Research Article Open Access
Significance and Outlook of Preoperative Serum Interleukin-1β and Interleukin-6 as Prognostic Factors in Esophageal Cancer
Yuka Hattori*, Natsumi Tomita, Kouhei Yoshino and Yoshiaki KajiyamaDepartment of Esophageal and Gastroenterological Surgery, Juntendo University Graduate School of Medicine, Tokyo, Japan
- *Corresponding Author:
- Yuka Hattori
Department of Esophageal and Gastroenterological Surgery
Juntendo University Graduate School of Medicine, Tokyo, Japan
E-mail: hattoriyukamail@yahoo.co.jp
Received date: September 16, 2017; Accepted date: September 28, 2017; Published date: October 05, 2017
Citation: Hattori Y, Tomita N, Yoshino K, Kajiyama Y (2017) Significance and Outlook of Preoperative Serum Interleukin-1β and Interleukin-6 as Prognostic Factors in Esophageal Cancer. J Gastroint Dig Syst 7: 531. doi: 10.4172/2161-069X.1000531
Copyright: © 2017 Hattori Y, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Gastrointestinal & Digestive System
Abstract
Objective: We evaluated various cytokines in patients with esophageal cancers to elucidate the relationship between the Th1/Th2 balance and the patients’ prognosis.
Materials and Methods: Various serum cytokines of preoperative 98 patients, who subsequently underwent radical esophagectomy between September 2005 and December 2006, were quantitatively measured by Cytometric Bead Array (CBA) system. The cytokines include Th1/Th2 balance related 14 cytokines-IL-1α, IL-1β, IL-2, IL-4, IL-5, IL-6, IL-7, IL-8, IL-10, IL-12p70, IL-13, IL-17A, INFγ and IgE. Multivariable analysis was performed using the Cox proportional hazard model. The survival rates based on CRP values were analyzed by the Kaplan-Meier method. Eight clinical pathological factors of the tumors, which are regarded as relevant prognostic factors: diameter, histological type, invasion type, vascular invasion, venous invasion, depth of the invation (T factor), regional lymph nodal status (N factor) and intramural metastasis (IM) and the 14 cytokines were evaluated by the multivariable analysis.
Results: The multivariable analysis showed that T factors (p=0.001), N factors (p=0.002), IL-1β (p=0.003) and IL-6 (p=0.021) were significantly related to the worse prognosis. The two cytokines are known as inflammatory cytokines and they theoretically induce the production of acute inflammatory proteins, including C-reactive protein (CRP). We hereby evaluated the preoperative serum CRP values of the same patients. By means of ROC analysis (Youden index), the cut off value of the serum CRP was determined as 0.25 mg/dl and the survival rates were analyzed by the Kaplan-Meier method. The high CRP group (CRP ≥ 0.25 mg/dl) showed significantly poorer prognosis than low CRP group (<0.25 mg/dl) (p=0.027; Log-rank test).
Conclusion: Out study showed that the inflammatory cytokines, IL-1β and IL-6, are significant prognostic factors of the patients with esophageal cancers and the Th1/Th2 balance in the patients of poorer prognosis may be shifted toward Th2-dominant side.
Keywords
Esophageal cancer; IL-1β; IL-6; Cytokine; Cytometric bead array; C-reactive protein; Prognostic factor
Introduction
Cytokines produced by helper T (Th) cells are classified into Th1 types that are involved in cell-mediated immunity and delayed hypersensitivity, or Th2 types that are involved in humoral immunity and immediate hypersensitivity. The Th1/2 paradigm was first proposed by Mosmann et al., and it has been demonstrated that a disturbance in this Th1/Th2 balance causes the onset of a variety of immunological diseases [1]. In addition, it has also been gradually elucidated that a disturbance in this Th1/Th2 balance is strongly related to the onset and prognosis of cancer patients [2].
Immunosuppressive substances produced by the tumor are thought to suppress Th1 cells and induce Th2 cells, and correcting Th2 dominance in cancer patients has been considered the basis of cancer immunotherapy [3]. There have been numerous studies recently that have detected these Th1/Th2 balance disturbances by measuring preoperative serum cytokines in cancer patients, and the results have been used to determine prognosis. The association between various serum cytokines and prognosis has been identified in multiple malignant tumors [4,5]. In the present study, we investigated the relationship between preoperative serum cytokines in esophageal cancer and the patient’s prognosis, and verified whether or not the Th1/Th2 balance is also disrupted in esophageal cancer patients.
Materials and Methods
Of the esophageal cancer patients who underwent surgery between September 2005 and December 2006 at our department, we investigated 98 patients with thoracic esophageal squamous cell carcinoma who underwent esophageal resection using a right thoracotomy and laparotomy and three-field lymphadenectomy as curative operation without receiving preoperative treatment. Adjuvant chemotherapy with docetaxel, cisplatin and 5-fluorouracil) or chemo radiotherapy (the same chemotherapy agents with radiation therapy, 2Gy for 5 days/ week: total 60Gy) was performed depend on final clinicopathological staging or curability of the operation according to our institutional protocol [6].
Various cytokines were measured in the preoperative serum samples of these patients using the Cytometric Bead Array (CBA) system. Fourteen cytokines that are known to be involved in the Th1/ Th2 balance were measured (interleukin (IL)-1α, IL-1β, IL-2, IL-4, IL- 5, IL-6, IL-7, IL-8, IL-10, IL-12p70, IL-13, IL-17A, interferon (IFN)-γ, and IgE). Survival or date of death of the patients was censored at the date of last follow up by chart review or telephone survey. Multivariate analysis (Cox regression analysis) was used to assess the association with prognosis and the relationship between preoperative C-reactive protein (CRP) levels and prognosis was also investigated.
Covariates that were entered into the Cox regression analysis were the following clinicopathological factors: (1) long diameter of the tumor, (2) histological grade, (3) infiltration pattern, (4) vascular invasion, (5) venous invasion, (6) pathological extent of tumor invasion (T), (7) lymph node metastasis (N) and (8) presence or absence of intramural metastasis, in addition to the levels of the 14 cytokines mentioned above. For cytokine measurements, serum samples that were isolated from the preoperative peripheral blood collected immediately after general anesthesia induction were used. These serum samples were stored at -80°C and were thawed at room temperature at the time of measurement. Subsequently, the quantities of the 14 aforementioned serum cytokines were measured using CBA system (Figure 1). For TNM classification, the sixth edition was used.
Figure 1: BD Cytometric Bead Array (CBA) System; [In the CBA system, the samples are coated with two types of fluorescent beads (capture antibody and PE-labeled detection antibodies), and because the fluorescence intensity of the capture antibody is specific for each measured protein, the system allows for the simultaneous analysis of multiple proteins].
CBA system
In the CBA system, the samples were first coated with two types of fluorescent beads, and because the fluorescence intensity of the captured antibody is different for each measured protein, the system allows for the simultaneous analysis of multiple proteins (Figure 1). By simultaneously measuring the phycoerythrin (PE) fluorescence intensity from PE-labeled detection antibody, this system can be used to perform simultaneous quantitative measurements (pg/mL) of cytokines using less sample volume and greater sensitivity compared to the traditional ELISA method.
Statistical analysis
Using IBM SPSS Statistics 19, analyses were performed using Cox regression analysis and the Kaplan Meier method, and the x2 test was used for univariate analysis. In all analyses, p<0.05 was considered statistically significant. The receiver operating characteristic (ROC) curve (Youden Index method) was used to determine the cut-off values.
Results
The clinicopathological background information of the 98 patients in the study is shown in Table 1. The mean age of the patients was 62.6 years, and there were 80 men (81.6%) in the study. The middle thoracic (Mt) area was the most common primary location of the tumor as seen in 56 patients (57.1%) and the mean length of the long diameter of the tumor was 58.4 mm. Regarding histological grades, there were 45 patients (45.9%) each with well-differentiated squamous cell carcinoma (well) and moderately-differentiated squamous cell carcinoma (mod). INF-β was the most common infiltration pattern as seen in 72 patients (73.5%) and vascular invasion was observed with high frequency as seen in 94 patients (95.9%) who were positive for lymphatic invasion (ly) and 93 patients (94.8%) who were positive for venous invasion (v). pT3 was most frequently observed pathological extent of tumor invasion (TNM classification) as seen in 54 patients (55.1%).
| Age | Mean | 62.6 (39-80) | ± 8.4 |
|---|---|---|---|
| Gender | Men | 80 | 81.6 |
| Women | 18 | 18.4 | |
| Primary location of tumor | Upper thoracic | 5 | 5.1 |
| Middle thoracic | 56 | 57.1 | |
| Lower thoracic | 37 | 37.8 | |
| Long diameter of tumor (mm) | Mean | 58.4 (10-325) | ±39.7 |
| Histological grade (SCC) | Well differentiated | 45 | 45.9 |
| Moderately differentiated | 45 | 45.9 | |
| Poorly differentiated | 8 | 8.2 | |
| Infiltration pattern (Japanese classification) | INF-α | 8 | 8.2 |
| INF-β | 72 | 73.5 | |
| INF-γ | 18 | 18.3 | |
| Lymphatic invasion (Japanese classification) | ly(-) | 4 | 4.1 |
| ly(+) | 94 | 95.9 | |
| Venous invasion (Japanese classification) | v(-) | 5 | 5.2 |
| v(+) | 93 | 94.8 | |
| Pathological extent of tumor invasion (TNM classification) | pT1 | 24 | 24.5 |
| pT1a | 3 | ||
| pT1b | 21 | ||
| pT2 | 18 | 18.4 | |
| pT3 | 54 | 55.1 | |
| pT4 | 2 | 2 | |
| Lymph node metastasis (TNM classification) | N0 | 27 | 27.6 |
| N1 | 47 | 48 | |
| M1 | 24 | 24.4 | |
| Intramural metastasis | IM0 | 86 | 87.8 |
| IM1 | 12 | 12.2 | |
| pStage (TNM classification) | I | 10 | 10.2 |
| II | 34 | 34.7 | |
| IIA | 17 | ||
| IIB | 17 | ||
| III | 30 | 30.6 | |
| IV | 24 | 24.5 | |
| IVA | 2 | ||
| IVB | 22 |
Table 1: Clinicopathological background of 98 patients with thoracic esophageal squamous cell carcinoma.
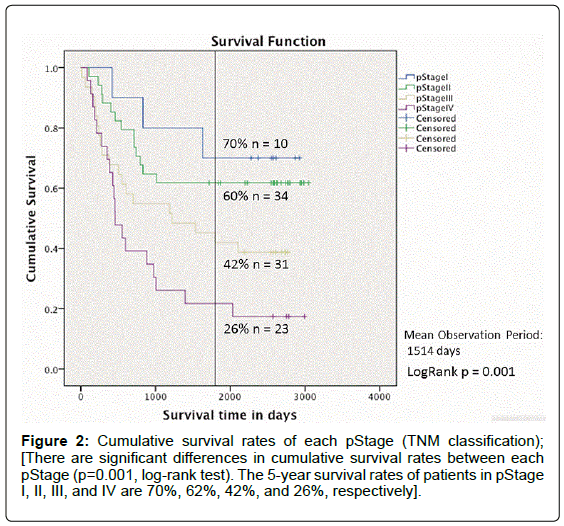
Regarding lymph node metastasis (TNM classification, sixth edition), there were 27 patients (27.6%) who did not exhibit lymph node metastasis (N0), 47 (48.0%) who exhibited regional lymph node metastasis (N1) and 24 (24.4%) who exhibited distant lymph node metastasis (M1). Twelve patients (12.2%) had intramural metastasis. Regarding the pStage (TNM classification), patients with pStage III were most frequently observed (30.6%), followed by pStage IVB (22.4%). The cumulative survival rate of the 98 patients in each pStage (TNM classification) was assessed using the Kaplan-Meier method (mean observation period: 1514 days) (Figure 2).
The results showed that significant differences were observed in cumulative survival rates between each pStage (p=0.001, log-rank test). The cumulative survival curve showed that prognosis worsened with progression from pStage I to pStage IV and that the 5 year survival rates of patients in pStage I, II, III, and IV were 70%, 62%, 42%, and 26%, respectively. Table 2 shows the mean values as well as the minimummaximum values of the 14 serum cytokines that were quantitatively measured using the CBA system in samples collected immediately prior to the operation. In all patients (n=98) cox regression analysis was performed with (1) long diameter of the tumor, (2) histological grade, (3) infiltration pattern, (4) vascular invasion, (5) venous invasion, (6) pathological extent of tumor invasion (T), (7) lymph node metastasis (N) and (8) presence or absence of intramural metastasis, as well as the levels of the 14 cytokines measured in the present study, as covariates and survival time in days as the dependent variable. Four factors-IL- 1β (p=0.003) and IL-6 (p=0.034) in addition to the T factor (p=0.001) and N factor (p ≤ 0.0001) were selected as significant prognostic factors (Table 3).
| Serum cytokine | Mean value | (Minimum-Maximum value) |
|---|---|---|
| IL-1α | 0.6059 | (0.00-26.26) |
| IL-1β | 1.8197 | (0.00-32.91) |
| IL-2 | 6.5929 | (0.00-135.34) |
| IL-4 | 0.4069 | (0.00-14.96) |
| IL-5 | 0.00 | (0.00-0.00) |
| IL-6 | 11.5137 | (0.00-234.51) |
| IL-7 | 0.8593 | (0.00-17.65) |
| IL-8 | 387.5142 | (0.00-16859.65) |
| IL-10 | 0.4255 | (0.00-18.98) |
| IL-12p70 | 0.1645 | (0.00-16.12) |
| IL-13 | 0.00 | (0.00-0.00) |
| IL-17A | 4.0802 | (0.00-56.44) |
| IgE | 133.3226 | (0.01-451.94) |
| INF-γ | 7.4718 | (0.00-40.96) |
Table 2: Quantitative values of the 14 measured serum cytokines; [all units: pg/ml, except IgE in ng/ml].
| Covariate | p-value | Hazard ratio | 95% CI |
|---|---|---|---|
| Pathological extent of tumor invasion (TNM classification) | 0.001 | (relative to T1) | |
| T2 | 0.81 | 1.169 | 0.326-4.200 |
| T3 | 0.017 | 2.945 | 1.214-7.147 |
| T4 | ≤0.0001 | 39.363 | 5.434-285.152 |
| Lymph node metastasis (TNM classification) | ≤0.0001 | (relative to N0) | |
| N1 | 0.334 | 1.553 | 0.636-3.794 |
| IL-1β | 0.003 | 1.184 | 1.057-1.325 |
| IL-6 | 0.034 | 1.023 | 1.002-1.045 |
Table 3: Prognostic factors in all patients (Cox regression analysis).
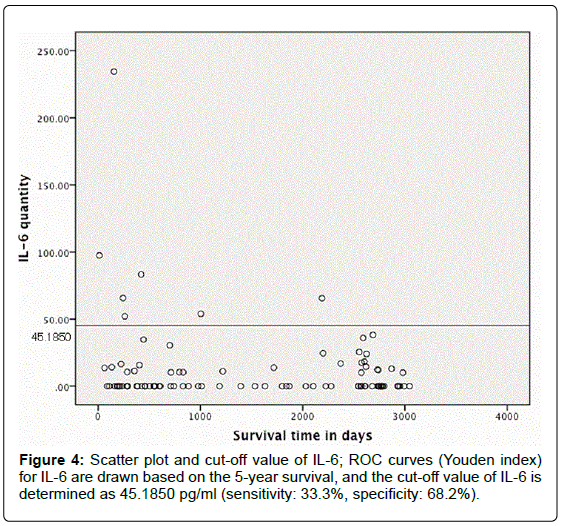
ROC curves (Youden index) for IL-1β and IL-6 were drawn based on the 5-year survival and the cut-off values were determined as IL-1β=16.9150 pg/ml and IL-6=45.1850 pg/ml (Figures 3 and 4). Subsequently, the patients were classified into those with high IL-1β (≥ 16.9150 pg/ml) and low IL-1β (<16.9150 pg/ml), as well as those with high IL-6 ( ≥ 45.1850 pg/ml) and low IL-6 (<45.1850 pg/ml) (Tables 4 and 5). Neither the IL-1β group nor the IL-6 group showed statistical significance (x2 test) in the number of patients in each pStage (IL-1β: p=0.502, IL-6: p=0.079). The survival curves (Kaplan-Meier method) between the high and low IL-1β and IL-6 groups were assessed. Although there were no differences between the two IL-1β groups (p=0.291, log-rank test), the high IL-6 group had significantly worse prognosis compared to the low IL-6 group (p=0.005, log-rank test).
| pStage (TNM classification) | High IL-1β group | Low IL-1β group |
|---|---|---|
| I | 1 | 9 |
| II | 1 | 33 |
| III | 1 | 30 |
| IV | 0 | 23 |
Table 4: pStage categorization of patients in the high and low IL-1β groups; [High IL-1β group: IL-1β ≥ 16.9150 pg/ml; Low IL-1β group: IL-1β <16.9150 pg/ml, X2 p=0.502].
| pStage (TNM classification) | High IL-6 group | Low IL-6 group |
|---|---|---|
| I | 1 | 9 |
| II | 0 | 34 |
| III | 5 | 26 |
| IV | 1 | 22 |
Table 5: pStage categorization of patients in the high and low IL-6 groups; [High IL-6 group: IL-6 ≥ 45.1850 pg/ml; Low IL-6 group: IL-6 <45.1850 pg/ml, X2 p=0.079].
Figures 5 and 6 show the cumulative survival curves of the IL 1β and IL-6 groups, respectively. Next, we investigated CRP, which is known to be associated with IL-6 and IL-1β. The cut-off value for the CRP levels that was preoperatively measured was set to 0.2500 mg/dl using the ROC curve (Youden index) based on 5-year survival. Patients were divided into a high CRP group (≥ 0.2500 mg/dl) and a low CRP group (<0.2500 mg/dl), and the survival rates between the two groups were compared using Kaplan-Meier method. The scatter plot of the CRP level and survival time in days in the 98 patients is shown in Figure 7.
Figure 5: Cumulative survival rates of the high IL-1β group and the low IL-1β group; [The survival curves (Kaplan-Meier method) between the high and low IL-1β groups are assessed. Although there is no significant difference between the two IL-1β groups (p=0.291, log-rank test), the high IL-1β shows tendency of the worse survival. The mean survivals of the high and low groups are 961 days and 1719 days, respectively].
Figure 6: Cumulative survival rates of the high IL-6 group and the low IL-6 group; [The high IL-6 group shows significantly worse prognosis compared to the low IL-6 group (p=0.005, log-rank test) with the 5-year survival rates being 14% and 48% for the high and low groups, respectively. The mean survivals are 611 days and 1776 days, respectively].
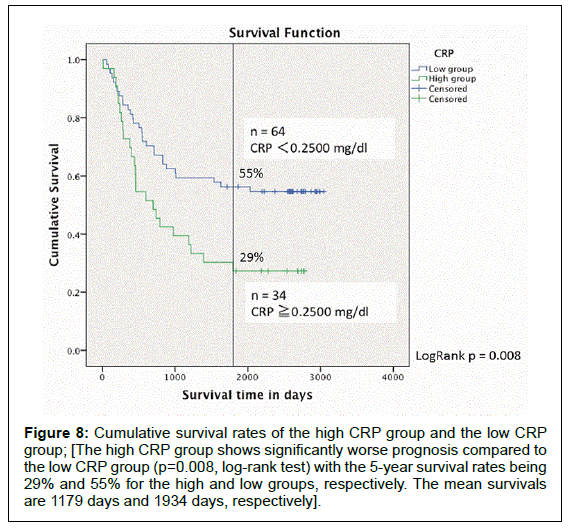
The high CRP group had significantly worse prognosis compared to the low CRP group (p=0.008, log-rank test) with the 5-year survival rates being 29% and 55% for the high and low groups, respectively (Figure 8). No statistical significance in the number of patients in each pStage was observed between the two groups (p=0.054, X2 test) (Table 6). Correlation analysis between the serum IL-6 concentration and the CRP level showed that there was a positive, albeit weak, correlation (correlation coefficient, r=0.21).
Figure 8: Cumulative survival rates of the high CRP group and the low CRP group; [The high CRP group shows significantly worse prognosis compared to the low CRP group (p=0.008, log-rank test) with the 5-year survival rates being 29% and 55% for the high and low groups, respectively. The mean survivals are 1179 days and 1934 days, respectively].
| pStage (TNM classification) | High CRP group | Low CRP group |
|---|---|---|
| I | 1 | 9 |
| II | 9 | 25 |
| III | 16 | 15 |
| IV | 8 | 15 |
Table 6: pStage categorization of patients in the high and low CRP groups; [High CRP group: CRP ≥ 0.2500 mg/dl; Low CRP group: CRP <0.2500 mg/dl, X2 p=0.054].
Discussion
According to the Th1/Th2 paradigm first proposed by Mosmann et al., cytokines produced by Th cells are classified into Th1 types that are involved in cell-mediated immunity and delayed hypersensitivity, or Th2 types that are involved in humoral immunity and immediate hypersensitivity, and it has been elucidated that a disturbance in this Th1/Th2 balance is mechanistically involved in the onset of various immunological diseases [1,7]. Additionally, it has also been gradually elucidated that this Th1/Th2 paradigm is greatly involved in tumor pathogenesis as well as prognosis, forming the basis of today’s tumor immunology [3].
Previous studies have proven the suppression of Th1 type cells and the overproduction of Th2 type cells in local tumors [8,9]. In recent years, with advancements in measurement techniques such as the CBA system, it has become feasible to evaluate the immune balance in the serum in addition to the immune balance in local tumors [9]. As a result of using such techniques, it has been recognized in multiple solid tumors that various cytokines influence prognosis independent of histopathological progression [10,11]. In the current study, we assessed the prognosis in esophageal cancer patients with multivariate analysis using the Cox proportional hazards model with (1) long diameter of the tumor, (2) histological grade, (3) infiltration pattern, (4) vascular invasion, (5) venous invasion, (6) pathological extent of tumor invasion (T), (7) lymph node metastasis (N), and (8) presence or absence of intramural metastasis, as well as the levels of the 14 cytokines measured as covariates.
Results showed that four factors, extent of tumor T factor (p=0.001), lymph node metastasis N factor (p ≤ 0.0001), IL-1β (p=0.003) and IL-6 (p=0.034), were selected as significant prognostic factors. However, regarding IL-1β and IL-6, in the comparison of prognosis between high and low groups (with cut-off values determined using ROC curves based on 5-year survival), although significant differences were observed between the high and low IL-6 groups (p=0.005, log-rank test), no differences were observed between the high and low IL-1β groups (p=0.291, log-rank test).
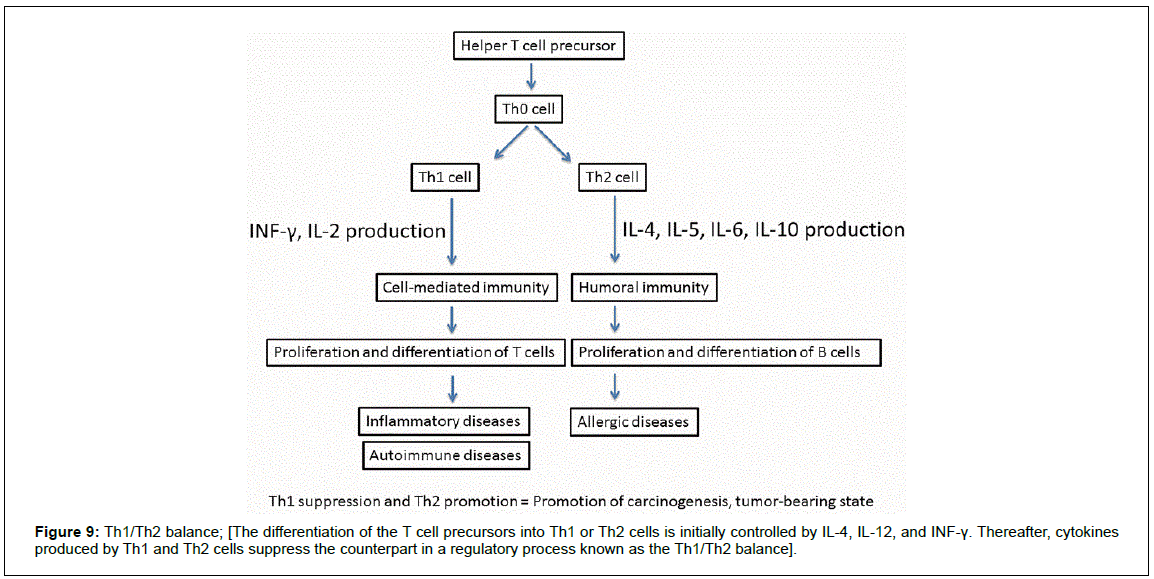
Th cells progress through the stages of naive T cell precursor to Th0 and functionally mature into Th1 or Th2 cells depending on the pattern of the cytokines they produce. Th1 cells produce IFN-γ, IL-2, and lymphotoxin and are involved in cell-mediated immunity responses, whereas Th2 cells produce IL-4, IL-5, IL-6, and IL-10, activate B cells and are involved in the humoral immunity response [1]. The differentiation into Th1 or Th2 cells is controlled by IL-4, IL-12, and INF-γ and cytokines produced by Th1 and Th2 cells suppress each other’s function in a process known as the Th1/Th2 balance (Figure 9) [12,13]. It has been previously reported that cancer patients are Th2- dominant in the Th1/Th2 balance [2].
It has also been reported that there is decreased IL-2 production and elevated IL-4, IL-6, and IL-10 production in colorectal cancer, decreased IL-2 and IFN-γ production in bladder cancer, prostate cancer, and renal cancer and increased IL-10 mRNA expression in lung cancer [8,14-17]. Expression of the Th2 type cytokine IL-6 within esophageal cancer tissue is known to be profoundly associated with the progression of esophageal cancer and poor prognosis [18,19]. In the present study, esophageal cancer patients with high IL-6 in the preoperative peripheral blood also had significantly worse prognosis compared to those with low IL-6, showing a similar trend in the levels of the cytokine in the serum and in tumor tissues.
IL-1 is a cytokine that is known to possess many functions upstream of IL-6 in the cytokine network (Figure 10). Although they have different protein sequences, IL-1α and IL-1β possess physiological actions that are essentially identical and IL-1β is known to promote IL-6 production by tumor cells and inflammatory cells in cancer patients [20,21]. From the multivariate analysis in the present study, IL-1β was selected as a significant prognostic factor together with IL-6 and this indicated the possibility that, of the many differentiationinducing functions of IL-1β, differentiation into Th2 types including IL-6 is promoted in esophageal cancer patients as well. IL-6 and IL-1β are termed “inflammatory cytokines” along with tumor necrosis factor (TNF)-α and are known to act on hepatocytes to promote acute-phase protein (CRP) production [20-22]. Previous reports have demonstrated that patients with solid tumors who show high preoperative serum CRP levels have poor prognosis, regardless of the disease stage [23-28].
Moreover, there are recent reports that indicated that an increase in the CRP level is correlated with the serum IL-6 concentration in gastric cancer and renal cell carcinoma [4,5]. We also investigated the correlation between the serum IL-6 concentration and CRP level in the present study, but did not observe a strong correlation (correlation coefficient r=0.21). However, when we compared the 5 year survival rates (Kaplan-Meier method) between the two groups that were divided into high and low CRP groups with a cut-off value of 0.2500 mg/dl, we found that the high CRP group had significantly worse prognosis (p=0.008, log-rank test) and that the 5 year survival rates were 29% and 55% for the high and low CRP groups, respectively.
Although a small bias (p=0.054) was observed in the number of patients in each pStage between the two groups, this was not statistically significant and high levels of preoperative serum CRP were also suggested to be a factor for poor prognosis in esophageal cancer patients regardless of the disease stage. In the present study, we did not observe elevations in other Th2 type cytokines represented by IL-4, IL- 5, and IL-10. Some of the potential reasons for this finding include the fact that there is detection limit due to low cytokine concentration in the serum, although quantitative measurement to the pg/ml level is feasible with BCA systems, and that the experimental environment could easily influence the results depending on the type of cytokine [29].
Regarding the Th1/Th2 balance, cancer immunotherapy using compounds such as lentinan, which corrects Th2 dominance in a tumor-bearing state, has already been clinically applied, and its effectiveness against esophageal cancer has also been reported [30,31]. The elucidation not only of the Th1/Th2 balance but also of the mechanism that induces specific cytokine elevation is important as it may become the foundation for the development of new drugs for cancer immunotherapy and molecular targeted therapy. In the future, measuring serum cytokines not only preoperatively but also postoperatively as well as before and after postoperative adjuvant therapy to assess the changes over time is considered to be necessary. As a result, ascertaining the tumor immunocompetence status and predicting prognosis may become feasible through the assessment of the Th1/Th2 balance and the time-dependent changes in specific cytokines.
Conclusion
The present study demonstrated that preoperative serum levels of the inflammatory cytokines IL-1β and IL-6 are significant prognostic factors in esophageal cancer patients, and also suggested that the Th1/ Th2 balance in the immune response of esophageal cancer patients with poor prognosis may be shifted towards Th2 dominance.
References
- Mosmann TR, Sad S (1996) The expanding universe of T-cell subsets: Th1, Th2 and more. Immunol Today 17: 138-146.
- Yoshino S (1999) Th1/Th2 balance in cancer patients. Biotherapy 13: 1079-1086.
- Yoshino S (1998) Immunotherapy based on Th1, Th2 theory. Biotherapy 12: 1435-1440.
- Mohri Y (2011) Prognostic significance of preoperative serum CRP level in patients with gastric cancer. J Japan Surgical Assoc 72: 2489-2495.
- Yoshida N (2001) Role of inflammatory cytokines in patients with renal cell carcinoma. J Osaka City Medical Assoc 50: 57-67.
- Hamuro J (2005) New trends in the application of Th1/Th2 theory. Surg Frontier 12: 8-18.
- Hashiguchi T (2014) Docetaxel, cisplatin and 5-fluorouracil adjuvant chemotherapy following three-field lymph node dissection for stage II/III N1, 2 esophageal cancer. Mol Clinical Oncol 1: 719-724.
- Asselin‐Paturel C (1998) Quantitative analysis of Th1, Th2 and TGF‐β1 cytokine expression in tumor, TIL and PBL of non‐small cell lung cancer patients. Int J Cancer 77: 7-12.
- Al‐Saleh W (1998) Correlation of T‐helper secretory differentiation and types of antigen‐presenting cells in squamous intraepithelial lesions of the uterine cervix. J Pathol 184: 283-290.
- Sato M (1997) Impaired production of Th1 cytokines and increased frequency of Th2 subsets in PBMC from advanced cancer patients. Anticancer Res 18: 3951-3955.
- Tabata T (1999) Th2 subset dominance among peripheral blood T lymphocytes in patients with digestive cancers. Am J Surg 177: 203-208.
- Szabo SJ (1997) Regulation of the interleukin (IL)-12R β2 subunit expression in developing T helper 1 (Th1) and Th2 cells. J Exp Med 185: 817-824.
- Mosmann TR, Moore KW (1991) The role of IL-10 in crossregulation of TH1 and TH2 responses. Immunol Today 12: A49-A53.
- Shibata M (1996) Serum levels of interleukin‐10 and interleukin‐12 in patients with colorectal cancer. Ann N Y Acad Sci 795: 410-412.
- Pellegrini P (1996) Disregulation in TH1 and TH2 subsets of CD4+ T cells in peripheral blood of colorectal cancer patients and involvement in cancer establishment and progression. Cancer Immunol Immunother 42: 1-8.
- Berghella AM (1997) The significance of an increase in soluble interleukin-2 receptor level in colorectal cancer and its biological regulating role in the physiological switching of the immune response cytokine network from TH1 to TH2 and back. Cancer Immunol Immunother 45: 241-249.
- Elsässer-Beile U (1998) Th1 and Th2 cytokine response patterns in leukocyte cultures of patients with urinary bladder, renal cell and prostate carcinomas. Tumor Biol 19: 470-476.
- Oka M (1996) Relationship between serum levels of interleukin 6, various disease parameters, and malnutrition in patients with esophageal squamous cell carcinoma. Cancer Res 56: 2776-2780.
- Wang LS, Chow KC, Wu CW (1999) Expression and up-regulation of interleukin-6 in oesophageal carcinoma cells by n-sodium butyrate. Br J Cancer 80: 1617-1622.
- Strassmann G (1993) Suramin interferes with interleukin-6 receptor binding in vitro and inhibits colon-26-mediated experimental cancer cachexia in vivo. J Clin Invest 92: 2152-2159.
- Strassmann G (1993) Mechanisms of experimental cancer cachexia: Local involvement of IL-1 in colon-26 tumor. J Immunol 150: 2341-2345.
- O'riordain MG (1999) Peripheral blood cells from weight-losing cancer patients control the hepatic acute phase response by a primarily interleukin-6 dependent mechanism. Int J Oncol 15: 823-830.
- Mcmillan DC, Canna K, Mcardle CS (2003) Systemic inflammatory response predicts survival following curative resection of colorectal cancer. Bri J Surg 90: 215-219.
- Hilmy M (2005) The relationship between the systemic inflammatory response and survival in patients with transitional cell carcinoma of the urinary bladder. Bri J Cancer 92: 625-627.
- Jamieson NB (2004) Systemic inflammatory response predicts outcome in patients undergoing resection for ductal adenocarcinoma head of pancreas. Bri J Cancer 92: 21-23.
- Zhao ZF (2016) Interleukin 6 as a potential molecular target in esophageal squamous cell carcinoma. Oncol Lett 1: 925-932.
- Chen MF (2012) Role of interleukin 1 beta in esophageal squamous cell carcinoma. J Mol Med 1: 89-100.
- Crumley ABC (2006) An elevated C-reactive protein concentration, prior to surgery, predicts poor cancer-specific survival in patients undergoing resection for gastro-oesophageal cancer. Bri J Cancer 94: 1568-1571.
- Ito T (2007) The flow beads array assay as an efficacy assessment of a clinical trial. Cytometry Res 17: 1-6.
- Yoshino S (1999) Immunoregulatory effects of lentinan on the balance between Th1 and Th2. Biotherapy 13: 783-787.
- Hayashi S (1993) The effect of lentinan on the immunological changes after operation for esophageal cancer. Jpn J Gastroenterol Surg 26: 1921-1928.
Relevant Topics
- Constipation
- Digestive Enzymes
- Endoscopy
- Epigastric Pain
- Gall Bladder
- Gastric Cancer
- Gastrointestinal Bleeding
- Gastrointestinal Hormones
- Gastrointestinal Infections
- Gastrointestinal Inflammation
- Gastrointestinal Pathology
- Gastrointestinal Pharmacology
- Gastrointestinal Radiology
- Gastrointestinal Surgery
- Gastrointestinal Tuberculosis
- GIST Sarcoma
- Intestinal Blockage
- Pancreas
- Salivary Glands
- Stomach Bloating
- Stomach Cramps
- Stomach Disorders
- Stomach Ulcer
Recommended Journals
Article Tools
Article Usage
- Total views: 3615
- [From(publication date):
October-2017 - Aug 18, 2025] - Breakdown by view type
- HTML page views : 2737
- PDF downloads : 878