Oropharyngolaryngeal Sarcocystosis and Histoplasmosis Co-Infection in a Patient with Systemic Lupus Erythematosus
Received: 30-Jun-2016 / Accepted Date: 20-Jul-2016 / Published Date: 22-Jul-2016 DOI: 10.4172/2476-2024.1000117
Abstract
Sarcocystosis is an uncommon zoonotic coccidian protozoal infection caused by Sarcocystis spp. Muscular sarcocystosis have been detected in patients with various types of cancer but co-infection with histoplasmosis has never been reported. We report a 39-year-old woman with active lupus nephritis who presented with hoarseness for eleven months. Physical examination and magnetic resonance imaging revealed an irregular exophytic mass at the left oropharynx and the left true vocal cord. The histopathologic diagnosis was oropharyngolaryngeal sarcocystosis and histoplasmosis. She was treated with oral itraconazole for histoplasmosis. The authors briefly review the clinicopathologic features and pathogenesis of oropharyngolaryngeal sarcocystosis and histoplasmosis and conclude the possibility of co-infection of oropharyngolaryngeal sarcocystosis and histoplasmosis, especially in immunocompromised patient.
Keywords: Sarcocystosis; Histoplasmosis; Systemic lupus erythematosus; Larynx; Oropharynx
6077Case Report
Sarcocystosis is an uncommon protozoal parasitic infection of human being caused by Sarcocystis spp. However, the outbreaks of muscular sarcocystosis have been recently documented [1,2]. In addition, we published the interesting case of sarcocystosis concomitant with laryngeal squamous cell carcinoma in September 2011 edition of Southeast Asia journal of tropical medicine and public health [3]. Herein, we supplementary report a rare case of concomitant oropharyngolaryngeal sarcocystosis and histoplasmosis in a patient with systemic lupus erythematosus (SLE). A combined coccidian parasitic and fungal infection has never been reported.
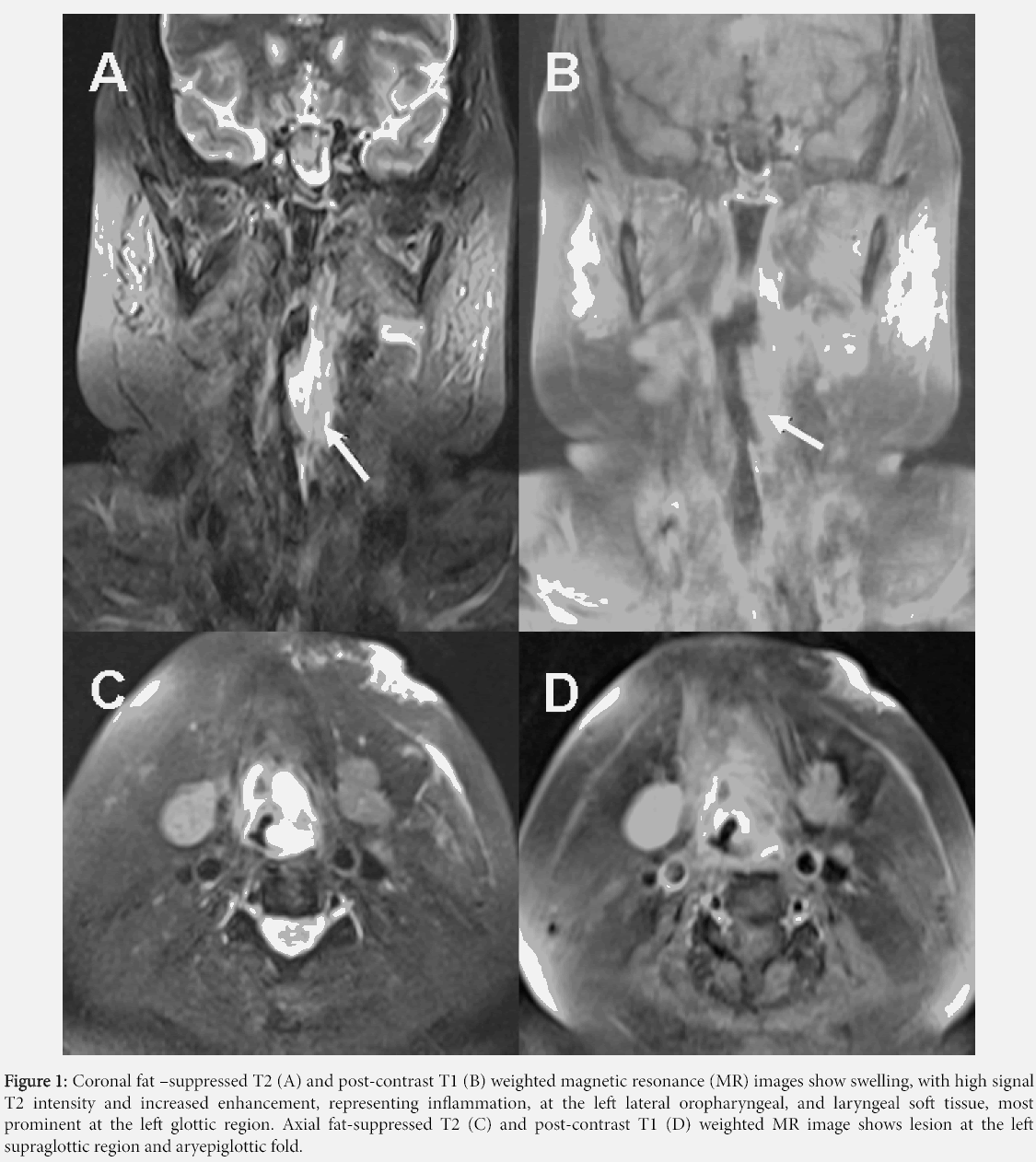
A 39-year-old woman presented with hoarseness for eleven months. Her underlying disease was systemic lupus erythematosus, diagnosed 8 years earlier with criteria of lupus nephritis (class III), arthritis, oral ulcer, and positive anti-nuclear antibody. She was treated with intravenous high-dose of cyclophosphamide and corticosteroids. A physical examination showed an irregular mucosal surface at the left true vocal cord. The left oropharynx showed an irregular ulcer. Magnetic resonance imaging exhibited a swelling lesion at the glottic region, left aryepiglottic fold, left lateral oropharyngeal wall, and left oral mucosa (Figure 1). The endoscopic biopsy was performed. Serological testing for IgG/IgM anti-Toxoplasma antibody was negative.
Figure 1: Coronal fat –suppressed T2 (A) and post-contrast T1 (B) weighted magnetic resonance (MR) images show swelling, with high signal T2 intensity and increased enhancement, representing inflammation, at the left lateral oropharyngeal, and laryngeal soft tissue, most prominent at the left glottic region. Axial fat-suppressed T2 (C) and post-contrast T1 (D) weighted MR image shows lesion at the left supraglottic region and aryepiglottic fold.
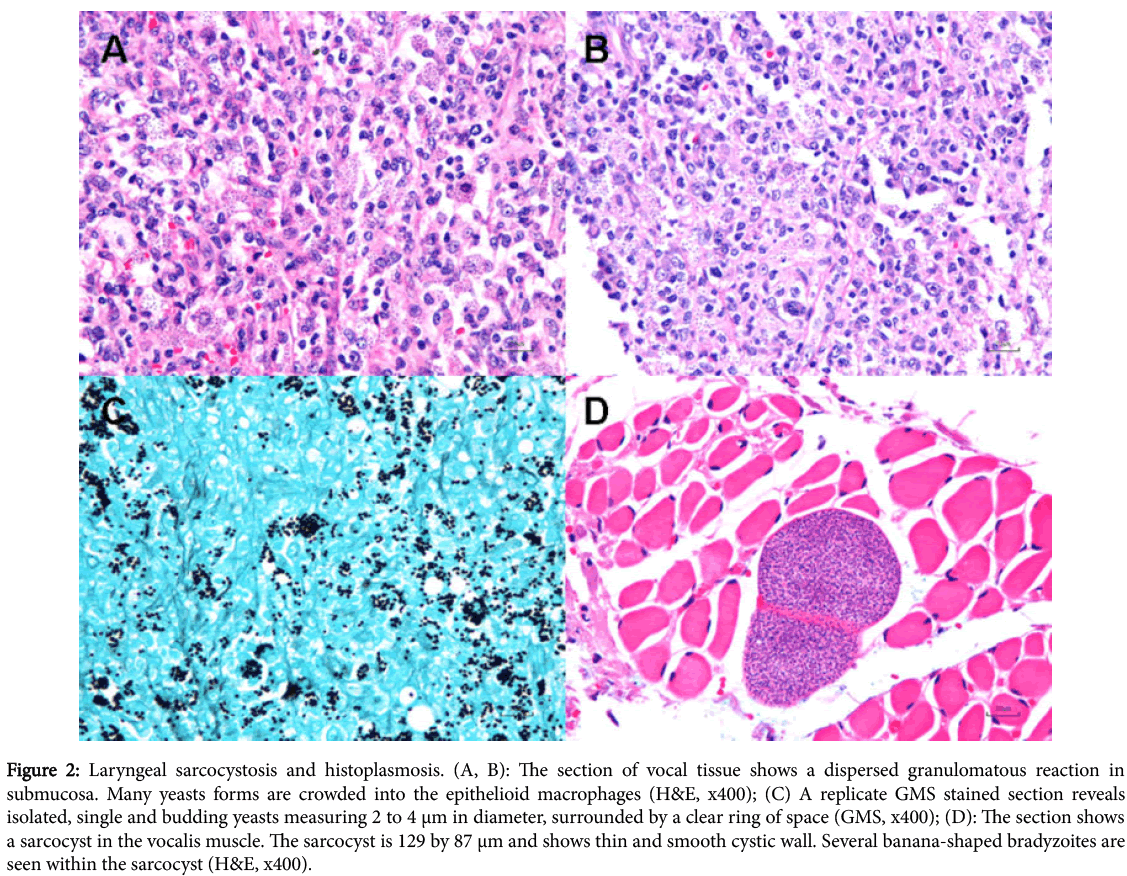
The histopathology revealed an acanthotic squamous mucosa with diffuse infiltration of a poorly-formed granuloma. There were multiple intracellular yeasts in macrophages (Figures 2A, 2B and 2C) with multiple sarcocysts in the skeletal muscle (Figures 2D). The pathologic diagnosis was oropharyngolaryngeal sarcocystosis and histoplasmosis.
Figure 2: Laryngeal sarcocystosis and histoplasmosis. (A, B): The section of vocal tissue shows a dispersed granulomatous reaction in submucosa. Many yeasts forms are crowded into the epithelioid macrophages (H&E, x400); (C) A replicate GMS stained section reveals isolated, single and budding yeasts measuring 2 to 4 μm in diameter, surrounded by a clear ring of space (GMS, x400); (D): The section shows a sarcocyst in the vocalis muscle. The sarcocyst is 129 by 87 μm and shows thin and smooth cystic wall. Several banana-shaped bradyzoites are seen within the sarcocyst (H&E, x400).
She had been treated with oral itraconazole for histoplasmosis. Later on, she was referred to our hospital due to progressive jaundice after seven weeks of therapy. Her liver function test shows elevated liver enzymes as shown in Table 1. Itraconazole-induced cholestasis was diagnosed. She developed renal failure that required dialysis. Multiple hyperpigmented papules at her right leg showed palisading granulomatous dermatitis.
| Liver enzyme | Pre-treatment with itraconazole | After treatment with itraconazole |
|---|---|---|
| Serum total bilirubin (mg/dL) | 2.54 | 30.8 |
| Gamma glutamyltransferase (U/L) | Not performed | 4476 |
| Alkaline phosphatase (U/L) | 122 | 527 |
Table 1: The detail of liver function test.
She was treated as disseminated histoplasmosis. Antifungal therapy was switched to liposomal amphotericin B. She subsequently developed hepatic failure, hepatic encephalopathy, status epilepticus, and septicemia. Finally, she developed multiorgan failure and succumbed to the complication of disease, three months after the diagnosis of oropharyngolaryngeal sarcocystosis and histoplasmosis. No autopsy was performed.
A variety of parasitic protozoan have been identified in the muscular tissue, but only Toxoplasma spp, Neospora spp, and Sarcocystis spp. have been identified as pathogenic for humans. Sarcocystosis is highly endemic in Southeast Asia [1-4]. Muscular sarcocystosis is acquired by ingestion of infected oocysts. Subsequent hematogenous dissemination leads to merozoites invading microcirculation into muscular tissue of intermediate hosts which harbor sarcocysts or zoitocysts in their muscles [1,2].
The histopathology shows sarcocyst measuring between 40 μm to 456 μm in length and 26 μm to 142 μm in width [3]. The cystic wall is composed of hyaline eosinophilic monomorphous material and is well demarcated from the surrounding muscular fibers. The bradizoites appears as crescentric “banana” shaped structures. The clinical and histopathological features of muscular sarcocystosis and intestinal sarcocystosis in humans are summarized in Table 2.
| Detail | Muscular Sarcocystosis | Intestinal Sarcocystosis |
|---|---|---|
| Definition | A muscular infection caused by ingestion of oocyst or sporocyst | An intestinal infection caused by sarcocyst or zoitocyst |
| Species | S. lindemanni (probably from dogs) S. nesbitti (from snakes) |
S. hominis (from cattle) S. suihominis (from pigs) |
| Caused by | Ingestion of contaminated oocyst or sporocyst in food or water | Ingestion of sarcocyst or zoitocyst in undercooked meat |
| Infected animal | Herbivore or Omnivore | Carnivore or Omnivore |
| Human being | Intermediate host | Definite host |
| Reproduction | Asexual multiplication (Schizogony) | Sexual reproduction (Gametogony followed by sporogony) |
| Incidence | 21% detected by histopathologic study of tongue tissue | 23.2% detected by stool examination |
| Clinical features | Myositis, myalgia, localized painful muscular swelling, fever, weakness, vasculitis | Nausea, vomiting, stomachache, diarrhea, enteritis, fever, chill, diaphoresis |
| Eosinophilia | Common | Common |
| Erythrocyte sedimentation rate (ESR) | Increase | Normal |
| C-reactive protein (CRP) | Increase | Normal |
| Creatine kinase | Increase | Normal |
| Adolase | Increase | Normal |
| Diagnosis | Histopathology of the muscle demonstrates sarcocyst or zoitocyst | - Histopathology of the intestine demonstrates oocyst or sporocyst - Microscopic examination of stool for oocyst or sporocyst |
| Size of organism | 40-456 × 26-142 µm | 12.3-14.6 × 18.5-20 µm |
| Pathological features | Sarcocyst or zoitocyst in muscular cells surrounded by an infiltration of mononuclear cells and eosinophils | Oocyst or sporocyst in epithelial or endothelial cells of the segment of necrotizing/eosinophilic enteritis |
| Facilitate staining identification | Periodic acid-Schiff (PAS) | Periodic acid-Schiff (PAS), Ziehl-Neelsen |
| Complication | Myositis, vasculitis, myocarditis | Dehydration, enteritis, false diverticulum, partial gut obstruction, |
| Treatment | - Corticosteroid use in vasculitis or eosinophilic myositis - Co-trimoxazole, albendazole, medendazole |
Supportive treatment |
Table 2: A clinicopathological feature of muscular sarcocystosis and intestinal sarcocystosis in humans.
Sarcocysts should be differentiated from the tissue apicomplexa cyst of Toxoplasma gondii . The bradyzoites of Toxoplasma spp. appear as dot-like structures, while the bradyzoites of Sarcocystis spp. are larger and appear as crescentric banana-shaped structures. The sarcocystic wall is thick and shows variable striation, cytophaneres.
However, additional diagnostic studies such as immunohistochemical stain for Toxoplasma gondii should be performed. Therefore, the histopathologic examination is of value for identifying unsuspected conditions and may reveal major unexpected findings that are of clinical importance and necessary for the diagnosis, treatment and prevention of the infectious disease, especially in immunocompromised host.
Oropharyngolaryngeal histoplasmosis should be differentiated between primary histoplasmosis and progressive disseminated histoplasmosis. Among immunosuppressed patient who is prone for development of dissemination should have thought evaluation for multiorgan involvement. Oropharyngolaryngeal histoplasmosis treated like subacute progressive disseminated histoplasmosis.
For moderately severe to severe progressive disseminated disease, liposomal amphotericin B is recommended for 1-2 weeks or longer depends on the severity of disease, followed by oral itraconazole for a total at least 1 year. The overall prognosis is good. However, our patient had the delayed diagnosis and treatment, which lead to progression of disease to presumptive disseminated histoplasmosis.
She also had the itraconazole-induced cholestasis and subsequently developed hepatic failure, hepatic encephalopathy, status epilepticus, and septicemia, which were the major complication and cause of death.
Muscular sarcocystosis has no specific treatment [1]. The overall prognosis is excellent. Proper disposal of human feces and careful animal husbandry are mandatory in the control of muscular sarcocystosis. An accurate diagnosis requires recognition of the clinicopathological finding and a high index of suspicion. Public health education on transmission, combined with proper diagnosis and treatment are crucial [3].
We would like to emphasize that oropharyngolaryngeal sarcocystosis may accompany with an infectious disease such as histoplasmosis, especially in the immunocompromised patient. The authors also would like to alert clinicians and pathologists to suspect the possibility of such concomitance and the necessity of early diagnosis.
Conflict of interest
Authors declare that there is no conflict of interest.
Consent
Written informed consent was obtained from the patient for publication of this Case Report and accompanying images.
References
- Fayer R, Esposito DH, Dubey JP (2015) Human infections with Sarcocystis species. Clin Microbiol Rev 28: 295-311.
- Poulsen CS, Stensvold CR (2014) Current status of epidemiology and diagnosis of human sarcocystosis. J Clin Microbiol 52: 3524-3530.
- Larbcharoensub N, Cheewaruangroj W, Nitiyanant P (2011) Laryngeal sarcocystosis accompanying laryngeal squamous cell carcinoma: case report and literature review. Southeast Asian J Trop Med Public Health 42: 1072-1076.
- Wong KT, Pathmanathan R (1992) High prevalence of human skeletal muscle sarcocystosis in south-east Asia. Trans R Soc Trop Med Hyg 86: 631-632.
Citation: Larbcharoensub N, Kanoksilp W, Wirojtananugoon C, Chitasombat MN, Chawengkiattikul W, et al. (2016) Oropharyngolaryngeal Sarcocystosis and Histoplasmosis Co-Infection in a Patient with Systemic Lupus Erythematosus. Diagn Pathol Open 1: 117. DOI: 10.4172/2476-2024.1000117
Copyright: ©2016 Larbcharoensub N, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Open Access Journals
Article Tools
Article Usage
- Total views: 12632
- [From(publication date): 7-2016 - Aug 17, 2025]
- Breakdown by view type
- HTML page views: 11664
- PDF downloads: 968