Case Report Open Access
Renal Failure and Hypocalcaemia Secondary to Cabazitaxel Treatment for Prostate Cancer: A Rare but Potentially Lethal Side Effect
Farrouq Mahmood1*, Mazan Matar2 and Andrew Davis3
1Specialty Doctor in Oncology at North Devon District Hospital, Barnstaple, UK
2Core Medical Trainee at North Devon District Hospital, Barnstaple, UK
3Consultant in Gastroenterology at North Devon District Hospital, Barnstaple, UK
- Corresponding Author:
- Farrouq Mahmood
Specialist Doctor in Oncology
North Devon District Hospital, UK
Tel: 07825290631
E-mail: farrouqmahmood@doctors.org.uk
Received date: January 15, 2014; Accepted date: May 30, 2014; Published date: June 06, 2014
Citation: Mahmood F, Matar M, Davis A (2014) Renal Failure and Hypocalcaemia Secondary to Cabazitaxel Treatment for Prostate Cancer: A Rare but Potentially Lethal Side Effect. J Clin Diagn Res 2:106. doi:10.4172/2376-0311.1000106
Copyright: © 2014 Mahmood F, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at JBR Journal of Clinical Diagnosis and Research
Introduction
Cabazitaxel is a Toxoid derivative that has been licensed for treatment of hormone refractory prostate cancer. It is used in combination with Prednisolone following Docetaxel treatment. Renal failure secondary to Cabazitaxel treatment is very rare, but there have been some reports in literature of patients developing nephrotoxicity. In most cases it was part of a multifactorial cause and occurred with sepsis, dehydration or obstructive uropathy. In some cases, renal failure occurred for no obvious reason. The following case demonstrates a patient developing renal failure and hypocalcaemia following treatment with Cabazitaxel.
Case Report
An 80 year old male, who had received chemotherapy treatment for hormone refractory metastatic prostate cancer two weeks prior to admission, presented to the medical assessment unit with leg cramps, generalised weakness and poor oral intake. He also complained of diarrhoea which occurred a few days after he received chemotherapy treatment. He had no fever but admitted to poor oral intake. He also mentioned of paraesthesia in both hands.
His past medical history included metastatic prostate cancer for which he received LHRH and Zoledronate treatments from 2009 until 2011. He then received LHRH and Docetaxel from January 2012 until April 2012. He went on to receive Abiraterone and Prednisolone treatment. Follow up staging CT had shown progression of disease and the patient was started on Cabazitaxel treatment two weeks prior to admission. He had also developed right mandible osteonecrosis as result of bisphosphonate treatment for which had maxillofacial surgery. His only other medical history was that he was a type 2 diabetic.
His preadmission medications were Metformin, Calcichew D3 Forte, Tamsulosin and Ondansetron. None of these medications had been recently started and the patient had not been taking any over the counter medication. He was unaware of the name of the chemotherapy agent he had received and it was discovered later on to be Cabazitaxel by the medical team, after contacting the patient’s oncologist.
He was a non-smoker and drank less than 21 units of alcohol per week. He lived with his wife and was independent with activities of daily living.
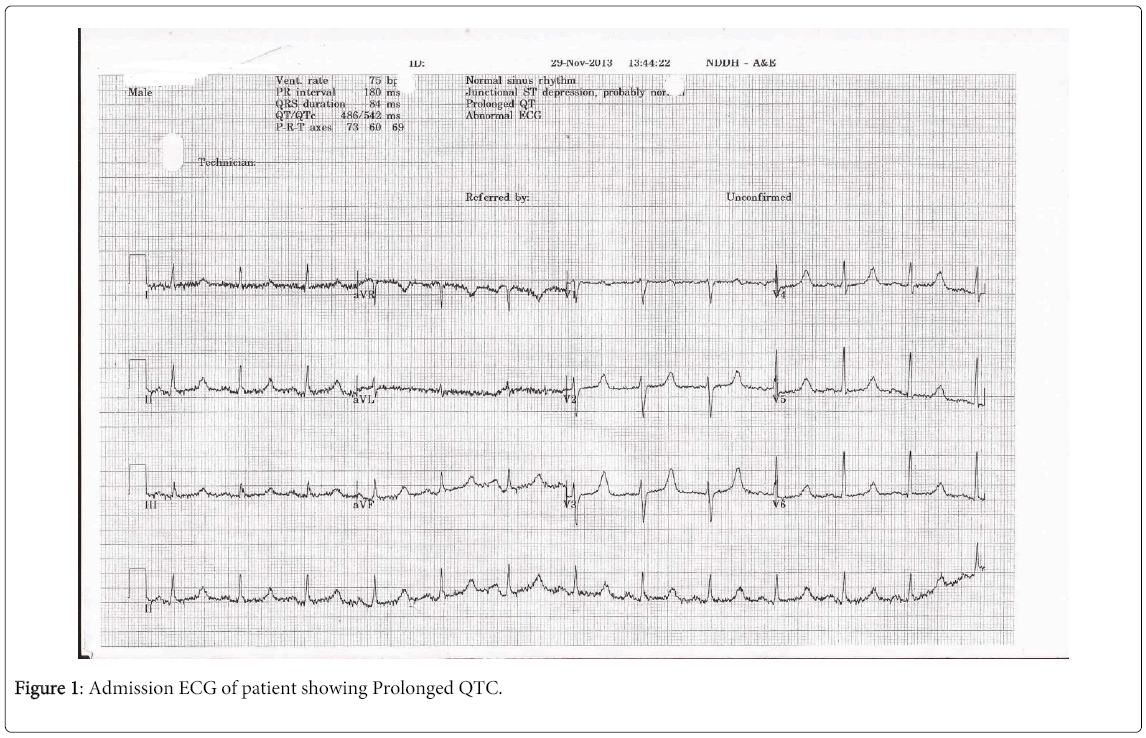
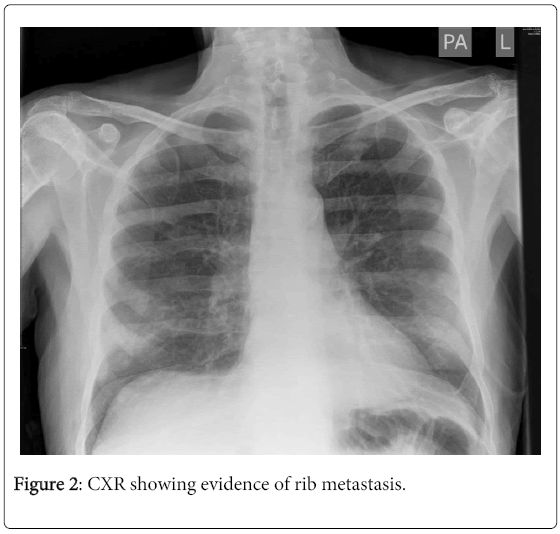
On admission, his observations were within normal limits and his cardio-respiratory examination was unremarkable. ECG showed prolonged QTc (Figure 1). CXR showed evidence of rib metastasis (Figure 2). Initial blood tests showed corrected calcium of 1.4 (2.2-2.6), Hb 8.7 (130-170), Urea 8.9 (3.8-8.3) and Creatinine 129 (70-110) with normal liver function tests and all other electrolytes were within normal range. His C-reactive protein was 53 (<8). A urine dipstick was positive for nitrites, protein, blood and pH was 6.
He was treated with Intravenous Calcium Gluconate to correct the hypocalcaemia. He also received intravenous Gentamicin and intravenous Amoxicillin for presumed urinary tract infection based on the positive urine dipstick test and raised inflammatory markers. His diarrhoea had settled by the fifth day in hospital and he was receiving intravenous fluid therapy for rehydration, with a positive fluid balance of 2 litres.
Despite this follow up blood tests showed worsening kidney function, and creatinine values changed from 85 mmol pre chemotherapy and 126 on admission to a worst value of 378 mmol. His calcium levels did not rise and corrected calcium was 1.37 following initial treatment. Ultrasound of kidney tract showed no obstruction with normal sized kidneys. Myeloma screen tests were negative, as were autoantibody testing for vasculitis. Following a discussion with our renal consultant, it was felt that the most likely cause for the acute kidney injury was dehydration secondary to diarrhoeal illness, and was taking time to respond to IV fluids. A source of infection was looked for but cultures were negative.
All nephrotoxic drugs were discontinued, and the patient continued to receive aggressive intravenous hydration to maintain a positive fluid balance. With diarrhoea settling and no obvious infection it was decided that the most likely cause of the kidney injury was Cabazitaxel therapy. The renal team advised that renal biopsy was not needed as it would not change management.
Patient was started on higher doses of Alfacalcidol and also received higher doses of IV calcium gluconate following discussion with our endocrinologists. Creatinine values began to improve after the third week and reached a plateau of 320, and calcium levels reached 2.17 after being maintained on 6 micrograms of Alfacalcidol daily and multiple IV calcium infusions. Patient was discharged home with outpatient follow up with the renal team. His oncologist was made aware of the events that occurred during the admission and has decided to stop Cabazitaxel treatment.
Discussion
Cabazitaxel was approved by FDA in 2010 for treatment of hormone refractory prostate cancer. It is a semi-synthetic antineoplastic agent belonging to the taxane class of drugs [1]. Adverse effects reported by manufacturers include neutropenia, anaemia, leukopenia, thrombocytopenia, diarrhoea, fatigue, nausea, vomiting, and constipation. Peripheral neuropathy and alopecia have also been reported.
The most common side effect reported in literature is febrile neutropenia [1,2]. Severe diarrhoea is reported as a common side effect in most cases, with fatalities in some patients due to a combination of diarrhoea, dehydration and electrolyte imbalances [1]. Our patient developed diarrhoea soon after starting therapy with Cabazitaxel. Interestingly our patient had severe hypocalcaemia which caused changes in his ECG; there was one case report of a patient on Cabazitaxel developing hypocalcaemia, although this was attributed to Zoledronic acid which the patient had also been receiving [3].
Renal failure, including four cases with fatal outcome, was reported in a randomized clinical trial according to the manufacturers’ information [4]. Most cases occurred in association with sepsis, dehydration, or obstructive uropathy [1]. Some deaths due to renal failure did not have a clear aetiology. It is unclear whether the renal failure could directly be linked to the drug as a direct nephrotoxic effect. In most cases it does seem to occur when in combination with some other pathology such as dehydration and diarrhoea [1]. Certainly our patient had severe diarrhoea which no doubt contributed to his renal failure and electrolyte imbalance.
It remains difficult to explain just why the renal failure and hypocalcaemia persists in our patient. More studies are needed to examine the exact nephrotoxic mechanism by which Cabazitaxel affects the kidney. This remains the most likely reason why the renal function has not improved in our patient as his diarrhoea had settled a few days into admission and he had received aggressive intravenous fluid therapy. How the drug has caused the hypocalcaemia and why this continues to persist also remains unexplained. Interestingly it was the only electrolyte that was affected in the patient. It is possible it could be due to renal wasting secondary to the poor renal function but none of the other electrolytes, including his magnesium, were affected.
Conclusion
An 80 year old male developed Acute Kidney Injury and Hypocalcaemia soon after starting Cabazitaxel for hormone refractory prostate cancer. This required treatment with intravenous fluids and calcium. His renal function remains poor but stable upon discharge. Further studies need to be done to look at the relationship between Cabazitaxel and acute kidney injury.
References
- Abidi A (2013) Cabazitaxel: A novel taxane for metastatic castration-resistant prostate cancer-current implications and future prospects. Journal of Pharmacology & Pharmocotherapeutics 4: 230-237.
- Abraham J (2010) Cabazitaxel and Prednisolone as second-line therapy of metastatic, castration-resistant prostate cancer. Community Oncology 7: 540-543.
- Colburn D (2012) Sequencing of Cabazitaxel in metastatic castrate-resistant prostate cancer: A Case Report. Case Reports in Oncology 5: 320-324.
- Bridgewater NJ: Sanofi-aventis US (2010).Jevtana (Cabazitaxel) injection prescribing information.
Relevant Topics
- Back Pain Diagnosis
- Cardiovascular Diagnosis
- Clinical Diagnosis
- Clinical Echocardiography
- COPD Diagnosis
- Diabetes Diagnosis
- Diagnosis Methods
- Diagnosis of cancer
- Diagnosis of CNS
- Diagnosis of Diabetes
- Diagnostic Products
- Diagnostics Market Analysis
- Heart diagnosis
- Immuno Diagnosis
- Infertility Diagnosis
- Medical Diagnostic Tools
- Preimplementation Genetic Diagnosis
- Prenatal Diagnostics
- Ultrasonography
Recommended Journals
Article Tools
Article Usage
- Total views: 15703
- [From(publication date):
December-2014 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 10986
- PDF downloads : 4717