Research Article Open Access
Production of Biosurfactant and their Role in Bioremediation
Uma Aulwar1* and Awasthi RS21Department of Botany, S P College, Tilak Road, Pune, India.
2Shivaji Mahavidyalay, Renapur, Latur, India.
- *Corresponding Author:
- Aulwar U
Department of Botany
S P College, Tilak Road
Pune, India
Tel: 9423274941
E-mail: aulwar_uma@rediffmail.com
Received Date: December 03, 2015; Accepted Date: August 03, 2016; Published Date: August 03, 2016
Citation: Aulwar U, Awasthi RS (2016) Production of Biosurfactant and their Role in Bioremediation. J Ecosys Ecograph 6:202. doi:10.4172/2157-7625.1000202
Copyright: © 2016 Aulwar U, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Ecosystem & Ecography
Abstract
The treatment of environmental pollution by microorganisms is a promising technology. Various genetic approaches have been developed and used to optimize the enzymes, metabolic pathways and organisms relevant for biodegradation. Naturally occurring microbial activities are and have been the starting point for all biotechnological applications. It is therefore necessary to isolate bacterial strains with novel metabolic capabilities and to biochemically and genetically elucidate degradative pathways. Microbial biosurfactants are extracellular compounds produced by microbes such as bacteria, fungi and actinomycetes when grown in culture medium containing hydrophobic/ hydrophilic substrates. Biosurfactants are surface active molecules having hydrophilic and hydrophobic moieties as their constituents which allow them to interact at interfaces and reduce the surface tension. As compared to their chemical/synthetic counterparts, biosurfactants have unique properties such as high activity, less/or no toxicity, biodegradability and ease of production from renewable resources. There is a useful diversity of biosurfactants due to the wide variety of producer microorganisms. Also their chemical compositions and functional properties can be strongly influenced by environmental conditions.
Keywords
Biosurfactants; Bioremediation; Oil pollution; Oildegrading micro organisms; Bioavailability; Hydrocarbons
Introduction
Increasing public awareness of environmental pollution influences the search and development of technologies that help in clean up of organic and inorganic contaminants such as hydrocarbons and metals. Petroleum hydrocarbons are an essential raw material in our current society, but they also constitute a major environmental pollutant that is very difficult to be bioremediated. Crude oils have very low water solubility, high adsorption onto soil matrix and present limited rate of mass transfer for biodegradation.
Oil-contaminated soil is especially difficult for bioremediation since oil excess forms droplets or films on soil particles, which is a powerful barrier against microbial degradation. Bioavailability of contaminants in soil to the metabolizing organisms is influenced by factors such as desorption, diffusion and dissolution. Biosurfactants are produced to decrease the tension at the hydrocarbon-water interface aiming to pseudo-solubilize the hydrocarbons, thus increasing mobility, bioavailability and consequent biodegradation.
Several biosurfactant are produced by a diversity of microorganisms in order to survive in an oil rich environment, and this adaptation process selected for surfactants with highly adaptable phycal-chemical properties. Biosurfactants are, therefore, very suitable for applications in the oil industry and this is reflected in the market, where the large majority of biosurfactants produced are in petroleum-related applications.
Surfactant molecules act by reducing interfacial energy (interfacial tension) and surface tension through the formation of an ordered molecular film at the interface [1].
Biosurfactants can improve the bioavailability of hydrocarbons to the microbial cells by increasing the area at the aqueous-hydrocarbon interface. This increases the rate of hydrocarbon dissolution and their utilization by microorganisms [2].
The main issue which directly influences the efficiency of biological treatment is the “bioavailability” of the pollutant. Possible sorption of molecules into the soil matrix, formation of non-aqueous phases, interactions with organic matter, biotransformation, and contaminant aging these naturally occurring processes often result in limited bioavailability, thus decreasing the efficiency of bioremediation. The most common intended role of biosurfactants is therefore enhancing the distribution of contaminants into the aqueous phase and increasing their bioavailability.
The success of biosurfactant production not only depends on right choice of producer microorganism but also on selection of low cost waste material as substrate.
Frying oil, molasses, whey, starch rich wastes, agricultural and industrial wastes are reported as substrate for biosurfactant production.
The Deproteinised Juice (DPJ) is rich in water soluble plant nutrients. It contains water soluble carbohydrates, minerals, free amino acids etc. DPJ supports to the growth of Rhizoctonia and also used for alcohol production by yeast. Screening of DPJ of plant leaves for production of biosurfactant by different fungi was carried out. DPJ was found supportive for growth and biosurfactant production by Rhizopusnigricans. Preliminary chemical analysis of biosurfactant was carried out. It was a heteropolymer consisting of protein, carbohydrate and lipid.
The objective of the study was to determine the biosurfactant production and its potential use in bioremediation processes, especially in the petroleum industry.
A promising method that can improve bioremediation effectiveness of hydrocarbon contaminated environments is the use of biosurfactants. They can enhance hydrocarbon bioremediation by two mechanisms. The first includes the increase of substrate bioavailability for microorganisms, while the other involves interaction with the cell surface which increases the hydrophobicity of the surface allowing hydrophobic substrates to associate more easily with bacterial cells.
In bioremediation applications biosurfactants play main role in promoting the accessibility/bioavailability of the pollutants to the microbes involved in biodegradation process. Application of biosurfactants in oil biodegradation experiments showed 20-30% increase in total degradation. The biosurfactant solution applied to the oil field contacts the small pockets of trapped oil and reduces the interfacial tension, mobilizes the trapped oil and thus the released oil can be recovered more efficiently.
The success of bioremediation is governed by three important factors: availability of microbes, accessibility of contaminants and a conducive environment. The efficiency of the bioremediation is dependent upon the microbial ability to degrade these complex mixtures and their rate-limiting kinetics. The intensity of biodegradation is influenced by several factors, such as nutrients, oxygen, pH value, composition, concentration, and bioavailability of the contaminants, chemical and physical characteristics, and the pollution history of the contaminated environment.
Materials and Methods
Microorganisms
Hydrocarbon-utilizing fungi were isolate from different soil samples and were identified as Aspergillus niger, Aspergillus flavus, Rhizopusni gricans. To confirm their ability to grow on hydrocarbons, a few filaments from fungal cultures were transferred in to 100 ml Erlenmeyer flasks containing 20 ml liquid medium supplemented with 2% hexadecane or n-paraffins and kerosene, respectively and cultivated at 28°C and 130 rpm. Fungal cultures were maintained on potato dextrose agar slants at 4°C.
Medium
Modified GN medium-(glucose-10 gm, KN03-2.5 gm, KH2PO4-1 gm, MgSO4-0.5 gm in 1000 ml distilled water).
DPJ (Deproteinised leaf juice)
Deproteinised leaf juice was prepared as method described by Davys and Pirie and Davys et al. Eucalyptus (Eucalyptus sp. L.), Castor (Ricinuscommunis L.), Soyabean (Glycine max L.), and cauliflower were used.
Method of inoculation
A loopful of spore suspension was standardize to contain 20-30 spores per field (10 × 10) was inoculated in 50 ml fermentation medium.
Incubation
In 250 ml Erlenmeyer flask 50 ml sterilized fermentation medium was inoculated with fungal culture and incubated in static and shake condition for 7 days.
Assessment of growth
Growth was assessed in terms of dry weight at the end of incubation period. (Gravimetric method).
Biosurfactant production
Biosurfactant production was determined by measuring surface tension by capillary rise method as described in Physical Chemistry Laboratory Manual, 2003.
Screening and identification of biosurfactant-producer
Initially, the screening of biosurfactant production by the isolated strain was carried out by oil-displacement test according to the method described elsewhere [3]. Further, emulsification activity of the cell-free culture supernatant again paraffin oil was carried out as mentioned by Paraszkiewicz et al. [4]. To confirm the biosurfactant production from the above mentioned tests, surface tension of the culture samples were measured by DuNouy Tensiometer (Table 1).
| DPJ of plants | Dry weight Mg/100ml | Emulsion Index |
|---|---|---|
| Eucalyptus | 347 | 25.02 |
| Castor | 271 | 15.17 |
| Soybean | 282 | 16.68 |
| Cauliflower | 233 | 11.48 |
| Modified GN medium(control) | 330 | 21.23 |
Table 1: Growth and emulsification activity of Rhizopusnigricans in DPJ media.
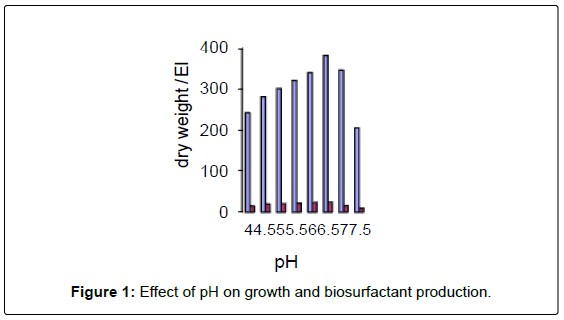
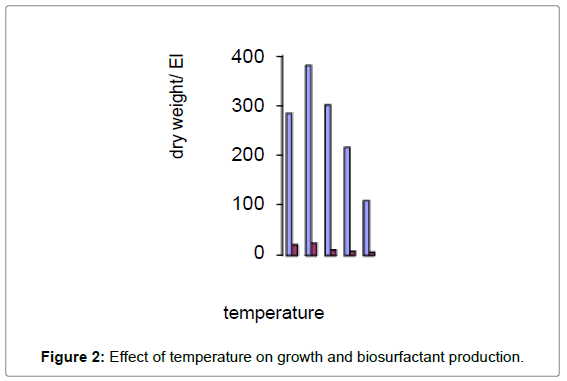
There are at least two ways in which biosurfactants are involved in bioremediation: increasing the surface area of hydrophobic waterinsoluble substrates and increasing the bioavailability of hydrophobic compounds. Biosurfactants are more effective than chemical surfactants in increasing the bioavailability of hydrophobic compounds (Figure 1). In addition, they are selective, environmentally friendly, and generally less stable than most synthetic surfactants. The high molecular weight bioemulsifier Alasan was recently shown to significantly increase the rate of biodegradation of several PAHs (Figure 2).
Extraction of biosurfactant
Biosurfactants released by Rhizopus nigricans at stationary phase mixed with 4 ml of water, 5 ml of chloroform and 10ml of methanol. After shaking for 5 minutes, the mixture was filtered through Whatman filter paper no.1 with slight suction. The residue on the filter paper was homogenised with 5 ml of chloroform and filtered once more. The residue on the filter paper was dissolved in water and freeze-dried.
For extraction of lipid, two filters were combined, 5 ml of water was added and the mixture was shaken thoroughly before separation of layer allowed. The upper methanol-water layer was removed by aspiration, retoevaporated until an aqueous solution was left and subsequently freeze-dried. The lower chloroform layer containing lipid fraction was retoevaporated resuspended in chloroform-methanol-water (4:3:1 vol/vol) and again retoevaporated until a cloudy aqueous emulsion was formed. This emulsion was freeze-dried and stored at -20°C. Different concentrations of the biosurfactant in the range of 10-80 mg/l were prepared from the stock solution of biosurfactant (0.1 g/l) in water. CMC was determined by measuring the surface tension of the dilutions at room temperature. The CMC was defined by plotting the surface tension as a function of biosurfactant concentration. These experiments were conducted in three independent experiments. The surface tension reaches its minimum value at a concentration of biosurfactant called critical micelle concentration (CMC) above which the molecules are associated, forming supermolecular structures.
Thin – layer chromatography (TLC)
For TLC, 20 mg of freeze-dried biosurfactant was dissolved in mixture of chloroform, methanol and water (1:1:0.3, v/v), while 2 mg of the chloroform fraction was dissolved in 1 ml of chloroform. TLC was carried out on Silica Gel 60 Plates (0.25 mm thick) with concentration zones (Merck) by use of mixture of chloroform, methanol and water (65:25:4 v/v) (Figure 3).
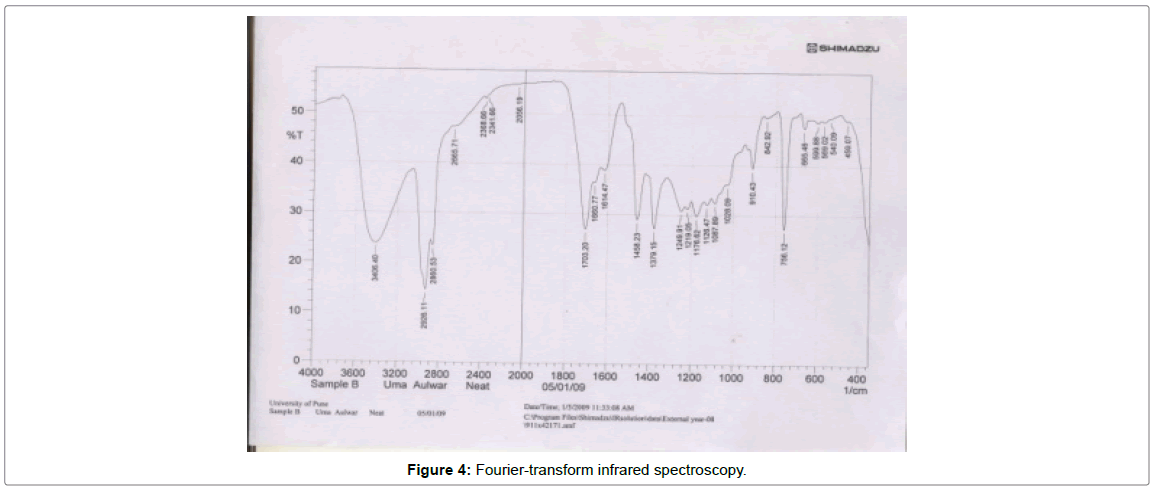
Fourier-transform infrared spectroscopy (FT-IR)
Fourier transform infrared spectroscopy (FT-IR) is an analytical technique used to identify organic (and in some cases inorganic) materials. This technique measures the absorption of various infrared light wavelengths by the material of interest. These infrared absorption bands identify specific molecular components and structures (Figure 4).
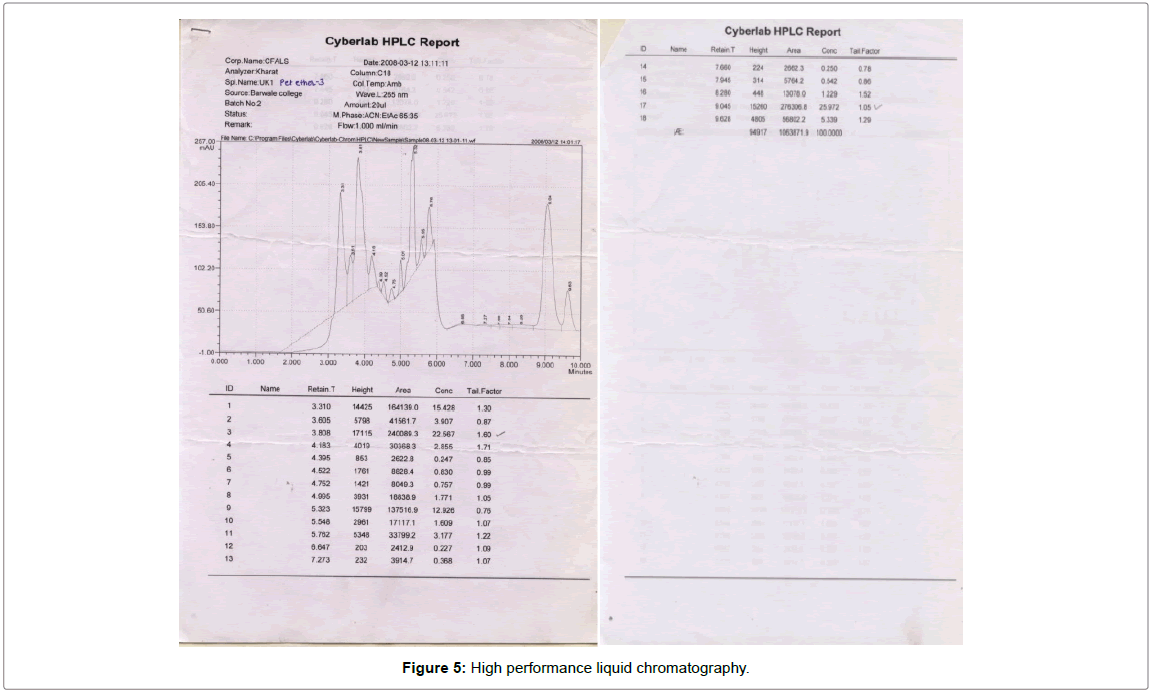
High performance liquid chromatography (HPLC)
C-18 reversed-phase high performance liquid chromatography (HPLC) of biosurfactant sample was carried out on a Perkin Elmer series 410 HPLC system. Sample was analysed on C-18 column maintained at ambient temperature. 20 ml amount was used. Mobile phase used was CAN: EtAc (65:35). Flow rate was 1 ml/min. chromatogram was detected on detector at 265 nm wavelength. Detailed report of HPLC analysis is shown in Figure 5.
Nuclear magnetic resonance spectroscopy (NMR)
After the hydrolysis of purified biosurfactants (about 100 mg) with HCl, the fatty acids were extracted with ether and purified by thin – layer chromatography on silica 60 with the solvent system hexaneether- acetic acid (90:10:1 [vol/vol/vol]). The fatty acids fraction (Rf=0.16) was recovered from the plate, and the sample (6.3 mg) was dissolved in 0.7 g of CDCl3. Tetramethylsilane was used as an internal standard. 1H-nuclear magnetic resonance (NMR) of this fatty acid was analysed by 400-MHz NMR (JNM-GSX-400; JEOL, Tokyo, Japan). The intact biosurfactant was also dissolved in dimethylsulfoxide- d6 and analysed by 1H-NMR.
Oil-degradation activity of biosurfactant
Natural oil and fats mostly consists of higher fatty acids and esters of glycerol. The basic hydrolysis of esters is known as saponification. The saponification value indicates about oil degradation. Oils viz., coconut, sunflower, castor bean, palm, soyabean and cottonseed oil were used. In 100 ml oil sample 10 ml crude biosurfactant solution was added and it was kept for 12 hours.
Analytical methods
Protein concentration in the isolated biosurfactant was determined by the Lowry method [5] using Bovine serum albumin as a standard. Carbohydrates were determined by the phenol-sulphuric method, using D-glucose as a standard [6]. The lipid composition of the crude biosurfactant was determined according to Manocha et al. [7].
Result and Discussion
Growth and optimization studies were carried out. On the basis of the growth and emulsification activity of all these isolates were measured in static condition and in suspension culture by using modified GN-medium. Amongst these isolates Aspergillusniger was found suitable for more biosurfactant production. Growth measured was 1.9 g/L and emulsion activity measured was 21.61. Aspergillusniger was selected for further studies. Optimum growth and maximum activity was measured in GN-medium.
Aspergillusniger when grown on modified GN- medium with pH 6.5, temperature 28°C, on orbital shaker at 120 rpm, on seven days of incubation produced 1.9 g/L of growth (dry weight basis) and 21.61 emulsification activity. In 1995 reported optimal yields of bioemulsifier are usually obtained when carbohydrate and vegetable oil are used as substrate. Liposan an extracellular emulsifier synthesized by Candidalipolytica was composed of 93% carbohydrate and 7% protein [8]. Other polymeric emulsifiers containing proteins carbohydrate and lipids were produced by Candidalipolytica when grown in babassu oil [9]. Preliminary analysis of the bioemulisifier produced by Aspergillusniger in modified GN-medium indicated that it was a hetropolymer, which consisted of 19% protein, 25% carbohydrate, and 5.5% lipid.
The chemical nature of biosurfactant produced was detected by HPLC, FTIR, IR, NMR and GC/MS. Joachim Vater et al. performed reverse phase HPLC to separate the isoforms of the lipopeptide biosurfactant complexes produced by Bacillus subtilis C-1. They found the lipopeptide products in 23 fractions.
The biosurfactant produced by Aspergillus niger was found heteropolymer made up of protein, carbohydrate and lipid. TLC revealed the presence of glycolipids. FT-IR, HPLC, NMR and GC/ MS analysis indicated presence of Pentadecanecarboxylic acid and D-xyletolpenta-acetate in biosurfactant. Further studies are required for detailed elucidation of structure and chemical nature of biosurfactant.
Oil degradation activity of biosurfactant was determined in terms of saponification value. Biosurfactant have capability of hydrolyzing the esters. Increase in saponification value is an indication of degradation of oil. Oil degradation activity of biosurfactant was checked against six different types of oils. Saponification activity was found most against soyabean and cottonseed oil. It indicates oil degradation by biosurfactant.
Masaaki Morikawa et al. [10] noted the similar report for biosurfactant produced by Arthrobacter sp. Strain MIS38.
A promising method that can improve bioremediation effectiveness of hydrocarbon contaminated environments is the use of biosurfactants. They can enhance hydrocarbon bioremediation by two mechanisms. The first includes the increase of substrate bioavailability for microorganisms, while the other involves interaction with the cell surface which increases the hydrophobicity of the surface allowing hydrophobic substrates to associate more easily with bacterial cells. By reducing surface and interfacial tensions, biosurfactants increase the surface areas of insoluble compounds leading to increased mobility and bioavailability of hydrocarbons [11-13]. In consequence, biosurfactants enhance biodegradation and removal of hydrocarbons. Addition of biosurfactants can be expected to enhance hydrocarbon biodegradation by mobilization, solubilization or emulsification.
Emulsification is a process that forms a liquid, known as an emulsion, containing very small droplets of fat or oil suspended in a fluid, usually water. The high molecular weight biosurfactants are efficient emulsifying agents. They are often applied as an additive to stimulate bioremediation and removal of oil substances from environments.
The effective microbiological method in bioremediation of hydrocarbon polluted sites is the use of biosurfactant producing microorganisms without necessarily characterizing the chemical structure of the surface active compounds [14]. The cell free culture broth containing the biosurfactants can be applied directly or by diluting it appropriately to the contaminated site. The other benefit of this approach is that the biosurfactants are very stable and effective in the culture medium that was used for their synthesis.
Bioremediation usually consists of the application of nitrogenous and phosphorous fertilizers, adjusting the pH and water content, if necessary, supplying air and often adding bacteria. The addition of emulsifiers is advantageous when bacterial growth is slow (e.g. at cold temperatures or in the presence of high concentrations of pollutants) or when the pollutants consist of compounds that are difficult to degrade. Bioemulsifiers can be applied as an additive to stimulate the bioremediation process; however, with advanced genetic technologies it is expected that the increase in bioemulsifier concentration during bioremediation would be achieved by the addition of microorganisms that overproduce bioemulsifiers [15].
Biosurfactants are produced by a variety of oil-degrading microorganisms. These biosurfactants can be of low molecular weight, acting by decreasing the oil–water interfacial tension, or high molecular weight and act as biodispersants by preventing coalescence of oil drops in water. The high molecular weight bioemulsifiers are heteropolysaccharides and the active components are lipids or proteins. The activity of bacterial biosurfactants in bioremediation stems from their ability to increase the surface area of hydrophobic waterinsoluble substrates and to increase the solubility and bioavailability of hydrocarbons [16,17]. They can be added to bioremediation processes as purified materials or in the form of bioemulsifier over producing micro-organisms. In either case, they can stimulate the growth of oildegrading bacteria and improve their ability to utilize hydrocarbons [18].
References
- Tan HM (2000) Biosurfactants and their roles in bioremediation. Environmental Biotechnology. pp: 1-12.
- Gerson DF, Zajic JE (1979) Microbial biosurfactants. ProcBiochem 14: 20-29.
- Ebrahimi A, Tashi N, Lotfalian S (2012) Isolation of biosurfactant producing bacteria from oily skin areas of small animals Jundishapour. J Microbiol 5: 401-404.
- Paraszkiewicz K, Kanwal A, Dugonski J (2002) Emulsifier production by steroid transforming filamentous fungus Curvularialunata. Growth and product characterization. J Biotechnol 92: 287-294.
- Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurment with Folin phenol reagent. J Bio Chem 193: 265-275.
- Hanson RS, Phillips JA (1981) Chemical composition. In: Gherhardt P (ed). Manual of methods for general bacteriology. American society of Microbiology, Washington, DC. pp: 328-364.
- Manocha MS, San-Blas G, Centeno S (1980) Lipid composition of Paracocodioidesbrasilienses: possible correlation with virulence of different strains. J Gen Micro 177: 147-154.
- Cirigliano MC, Carman GM (1984) Isolation of a bioemulsifier from Candida lipolytica. Appl Environ Microbiol 48: 747-750.
- Surubbo LA, de Luna JM, de Campos-Takaki GM (2006) Production and stability studies of the bioemulsifier obtained from a new strain Candida glabrataUCP 1002. Environmental Biotechnilogy 9: 1-7.
- Morikawa M, Daido H, Takao T, Murata S, Shimonishi Y, et al. (1993) Anew lipopeptidebiosurfactant produced by Arthrobacter sp. strain MIS38. J Bacteriol175: 6459-6466.
- Bognolo G (1999) Biosurfactantsasemulsifyng agents for hydrocarbons.ColloidsandSurfaces A: Physicochemical and Engineering Aspects152: 41-52.
- Cha DK (2000) The effect of biosurfactants on the fate and transport of nonpolar organic contaminants in porous media. Environ Eng20: 1-17.
- DesaiJD,BanatIM (1997) Microbial production of surfactants and their commercial potential. MicrobiolMolBiolRev61: 47-64.
- KaranthNG,DeoPG, VeenanadigNK (1999) Microbial production of bio surfactants and their importance. CurrSci77:116-123.
- KangSW, KimYB, ShinJD, KimEK (2010) Enhanced biodegradation of hydrocarbons in soil by microbial biosurfactant, sophorolipid. ApplBiochemBiotechnol160: 780-790.
- RodriguesLR, TeixeiraJA, Van der MeiHC, OliveiraR(2006)Physiochemical and functional characterization of a biosurfactant produced by Lactococcuslactis53. Colloids Surf B Biointerfaces49: 79-86.
- Abu-RuwaidaAS, Banat IM,Haditirto S,Khamis A (1991) Nutritional requirements and growth characteristics of a biosurfactant producing Rhodococcus bacterium. World JMicrobiolBiotechnol7:53-61.
- BanatIM (1993) The isolation of a thermophilicbiosurfactant producing Bacillus sp. BiotechnolLett15:5 91-594.
Relevant Topics
- Aquatic Ecosystems
- Biodiversity
- Conservation Biology
- Coral Reef Ecology
- Distribution Aggregation
- Ecology and Migration of Animal
- Ecosystem Service
- Ecosystem-Level Measuring
- Endangered Species
- Environmental Tourism
- Forest Biome
- Lake Circulation
- Leaf Morphology
- Marine Conservation
- Marine Ecosystems
- Phytoplankton Abundance
- Population Dyanamics
- Semiarid Ecosystem Soil Properties
- Spatial Distribution
- Species Composition
- Species Rarity
- Sustainability Dynamics
- Sustainable Forest Management
- Tropical Aquaculture
- Tropical Ecosystems
Recommended Journals
Article Tools
Article Usage
- Total views: 17019
- [From(publication date):
September-2016 - Aug 30, 2025] - Breakdown by view type
- HTML page views : 15456
- PDF downloads : 1563