Make the best use of Scientific Research and information from our 700+ peer reviewed, Open Access Journals that operates with the help of 50,000+ Editorial Board Members and esteemed reviewers and 1000+ Scientific associations in Medical, Clinical, Pharmaceutical, Engineering, Technology and Management Fields.
Meet Inspiring Speakers and Experts at our 3000+ Global Conferenceseries Events with over 600+ Conferences, 1200+ Symposiums and 1200+ Workshops on Medical, Pharma, Engineering, Science, Technology and Business
Research Article Open Access
Long Term Outcome of CT-Based Image-Guided Brachytherapy for Cervix Cancer Using the Tandem-Ring Applicator
| Tan LT*, Diane Whitney D and Coles CE | |
| Oncology Centre, Box 193, Cambridge University Hospitals NHS Foundation Trust, Cambridge, UK | |
| Corresponding Author : | Tan LT Oncology Centre, Box 193 Cambridge University Hospitals NHS Foundation Trust, Cambridge, UK Tel: (01223)596182 Fax: (01223)763120 E-mail: litee.tan@addenbrookes.nhs.uk |
| Received April 23, 2014; Accepted July 07, 2014; Published July 10, 2014 | |
| Citation: Tan LT, Diane Whitney D, Coles CE (2014) Long Term Outcome of CTBased Image-Guided Brachytherapy for Cervix Cancer Using the Tandem-Ring Applicator. OMICS J Radiol S1:003. doi: 10.4172/2167-7964.S1-003 | |
| Copyright: © 2014 Tan LT, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Visit for more related articles at Journal of Radiology
| Keywords |
| Cervix cancer; Image-guided; Brachytherapy; CT |
| Abbreviations |
| BT: Brachytherapy; CSS: Cancer-specific survival; CT: Computed Tomography; EBRT: External Beam Radiotherapy; EQD2: Equivalent Dose in 2 Gy fractions; EUA: Examination Under Anaesthesia; GEC-ESTRO: Groupe Européen de Curiethérapie European Society for Therapeutic Radiology and Oncology; HDR: High Dose Rate; HR-CTV: High Risk Clinical Target Volume; IGBT: Image- Guided Brachytherapy; IMRT: Intensity-Modulated Radiotherapy; LC: Local Control; MRI: Magnetic Resonance Imaging; OAR: Organs At Risk; OS: Overall Survival; PDR: Pulsed Dose Rate; PMB: Parametrial Boost; RT: Radiotherapy |
| Introduction |
| Image-guided brachytherapy (IGBT) is the new gold standard for cervix cancer brachytherapy (BT). In 2005, the Groupe Européen de Curiethérapie European Society for Therapeutic Radiology and Oncology (GEC-ESTRO) published recommendations on contouring of tumor target and organs at risk (OAR) and reporting of dose volume parameters for magnetic resonance imaging (MRI)-based IGBT [1,2]. This was followed by the first single institution report from Vienna in 2007 [3] showing a 3-year local control (LC) rate of 87% (88% for tumors 2-5 cm, 81% for tumors >5 cm) and a 3-year rate of G3-4 bowel and urinary toxicity of 2%. In an update published in 2011 [4], the 3-year LC rate was 91% (95% for tumors 2-5 cm, 90% for tumors >5 cm) with an incidence of G3-4 bowel and urinary toxicity of 5%. The GEC-ESTRO guidelines have subsequently been adopted by the American Brachytherapy Society [5]. |
| While the gain in therapeutic index with MRI-based IGBT is potentially large, many radiotherapy (RT) centres do not have access to MRI for RT planning. In 2009, the Royal College of Radiologists published a guidance document to facilitate implementation of IGBT for cervix cancer in the UK [6]. A questionnaire survey [7] showed that by March 2011, the number of UK centres offering IGBT for cervix cancer had increased to 32/45 (71%) compared to 12/45 (26%) in September 2008. However, the vast majority (23/32=72%) were offering computed tomography (CT)-based IGBT rather than MRIbased IGBT. |
| At Addenbrooke’s Oncology Centre, Cambridge, CT-based IGBT was used for the treatment of cervix cancer between February 2005 and December 2008 (since January 2009, MRI-based IGBT has been used). Analysis of our first 3 years’ experience of 28 patients with a median follow-up of 23 months showed a 3-year LC rate of 96% [8] compared to a historical series of 49 patients treated with conventional BT (76%) [9] (p=0.04) However, only 4 patients (14%) had tumors >5 cm in diameter; the 3-year LC in these patients was 75%. This paper is reports the long term outcome of a larger group of patients with mature follow-up. |
| Materials and Methods |
| Study |
| A retrospective audit of 40 patients treated with high dose rate (HDR) CT-based IGBT between February 2005 and December 2008 was carried out. All cervix cancer patients treated radically with primary radiotherapy ± concomitant chemotherapy during the study period were included. Patients who received post-operative or palliative radiotherapy were excluded. Written informed consent was obtained from all patients prior to commencing treatment. |
| Patients |
| The median age of the patients was 52 years (range: 24-80 years). All the patients underwent examination under anaesthesia (EUA) prior to commencing RT. 39 patients had staging MRI; one patient who declined MRI due to claustrophobia had a staging CT. 30 (75%) patients had squamous cell carcinomas, 9 (23%) adenocarcinomas and 1 adenosquamous carcinoma. The FIGO stage distribution was Ib=8, Ib2=4, IIa=4, IIb=17, IIIb=5, IVa=2. 14 (35%) patients had tumors >5 cm. 14 (35%) patients had pelvic node involvement on imaging (≥ 1 cm) or histology. 4 (10%) patients had paraaortic node involvement. |
| Treatment |
| All the patients received initial external beam RT (EBRT) using 15 MV photons. In 33 (83%) patients, a conformal 4-field pelvic plan was used. Seven (18%) patients received extended-field RT; 6 patients were treated with opposed anterior-posterior fields while one was treated with intensity-modulated RT (IMRT). The planned EBRT dose was 45 Gy in 25 fractions over 5 weeks in 39 patients. The IMRT patient received 60 Gy in 30 fractions over 6 weeks to the pelvis and paraaortic nodes. 4 (10%) patients received parametrial boosts (PMB) after BT for significant residual disease in the parametrium, 8 Gy in 4 fractions, using opposed fields and a half beam blocking isocentric technique. The sagittal plane through the isocentre was matched to the 80% isodose line of the BT dose distribution. 39 (98%) patients received weekly cisplatin chemotherapy 40 mg/m2 during EBRT. One patient did not receive chemotherapy as she had microscopic Stage Ib1 disease but was deemed not suitable for surgery. Two patients underwent nodal debulking prior to EBRT. |
| On completion of EBRT, all the patients proceeded to HDR BT using a tandem-ring applicator (Nucletron, an Elekta company, Elekta AB, Stockholm, Sweden); interstitial BT was not used. The median time between EBRT and first BT was 3 days (range: 1-11 days). In 38 patients, BT was planned to be delivered in 3 fractions over 8 days. Two patients were planned for 2 fractions over 8 days, one because of logistical reasons and the other because the patient had received a higher EBRT dose using IMRT. |
| For each fraction, a CT scan was obtained with the BT applicator in situ. The high risk clinical target volume (HR-CTV) (whole cervix +visible and palpable tumor at EUA) and OAR (bladder, rectum, sigmoid) were contoured on the CT images. The following dose parameters were evaluated: dose to point A, D90 (minimum dose to 90% of the HR-CTV), V100 (percentage of HR-CTV receiving 100% of the prescribed dose) and D2cc (minimum dose in the most exposed 2 cm3 volumes) for bladder, rectum and sigmoid. The BT dose was added to the EBRT dose to produce the total equivalent dose in 2 Gy fractions (EQD2) using a α/β ratio of 10 (Gy10) and 3 (Gy3) for tumor and normal tissues. The PMB dose was not included in the calculations. |
| An individualized dosimetry plan (PLATO v.14.3.5, Nucletron) was created for each fraction. The standard loading pattern used was as recommended by the Vienna group [10]. Manual optimization of source positions and dwell times was carried out if the dosimetry achieved with the standard loading pattern did not meet our planning aims. Prior to 2008, our planning aims were to achieve a point A dose of ≥ 74 Gy10, a D90 ≥ 74 Gy10, V100 of 100% and a D2cc rectum of ≤ 5 Gy per fraction. In 2008, our aims were amended to point A ≥ 74 Gy10, D90 ≥ 74 Gy10, V100 ≥ 95%, D2cc rectum and sigmoid ≤ 75 Gy3 and D2cc bladder ≤ 95 Gy3. |
| Treatment failures were classified as local (including pelvic node relapse) or distant (including paraaortic node relapse). Late RT toxicity, defined as complications present at or after 6 months after RT, was prospectively graded using Common Terminology Criteria for Adverse Events version 3.0. Kaplan-Meier curves for overall survival (OS), cancer-specific survival (CSS), LC and late RT toxicity were computed using MedCalc for Windows, version 12.3.0.0 (MedCalc Software, Mariakerke, Belgium). Time intervals for survival and LC were calculated from the date of biopsy to date of event or last appointment. Time intervals for toxicity were calculated from the date of completion of RT and were censored for disease recurrence or patient death. |
| Results |
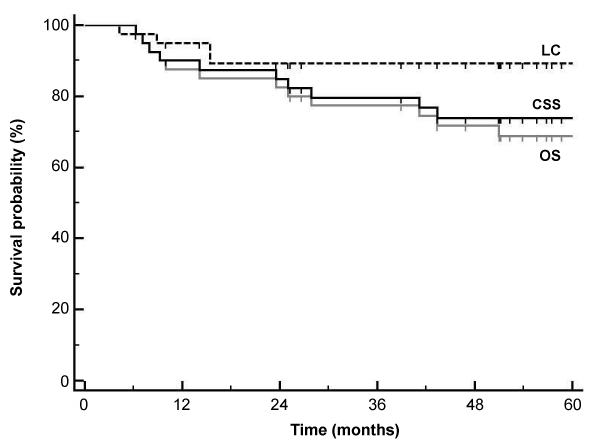
| Median follow-up for surviving patients was 61 months (range: 25- 85 months). 12 patients developed recurrent disease (Table 1), 2 with pelvic recurrence only, 8 with distant metastases only and 2 with both pelvic and distant relapse. The 3-year and 5-year OS was 77% and 69% with a CSS of 80% and 74% respectively (Figure 1). The 3-year and 5-year LC was the same at 89%. For tumors ≤ 5 cm, the 3-year and 5-year OS, CSS and LC was 84%, 87% and 96% respectively. For tumors >5 cm, the OS and CSS was 52% and 59% at 3 years and 44% and 52% at 5 years while LC was 76% at both 3 years and 5 years. The CSS for node-negative patients was 81% at 3 years and 5 years compared to 77% and 66% for node-positive patients (p=0.4). |
| Seven patients developed recurrent disease within 6 months and were excluded from the toxicity analysis. The incidence of late bowel toxicity was G0=17, G1=4, G2 =8, G3=4 and G4=0. For urinary toxicity, the incidence was G0=19, G1=2, G2=10, G3=2 and G4=0. Four patients had G3 complications (Table 2). The 3-year and 5-year rate of G3 complications was 10% and 15%. |
| 38 patients (95%) completed planned EBRT without any interruptions. Four patients did not complete planned BT. One patient refused further BT after 1 fraction and was given an EBRT boost. Two patients developed septicaemia after 2 fractions and the third fraction was abandoned. In one patient, there was technical difficulty in applicator placement and only 2 fractions could be delivered. The median overall treatment time (EBRT+BT+PMB) was 43 days (range: 41-69 days). |
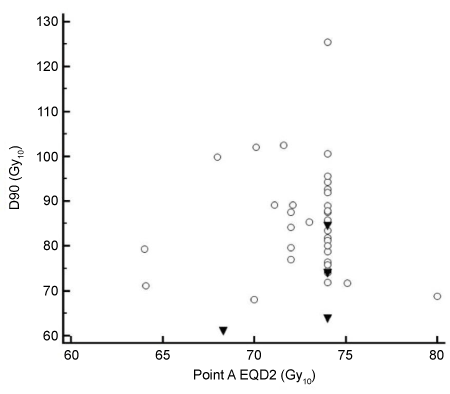
| A total of 113 BT fractions were delivered during the study period. The mean HR-CTV volume was 20.5 cc (standard deviation SD=10.1 cc). In 96 (85%) fractions, manual optimization did not improve the dosimetry compared to the standard plans. Figure 2 shows D90 plotted against point A EQD2. A point A EQD2 of ≥ 74 Gy10 was achieved in 26 (65%) patients. The D90 was ≥ 74 Gy10 in 33 (83%) patients. A V100 of >95% was achieved in 81 (72%) fractions. |
| Table 1 shows the D90 for the patients with relapse. The mean D90 (± SD) for the patients with local recurrence was 70.8 ± 9.3 Gy10 (range: 61.0-84.5 Gy10) compared to 85.2 ± 11.5 Gy10 (range: 68.0-125.4 Gy10) for those without local recurrence (p=0.02, Student’s t-test). |
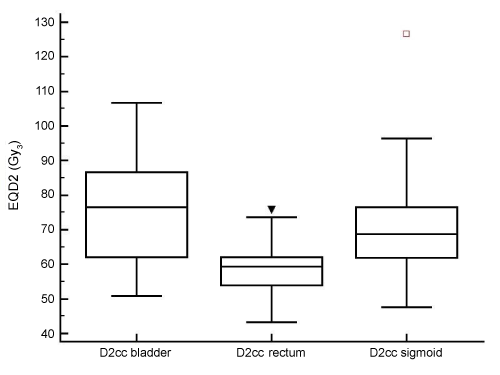
| Figure 3 shows the distribution of D2cc bladder, rectum and sigmoid. The D2cc rectum was lower than for the other OAR as it was the only dose constraint we were using before 2008. The D2cc for the patients with G3 toxicity are shown in Table 2. The OAR doses for the only patient who had both bowel and urinary G3 toxicity were all below the planned dose constraints; this patient is the only patient who did not receive chemotherapy suggesting that she may have abnormal normal tissue radiosensitivity. Two of the patients with G3 bowel toxicity had high D2cc sigmoid (83.4 and 83.7 Gy3). In comparison, 4 of 6 other patients who had D2cc sigmoid ≥80 Gy3 had no bowel toxicity at all. |
| Discussion |
| Our present analysis confirms that CT-based IGBT has a beneficial impact on long term local tumor control. The 3-year LC rate was 89% compared to 76% for a historical series treated with conventional BT [8,9] (Table 3). For tumors ≤ 5 cm, the 3-year LC rate was 96% which is comparable to the Vienna MRI-based technique [4]. For tumors >5 cm, the improvement in 3-year LC seen in our initial analysis was maintained at 76% even though there were more patients with bulky tumors (35% vs. 14%). While this is superior to our historical series (3- year LC = 59%), it is inferior to the Vienna series. The 3-year rate of G3 bowel and urinary toxicity was lower than our conventional BT series (10% vs. 16%) but was higher than the Vienna series. |
| Table 3 shows the results of CT-based IGBT reported by other groups. Kang et al. [11] from Seoul compared the outcome of patients treated with HDR BT and found no significant difference in 3-year LC of those who underwent IGBT (97%) and those planned conventionally (91%). For tumors >4 cm, however, the LC was 98% with IGBT compared to 81% with conventional BT (p=0.02). The incidence of G3-4 late rectal bleeding was lower with IGBT (2%) compared to conventional BT (13%) (p=0.02). The French STIC prospective study [12] reported the outcome of 117 patients treated with mainly CTbased pulsed dose rate (PDR) BT. There was a significant increase in the 2-year LC rate (IGBT 69.6%, standard BT 61.2%, p=0.001) with a reduction in G3-4 toxicity (IGBT 2.6%, standard BT 22.7%, p=0.002). Tharavichitkul et al. [13] from Chiang Mai reported the outcome of 47 patients treated with IMRT and HDR IGBT (15 patients had MRI at the first fraction, the remainder had CT-based planning alone). The 3-year LC rate was 97.9%; only one patient developed G3 bowel and urinary toxicity. |
| The lower LC rates seen in the French series compared to the other CT-based IGBT series raises some interesting questions. It is unlikely that this difference is because PDR BT was used. A more plausible explanation may be the doses delivered (Table 3). The mean point A dose for the French series was 70 ± 10 Gy10 compared to 73 ± 3 Gy10 (median: 74 Gy10, range: 64-80 Gy10) for our series and a median dose of 82 Gy10 (range: 63–98 Gy10) for the Seoul series. The mean HR-CTV D90 in the French series was 73 ± 11 Gy10 compared to 84 ± 12 Gy10 for our series, 93 ± 8 Gy10 for the Chiang Mai series and 93 ± 13 Gy10 for the Vienna MRI series. The Seoul paper did not evaluate the D90 HR-CTV but mentioned that a tumor EQD2 of >85 Gy10 was achieved in 50% (48/97) of patients. This is similar to our series in which 43% (17/40) of patients had a D90 HR-CTV of >85 Gy10. |
| The optimum dose for local tumor control in cervical cancer remains to be established. Dimopoulos et al. [14] showed that a D90 HR-CTV of ≥ 88 Gy10 was associated with a 90% control rate in the true pelvis. Our patients with local recurrence had a D90 HR-CTV of 61.0- 84.5 Gy10, which is in keeping with this finding. For smaller tumors, lower doses may be sufficient although the minimum threshold is unknown. Toita et al. [15] reported a 2-year LC rate of 96% for patients with tumors <4 cm treated with chemo-RT+conventional BT to a total point A EQD2 of only 52 Gy10. There were three patients with local recurrences who probably would have been cured with higher doses, but the paper illustrates that it is possible to cure a proportion of low bulk cervical cancers with relatively low doses. |
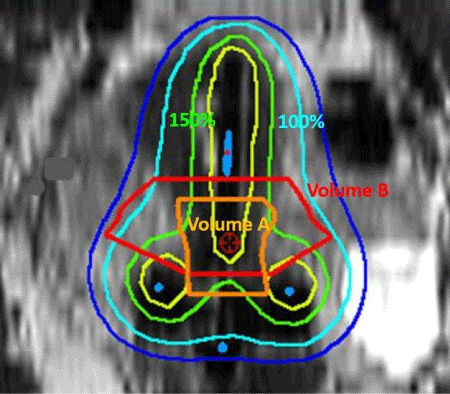
| It may perhaps not be widely appreciated that the D90 HR-CTV is influenced by variation in contouring. This is illustrated in Figure 4. The D90 in our series may have been artificially high as our mean HR-CTV volume was only 21 cc compared to 30-35 cc for other series [10,12,16]. In the French STIC study, while the majority of recurrences may have occurred in larger tumors, it is possible that there were recurrences in patients with small tumors as well as 75% of patients had Stage Ib1- IIb tumors. With a mean point A dose of 70 Gy10 and a SD of 10 Gy10, it is likely that a significant proportion of patients received <65 Gy10 (or even <60 Gy10) to point A; this may have contributed to the lower LC rates with IGBT, even if the D90 HR-CTV appeared satisfactory. In the future, it is hoped that the ongoing EMBRACE study (www.embracestudy.dk) will provide information about the doses (including D90 HR-CTV and point A dose) necessary for optimum control of both small and large tumors. Until then, it may be prudent for users of IGBT to have minimum thresholds for the point A dose, particularly for small tumors, to avoid inadvertent underdose due to contouring error which could compromise local tumor control. For large tumors, the D90 HR-CTV is usually lower than the point A dose; a combined intracavitary and interstitial technique has been shown to improve tumor coverage in these patients with improvement in local tumor control [3]. This is supported by the lower LC for bulky tumors in our series as interstitial BT was not used. |
| The rate of G3 toxicity in our series was higher than in the Vienna, French, Seoul and Chiang Mai series. The mean D2cc bladder, rectum and sigmoid in our series was 75.5 ± 14.8 Gy3, 58.5 ± 6.9 Gy3 and 69.1 ± 14.4 Gy3 respectively compared to the Vienna (95 ± 22 Gy3, 65 ± 12 Gy3 and 62 ± 12 Gy3) [17], French (64.2 ± 11.9 Gy3, 67.2 ± 20 Gy3 and 58.1 ± 8.9 Gy3) and Chiang Mai (88.2 ± 7.2 Gy3, 69.6 ± 6.6 Gy3 and 72.0 ± 6.9 Gy3) series. Possible explanations for our high toxicity rate include our lack of dose constraints for the sigmoid and bladder prior to 2008 (although 3 of the 4 patients with G3 toxicity were treated in 2008), our policy to give priority to optimization of tumor target dose and coverage compared to meeting OAR dose constraints, and variation in individual patient normal tissue radiosensitivity. Moreover, we found that the scope for dose adaptation with the CT-based intracavitary technique rather limited; 19 (48%) of our patients had OAR doses which exceeded at least one of the constraints despite optimization. |
| In January 2009, we implemented MRI-based IGBT for all patients with cervix cancer, combined with interstitial BT for selected patients. More recently, we have implemented a policy of differential doses depending on tumor size and stage. Patients in whom the HR-CTV can be adequately covered with intracavitary BT alone are given lower doses (3 fractions, D90 HR-CTV ≥ 80 Gy10, point A dose ≥ 70 Gy10) with lower OAR dose constraints (D2cc bladder ≤ 90 Gy3, rectum ≤ 70 Gy3 and sigmoid ≤ 70 Gy3). Those who require additional interstitial BT for adequate tumor coverage are given higher doses (4 fractions, D90 HR-CTV ≥ 90 Gy10, point A dose ≥ 84 Gy10) with higher OAR constraints (D2cc bladder ≤ 95 Gy3, rectum ≤ 75 Gy3 and sigmoid ≤ 75 Gy3). The priority for the first group is to meet OAR dose constraints while for the second group, it is to ensure adequate tumor coverage. It is hoped that this differential approach will reduce the late toxicity rate in our patients while maintaining the high LC rate. |
| Conclusion |
| CT-based IGBT improves local control compared to conventional BT, particularly for small tumors. For large tumors, the results of a CTbased intracavitary technique are inferior to MRI-based IGBT with a combined intracavitary and interstitial technique as reported in the literature. This is most likely because the scope for dose adaptation with a CT-based intracavitary technique is limited. |
References |
|
Tables and Figures at a glance
| Table 1 | Table 2 | Table 3 |
Figures at a glance
 |
 |
 |
 |
| Figure 1 | Figure 2 | Figure 3 | Figure 4 |
Post your comment
Relevant Topics
- Abdominal Radiology
- AI in Radiology
- Breast Imaging
- Cardiovascular Radiology
- Chest Radiology
- Clinical Radiology
- CT Imaging
- Diagnostic Radiology
- Emergency Radiology
- Fluoroscopy Radiology
- General Radiology
- Genitourinary Radiology
- Interventional Radiology Techniques
- Mammography
- Minimal Invasive surgery
- Musculoskeletal Radiology
- Neuroradiology
- Neuroradiology Advances
- Oral and Maxillofacial Radiology
- Radiography
- Radiology Imaging
- Surgical Radiology
- Tele Radiology
- Therapeutic Radiology
Recommended Journals
Article Tools
Article Usage
- Total views: 14591
- [From(publication date):
specialissue-2014 - Aug 19, 2025] - Breakdown by view type
- HTML page views : 9954
- PDF downloads : 4637
