The “Calcium Paradox” due to Ca2+/cAMP Interaction and Antihypertensive Pharmacotherapy: New Avenues for Drug Development
Received: 09-Mar-2017 / Accepted Date: 21-Mar-2017 / Published Date: 28-Mar-2017
Abstract
In 2013, we showed that the paradoxical effects (sympathetic hyperactivity) induced by L-type voltage-activated Ca2+ channels (VACC) blockers, named by us “calcium paradox” phenomenon, were potentiated by drugs which increase cytosolic cAMP concentration ([cAMP]c-enhancers), for example rolipram, IBMX and forskolin, indicating that the sympathetic hyperactivity induced by VACC blockers is due to interaction of the Ca2+/cAMP intracellular signaling pathways (Ca2+/cAMP interaction). Then, the pharmacological handling of this interaction produced by combined use of the L-type VACC blockers prescribed in the antihypertensive therapy, and [cAMP]c-accumulating compounds prescribed in the antidepressive therapy, could represent a potential cardiovascular deleterious effect for hypertensive patients due to stimulation of sympathetic hyperactivity. Then, we discussed the role of Ca2+/cAMP interaction for antihypertensive pharmacotherapy. In conclusion, this interaction could be a novel therapeutic target for drug development.
Keywords: Ca2+/cAMP interaction; Sympathetic neurotransmission; Hypertension
78803Introduction
Since 1970´s, many medical studies have described that prescription of Ca2+ channels blockers (CCBs) for hypertensive patients, such as verapamil and nifedipine, produces reduction in peripheral vascular resistance and arterial pressure due to vasodilation, resultant of reduction of Ca2+ influx through plasmalemal L-type voltage-activated Ca2+ channels (VACC) in smooth muscle cells, but also produces increase in plasma catecholamines levels and heart rate, typical symptoms of sympathetic hyperactivity [1]. Despite the risks of antihypertensive therapy, these adverse effects of L-type VACC blockers were initially attributed to autonomic adjust reflex of arterial pressure. However, several experimental studies performed in 1970´s, by studying the effects of L-type VACC blockers in smooth muscle richly inervated by sympathetic neurons (rodent vas deferens) submitted to electrical field stimulation (EFS), demonstrated that these blockers could produce dual effect on sympathetic neurotransmission. In high concentrations (>1 μM), L-type VACC blockers completely inhibited sympathetic EFS-contractions due to blockade of Ca2+ influx in smooth muscle cells, but paradoxically increased these contractions in low concentrations (<1 μM), suggesting that other working principle than only autonomic adjusting reflex is involved in this paradoxical event. However, the fundamental mechanisms involved in this paradoxical effect of L-type VACC blockers remained unclear during almost forty years [2-5].
In a study published in 2013 in Cell Calcium, we demonstrated that the paradoxical sympathetic hyperactivity produced by L-type VACC blocker, named by us as “calcium paradox”, is resultant of interference produced by these blockers on the signaling pathways interaction mediated by Ca2+ and cAMP (Ca2+/cAMP interaction) [6]. We showed that simultaneous administration of L-type VACC blockers (verapamil) with drugs which produce increase of cytosolic cAMP concentration ([cAMP]c-enhancers), such as rolipram, IBMX and forskolin, enhanced sympathetically-mediated contractions of vas deferens [6]. In addition, this enhancement of contraction was reduced by decreasing [cAMP]c caused by adenylyl cyclase (AC) inhibition (SQ 22536), or diminution of Ca2+ storage from endoplasmic reticulum (ER) by thapsigargin, revealing the involvement of Ca2+ mobilization from ER in this response [6].
Our studies showed that paradoxical increase of sympathetic activity produced by Ca2+ channels blockers, used in anti-hypertensive therapy, results from increment of neurotransmitters release from secretory response of postganglionic sympathetic neurons due to its interference on the Ca2+/cAMP interaction [6-8]. Thus, reduction of Ca2+ influx in these cells by verapamil and other Ca2+ channels blockers (in low concentration) increases AC activity, and [cAMP]c, due to decrease of [Ca2+]c [6-8]. It was showed that 5 and 6 isoforms of AC are involved in this AC-inhibition by Ca2+. The [cAMP]c increment causes increase of cAMP-stimulated Ca2+-release from ER, that in turn, stimulates exocytotic process due to enhance of secretory vesicles recruitment, and docking, to the plasma membrane, priming of fusion machinery, and fusion of vesicles with the plasma membrane [6-8].
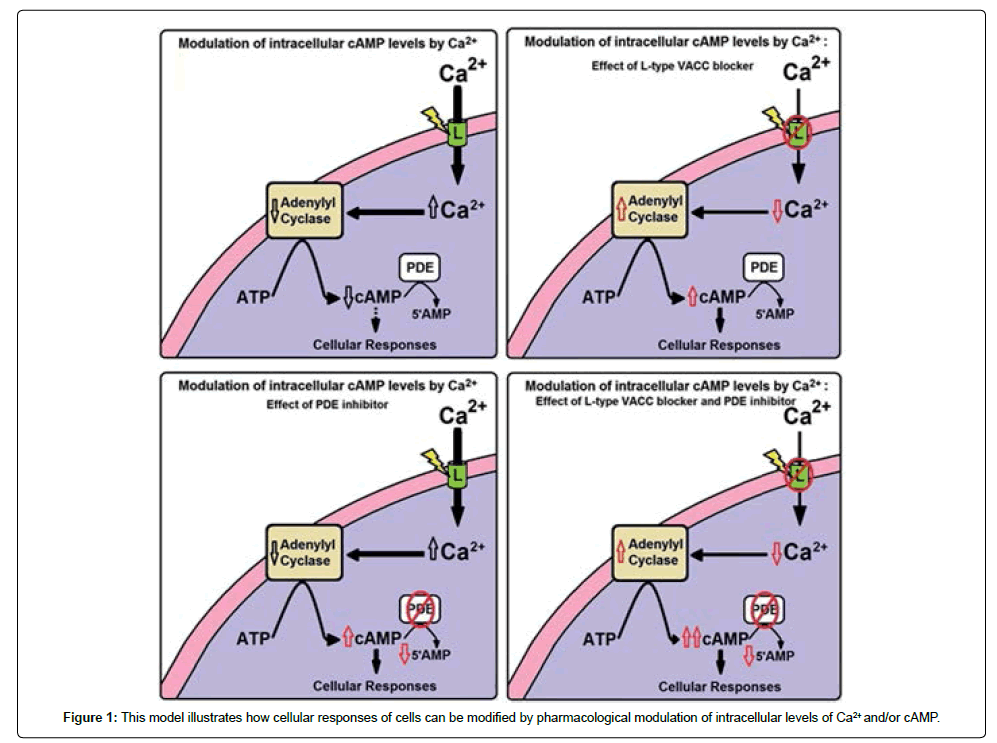
Theoretical model of Ca2+/cAMP interaction in secretory cells proposed by Bergantin et al. [7] and Caricati-Neto et al. [8] illustrates how secretory responses of the neurons and neuroendocrine cells can be modified by pharmacological modulation of intracellular levels of Ca2+ and/or cAMP (Figure 1). This proposement is reforcing the hypothesis that Ca2+/cAMP interaction plays an important role in the sympathetic activity, by regulating secretory response of sympathetic neurons, and adrenal chromafin cells [6]. In addition, these pharmacological manipulations of the Ca2+/cAMP interaction could have important implications for antihypertensive pharmacotherapy [7,8].
Implications of Ca2+/cAMP Interaction for Antihypertensive Pharmacotherapy: New Avenues for Drug Development
Grossman and Messerli [1] by analyzing Medline database, from 1975 to 1996, found 63 clinical studies involving 1,252 hypertensive patients highlighting sympathetic hyperactivity induced by acute and chronic prescrition of L-type VACC blockers, and other publications in some hypertensive patients that nifedipine has been reported to induce sympathetic activation, and a paradoxical increment of blood pressure [9,10]. Then, whether this “calcium paradox” due to Ca2+/cAMP interaction is involved in this sympathetic hyperactivity in hypertensive patients deserves special attention.
Indeed, L-type VACC blockers like verapamil, and analogous, have been extensively prescribed to reduce blood pressure in hypertensive patients, especially in combination with other drugs for alleviating angina or cardiac arrhythmias [11]. In the field of drug interaction, we could also conclude that a therapy involving the combined prescription of VACC blockers with drugs which increase [cAMP]c should be done carefully in hypertensive patients with neurological/psychiatric disorders [12], considering the participation of sympathetic transmission in modulating vascular tone by discharging neurotransmitters into the vasculature. Then, the pharmacological handling of the Ca2+/ cAMP interaction could represent a potential deleterious effect for antihypertensive therapy due to increase in sympathetic hyperactivity of the cardiovascular system.
Conclusion
Ca2+/cAMP interaction is implicated on the regulation of secretory response from sympathetic neurons and adrenal chromafin cells, and changes of this interaction could be implicated in sympathetic hyperactivity of hypertension due to exacerbation of catecholamines, and purines release, from sympathetic neurons and adrenal chromafin cells, which stimulates vasconstriction and reduces vasodilation, dramatically increasing peripheral vascular resistance, and arterial pressure. These findings may have important impact for antihypertensive pharmacotherapy due to multiple mechanisms involved in sympathetic hyperactivity associated to arterial hypertension, and potential risk involved in drug interaction between VACC blockers and drugs which increase [cAMP]c. New antihypertensive pharmacotherapy strategies could be developed in future to attenuate the cardiovascular risk associated with drug interference on the Ca2+/cAMP interaction in sympathetic neurons, and adrenal chromafin cells. In conclusion, this interaction could be a novel therapeutic target for drug development.
Disclosure
Caricati-Neto and Bergantin thank the continued financial support from CAPES, CNPq and FAPESP (Bergantin´s Postdoctoral Fellowship FAPESP #2014/10274-3).
Conflict of Interest
The authors declare no conflict of interest.
Authorship Contribution
Not applicable to reviews.
References
- Grossman E, Messerli FH (1998) Effect of calcium antagonists on sympathetic activity. Eur Heart J 19: 27-31.
- Kreye VA, Luth JB (1975) Proceedings: Verapamil-induced phasic contractions of the isolated rat vas deferens. NaunynSchmiedebergs Arch Pharmacol 287: 43.
- French AM, Scott NC (1981) A comparison of the effects of nifedipine and verapamil on rat vas deferens. Br J Pharmacol 73: 321-323.
- Moritoki H, Iwamoto T, Kanaya J, Maeshiba Y, Ishida Y, et al. (1987) Verapamil enhances the non-adrenergic twitch response of rat vas deferens. Eur J Pharmacol 140: 75-83.
- Rae GA, Calixto JB (1989) Interactions of calcium antagonists and the calcium channel agonist Bay K 8644 on neurotransmission of the mouse isolated vas deferens. Br J Pharmacol 96: 333-340.
- Bergantin LB, Souza CF, Ferreira RM, Smaili SS, Jurkiewicz NH, et al. (2013) Novel model for "calcium paradox" in sympathetic transmission of smooth muscles: role of cyclic AMP pathway. Cell Calcium 54: 202-212
- Bergantin LB, Jurkiewicz A, GarcÃa AG, Caricati-Neto A (2015) A Calcium paradox in the context of neurotransmission. J Pharm Pharmacol 3: 253-261.
- Caricati-Neto A, GarcÃa AG, Bergantin LB (2015) Pharmacological implications of the Ca2+/cAMPsignalling interaction: from risk for antihypertensive therapy to potential beneficial for neurological and psychiatric disorders. Pharmacol Res Perspect 3:e00181.
- Pohar B, Grad A, Mozina M, Rakovec P, Horvat M (1989) Paradoxical elevation of pulmonary vascular resistance after nifedipine in primary pulmonary hypertension. A case study. Corvasa 31:238–241.
- Ruzicka M, Coletta E, Floras J, Leenen FH (2004) Effects of low-dose nifedipine GITS on sympathetic activity in young and older patients with hypertension. J Hypertens 22:1039-1044.
- Elliott WJ, Ram CV (2011) Calcium channel blockers. J ClinHypertens13:687- 689.
- Bergantin LB, Caricati-Neto A (2016) Challenges for the pharmacological treatment of neurological and psychiatric disorders: Implications of the Ca2+/cAMP intracellular signalling interaction. Eur J Pharmacol 788:255-60.
Citation: Bueno LB, Neto CA (2017) The “Calcium Paradox” due to Ca2+/cAMP Interaction and Antihypertensive Pharmacotherapy: New Avenues for Drug Development. Cardiovasc Ther 2: 117.
Copyright: © 2017 Bueno LB, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Open Access Journals
Article Usage
- Total views: 4211
- [From(publication date): 0-2017 - Aug 29, 2025]
- Breakdown by view type
- HTML page views: 3298
- PDF downloads: 913