Pathogenesis of Orderly Loss of Nerve Fibers in Glaucoma
Received: 26-Apr-2016 / Accepted Date: 06-May-2016 / Published Date: 09-May-2016 DOI: 10.4172/2476-2075.1000110
Abstract
Chronic Glaucoma, commonly known as glaucoma, is a misunderstood disease since given a separate entity in the 1850s. Currently, there are many conflicting views of glaucoma but on one issue we have complete agreement: the million or so densely packed nerve fibers (NFs) in the optic nerve head (ONH) are being destroyed in an orderly tandem fashion from peripheral to central, never randomly–a pathognomonic feature. The orderly destruction of NFs is perhaps the only lead we have in solving the mystery of glaucoma, but rarely discussed. If the NFs were not destroyed in a predictable orderly sequence, the role of perimetry in glaucoma would be meaningless.
Various theories have been postulated regarding the pathogenesis of glaucoma, but none of them have addressed the crucial question of orderly destruction of NFs, which can’t be ignored. For any glaucoma theory to prevail, it must incorporate the issue of the orderly destruction of NFs otherwise it will be of no value. The ‘cupping’ theory dating back 160 years and the more recent theories like apoptosis, neurodegeneration, vascular pressure and others fail to answer the question of orderly loss of NFs in glaucoma.
This article examines the prevalent theories of glaucoma in the context of orderly loss of NFs and attempts to describe the hypothesis of the sinking disc and severance of nerve fibers corroborating with orderly loss in glaucoma.
Keywords: Nerve fibers; Glaucoma; Optic nerve head; Orderly loss
6401Introduction
What is the orderly loss of NFs occurring in glaucoma?
Before we discuss the visual fields (VF) in glaucoma, it is important to describe the arrangement of NFs in the retina and ONH. First, one million or so nerve fibers in the retina are arranged in layers superficial to deep. Second, the most central vision fibers originate closest to the disc, lie most superficial (closest to vitreous) and exit from the most central part of the disc. In contrast, the most peripheral nerve fibers originate from the most distant retina or farthest from the optic disc, lie deepest (closest to sclera) and exit closest to the edge of the scleral opening.
Third, the nerve fibers originating from the nasal retina proceed directly to the nasal part of the optic disc. However, the situation is different in the temporal retina because of the presence of the macular fibers. The nerve fibers originating from the nasal aspect of the macular area proceed directly to the temporal part of the disc. The fibers originating from the temporal macular and peripheral retina arch above and below the macular fibers to reach the superior and inferior poles of the optic disc respectively. They are hence known as the arcuate fibers [1].
According to glaucomatous visual fields, the NFs in glaucoma are invariably being destroyed in a predictable orderly sequence, never haphazardly. The peripheral fibers are destroyed first; however peripheral VF loss is not helpful in diagnosis due to normal variation in the extent of peripheral VF. Furthermore, VF constriction can occur in other diseases as well such as cataract. The most diagnostic feature of glaucomatous VF is the occurrence of isolated scotomas in the paracentral area (10 to 20 degrees). Isolated scotomas become enlarged and also increase in number and ultimately coalesce to form superior and inferior arcuate scotomas – together known as the ring scotoma.
Why do scotomas appear early in the paracentral area? Although the 360 degrees of NFs are being destroyed simultaneously, the arcuate fibers lying in the paracentral area are fewer in number compared to other sites; therefore their loss is manifested earlier. Also, their characteristic arcuate shape arrangement result is easily recognizable as arcuate scotomas. After the peripheral VF is lost, the remaining 10 degrees of central vision field is retained until the end stage of glaucoma. Since orderly loss of NFs is a very pertinent feature of glaucoma, it will be our basis in analyzing the prevalent theories of glaucoma in this presentation.
Can cupping of ONH cause orderly destruction of nerve fibers?
The term cupping was given 160 years ago when ophthalmologists, with help of the newly invented ophthalmoscope by Von Helmholtz, found the ONH of glaucoma subjects were cupped instead of normally being flat [2]. It was assumed the ONH had become cupped due to atrophy and shrinkage of the NFs resulting from the force of raised intraocular pressure (IOP). Since then, the term ‘cupping’ has become synonymous with glaucoma – this term was never questioned. Instead, one hundred years later, the term cup-to-disc ratio was introduced, thus giving further validity to the cupping concept.
What are physiological cups?
The physiological cups of various sizes are produced due to varying degree of atrophy of Bergmeister’s papilla - a tuft of hyaloid vessels giving nutrition to the lens in fetal life [3]. This remnant tissue is identified histologically as the central connective tissue meniscus (CCTM) lying superficially on the surface of the NFs of the ONH. By ophthalmoscope, the CCTM appears as a central whitish depressed area, forming the base of the physiological cup. Bigger the size of the CCTM, larger the cup/disc ratio.
Some subjects have no CCTM, so they don’t have physiological cups. Some researchers describe ONH with physiological cups as doughnut shaped, meaning all the NFs are present only in the so-called peripheral ‘neuroretinal rim’ whereas the central cupped area is a hole, devoid of nerve fibers. The aforementioned doughnut shaped configuration of the ONH is a misconception since there is no histology available depicting such an arrangement of NFs in any ONH. The histology of all ONHs reveals that NFs are present all the way to the center of LC and there is no empty space. If we review the histology of any ONH, the NFs are present underneath the CCTM as well.
As to the physiological cup enlarging: why should a fibrous tissue enlarge in response to raised IOP, especially concentrically. The fibrous base of the physiological cup is not enlarging but breaking as we will discuss later.
What is posterior bowing of the lamina cribrosa?
It is hypothesized that in response to raised IOP, the LC begins to bow posteriorly.
Bowing causes distortion of the holes of LC, resulting in pinching of NFs and impediment of the axoplasmic flow, thus leading to the death of retinal ganglion cells (RGCs) [4].
There are many arguments against bowing of LC. First, it is difficult to comprehend that a multilayered rigid connective tissue plate, densely packed with NFs is so flimsy that it will bow posteriorly with a rise of just 10 mmHg of IOP. However, not bow in cases of acute glaucoma where IOP becomes extremely elevated to 70 and above. There is no acute cupping occurring in acute glaucoma.
If the LC is bowing in response to raised IOP then why is the LC bowing in response to normal range IOP as in normal-tension glaucoma (NTG)? It is hypothesized that NTG subjects have unduly sensitive or weak LC so it will bow even with the force of normal range IOP level.
An argument against above scenario: if someone is born with unduly sensitive LC, then that subject should have developed NTG in early childhood, not later in life. Furthermore, NTGs are usually associated with systemic problems like chronic hypotension and sleep apnea so structural weakness of LC cannot be a factor. Furthermore, there is no histological proof of posterior bowing of LC, however it has been well documented by EDI-SD-OCT that LC starts migrating posteriorly from the early stages of glaucoma [5,6]. Therefore, a loosened and detached LC, while migrating posteriorly, cannot become bowed as well, thus the findings of posterior migration of LC refutes the concept of cupping of LC.
Can bowing of LC cause orderly destruction of nerve fibers?
Even if agreed that LC is bowing and causing distortion of the pores and pinching of the NFs, it is inconceivable that pinching and destruction of the NFs will be so precise and orderly; one by one, starting with most peripheral fibers and ending with most central. Furthermore, the NFs are fastened in bundles in the pores of LC where their individual separation is not possible for orderly destruction of NFs to occur.
If the LC was bowing, then the most central NFs should be pinched and destroyed first. In actuality, the most peripheral fibers are being destroyed first in glaucoma. Therefore, bowing of LC, even if occurring, cannot corroborate with orderly destruction of NFs from peripheral to central fibers - a pathognomonic feature of glaucoma.
Can neurodegeneration cause the orderly destruction of NFs?
Similar to Parkinson’s and Alzheimer’s disease, it is hypothesized that glaucoma is a neurodegenerative disease. However, in every neurodegenerative disease, the neurons are being destroyed randomly, but in glaucoma, the nerve fibers or their RGCs are invariably being destroyed in a specific sequence.
For glaucoma to be a neurodegenerative disease, RGCs will have to first predict the impending glaucoma so they could start apoptosis (self-destruction) in an orderly sequence. The sequence beginning with RGCs, serving the peripheral vision and ending with those serving the central, but never randomly - an unlikely scenario. Therefore, in view of orderly destruction of NFs, glaucoma cannot be a neurodegenerative disease [7].
Is the cerebrospinal fluid pressure (CSF) playing a role in glaucoma?
It has been hypothesized that glaucoma subjects have lower CSF pressure behind the LC so IOP, being comparatively higher, will cause bowing of the LC thus initiating glaucoma. The CSF pressure at lumbar level is 8-15 mmHg (lying down), thus we should expect the CSF pressure behind the LC be much lower due to negligible subarachnoid space. If this were true, everyone would develop glaucoma. Even if the CSF hypothesis is agreed, posterior bowing of the LC cannot result in the orderly destruction of NFs as discussed earlier.
Why are the nerve fibers being destroyed in an orderly fashion?
There is no biological mechanism which acting directly on the nerve fibers or their RGCs can result in the orderly destruction of NFs. Therefore, there must be a mechanical way causing the orderly loss of NFs. It is proposed the LC is sinking in the scleral canal (mechanical scenario) due to atrophy of the border tissue of Elschnig (BT) which is solely supplied by ciliary circulation- a lower circulatory pressure compared to central retinal artery (CRA). The systemic perfusion pressure supplying the BT and IOP are opposing forces. Normally, the perfusion pressure supplying the BT should be higher than IOP for healthy maintenance of the BT.
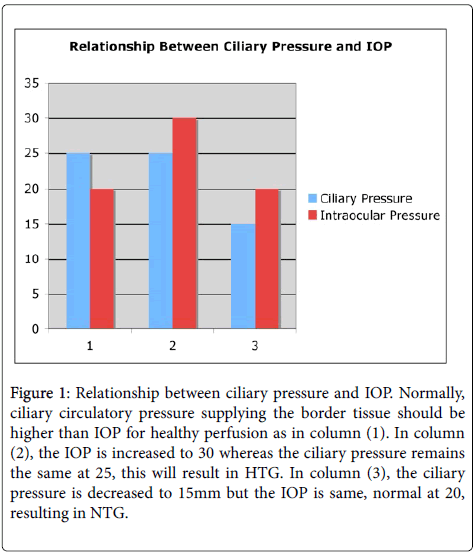
However, if the situation is reversed, either by IOP becoming elevated due to an ocular problem or the perfusion pressure supplying the BT becomes lower than the IOP due to systemic problems such as chronic hypotension, then even normal range IOP (10 to 21 mmHg) will act as high IOP for that subject and compress perfusion of the BT. This will induce chronic ischemia and atrophy of BT so NTG will ensue. Therefore, it is the IOP becoming higher than the perfusion pressure of the BT, resulting in both HTG and NTG (Figure 1).
Figure 1: Relationship between ciliary pressure and IOP. Normally, ciliary circulatory pressure supplying the border tissue should be higher than IOP for healthy perfusion as in column (1). In column (2), the IOP is increased to 30 whereas the ciliary pressure remains the same at 25, this will result in HTG. In column (3), the ciliary pressure is decreased to 15mm but the IOP is same, normal at 20, resulting in NTG.
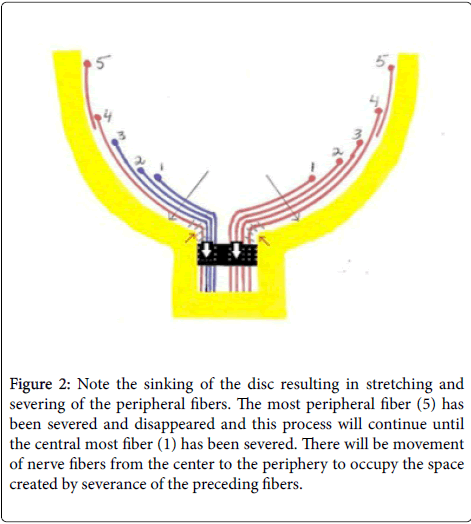
As the LC sinks, the peripheral NFs, being closest to the scleral edge are stretched and broken first. As a result, the next in line fiber will move towards the scleral edge and get severed (Figure 2). The severance of NFs leads to further LC sinking due to loss of anchorage provided by the NFs as roots anchor a tree. The cascade of severance of NFs and sinking of LC would become self-propagated and continue until all the NFs have moved in an orderly tandem fashion to the scleral edge and become severed. This may explain the unstoppable nature of glaucoma despite maximum lowering of IOP. The severed segments undergo phagocytosis, thus creating excavation (empty spaces) that we may be interpreting as cupping. The sinking of the LC and severance of nerve fibers can explain their orderly loss in glaucoma.
Figure 2: Note the sinking of the disc resulting in stretching and severing of the peripheral fibers. The most peripheral fiber (5) has been severed and disappeared and this process will continue until the central most fiber (1) has been severed. There will be movement of nerve fibers from the center to the periphery to occupy the space created by severance of the preceding fibers.
Do we have evidence of sinking LC and severance of nerve fibers?
It has been well documented that LC starts migrating posteriorly from the early stages of glaucoma. The progressive thinning of the RNFL and retinal arcuate empty spaces are due to severance of NFs. The notching at the poles of the ONH is due to severance and depletion of the arcuate fibers at the point of their entry. All the temporal fibers (macular, superior and inferior arcuate) are being severed simultaneously. However, the arcuate fibers, being fewer in number, are depleted earlier resulting in the sharply defined arcuate scotoma [8,9].
Notching at the poles of ONH, is the excavation between the NFs. At this time, the arcuate scotoma will appear on the perimetry - a confirmatory sign of glaucoma. Notching will break the physiological cup if present. The so called “floor effect” when OCT cannot measure any further thinning of RNFL is due to fact that the entire RNFL has been severed and disappeared. The histology of the end-stage glaucomatous disc reveals an empty crater left over after the severance and phagocytosis of NFs, and not 100% cupped LC [10]. The splinter hemorrhages on the margin of ONH are due to severance of vasculature, meeting the fate of NFs.
Conclusion
The orderly loss of NFs can be corroborated with the sinking LC and severance of NFs - a mechanical scenario due to the biological effect of raised IOP on the border tissue of Elschnig; resulting in chronic ischemia and its atrophy. The posterior migration or sinking of the LC from the initial stages of glaucoma has been well documented.
The histology of end-stage glaucomatous disc, an empty crater, is proof that severance of NFs and vasculature is occurring. In summary, the NFs are not being atrophied, but severed in glaucoma [11,12].
References
- Shields MB(1992) Textbook of Glaucoma(3rd edn.) Williams &Wilkens, Baltimore, USA.
- Duke-Elder S, Barrie J (1969)In Diseases of the lens and vitreous, glaucoma and hypotony, System, Vol. X1. London: Henry Kimpton 385.
- Wolff E (1968)In Anatomy of the Eye and Orbit. Revised by Last RJ. 6th ed. London: H.K. Lewis & Co pp 438.
- Quigley HA, AddicksEM (1981) Regional differences in the structure of the lamina cribrosa and their relation to glaucomatous optic nerve damage. Arch Ophthalmol 99: 137-143.
- Yang H, Williams G, Downs J, Sigal I, Roberts M (2010) Optic Nerve Head Lamina Cribrosa Insertion Migration and Pialization in Early Non-Human Primate Experimental Glaucoma. Invest Ophthalmol Vis Sci 51: 1631.
- Yang H, Williams G, Downs JC, Sigal IA, Roberts MD, et al. (2011) Posterior (outward) migration of the lamina cribrosa and early cupping in monkey experimental glaucoma. Invest Ophthalmol Vis Sci 52: 7109-7121.
- Hasnain SS (2014) Is Glaucoma a Neurodegenerative Disease? Axon Severance Paradigm Project 12: 4.
- Hasnain SS, Iqbal S, Iqbal MA, Hasnain A (2012) Pathogenesis of Arcuate Field Defects in Glaucoma. Highlights of Ophthalmology.
- Hasnain SS (2006) Scleral edge, not optic disc or retina, is the primary site of injury in chronic glaucoma. Med Hypotheses 67: 1320-1325.
- YanoffM, Fine BS (1982)Ocular pathology: A text and atlas, 2nd edition. Harper & Row 27: 209.
- Hasnain SS (2016) The Missing Piece in Glaucoma? Open Journal Of Ophthalmology 6: 56-62.
- Zeried FM,Osuagwu UL (2013) Changes in retinal nerve fiber layer and optic disc algorithms by optical coherence tomography in glaucomatous Arab subjects. Clinical Ophthalmology 7: 1941-1949.
Citation: Hasnain SS (2016) Pathogenesis of Orderly Loss of Nerve Fibers in Glaucoma. Optom open access 1: 110. DOI: 10.4172/2476-2075.1000110
Copyright: © 2016 Hasnain SS. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 17477
- [From(publication date): 6-2016 - Jun 15, 2025]
- Breakdown by view type
- HTML page views: 16461
- PDF downloads: 1016