Research Article Open Access
Poly-γ-Glutamic Acid from Bacillus Horneckiae Strain APA of Shallow Marine Vent Origin with Antiviral and Immunomodulatory Effects against Herpes Simplex Virus Type-2.
Annarita Poli1, Concetta Gugliandolo2, Antonio Spanò2, Valentina Taurisano1, Paola Di Donato1, Teresa L Maugeri2, Barbara Nicolaus1* and Adriana Arena2,31Consiglio Nazionale delle Ricerche (C.N.R.), Institute of Biomolecular Chemistry (I.C.B.), Via Campi Flegrei 34, 80078 Pozzuoli, (Na), Italy
2Research Centre for Extreme Environments and Extremophiles, Department of Biological and Environmental Sciences, University of Messina, V. le F. Stagno d’Alcontres 31, 98166 Messina, Italy
3Department of Human Pathology, Unit of Microbiology, Policlinico Universitario “G. Martino”, Via Consolare Valeria, 98125 Messina, Italy
- *Corresponding Author:
- Barbara Nicolaus
Consiglio Nazionale delle Ricerche (C.N.R.)
Institute of Biomolecular Chemistry (I.C.B.)
Via Campi Flegrei 34, 80078 Pozzuoli, (Na), Italy
Tel: +390818675245
Fax: +390818041770
E-mail: bnicolaus@icb.cnr.it
Received date: September 22, 2015; Accepted date: November 20, 2015; Published date: November 25, 2015
Citation: Poli A, Gugliandolo C, Spanò A, Taurisano V, Donato PD, et al. (2015) Poly-γ-Glutamic Acid from Bacillus Horneckiae Strain APA of Shallow Marine Vent Origin with Antiviral and Immunomodulatory Effects against Herpes Simplex Virus Type-2. J Marine Sci Res Dev 5:173. doi:10.4172/2155-9910.1000173
Copyright: © 2015 Poli A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Marine Science: Research & Development
Abstract
The shallow vents of the Eolian Islands (Italy) are sources of almost unexplored microorganisms able to produce biomolecules with promising pharmaceutical applications. This work describes the characterization of an extracellular poly-γ-glutamic acid (γ-PGA) from the new Bacillus horneckiae strain APA, and its antiviral and immunomodulatory effects against Herpes simplex virus type 2 (HSV-2). The γ-PGA-APA was able to hinder HSV-2 replication in human peripheral blood mononuclear cells (PBMC), involving the cell-mediated immunity. The exopolymer induced the expression of high levels of Th1–type cytokines in PBMC, while it had no effects on Th2-type cytokines production. This is the first report on a γ-PGA produced by a marine thermotolerant B. horneckiae strain with antiviral and immunomodulator activities. As stimulator of Th1 cell-mediated immunity, this biopolymer could be potentially used in the treatment of immunocompromised hosts as well as in patients affected by herpes virus infections.
Keywords
Antivirals; Bacillus horneckiae; γ-PGA; Immunomodulator; Microbial diversity; Shallow marine vents.
Abbreviations
ANOVA: One-Way Analysis of Variance; ELISA: Enzyme-Linked ImmunoSorbent Assay; EPS: Exopolysaccharides; FT-IR: Fourier Transform InfraRed; 1H-NMR: Proton Nuclear Magnetic Resonance; HSV-2: Herpes Simplex Virus Type 2; IFN: Interferon; IL: Interleukins; MOI: Multiplicity of Infection Index; MTT: 3-(4,5-Dimethylthiazol-2-Yl)-2,5-Diphenyltetrazolium Bromide; PBMC: Human Peripheral Blood Mononuclear Cells; PCR: Polymerase Chain Reaction; PFU: Plaque Forming Unit Per ml; PGAs: Poly-Glutamic Acids; γ-D-PGA: γ-D-Glutamic Acid; γ- L- PGA: γ-L-Glutamic Acid; RPMI medium: Culture Medium Developed At Roswell Park Memorial Institute; Th cells: T Helper Cells; TNF α: Tumour Necrosis Factor α; WISH: Human Amniotic Cells
Introduction
The shallow marine vents occur near active volcanic areas over a range of depth less than 200 m and are characterised by unusual chemical and physical conditions (such as high temperatures, low pH, high concentrations of CO2, H2S, hydrocarbons, heavy metals, etc.,) that are adverse to the majority of organisms, but not to extremophiles (i.e., a group of living organisms and microbes that are able to thrive in extreme conditions). Extremophilic microorganisms of marine origin are able to produce a wide range of biomolecules (e.g., enzymes, lipids, polymers and compatible solutes) which have useful functions in situ besides possessing a wide variety of properties that may not be found in their terrestrial counterparts. For these reasons they are the object of growing industrial interest in relation to the vast diversity of their products that are almost unexploited for their biotechnological and biological properties.
The shallow hydrothermal systems of the Eolian Islands (Italy) provide an interesting example of extreme marine environment from which a variety of novel thermophilic bacteria have been isolated [1-3]. Many of these species, under laboratory conditions, are able to produce enzymes and exopolysaccharides (EPS) [4-8], with attractive biological and chemical properties potentially useful in several biotechnological sectors [9-13].
Poly-glutamic acids (PGAs) are a group of non-toxic, microbial extracellular biopolymers with interesting properties, such as biodegradability and edibility that make them useful for applications in the food and pharmaceutical industries. Indeed, PGAs have been studied as thickeners and cryoprotectants for drug delivery, and also as biological adhesives, bioflocculants, and heavy metals absorbers for bioremediation purposes [14]. PGAs are predominantly produced by Bacteria belonging to the genus Bacillus, although they have been also identified from Archaea, such as the halophilic species of the genus Natrialba [15]. Several species of the genus Bacillus, mainly represented by B. subtilis and B. licheniformis, produce poly-γ-glutamic acid (γ-PGA), an unusual macromolecular anionic polypeptide consisting of D-and L-glutamic acid units linked by γ-amide bonds [16-21]. The molecular weight of bacterial γ-PGA molecules varies from 10 to 1000 kDa, and their stereochemical structure is of three types: i) homopolymers of D-glutamic acid (γ-D-PGA), ii) homopolymers of L-glutamic acid (γ-L-PGA), and iii) copolymers of random combinations of D-/L-glutamic acid (γ-DL-PGA) [22]. The γ-D-PGA is almost exclusively produced by B. anthracis [22], the first bacterium identified as PGA producer. The γ-L-PGA has been obtained from halotolerant Bacteria (B. megaterium and B. halodurans) and extremely halophilic Archaea (Natrialba aegyptiaca and Natronococcus occultus) [15]. The γ-DL-PGA is produced by different strains of several Bacillus species, such as Bacillus subtilis IFO 3335, B. subtilis MR-141, B. subtilis F-2-01, B. licheniformis ATCC 9945A and B. licheniformis A35 [15]. The presence of several free carboxyls in the main chain of γ-PGA is responsible for the polymer’s characteristics, e.g., super absorbency and moisture retention. Some chemical modifications, including crosslinking, derivative and chelate reactions, usually yield γ-PGA products that are used as hydrogels, flocculants, thickeners, dispersing agents, drug deliveries, cosmetics, and feed additives [23].
Nevertheless, only few examples of PGA producing extremophiles have been described in literature. This work describes the characterization of an extracellular poly-γ-glutamic acid (γ-PGA) from the novel Bacillus horneckiae strain APA, isolated from a shallow hydrothermal vent off Panarea Island (Eolian Islands, Italy), and its antiviral and immunomodulatory effects against Herpes simplex virus type 2 (HSV-2).
HSV-2 is the cause of common and ever-increasing, viral infections in humans, ranging from gingivostomatitis, keratoconjunctivitis, genital diseases, encephalitis, to infections of newborns and immunecompromised patients. Few drugs like cyclovir and congeners have been licensed for the treatment of HSV infections; however their extended use has led to the emergence of resistant viral strains, especially in the prophylaxis treatment of transplanted patients and in immuno-compromised hosts. In this frame, the search for new antiviral and immunomodulatory compounds of natural origin represents an emerging field of research.
Materials and Methods
Isolation and characterization of the strain APA
Strain APA was isolated from a thermal fluid sample collected at 21.3 m depth by SCUBA divers, from the shallow submarine vent named Campo 7 (Lat. 38°37’59”N–Long. 15°06’597”E) off Panarea Island (Eolian Islands, Italy) [1]. The characteristics of the thermal fluid emitted from the sampling site were: temperature 60C, pH 4.9 and conductivity 49.2 mS/cm. A preliminary characterization of strain APA, based on the partial 16S rRNA sequence analysis, revealed that strain APA was related to Geobacillus stearothermophilus strain mt-7 (94% similarity) [1]. However, the exhibited sequence similarity was below the 95% level, indicating that strain APA might represent a novel species of bacilli.
In this study the phenotypic and genotypic characteristics of strain APA have been screened in more details in order to clarify its taxonomic position. Biochemical characteristics were studied by test strips of API 20E, API 20NE and API50 CH (bioMérieux) systems, according to Maugeri et al. [2]. The complete 16S rRNA gene sequence was analysed in this study via polymerase chain reaction (PCR) by using the universal bacterial primers 27f and 1525r. The PCR product was purified with the Wizard Genomic DNA Purification kit (Promega, Madison, WI, USA), according to the manufacturer’s directions, and sequenced by BIOFAB (Rome, Italy). In order to obtain sequences with the most significant alignment, a nucleotide BLAST search (http://blast.ncbi.nlm.nih.gov/Blast.cgi) was performed. The sequence assigned to strain APA and selected reference sequences were used for the construction of the phylogenetic tree generated by the neighbour joining method, using the Kimura 2-parameter model in MEGA 4.0 (Biodesign Institute, AZ, USA). Distance matrix tree was generated by the neighbour-joining (NJ) method with the Felsenstein correction as implemented in the PAUP 4.0B software (Sinauer, Sunderland, MA, USA). The NJ calculation was subjected to bootstrap analysis (1000 replicates).
Biopolymer production and characterization
Strain APA was grown in 200 ml of a minimal medium containing yeast extract (0.01% w/v) in seawater (SWY), and sucrose (1% w/v) under its optimal growth conditions i.e. temperature 45°C, pH 7 and NaCl 3% w/v [1]. The biopolymer was recovered from the culture medium by centrifugation (9,800 × g, 20 min at 4°C) after 48 h, at the stationary phase of growth. The supernatant was treated with an equal volume of cold absolute ethanol added dropwise under stirring in an ice bath. The alcoholic solution was kept at -20°C overnight and then centrifuged at 13,000 × g for 30 min. The pellet was washed twice with ethanol, dissolved in hot water (80-90°C), dialysed against distilled water, lyophilised and weighed. The crude biopolymer was tested for carbohydrate content, according to the phenol-sulphuric acid method using glucose as standard [24]. Total protein content was estimated according to Bradford [25]. Uronic acid content was determined according to Blumenkrantz and Asboe-Hansen [26]. The molecular weight was determined by means of gel filtration chromatography on a Sepharose CL-6B column (GE Healthcare Bio-Sciences). The elution was performed with H2O at a flow rate of 0.3 ml/min. The fractions were analyzed by thin-layer chromatography (Silica GelF60, Merck), spraying plates with a ninhydrin solution (0.1% acetone). The calibration curve was derived by eluting a mixture of dextran standards, namely (Fluka) 10,000 Da, 100,000 Da, 670,000 Da and 1,000,000 Da, under the same condition used for the biopolymer.
UV-Vis Absorption spectra (1 mg/ml in water, at pH 7.19) were recorded with a Beckman Coulter® DU®730 Life Science Spectrophotometer. Nuclear Magnetic Risonance (NMR) spectra of biopolymer samples (5 mg/ml in D2O) were registered by means of a Bruker AMX-300 MHz (1H-NMR) instrument at 30°C. Fourier Transform InfraRed (FT-IR) spectra were recorded by a Perkin- Elmer Spectrum 100 single-beam spectrophotometer. A disc with the powdered sample was placed under the beam and the spectra were collected after 16 scans under nitrogen.
Anti-herpes virus activity
Isolation of human peripheral blood mononuclear cells (PBMC): blood donors (Centro Trasfusionale, Policlinico Universitario “G. Martino”, Messina, Italy) after centrifugation over Ficoll-Hypaque gradient. PBMC were then washed three times in RPMI 1640 medium (Sigma, Italy), and cultured at 37°C under 5% CO2 in 24-well plates at a concentration of 2 × 106 cells/ml per well. The RPMI 1640 medium was supplemented with 50μg/ml gentamicin and 5% fetal calf serum (FCS, Sigma, Italy).
Cytotoxicity test: The cytotoxicity test was performed on both PBMC and WISH (Wistar Institute Susan Hayflic) cells by using the colorimetric assay according to Mosmann [27]. In this assay, the pale yellow substrate tetrazolium salt 3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide (MTT) is cleaved by active mitochondria to produce a dark blue formazan product. Briefly, PBMC or WISH cells were seeded onto 96-well culture plates at a number of 104 per well. After 4 h of incubation to allow cell seeding, the biopolymer from APA (1mg/ml in apyrogenic sterile water) was added into each well up to the following concentrations: 200, 300, 400, 500, 600 and 700 μg/ml. Plates were incubated at 37°C under 5% of CO2 for 24 h. The medium was then discarded and the MTT reagent added. Plates were re-incubated at 37°C for 3h to allow for formazan formation, and read with a MicroELISA reader at a wavelength of 570 nm. The cytotoxicity percentage was calculated as follows: 1_ [(experiment OD -lyses control OD)/(cell control OD - lyses control OD)] × 100. Biopolymer’s concentrations that resulted not cytotoxic were further analysed.
Limulus test: Culture media and reagents tested for the presence of endotoxin by E-Toxate kit (Sigma, Milan) were found to contain ≤ 10 pg of endotoxin per ml.
HSV-2 infection: PBMC and WISH cells were seeded onto 24- well culture plates at a density of 2 × 106 cells per well. Both cell lines were treated with the APA biopolymer at different concentrations (200, 300 and 400 μg/ml), infected with HSV-2 at a multiplicity of infection (MOI) of 0.1, and incubated for further 24 h at 37°C under 5% CO2. The plates were frozen and thawed three times in order to release the intracellular virus. Cell lysates and supernatants were kept at –80°C until virus titration.
HSV-type 2 strain G was used in this study. HSV-2 infection and viral titre were assessed on WISH cells and expressed as plaque forming unit (PFU) per ml as previously described [11].
4.3.5. Cytokine evaluations: Th1-type cytokines interferon (IFN), including IFN-α and IFN-γ, tumour necrosis factor (TFN-α), interleukin (IL-12 and IL-18) and Th2-type cytokines (IL-4 and IL- 10) were determined by an ELISA immunoenzymatic method (DRG diagnostic, Milan, Italy). The detection limits were 3.1, 0.13, 2.1, 9.2, 0.06, 0.1 and 0.05 pg/ml, respectively.
Statistical analysis: Data, expressed as the means of three experiments ± standard deviation (SD), were analysed by one-way analysis of variance (ANOVA) and the Student-Newman-Keuls test and differences were considered statistically significant for p-value <0.05.
Results and Discussion
Characterization of Bacillus horneckiae strain APA
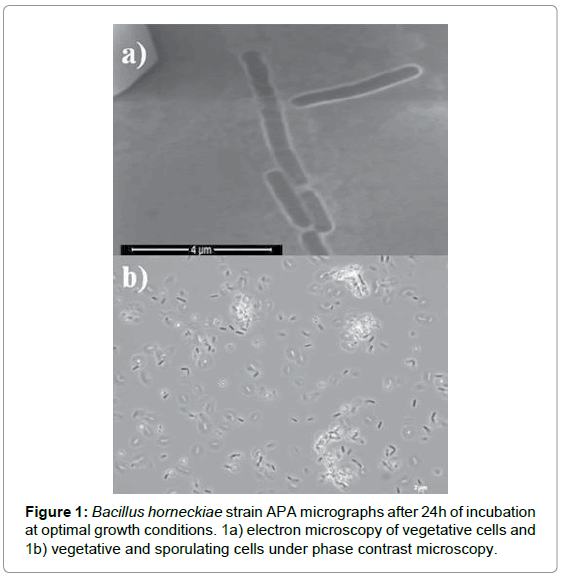
Cells of strain APA were Gram-positive, endospore-forming, and rod-shaped (Figure 1). This strain grew aerobically in Bacto Marine Broth 2216 (Difco) from 25°C to 70°C, and its optimal temperature occurred at 45°C. Colonies formed after 24 h of growth on Bacto Marine Agar 2216 (Difco) at 45°C are yellowish, plain, 3 mm in diameter, and possess regular edges. Phylogenetic analysis based on the full 16S rRNA gene sequence showed that Bacillus strain APA was strictly related (similarity 99%) to Bacillus horneckiae DSM 23495T, an extremophilic ultraviolet radiation resistant strain isolated from a clean room of the Kennedy Space Center where the Phoenix spacecraft was assembled [28]. Figure 2 shows the phylogenetic tree based on the almost complete 16S rRNA sequence of strain APA and related species of Bacillus. The 16S rRNA gene sequence of strain APA was deposited at GenBank/EMBL/DDBJ with accession number KC753180. Cells of strain APA was also deposited at the Deutsche Sammlung von Mikroorganismen und Zellkulturen (DSMZ), Braunschweig, Germany with number DSM 28882.
Figure 2: Phylogenetic neighbour-joining tree obtained with the 16S rRNA sequence of the strain APA and related Bacillus spp. The neighbourjoining tree was constructed from a data set using the Kimura 2 parameters model in MEGA 4.0 (Biodesign Institute, AZ, USA). The bootstrap tree was constructed using 1000 replicates.
Main phenotypic characteristics of strain APA are reported in Table 1, in comparison with those of the closest genotypically related B. horneckiae DSM 23495T. Temperature growth range and optimal temperature value of strain APA greatly differed from B. horneckiae DSM 23495T and indicated that the new isolate was more thermotolerant than its closest phylogenetically related strain. Therefore, despite the high genetic similarity with B. horneckiae DSM 23495T, the strain APA showed phenotypic characteristics that supported the hypothesis that it could be considered as a novel strain of this species. This finding confirm that the hydrothermal system off Panarea Island represents a source of novel thermophilic and thermotolerant bacilli, which play an active role in the biogeochemical cycles at shallow hydrothermal vents [2,6]. As inhabitant of an extreme environment, the novel thermotolerant B. horneckiae strain possesses a great physiological versatility that allows it to survive to the severe physico-chemical conditions.
| Bacillus horneckiae | ||
|---|---|---|
| Strain APA DSM 28882 | DSM 23495T (a) | |
| Temperature range (°C) | 25-70 | Apr-32 |
| Optimum temperature (°C) | 45 | 30 |
| pH range | 9-May | 7-10.8 |
| NaCl range | 10-Feb | 0-10 |
| β-galactosidase | + | - |
| Oxidase | + | - |
| Assimilation of: | ||
| Adipic acid | - | + |
| L-arabinose | + | - |
| D-glucose | + | - |
| D-mannitol | + | - |
| D-mannose | + | - |
| N-acetyl-D-glucosamine | + | - |
| Hydrolysis of: | ||
| Aesculin | + | - |
| Gelatin | + | +/w |
| Acid production from: | ||
| D-fructose | + | - |
| Amygdalin | + | - |
| Arbutin | + | - |
| Salicin | + | - |
| D-cellobiose | + | - |
| D-sucrose | + | - |
| D-trehalose | + | - |
aData from Vaishampayan et al. [28].
Table 1: Differential characteristics of Bacillus horneckiae strain APA DSM 28882 and its closest related strain Bacillus horneckiae DSM 23495T.
Biopolymer production and characterisation
After 48 h of incubation in its optimal growth conditions, strain APA yielded 85 mg/l of crude extracellular polymer that was chemically and spectrally characterised.
Chemical analysis showed that the biopolymer contained low percentages of carbohydrates (14%), proteins (1.8%), nucleic acids (<1%) and uronic acids (0.3%). Its relative molecular weight was found to be between 890 and 1000 kDa as determined by gel permeation chromatography.
With regard to the spectral features, the UV absorption spectrum of the isolated exopolymer exhibited a maximum absorption at wavelength 216 nm typical of PGAs absorption in aqueous solutions.
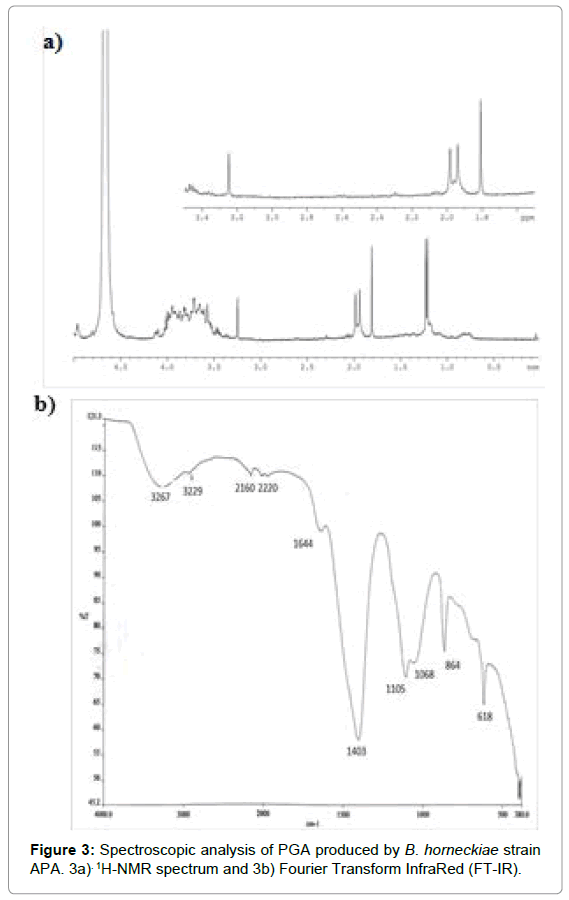
The 1H-NMR spectrum in D2O of the biopolymer showed the presence of signals in the aliphatic carbons region at δ 3.25, at δ 1.98 and δ 1.94, and at δ 1.81 that, according also to other previous studies [29], were attributable to α-CH, β-CH2 and γ-CH2 protons of PGA polymers, respectively (Figure 3a).
The IR spectrum of the isolated product showed some key features of PGAs [29] like for example a signal at 3267 cm-1 attributable to the OH (α-carboxylic group) stretching vibrations, an N-H bending signal at 1644 cm-1, a C=O stretching signal at 1403 cm-1 and a band at 1105 cm-1 relative to the C-N stretching (Figure 3b).
Overall, the chemical and spectral data suggested a γ-polyglutamic structure for the isolated exopolymer that was named γ-PGA-APA. Although other bacilli -e.g., Bacillus licheniformis and Bacillus subtilishave been reported to be PGA producers, this was the first evidence of the production and characterisation of a γ-PGA yielded by a marine strain of B. horneckiae.
Cytotoxicity and antiviral effects of γ-PGA-APA
The γ-PGA-APA showed a dose-dependent cytotoxicity towards PBMC (Table 2), comparable to that obtained on WISH cells (data not shown). When added in concentrations from 200 to 400 μg/ml the biopolymer was not cytotoxic and therefore this concentration range was used for the subsequent assays.
| γ-PGA-APA (μg/ml) | Citotoxicity (%) |
|---|---|
| 700 | 58 ± 10.0 |
| 600 | 25 ± 4.3 |
| 500 | 9 ± 2.1 |
| 400 | 0 |
| 300 | 0 |
| 200 | 0 |
Table 2: Cytotoxicity percentage on PBMC cells 48-h post γ-PGA-APA treatment. Values are the mean of three experiments ± SD.
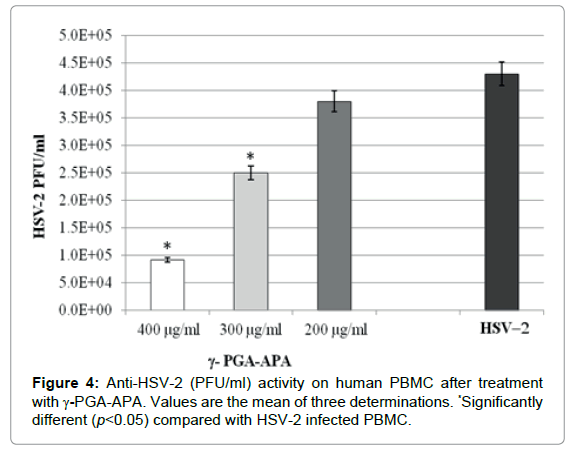
The potential antiviral properties of γ-PGA-APA were investigated by assaying its inhibitory effect on replication of herpes simplex virus type 2 (HSV-2). As shown in Figure 4, γ-PGA-APA antiviral effect was concentration dependent: indeed, when added at concentrations of 400 μg/ml, it significantly hindered HSV-2 replication in PBMC cells (p<0.05); on the other hand no significant antiviral effect was observed at lower concentration (300 and 200 μg/ml), when the PFU value was comparable to that observed in PMBC not treated with the biopolymer. Moreover, γ-PGA-APA treatment on WISH cells showed no significant inhibition of HSV-2 replication at any tested concentration (data not shown).
The antiviral effect observed on PBMC, but not on WISH cells, prompted us to investigate whether the antiviral activity of γ-PGA-APA could be related to the immune response involved in the control of viral replication. Therefore, the production of Th1 cytokines (IFN-γ, IFN-α, TFN-α, IL-12 and IL-18), that stimulate the immune cell responses, was evaluated. High levels of Th1 cytokines were produced by PBMC at 48 hours post γ-PGA-APA treatment, under the effect of HSV-2 infection in the supernatants of γ-PGA-APA (Table 3). It is noteworthy that 48 h incubation with the APA polymer, added at a concentration of 400 μg/ml, induced the production of higher amounts of all the tested cytokines. PBMC treated with γ-PGA-APA and infected with HSV-2 determined a down-regulation in the cytokine production in all the experimental conditions (p <0.05) (Table 3). Cytokine production was not affected by the addition of 200 μg/ml of γ-PGA-APA, whereas it was markedly reduced at 300 μg/ml, showing a dose-dependent activity (data not shown).
| Inducer | IFN-γ | IFN-α | TFN-α | IL-12 | IL-18 | |
|---|---|---|---|---|---|---|
| None | < 0.06 | < 3.1 | < 0.13 | < 2.1 | < 9.2 | |
| HSV-2 | < 0.08 | < 3.6 | < 0.12 | < 2.2 | < 8.9 | |
| γ-PGA-APA 400 μg/ml | 142 ± 1.8* | 186 ± 9.4* | 1478 ± 71.0* | 919 ± 84.0* | 57 ± 6.9* | |
| γ-PGA-APA 400 μg/ml + HSV-2 | 102 ± 2.8° | 106 ± 5.4° | 980 ± 89.0° | 481 ± 52.2° | 37 ± 5.9° | |
| *Significantly different (p< 0.05) compared with untreated control. | ||||||
| °Significantly different (p< 0.05) compared with γ-PGA-APA treated PBMC. | ||||||
Table 3: Production of Th1 cytokines (pg/ml) at 48 h post γ-PGA-APA treatment by PBMC and under the effect of HSV-2 infection (in grey).Values are the mean of four experiments ± SD.
In order to clarify the mechanism involved in the observed downregulation, the production of Th2-type anti-inflammatory cytokines, IL-4 and IL-10, was investigated in PBMC supernatants. In Table 4 are reported the results of IL-4 and IL-10 production after 48 hours of γ-PGA-APA treatment of PBMC, infected or not with HSV-2.
| Inducer | IL-4 | IL-10 |
|---|---|---|
| None | < 0.1 | < 0.5 |
| HSV-2 | 41 ± 6.1 | 35 ± 4.8 |
| γ-PGA-APA 400 μg/ml | < 0.1 | < 0.5 |
| γ-PGA-APA 400 μg/ml+ HSV-2 | 14 ± 4.1* | 7 ± 3.9* |
*Significantly different (p<0.05) compared with HSV-2 infected PBMC.
Table 4: Production of Th2 cytokines (IL-4 and IL-10) (pg/ml) at 48 h post γ-PGAAPA treatment by PBMC and under the effect of HSV-2 infection (in grey). Values are expressed as the mean of four experiments ± SD.
The γ-PGA-APA treatment did not trigger uninfected PBMC to release IL-4 or IL-10, which are strong hallmarks of Th2 response. Conversely, the HSV-2 infection of untreated PBMC induced the production of substantial amounts of IL-4 and IL-10. The HSV- 2 infection of PBMC treated with γ-PGA-APA resulted in the production of lower amounts of IL-4 and IL-10, in comparison with the levels induced by HSV-2 alone (14 ± 4.1 vs 41 ± 6.1 and 7 ± 3.9 vs 35 ± 4.8, respectively) (p<0.05), indicating that the γ-PGA-APA partially counteracted the effect of the virus by down-regulating the production of the Th2 cytokines.
In previous studies, we demonstrated that HSV-2 down-regulates the production of Th1 cytokines in PBMC, which represents the first line of defense against viral infections or the so-called “proinflammatory host response”, and stimulates Th2 cytokines in order to evade the immune response of the host [9-11].
The ability of γ-PGA-APA to inhibit HVS-2 replication in PBMC (expressed as logarithm) was lower (0.67) than that of the previously reported EPS produced by B. licheniformis strain B3-15 (0.82) [1], but it was higher than that of the EPSs produced by Geobacillus thermodenitrificans strain B3-72 (0.49) [2] and similar to that reported by B. licheniformis strain T14 (0.63) [11].
The antiviral effect of γ- PGA-APA on PBMC was related to the production of all tested Th1 cytokines. The γ-PGA-APA treatments induced PBMC to produce high amounts of Th1 cytokines (IFN-γ, IFN-α, TFN-α, IL-12, and IL-18), restricting viral infection via the induction of an antiviral state in neighbouring cells (i.e. IFNs) or destroying virus-infected cells (i.e. TNF-α and IL-18). Our findings suggest that γ-PGA-APA can activate host innate immune responses to display antiviral activity by the induction of both type I (α) and type II (γ) IFNs, similarly to the results previously reported by Lee et al. [30].
Moreover, HSV-2 infection of PBMC induced substantial amounts of Th2 cytokines IL-4 and IL-10, both strong hallmarks of Th2 response. PBMC treated with γ-PGA-APA counteracted the virus, reducing significantly the release of IL-4 and IL-10 cytokines in PBMC supernatants.
This is the first report on a γ-PGA produced by a marine thermotolerant B. horneckiae strain exhibiting antiviral and immunomodulator activities. As stimulator of cell-mediated immunity responses, this biopolymer could be potentially used in the treatment of immunocompromised hosts, as well as in patients affected by herpes virus infections.
References
- Gugliandolo C, Lentini V, Spanò A, Maugeri TL (2012) New bacilli from shallow hydrothermal vents of Panarea Island (Italy) and their biotechnological potential. J ApplMicrobiol112: 1102-1112.
- Maugeri TL, Gugliandolo C, Caccamo D, Stackebrandt E (2001) Apolyphasic taxonomic study of thermophilic bacilli from shallow, marine vents. Syst Appl Microbiol 24: 572-587.
- Maugeri TL, Bianconi G, Canganella F, Danovaro R, Gugliandolo C, et al. (2010) Shallow hydrothermal vents in the southern Tyrrhenian Sea. ChemEcol26: 285-298.
- Lentini V, Gugliandolo C, Maugeri TL (2007) Identification of enzyme-producing thermophilic bacilli isolated from marine vents of Eolian Islands (Italy). Ann Microbiol 57: 355-361.
- Maugeri TL, Gugliandolo C, Caccamo D, Panico A, Lama L, et al. (2002) AhalophilicthermotolerantBacillus isolated from a marine hot spring able to produce a new exopolysaccharide. BiotechnolLett 24: 515-519.
- Maugeri TL, Gugliandolo C, Caccamo D, Stackebrandt E (2002) Three novel halotolerant and thermophilicGeobacillus strains from shallow marine vents. Syst Appl Microbiol 25: 450-455.
- Nicolaus B, Panico A, Manca MC, Lama L, Gambacorta A, et al. (2000)AthermophilicBacillus isolated from an Eolian shallow hydrothermal vent, able to produce exopolysaccharides. Syst Appl Microbiol 23: 426-432.
- Spanò A, Gugliandolo C, Lentini V, Maugeri TL, Anzelmo G, et al. (2013) A novel EPS-producing strain of Bacillus licheniformis isolated from a shallow vent off Panarea Island (Italy)CurrMicrobiol67: 21-29.
- Arena A, Maugeri TL, Pavone B, Iannello D, Gugliandolo C, et al. (2006) Antiviral and immunomodulatory effect of a novel exopolysaccharide from a marine thermotolerantBacillus licheniformis. Int Immunopharmacol 6: 8-13.
- Arena A, Gugliandolo C, Stassi G, Pavone B, Iannello D, et al. (2009) Anexopolysaccharide produced by Geobacillusthermodenitrificans strain B3-72: antiviral activity on immunocompetent cells. ImmunolLett 123: 132-137.
- Gugliandolo C, Spanò A, Lentini V, Arena A, Maugeri TL (2014) Antiviral and immunomodulatory effects of a novel bacterial exopolysaccharide of shallow marine vent origin. J ApplMicrobiol 116: 1028-1034.
- Laurienzo P (2010) Marine polysaccharides in pharmaceutical applications: an overview. Mar Drugs8: 2435-2465.
- Poli A, Anzelmo G, Nicolaus B (2010) Bacterial exopolysaccharides from extreme habitat: production, characterization and biological activities. Mar Drugs 8: 1779-1802.
- Bajaj I, Singhal R (2011) Poly (glutamic acid) – An emerging biopolymer of commercial interest.BioresourTechnol 102: 5551-5561.
- Ashiuchi M (2013) Microbial production and chemical transformation of poly-γ-glutamate. MicrobBiotechnol 6: 664-674.
- Ashiuchi M, Misono H (2001) Isolation of Bacillus subtilis(chungkookjang), a poly-γ-glutamate producer with high genetic competence. ApplMicrobiolBiotechnol 57: 764-769.
- Du GC, Yang G, Qu YB, Chen J, Lun SY (2005) Effects of glycerol on the production ofpoly (γ-glutamic acid) by Bacillus licheniformis. Process Biochem 40: 2143-2147.
- Kambourova M, Tangney M, Priest FG (2001) Regulation of polyglutamic acid synthesis by glutamate in Bacillus licheniformis and Bacillus subtilis.Appl Environ Microbiol 67:1004-1007.
- Mingfeng C, Weitao G, Li L, Cunjiang S, Hui X, et al. (2011) Glutamic acid independent production of poly-γ-glutamic acid by Bacillus amyloliquefaciensLL3 and cloning of pgsBCAgenes. BioresourTechnol 102: 4251-4257.
- Wu Q, Xu H, Liang JF, Yao J (2008) Contribution of glycerol on production of poly (gamma-glutamic acid) in Bacillus subtilisNX-2. ApplBiochem Biotech 160: 386-392.
- Xuetuan W,Zhixia J, Shouwen C (2010) Isolation of halotolerantBacillus licheniformis WX-02 and regulatory effects of sodium chloride on yield and molecular sizes of poly-γ-glutamic acid. ApplBiochemBiotechnol 160: 1332-1340.
- Ashiuchi M, Misono H (2002) Biochemistry and molecular genetics of poly-γ-glutamate. ApplMicrobiolBiotechnol 59: 9-14.
- Shih IL, Van YT (2001) The production of poly (γ-glutamic acid) from microorganisms and its various applications. BioresourTechnol 79: 207-225.
- Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 3: 350-356.
- Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Chem 72: 248-254.
- Blumenkrantz N, Asboe-Hansen G (1973) New method for quantitative determination of uronic acids. Anal Chem 54: 484-489.
- Mosmann T (1989) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65: 55-63.
- Vaishampayan P, Probst A, Krishnamurthi S, Ghosh S, Osman S et al. (2010) Bacillus horneckiae sp. nov., isolated from a spacecraft-assembly clean room. Int J Syst Evol Microbiol 60: 1031-1037.
- Ho Y, Tzou DLM, Chu FI (2006) Solid-state NMR studies of the molecular structure of Taxol. Magn Reson Chem 44: 581-585.
- Lee W, Lee S-H, Ahn DG, Cho H, Sung M-H, et al. (2013) The antiviral activity of poly-γ-glutamic acid, a polypeptide secreted by Bacillus sp., through induction of CD14-dependent type I interferon responses. Biomaterials 34: 9700-9708.
Relevant Topics
- Algal Blooms
- Blue Carbon Sequestration
- Brackish Water
- Catfish
- Coral Bleaching
- Coral Reefs
- Deep Sea Fish
- Deep Sea Mining
- Ichthyoplankton
- Mangrove Ecosystem
- Marine Engineering
- Marine Fisheries
- Marine Mammal Research
- Marine Microbiome Analysis
- Marine Pollution
- Marine Reptiles
- Marine Science
- Ocean Currents
- Photoendosymbiosis
- Reef Biology
- Sea Food
- Sea Grass
- Sea Transportation
- Seaweed
Recommended Journals
Article Tools
Article Usage
- Total views: 11384
- [From(publication date):
December-2015 - Aug 17, 2025] - Breakdown by view type
- HTML page views : 10409
- PDF downloads : 975