phospholipids are dispersed in water. They are microscopic vesicles in which an aqueous volume is entirely enclosed by a membrane composed of lipid bilayers. NDDS aims to deliver the drug at a rate directed by the needs of the body during the period of treatment and direct the place of action. Liposomes are colloidal spheres of cholesterol non-toxic surfactants, sphingolipids, glycolipids, long chain fatty acids and even membrane proteins and drug molecules or it is also called vesicular system. It differs in size, composition and charge and drug carrier loaded with variety of molecules such as small drug molecules, proteins, nucleotides or plasmids etc. Few drugs are formulated as liposomes to improve their therapeutic index. Hence a number of vesicular drug delivery systems such as liposomes, niosomes, transfersomes and pharmacosomes are developed. The focus of this chapter is to the various method of preparation, characterization of liposomes, advantages and applications, etc.
Keywords: Liposomes; Characterization; Drug delivery; Medicine; Stability
Introduction
In 1965, some researchers published the first description of swollen phospholipid systems. Within a few years, a variety of enclosed phospholipid bilayer structures consisting of single bilayers, initially ‘bangosomes’ and then ‘liposomes’, were described. The early pioneers such as Gregoriadis and Perrie have established the concept that liposomes can entrap drugs and can be used as drug delivery systems [1]. In vivo activity of liposome-entrapped drugs in animal models were first demonstrated by using the anti-cancer drug cytosine arabinoside to demonstrate significant increases in the survival times of mice bearing L1210 leukemia [2]. From then it became a popular ‘model system’ for testing the effects of a wide variety of liposome characteristics on therapeutic outcomes.
Designing of liposomes is done to achieve the following optimized properties
1. Drug loading and control of drug release rate
2. Overcoming the rapid clearance of liposomes
3. Intracellular delivery of drugs
4. Receptor-mediated endocytosis of ligand-targeted liposomes
5. Triggered release
6. Delivery of nucleic acids and DNA
Mechanism of liposome formation
Liposomes are formed by phospholipids (amphiphilic molecules having a hydrophilic head and hydrophobic tail). The hydrophilic part is mainly phosphoric acid bound to a water soluble molecule whereas the hydrophobic part consists of two fatty acid chains with 10-24 carbon atoms and 0-6 double bonds in each chain. They form lamellar sheets when dispersed in aqueous medium by aligning themselves in such a way that the polar head group faces outwards the aqueous region while fatty acid groups face each other forming a spherical, vesicle like structures called as liposomes [3]. The polar fraction remains in contact with the aqueous region along with the shielding of the non-polar part. When phospholipids are hydrated in water, along with the input of energy like sonication, shaking, heating, homogenization, etc. It is the hydrophilic/ hydrophobic interactions between lipid-lipid, lipid-water molecules that lead to the formation of bilayered vesicles in order to achieve a thermodynamic equilibrium in the aqueous phase. Phospholipids are the main components of the cell membrane hence they possess excellent biocompatibility with amphiphilic properties. The amphiphilicity provide it the property of self assembly, emulsifying and wetting characteristics. When phospholipids introduced into aqueous milieu, they self assemble and it generates different structures with specific properties at different conditions. For example, phospholipids have a natural tendency to form liposomes, which can be employed as drug targeting molecules [4]. They also have good emulsifying property to stabilize the emulsions. In addition of wetting property, this can be used in coating of drug to provide hydrophilicity to hydrophobic drugs [5]. These three properties are employed in various drug designing. Variation in aliphatic chains and alcohols lead to the occurrence of varieties of phospholipids. Additionally different sources of phospholipids enhance varieties of phospholipids.
The reason for bilayer formation includes:
• The unfavorable interactions created between hydrophilic and hydrophobic phase which can be minimized by folding into closed concentric vesicles.
• The large free energy difference existing between the hydrophilic and hydrophobic environment is reduced by the formation of large vesicle formation. Since spherical structures have minimum surface tension and maximum stability. Hence there is maximum stability of self assembled structure by forming vesicles.
At neutral pH, the fatty acid carboxyl ions provide better electrostatic interaction with repulsion which makes the liposomes stable at lamellar phase. At acidic pH, fatty acid carboxyl groups are protonated leading the formation of HII phase. This in turn leads to unstable liposomes and easy to aggregate, fuse and of releasing the contents [4]. Hence pH sensitive liposomes are prepared for different usage in drug delivery.
Liposomes have a characteristic TC (transition temperature), at which they transit from gel phase to liquid crystalline phase. The encapsulated rugs are release at crystalline phase [4]. Liposomes formation is possible only if the temperature is above transition temperature. Liposome of pure lipid’s transition temperature is 41.4°C, whereas lipids from natural origin such as lecithin exhibit broad transition temperature [6] The upper limit of temperature for liposome formation is the Krafft point of lecithin, at 58°C and therefore temperature range between 41.4°C and 58°C is ideal for liposome formation eg thermosensitive liposomes [7]. While size of vesicles of the liposomes is dependent on the concentration of the phospholipid.
There is an important role of degree of fatty acid saturation; it affects the permeability of lipid bilayers to specific molecules (selective permeability). Saturated fats have higher chemical energy than the unsaturated fats because C-H bonds have more energy than C=C bonds. These double bonds create spaces for other molecules to pass through its tightly packed tails. Therefore lipid bilayers containing many unsaturated fatty acids have more gaps and more permeability than bilayers with fewer unsaturated fatty acids [8]. Generally unsaturated fatty acids occur in natural phospholipids. The length and degree of unsaturation of the lipid chain affects the gel liquid crystalline phase (transition temperature). Phospholipids with shorter and compact tails form micelles while phospholipids with longer tails tend to form liposomes.
Liposomes which are targetted to specific tissues are developed by stabilization of phosphotidylethanolamine into bilayer with antibody derivatives of fatty acids like palmitic acid. After binding to the target’s cell surface, immunoglobulin molecules concentration at contact point leads to the destabilization of bilayers. Finally, liposomal content is released at this site. Specific sub-cellular targeting is still in novice and is in in vitro research phase. For example, mitochondrial targeting is achieved by using polymer (Rh123)-PEG-DOPE (Rhodamine 123-Polyethylene glycol-1,2-dioleoyl-sn-glycero-3-phosphoethanolamine) which contains mitochondriotropic dye rhodamine. Liposomal surface modifications with rhodamine-123-conjugated polymer results in enhanced mitochondrial targeting [9].
Classification Of Liposomes
Liposomes can be classified on the basis of size and number of bilayers. They are classified as multilamellar vesicles (MLV), large unilamellar vesicles (LUV) and small unilamellar vesicles (SUV). Based on composition, they are classified as conventional liposomes (CL), pH-sensitive liposomes, cationic liposomes, long circulating liposomes (LCL) and immuno-liposomes. Based on the method of preparation, they are classified [10] as reverse phase evaporation vesicles (REV), French press vesicles (FPV) and ether injection vesicles (EIV) as given in Table 1.
| S. No. | Classification based on structural features | Classification based on method of liposome preparation | Classification based on targeting concepts of liposomes |
|---|---|---|---|
| 1 | Multilamellar large vesicles | Single or oligolamellar vesicle made by reverse phase evaporation method. | PEGylated liposomes |
| 2 | Oligolamellar vesicles | Multilamellar vesicles made by reverse phase evaporation method. | Immunoliposomes |
| 3 | Unilamellar vesicles | Stable plurilamellar vesicles. | Cationic liposomes |
| 4 | Small unilamellar vesicles | Frozen and thawed MLV | Thermosensitive liposomes |
| 5 | Medium sized unilamellar vesicles | Vesicles prepared by extrusion method. | |
| 6 | Large unilamellar vesicles | Vesicles prepared by fusion | |
| 7 | Giant unilamellar vesicles | Vesicles prepared by French press | |
| 8 | Multivesicular vesicles | Dehydration-rehydration vesicles | |
| 9 | Bubblesomes |
Table 1: Liposome classification.
Stability Of Liposomes
The therapeutic efficacy of the drug molecule is governed by the stability of the liposomes involving manufacturing steps, storage and delivery. A stable dosage form maintains the physical stability and chemical integrity of the active molecule during its developmental procedure and storage. Stability study with designing includes the evaluation of its physical, chemical and microbial parameters along with the assurance of integrity of the product during its storage [11] (Table 2).
| S. No. | Parameter | Technique |
|---|---|---|
| 1 | Size | Electron microscopy, light scattering ultracentrifugation, Coulter counter |
| 2 | Number of Lamellae | NMR spectroscopy, Small angle x-ray scattering, Electron microscopy |
| 3 | Bilayer Fluidity | Fluorescence polarization |
| 4 | Charges | Microelectrophoresis |
Table 2: Technique for physical characterization of liposomes.
Physical stability
The vesicles obtained during the liposomal formation processes are of different sizes. During its storage, vesicles tend to aggregate and increase in size to attain a thermodynamically favourable state. While storage, leakage of the drug from the vesicles can cause its fusion and breaking. This deteriorates the physical stability of the liposomal drug product. Therefore, morphology and size distribution of the vesicles are important parameters for assessing the physical stability [12]. A variety of techniques are employed to estimate the visual appearance (morphology and size of the vesicles like light scattering and electron microscopy. Cholesterol makes the lipid membrane stronger but its concentration cannot be more than 50% in the liposome structure. It is necessary for the stabilization and maintenance of the bioactive molecule at the core of the liposome. Physical stability can be maintained by avoiding the excess unsaturation in the phospholipids as they are subjected to mere peroxidation and by maintaining the pH conditions. They must be stored at 4°C with no freezing and light exposure.
Chemical stability
Phospholipids are chemically unsaturated fatty acids, prone to oxidation and hydrolysis, which may alter the stability of the drug product. pH, ionic strength, solvent system and buffered species also play a major role in maintaining a liposomal formulation. Oxidation deterioration involves the formation of cyclic peroxides and hydroxyperoxidases due to the result of free radical generation in the oxidation process. Liposomes can be prevented from oxidative degradation by protecting them from light, by adding anti-oxidants such as α-tocopherol or butylated hydroxyl toluene (BHT), producing the product in an inert environment (presence of nitrogen or Argon) or by adding EDTA to remove trace heavy metals [3]. Hydrolysis of the ester bond at C-4 position of the glycerol moiety of phospholipids leads to the formation of lyso-phosphatidylcholine (lysoPC). This will enhance the permeability of the liposomal contents. Hence, control of lysoPC limit within the drug product of lysosomes becomes important. It can be achieved by formulating lysoPC free liposomes with phosphatidylcholine [13].
Mechanism Of Action Of Liposomes
A liposome consists of a region of aqueous solution inside a hydrophobic membrane. Hydrophobic chemicals can be easily dissolved into the lipid membranes; in this way liposomes are able to carry both hydrophilic and hydrophobic molecules. While the extent of location of the drug will depend upon its physiochemical characteristics and composition of lipid. For the deliverance of necessary drug molecules to the site of action, the lipid bilayers fuse with other bilayers of the cell (cell membrane) to release the liposomal content.
Steps involved in liposome action of drug delivery:
1. Adsorption: Adsorption of liposomes to cell membranes causes its contact on the cell membrane.
2. Endocytosis: Adsorption of liposomes on the cell surface membrane followed by engulfment and internalization into the liposomes
3. Fusion: fusion of lipid bilayers of liposomes with the lipoidal cell membrane by lateral diffusion and intermingling of lipids results in direct delivery of liposomal contents in the cytoplasm.
4. Lipid exchange: Due to the similarity of liposomal lipid membrane with cell membrane phospholipids, lipid transfer proteins in the cell membrane easily recognize liposomes and cause lipid exchange.
For example, in case of cancer cells; they consume large amounts of fats to fill the requirement of rapid growth, they recognize the liposomes (loaded with anti-cancer drug) as a potential source of nutrition. When they are targeted by liposome, they get absorbed. Once the anti-cancer drugs are released from the liposome into the site, cancer cells are killed by the drug.
Therapeutic Applications Of Liposomes
Liposomes provide superior therapeutic efficacy and safety in comparison to existing formulations. Some of the major therapeutic applications of liposomes in drug delivery include:
Site-avoidance delivery
The cytotoxicity of anti-cancer drugs to normal tissues is attributed to their narrow therapeutic index (TI). Under such circumstances, the TI can be improved by minimizing the delivery of drug to normal cells by encapsulating in liposomes. For eg doxorubicin has a severe side effect of cardiac toxicity, but when formulated as liposomes, the toxicity was reduced without any change in the therapeutic activity [14].
Site specific targeting
Delivery of a larger fraction of the drug to the desired (diseased) site, reducing the drug’s exposure to normal tissues can be achieved by site specific targeting. On systemic administration, long circulating immunoliposomes are able to recognize and bind to target cells with greater specificity [15]. For e.g. in patients with recurrent osteosarcoma, there was an enhanced tumoricidal activity of monocytes, when muramyl peptide derivatives were formulated as liposomes and administered systemically.
Intracellular drug delivery
Increased delivery of potential drugs to the cytosol (where drug receptors are present) can be accomplished by using LDDS. N-(phosphonacetyl)-L-aspartate (PALA) is normally poorly taken up into cells. Such drugs when encapsulated within liposomes, showed greater activity against ovarian tumor cell lines in comparison to free drug [16].
Sustained release drug delivery
To achieve the optimum therapeutic efficacy, which requires a prolonged plasma concentration at therapeutic levels, liposomes provide sustained release of target drugs [17]. Drugs like cytosine Arabinoside can be encapsulated in liposomes for sustained release and optimized drug release rate in vivo.
Intraperitoneal administration
Tumors that develop in the intra-peritoneal (ip) cavity can be treated by administering the drug to ip cavity. But the rapid clearance of the drugs from the ip cavity results in minimized amount of drug at the diseased site. However, liposomal encapsulated drugs have lower clearance rate, when compared to free drug and can provide a maximum fraction of drug in a prolonged manner to the target site [18].
Immunological adjuvants in vaccines
Liposomes can be used for enhancing the immune response by encapsulating the adjuvants. Depending on the lipophilicity of antigens, the liposome can accommodate antigens in the aqueous cavity or incorporate within the bilayers. To enhance the immune response of diphtheria toxoid, liposomes were first used as immunological adjuvants (Table 3).
| S. No | Liposomal formulation | Drug loaded | Target | Activity | References |
|---|---|---|---|---|---|
| 1 | Stearyl amine and diacetyl phosphate | Zidovudine | HIV | Targeting to ZDV lymphatics is enhanced | [19] |
| 2 | Hydrogenated soy phosphatidycholine cholesterol and distearoylphosphatidyl glycerol | Amphotericin B | Aspergillus fumigates | Targeted delivery of drug at infection site | [20] |
| 3 | Partially hydrogenated egg phosphatidycholine and cholesterol | Gentamycin | Klebsiella pneumonia | Drug showed increased survival rate of animal model and increased therapeutic efficacy | [21] |
| 4 | 1,2-dipalmitoyl-sn-glycero-phosphatidylcholine and cholesterol | Polymixin B | Pseudomonas aeroginosa | Drug showed decreases bacteria count in lung, increased bioavailability and decreased lung injury caused by bacteria | [22] |
| 5 | Cholesterol Phosphatidylserine, phosphatidylglycerol or cardiolipin. Saturated or unsaturated Phospholipid acyl chains | Doxorubicin | Anti-cancerous | Reduction in cardiotoxicity and enhanced antitumor activity | [23] |
| 6 | Soya lecithin, cholesterol tocopheryl acetate | Ketaconazole | Candida spp., Microsporum | Improve therapeutic response and reduce adverse effects | [24] |
| 7 | Egg phosphotidyl choline, cholesterol (steryl amine and diacetyl phosphate as + or – charge inducers) | Acetazolamide | Used in glaucoma and epilepsy | Increasing the stability and reducing the intraocular pressure | [25] |
| 8 | Dimyristoyl phosphatidylcholine cholesterol or ergosterol | N-Methyl-N-D-fructosyl amphotericin B methyl ester (MFAME) | Anti-fungal | Reduction in toxicity of Amphotericin | [26] |
| 9 | Egg lecithin, 10% (mol/mol) cholesterol and 10% (mol/mol) Tween 80. Ascorbic acid | Ferrous sulphate | Anemia | Increased electrostatic and steric stability | [27] |
| 10 | L-a-phosphatidylcholine 95% (PC), cholesterol | Hydroxyzine | Used as sedative | Increase in drug concentration in skin and enhanced efficacy | [28] |
| 11 | Soyabean phosphatidylcholine or hydrogenated soybean phosphatidylcholine and cholesterol (PEG Ligated) | Topotecan HCl | Anti-cancerous | Improved stability and enhanced efficacy by accumulation in tumor cells | [29] |
| 12 | Soyabean phosphatidyl choline (S100PC) and 1,2-distearoyl-sn-glycero-3-phosphoethanolamine [methoxy (polyethyleneglycol)-Cholesterol (CH)4 | Paclitaxel | Anti-cancerous | Increased aqueous solubility | [30] |
Table 3: Liposomal drug delivery system.
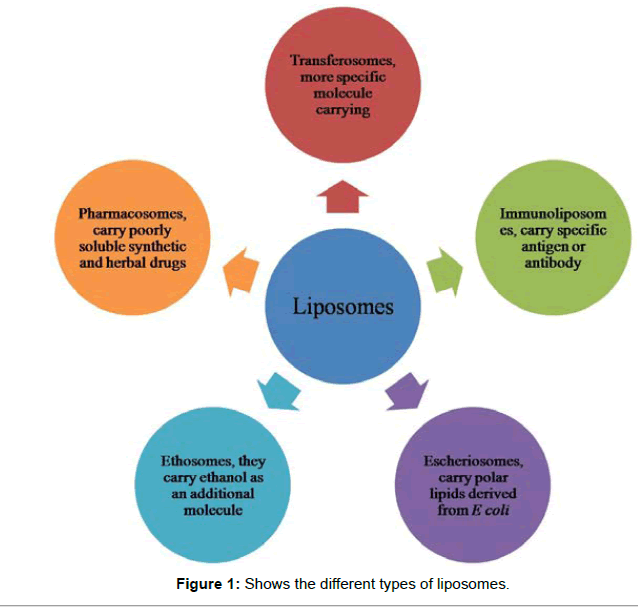
Stealth liposomes contain few biological species as a ligand to enable binding with specific expression on the drug delivery site (targeted site) in addition to PED coating. These targeting ligands could be, vitamins, specific antigens or monoclonal antibodies (making an immuno-liposome), but it must be available. Naturally toxic drugs can be less toxic systemically if delivered to the diseased tissues or site. Ligands used for targeting to lungs for treatment of tuberculosis include maleylated bovine serum albumin (MBSA) and O-steroyl amylopectin. Transfersomes (a type of liposomes) are highly deformable vesicles, used for transdermal material delivery (non-invasive method) [31]. Doxorubicin (Doxil) and Daunorubicin (anticancer drugs) may be given via liposomes. Liposomes is used in cancer therapy, since cancer cells have overexpressed folate and transferrin receptors, making transferrin and folic acid as suitable ligands [32]. Other ligands used in cancer therapy are peptides and antibodies against VEGF, VCAM, matrix metalloproteases (MMPs), integrins etc. A recent Phase I/II study evaluating the safety and efficacy of a novel neoadjuvant combination treatment of paclitaxel, pegylated liposomal doxorubicin, and hyperthermia to treat locally advanced breast cancer. A phase II clinical trial of pegylated liposomal doxorubicin and carboplatin in Japanese patients with platinumsensitive recurrent ovarian, fallopian tube or primary peritoneal cancer. Its combination chemotherapy is done to treat patients with platinum-sensitive recurrent ovarian cancer. The thermosensitive liposomal formulation ThermoDox® (Celsion Corporation, Lawrenceville, NJ), which contains lysophosphatidylcholine and is employed in the treatment of various cancers including primary liver cancer, recurrent chest wall breast cancer, colorectal, pancreatic and metastatic liver cancer, is currently in various stages of human clinical trials (Phase II/III) [33]. Lipoplatin is developed recently by using cisplatin as a carrier for treating cancer [34]. Escheriosomes (a type of liposomes) prepared from lipids (polar) {obtained from the Escherichia coli} have shown high cytotoxic T lymphocyte (CTL) responses to deliver their entrapped molecules right into the cytosol of the APCs (Antigen Presenting Cells) [35].
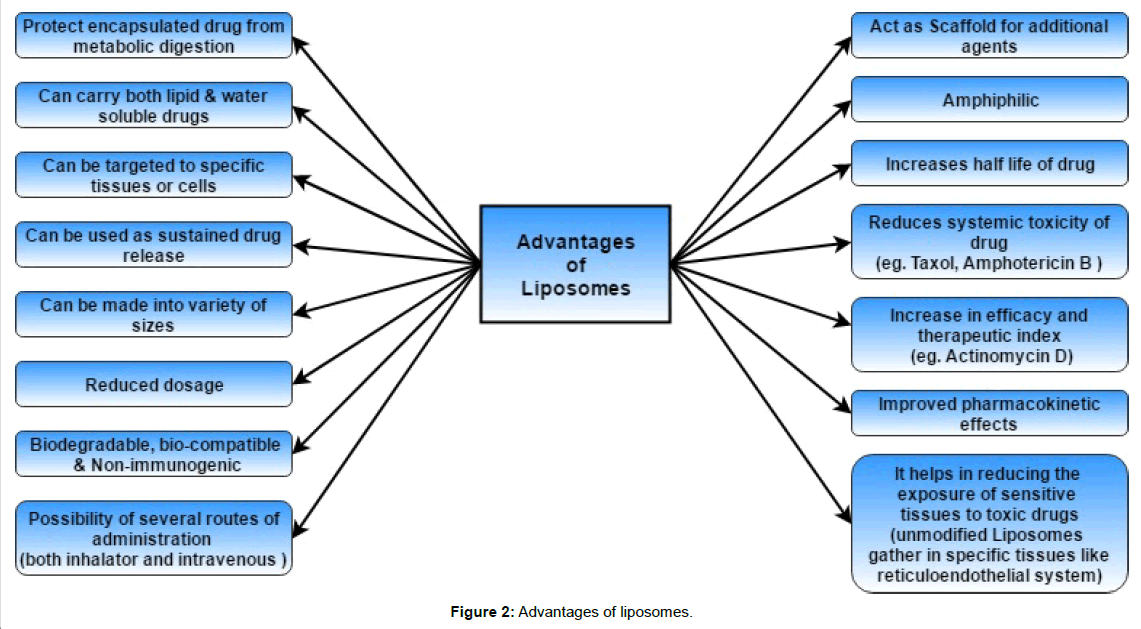
Advantages Of Liposomal Drug Delivery
Drug encapsulation in a liposomal or lipid drug delivery system improves the pharmacokinetic and pharmacodynamic properties to such an extent that the drugs can be brought into regular use [36].
Advantages of liposomes as a drug delivery system for antimicrobials are:
• Improvement and control over pharmacokinetics and pharmacodynamics
• Decreased toxicity
• Enhanced drug activity against intracellular pathogens
• Liposomes used as target selective
• Enhanced activity against extracellular pathogens [37]
Improvement of pharmacokinetics and pharmacodynamics
Many drugs require regular doses when given without a drug delivery system. While liposomes can be formulated to have a long circulating time in the body, keeping the constant level of the drug for a longer time. Factors that influence the circulation time are: the fluidity of the membrane, the size of the liposomes (smaller liposomes circulate longer) and the charge on the liposomes (neutral liposomes have a long circulation time). Liposomes protect some drugs against chemical and immunological breakdown, as well as protecting them against the effect of enzymes. Liposomes give lowered toxicity and lower dosing because of sustained drug levels, especially when so called “stealth” liposomes are used [38].
Liposomes used as target selective
Structure and make-up of the cell membranes is an important factor in reaction of cells. This can be exploited for drug targeting by inducing specific cells to react to and absorb the liposomes. The membrane surface structure can be modified for specific drug targeting; either by changing the charge on the membrane, or adding specific proteins, antibodies or immunoglobulins. It increases the affinity of liposomes to specific cells. Experimenting with liposomes include: creating liposomes that react to specific pH’s or temperatures, before releasing the drug. Liposomes can be made to just interact with specific organisms [5]. In order to lower toxicity, liposomes are made to avoid certain areas, also known as siteavoidance- therapy.
Enhanced drug activity against intracellular pathogens
Various formulations of liposomes have been tested for many years against the entire host of intracellular parasites and other pathogens. It has been used to great success in the treatment of leishmaniasis as they are actively removed in vivo by the macrophages which are infected with Leishmania. The modified encapsulated drug is 700 times more effective than the free drug in the treatment of Leishmania in hamsters [39]. These findings were followed by other studies confirmed these types of successes with leishmaniasis. In studies of anti-tuberculosis drugs like clarithromycin, isoniazid and rifampicin, the efficacy of each drug was significantly higher when compared to the free form of the drug [40]. Other studies showed lowered toxicity to surrounding tissues. This lowered toxicity is especially important with drugs that are in themselves very toxic, like the drugs that are currently available to treat Trypanosoma brucei and Trypanosoma cruzi. Even though the in vivo effect against the parasite is not significantly better than the free drug, the liposomes did have a protective effect against the toxicity of the drugs used in certain instances. The main objective of encapsulating anti-malarial drugs in liposomes is to lower the adverse effects, providing sustained release and protecting the drug from being broken down. In a study, the maximum plasma concentration was increased in a shorter time for encapsulated liposomes. The time taken for the drug concentration to reach its maximum level get shortened. In an important experiment, the arteether’s bioavailability was close to 98% when formulated in liposomes, compared to arteether in suspension with a bioavailability of approx. 32%. Antimalarials used earlier that have been used for entrapment in liposomes include chloroquine and primaquine. It becomes quite clear that liposomes can become a great asset in the battle against malaria. It has been proved that liposomes are able to overcome bacterial resistance in some cases. Other studies even claim lowering the required dose necessary for effective treatment by between 4-16 times against Pseudomonas (Figure 1).
Disadvantages Of Liposomes
Lipid based drug delivery system are expensive to produce, hence the production cost is high. The cost is high because of high costs associated with the raw materials used in lipid excipients as well as expensive equipment needed to increase manufacturing [41].
Sterilization
The sterilization of liposomes is a complicated conundrum, as liposomes are sensitive to high temperatures and certain methods of radiation. Sterilizing with chemicals is not a viable option either, as it may affect the stability of the liposomes. For the production of sterile liposomes, there is only method of filteration through 0.22 μm membrane filter. This method is not suitable if the liposomes are bigger than 0.2 μm in diameter and it does not remove viruses [42]. Another option is filtering the initial solutions through 0.45 μm regenerated cellulose filters and glass fiber filters before starting production, thereafter the entire production process must be done under aseptic conditions.
Short shelf life and stability
For a pharmaceutical product to be viable in the market, it requires the product to be stable in some form or another for at least a year and a half to two years. To achieve this, is very difficult if the liposomes remain in suspension. After production, freeze drying can be used to increase the selflife of liposomes. Two factors play a major role in the stability of liposomes namely, chemical and physical degradation. They degrade chemically through oxidation and hydrolysis. To decrease oxidation and hydrolysis, only fresh and new reagents of the highest quality are used, avoid methods that have high temperatures, use inert atmosphere to store liposomes, deoxygenate aqueous solutions and do all manufacturing in the absence of oxygen. Lastly an antioxidant such as α-tocopherol may be added [43]. Physical degradation is most often attributed to the difference in the packing density of the lipids in the bilayer structure. This can be fixed by incubating the liposomes at a temperature close to the phase transition temperature, until the arrangement of the lipids equalizes. Fusion phenomenon is very common between liposomes which are the reason behind its instability. The instability is curbed by addition of cholesterol into the mixture of lipid to raise its transition temperature [44]. Different types of liposomes have different issues when the composition of the membrane is changed. This phenomenon is exploited in the creation of thermosensitive liposomes. The liposomes release the drug as soon as the temperature is high enough. Physical degradation is an important factor when the synthesized formulations are freeze-dried. For the preparation of freeze-dried products cryoprotector is added to ensure its stability at the time of reconstituted.
Encapsulation efficacy
Liposome is an ideal drug carrier and must be able to entrap the drug in a therapeutic dose. Otherwise the amount of lipids and or other constituents of the liposomes can become toxic. Therefore, the method of entrapment is of utmost importance. When entrapment of the drug is low, methods like active loading is used to improve the entrapment since liposomes entrap low amount of drug [45]. This method involves using an uncharged drug that can easily cross the lipid bilayer in uncharged form, but changes in the charged species once inside the liposome (the drug is unable to escape the interior of the liposome in the charged form). The effect can be created by entrapping a low pH environment inside the liposome and suspending the vesicles in a neutral pH environment, which contains the drug.
Removal from circulation by the reticulo-endothelial system (RES)
One of the major obstacle of liposome drug delivery system is its rapid clearance from the bloodstream by phagocytic cells of the RES or mononuclear phagocyte system (MPS) [46]. The speed by which uptake takes place is dependent on few factors, including the size and charge of the liposomes as larger ones are eliminated from circulation faster than smaller. The liposomes accumulate in the liver and the spleen, because of the rich blood supply as well as the amount of macrophages that accumulate there. Some strategies to prolong circulating time have come to light, such as the formulation of long circulating liposomes or so called LCL’s. These liposomes are formulated in different ways, such as coating the liposome with a polymer, with the most popular one being; coating of existing liposomes with polyethylene glycol (PEG). These types of liposomes are referred as “stealth” liposomes [38]. Another alternative is including cholesterol and sphinomyelin (SM) in the formulation to increase the TC of the formulation. It increases the stability in plasma and reduces the uptake by the RES (Figure 2).
Conclusion
The application of liposomes has been broadened since its discovery in 1964. Liposomes composed of a whole host of different lipids, manmade or naturally occurring, each having their own uses, advantages and disadvantages. The widely used lipid component is phosphatidycholine, because of it is neutral and relatively low in cost. Liposomes are classified according to production method, composition as well as size and shape. Liposomes as a drug delivery system include benefits like improved pharmacokinetics and pharmacodynamics, decreased toxicity, enhanced therapeutic efficacy against pathogens and improved drug-target selectivity. The disadvantages, on the other hand are: certain lipids, especially charged lipids, become toxic in increased doses, sterilization is a huge obstacle, problems with short shelf-life and stability, and problems with encapsulation efficacy. The interaction of liposomes with cells is very important as they influence delivery of drug. Liposomes have been in use as drug delivery systems in the recent years with a few formulations commercially available, which show greater effectivity. Liposomes based drug delivery has a great promise in the future.
References
- Mayhew E, Rustum YM, Szoka F, Papahadjopoulos D (1979) Role of cholesterol in enhancing the antitumor activity of cytosine arabinoside entrapped in liposomes. Cancer Treat Rep 63: 1923-1928.
- Çagdas M, Sezer AD, Bucak S (2014) Liposomes as potential drug carrier systems for drug delivery. Nanotechnol Nanomater 2014: 1-50.
- Li J, Wang X, Zhang T, Wang C, Huang Z, et al. (2014) A review on phospholipids and their main applications in drug delivery systems. Asian J Pharm Sci 10: 81-98.
- Bozzuto G, Molinari A (2015) Liposomes as nanomedical devices. Int J Nanomed 10: 975-999.
- Taylor KMG, Morris RM (1995) Thermal analysis of phase transition in liposomes behaviour. Thermochim Acta 248: 289-301.
- Dölle S, Lechner BD, Park JH, Schymura S, Lagerwall JPF, et al. (2012) Utilizing the Krafft phenomenon to generate ideal micelle-free surfactant-stabilized nanoparticle suspensions. Angew Chemie 51: 3254-3257.
- Monteiro N, Martins A, Reis RL, Neves NM (2014) Liposomes in tissue engineering and regenerative medicine. J R Soc Interface 11: 20140459.
- Biswas S, Dodwadkar NS, Sawant RR, Koshkaryev A, Torchilin VP (2011) Surface modification of liposomes with rhodamine-123-conjugated polymer results in enhanced mitochondrial targeting. J Drug Target 19: 552-561.
- Akbarzadeh A, Rezaei-Sadabady R, Davaran S, Joo SW, Zarghami N, et al. (2013) Liposome: classification, preparation and applications. Nanoscale Res Lett 8: 102.
- Yadav AV, Murthy MS, Shete AS, Sakhare S (2011) Stability aspects of liposomes. Indian J Pharm Educ Res 45: 402-413.
- Grit M, Crommelin DJ (1993) Chemical stability of liposomes: Implications for their physical stability. Chem Phys Lipids 64: 3-18.
- Cruz-Leal Y, Machado Y, López-Requena A, Canet L, Laborde R, et al. (2014) Role of B-1 cells in the immune response against an antigen encapsulated into phosphatidyl choline-containing liposomes. Int Immunol 26: 427-437.
- Alyane M, Barratt G, Lahouel M (2016) Remote loading of doxorubicin into liposomes by transmembrane pH gradient to reduce toxicity toward H9c2 cells. Saudi Pharm J 24: 165-175.
- Paszko E, Senge MO (2012) Immunoliposomes. Curr Med Chem 19: 5239-5277.
- Krieger ML, Eckstein N, Schneider V, Koch M, Royer HD, et al. (2010) Overcoming cisplatin resistance of ovarian cancer cells by targeted liposomes in vitro. Int J Pharm 0389: 10-17.
- Loira-Pastoriza C, Todoroff J, Vanbever R (2014) Delivery strategies for sustained drug release in the lungs. Adv Drug Del Rev 75: 81-91.
- Eloy JO, Claro de Souza M, Petrilli R, Barcellos JPA, Lee RJ, et al. (2014) Liposomes as carriers of hydrophilic small molecule drugs: Strategies to enhance encapsulation and delivery. Colloids and Surf B Biointerfaces 123: 345-63.
- Kaur CD, Nahar M, Jain NK (2008) Lymphatic targeting of zidovudine using surface-engineered liposomes. J Drug Target 16: 798-805.
- Takemoto SK, Zeevi A, Feng S, Colvin RB, Jordan S, et al. (2004) National conference to assess antibody-mediated rejection in solid organ transplantation. Am J Transplant 4: 1033-1041.
- Cabanes A, Reig F, Garcia-Anton JM, Arboix M (1998) Evaluation of free and liposome-encapsulated gentamycin for intramuscular sustained release in rabbits. Res Vet Sci 64: 213-217.
- Omri A, Suntres ZE, Shek PN (2002) Enhanced activity of liposomal polymyxin B against Pseudomonas aeruginosa in a rat model of lung infection. Biochem Pharmacol 64: 1407-1413.
- Xiong XB, Huang Y, Lu WL, Zhang H, Zhang X, et al. (2005) Enhanced intracellular uptake of sterically stabilized liposomal doxorubicin in vitro resulting in improved antitumor activity in vivo. Pharm Res 22: 933-939.
- Shirsand S, Kanani K, Keerthy D, Nagendrakumar D, Para M (2012) Formulation and evaluation of Ketoconazole niosomal gel drug delivery system. Int J Pharm Investig 2: 201.
- Hathout Y (2007) Approaches to the study of the cell secretome. Expert Rev Proteomics 4: 239-248.
- Angra PK, Oettinger C, Balakrishna Pai S, D’Souza MJ (2009) Amphotericin B microspheres: A therapeutic approach to minimize toxicity while maintaining antifungal efficacy. J Microencapsul 26: 580-587.
- Xia S, Xu S (2005) Ferrous sulfate liposomes: Preparation, stability and application in fluid milk. Food Res Int 38: 289-96.
- Elzainy AAW, Gu X, Simons FER, Simons KJ (2003) Hydroxyzine from topical phospholipid liposomal formulations: Evaluation of peripheral antihistaminic activity and systemic absorption in a rabbit model. AAPS PharmSci 5: 41-48.
- Blanco E, Kessinger CW, Sumer BD, Gao J (2009) Multifunctional micellar nanomedicine for cancer therapy. Exp Biol Med (Maywood) 234: 123-131.
- Xing H, Hwang K, Lu Y (2016) Recent developments of liposomes as nanocarriers for theranostic applications. Theranostics 6: 1336-1352.
- Chaudhary H, Kohli K, Kumar VK (2013) Nano-transfersomes as a novel carrier for transdermal delivery. Int J Pharm 454: 367-380.
- Sriraman SK, Salzano G, Sarisozen C, Torchilin V (2016) Anti-cancer activity of doxorubicin-loaded liposomes co-modified with transferrin and folic acid. Eur J Pharm Biopharm 105: 40-49.
- Zylberberg C, Matosevic S (2016) Pharmaceutical liposomal drug delivery: A review of new delivery systems and a look at the regulatory landscape. Drug Deliv 23: 3319-3329.
- Vhora I, Khatri N, Desai J, Thakkar HP (2014) Caprylate-conjugated cisplatin for the development of novel liposomal formulation. AAPS PharmSciTech 15: 845-857.
- Syed FM, Khan MA, Nasti TH, Ahmad N, Mohammad O (2003) Antigen entrapped in the escheriosomes leads to the generation of CD4(+) helper and CD8(+) cytotoxic T cell response. Vaccine 21: 2383-2393.
- del Amo EM, Rimpelä AK, Heikkinen E, Kari OK, Ramsay E, et al. (2017) Pharmacokinetic aspects of retinal drug delivery. Prog Retin Eye Res 57: 134-185.
- Zou Y, Lee HY, Seo YC, Ahn J (2012) Enhanced antimicrobial activity of nisin-loaded liposomal nanoparticles against foodborne pathogens. J Food Sci 77: 165-170.
- Sur S, Fries AC, Kinzler KW, Zhou S, Vogelstein B (2014) Remote loading of pre-encapsulated drugs into stealth liposomes. Proc Natl Acad Sci USA 111: 2283-2288.
- Flanigen EM, Bennett JM, Grose RW, Cohen JP, Patton RL, et al. (1978) Silicalite, a new hydrophobic crystalline silica molecular sieve. Nature 271: 512-516.
- Rodrigues C, Gameiro P, Prieto M, De Castro B (2003) Interaction of rifampicin and isoniazid with large unilamellar liposomes: Spectroscopic location studies. Biochim Biophys Acta 1620: 151-159.
- Altaf SBM, Yada V, Mamatha Y, Prasanth VV (2012) liposomes? An overview. J Pharm Sci Innov 1:13-21.
- Toh MR, Chiu GNC (2013) Liposomes as sterile preparations and limitations of sterilization techniques in liposomal manufacturing. Asian J Pharm Sci 8: 88-95.
- Gibis M, Rahn N, Weiss J (2013) Physical and oxidative stability of uncoated and chitosan-coated liposomes containing grape seed extract. Pharmaceutics 5: 421-433.
- Lee DE, Lew MG, Woodbury DJ (2013) Vesicle fusion to planar membranes is enhanced by cholesterol and low temperature. Chem Phys Lipids 166: 45-54.
- Pattni BS, Chupin VV, Torchilin VP (2015) New developments in liposomal drug delivery. Chem Rev 115: 10938-10966.
- Ishida T, Harashima H, Kiwada H (2002) Liposome clearance. Biosci Rep 22: 197-224.
Citation: Yadav D, Sandeep K, Pandey D, Dutta RK (2017) Liposomes for Drug Delivery. J Biotechnol Biomater 7: 276. DOI: 10.4172/2155-952X.1000276
Copyright: 2017 Yadav D, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 66106
- [From(publication date): 0-2017 - Dec 04, 2025]
- Breakdown by view type
- HTML page views: 62551
- PDF downloads: 3555