Review Article Open Access
Use and Interpretation of the National Comprehensive Cancer Network- Functional Assessment of Cancer Therapy Symptom Indexes in Palliative Research and Treatment: Special Considerations in Ovarian Cancer
Sally E Jensen1 and David Cella2*1Department of Medical Social Sciences, Department of Surgery (Division of Organ Transplantation), Northwestern University Feinberg School of Medicine, USA
2Department of Medical Social Sciences, Robert H. Lurie Comprehensive Cancer Center, Northwestern University Feinberg School of Medicine, USA
- *Corresponding Author:
- David Cella
Department of Medical Social Sciences
Department of Surgery (Division of Organ Transplantation)
Northwestern University Feinberg School of Medicine, USA
Tel: 312-503-1725
Fax: 312-503-9800
E-mail: d-cella@northwestern.edu
Received date February 01, 2013; Accepted date May 02, 2013; Published date May 04, 2013
Citation: Jensen SE, Cella D (2013) Use and Interpretation of the National Comprehensive Cancer Network-Functional Assessment of Cancer Therapy Symptom Indexes in Palliative Research and Treatment: Special Considerations in Ovarian Cancer. J Palliative Care Med 3:145. doi:10.4172/2165-7386.1000145
Copyright: © 2013 Jensen SE, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Palliative Care & Medicine
Keywords
Health-related quality of life; Patient-reported outcomes; Ovarian cancer; Advanced cancer
Introduction
Interpretation of oncology clinical trials has become complicated by the frequent absence of a survival advantage associated with a new therapy, despite evidence suggesting there may be some value to treatment. Surrogate endpoints such as time to disease progression and progression-free survival may be improved with new treatments, but may not correlate with overall survival [1]. This can occur because the array of post-study treatments available to patients after they discontinue study medication may provide further benefit, because of uneven crossover after study treatment, or because these surrogate endpoints are actually not good surrogates for overall survival. Often, this begs the question of whether or not there is a value to the patient of extending time without disease progression. One can assess this value by asking patients about the effect that cancer is having upon their lives. This effect is typically estimated by asking about disease-related and treatment-related symptoms, and the effects that those symptoms have upon patients’ function and well-being.
Cancer symptom assessment, whether symptoms of disease or treatment side effects, can provide an early indication of benefit that is directly relevant to patients’ lives. Cancer symptom assessment can also predict long-term outcomes—including tumor response [2], disease progression [2-5] and survival [2,4-9]. Understanding the most important symptoms and related concerns associated with advanced solid tumors can be crucial to fully appreciating the value of new treatments. In this paper, we review an approach to efficiently measuring the most important symptoms and concerns of people being treated for advanced cancer, with special emphasis on ovarian cancer as a model disease that illustrates the importance of this assessment as a major component to determine treatment value.
Overview of the National Comprehensive Cancer Network Functional Assessment of Cancer Therapy (NCCN-FACT) Indexes
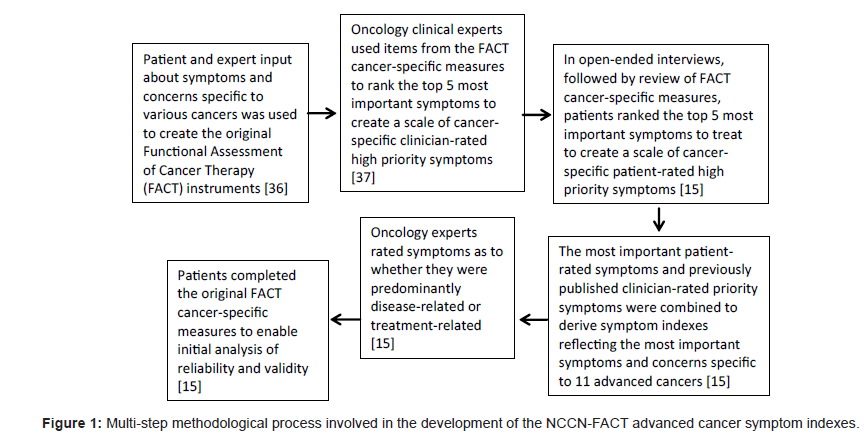
Health-related quality of life (HRQOL) has increasingly become an accepted outcome in clinical trials; however, concern about the use and interpretability of multi-item, multi-dimensional HRQOL measures has led to some reluctance on the part of clinicians and regulatory agencies in the implementation of HRQOL assessment in clinical research and practice [10-13]. In response to the concerns about HRQOL measurement in clinical trials, the Food and Drug Administration Oncology Drug Advisory Committee‘s Quality of life Subcommittee stated that pharmaceutical company claims of improved HRQOL must be specific to the QOL domain measured, with the recommendation that assessment of specific symptoms serve as a starting point for improved measurement of HRQOL domains [14]. In recognition of the importance of assessing HRQOL and symptom improvement, particularly in the case of advanced cancer, recent research sought to improve upon existing validated cancer-related HRQOL and symptom measures to develop clinically-relevant symptom-specific measures that are sensitive to intervention-related changes and which reflect the symptoms considered most important to measure by both oncology clinicians and patients [15]. Patients provided input on the most important symptoms for 11 different types of advanced cancer and physician experts rated whether the symptoms were predominately disease-related or treatment-related. These results were reconciled with previously published indexes which measured the impost important symptoms to measure as determined by oncology clinicians. This multi-step process resulted in the development and initial validation of 11 advanced cancer symptom indexes that reflect the symptoms rated highest priority by patients and clinicians [15]. Figure 1 illustrates the methodological steps involved in the development of these advanced cancer symptom indexes.
In addition to providing clinically-meaningful tools for the assessment of the most important symptoms to measure across 11 different types of advanced cancer (Table 1), the development of these symptom indexes also demonstrates improved adherence to FDA guidance on patient-reported outcomes. It does this by ensuring content validity of the final questionnaire. Inferences regarding other aspects of validity can be drawn from the published performance of highly-related, precursor instruments that bear significant content overlap. For illustration, note in table 1 that the number of new items added to existing FACT-specific questionnaires based on the FDA-recommended approach, ranged from 0-4, with new content never exceeding 20% of the final index.
| Cancer Type | NCCN/FACT Index Name | Number of items (New items) |
|---|---|---|
| Bladder | NCCN-FACT Bladder Symptom Index (NFBlSI-18) [38] | 18 (2) |
| Brain | NCCN-FACT Brain Symptom Index (NFBrSI-24) | 24 (3) |
| Breast | NCCN-FACT Breast Symptom Index (NFBSI-16) [39] | 16 (3) |
| Colon/rectum | NCCN-FACT Colorectal Symptom Index (NFCSI-19) [40,41] | 19 (4) |
| Head and neck | NCCN-FACT Head and Neck Symptom Index (NFHNSI-22) [42] | 22 (4) |
| Hepatobiliary | NCCN-FACT Hepatobiliary-Pancreatic Symptom Index (NFHSI-18) [43-,44] | 18 (0) |
| Kidney | NCCN-FACT Kidney Symptom Index (NFKSI-19) | 19 (2) |
| Lung | NCCN-FACT Lung Symptom Index (NFLSI-17) [45] | 17 (2) |
| Lymphoma | NCCN-FACT Lymphoma Symptom Index (NFlymSI-18) | 18 (2) |
| Ovary | NCCN-FACT Ovarian Symptom Index (NFOSI-18) [23] | 18 (2) |
| Prostate | NCCN-FACT Prostate Symptom Index (NFPSI-17) [46] | 17 (2) |
Table 1: Overview of NCCN/FACT Symptom Indexes.
Use and Interpretation of NCCN-FACT Symptom Indexes in Palliative Oncology
The often limited availability of curative treatment options in advanced stage cancer highlights the importance of patient-reported HRQOL as an endpoint when evaluating the success of treatment. Among individuals with advanced cancer, HRQOL may be adversely affected by numerous physical and mental symptoms. As such, primary goals of clinical trials in advanced cancer involve symptom management, the preservation of functionality, and the maintenance or improvement of HRQOL. The emphasis on such endpoints in palliative interventions reinforces the need for psychometrically sound HRQOL assessment that yields clinically meaningful information, but also HRQOL measurement that reflects regulatory guidance in that it focuses specifically on the symptoms of importance for that condition. The rigorous multi-step methodology employed to develop the NCCNFACT symptom indexes uniquely positions them for implementation in clinical research and practice involving palliative treatments for advanced cancer.
When to use the NCCN-FACT symptom indexes
The NCCN-FACT symptom indexes provide clinical providers and researchers with a new option for assessing patients’ symptom- specific responses to treatment. The NCCN-FACT symptom indexes offer the unique combination of clinical relevance and brevity. Thus, they may be especially well-suited for situations and settings that require brief, clinically meaningful assessment of HRQOL that is also sensitive to change. Each index is between 16-24 items in length, which may help to overcome traditional concerns about implementation of HRQOL in clinical practice and research settings, including concerns about patient burden, interruption of clinic flow, and interpretability [15]. Minimizing patient burden may be of particular importance in the palliative context, given that patients may be more bothered by fatigue and other symptoms which could limit their ability to complete more lengthy HRQOL assessments. The brevity of the NCCN-FACT indexes highlights an improvement over the original FACT cancerspecific measures, which are longer in length and require more effort to complete.
The NCCN-FACT symptom indexes were developed specifically to measure the most important symptoms for patients with advanced (stages III and IV) cancer who are undergoing chemotherapy. Consequently, they are especially well suited for clinical practice or clinical trials which seek to assess the focused symptom experience of individuals with advanced disease, as well as the effect of disease and treatment on the symptom experience. In situations in which clinical providers and researchers seek to examine the multidimensional HRQOL experience of individuals with advanced cancer, or HRQOL in individuals with early stage cancer, the original FACT cancer-specific measures may offer a better alternative.
Finally, the NCCN-FACT symptom indexes were developed with special consideration of the FDA guidance on patient-reported outcomes [14] and thus offer potential advantage in terms of their acceptability in the regulatory setting. They provide a reasonable choice for measuring HRQOL in clinical research involving regulatory submission in the process of evaluating the effect of new treatments on the symptoms rated most important across cancer types. In ovarian cancer, for example, there is no other instrument or index that is more responsive to the FDA Patient-Reported Outcome (PRO) Guidance. Several valid and reliable ovarian cancer-specific HRQOL measures, including the Functional Assessment of Cancer Therapy- Ovarian Cancer (FACT-0) [16], and the Quality of Life Instrument—Ovarian Cancer Patient Version (QOL-OVCA) [17], are currently available. Although patients’ input was included in the development of these measures, patients had no direct input in the selection of items included on the scale. Consequently, the FACT-O and QOL-OVCA may not fully reflect symptoms prioritized by patients and therefore do not meet the FDA regulatory standard. The European Organization for Research and Treatment of Cancer Quality of Life Questionnaire Ovarian Cancer Module (EORTC-QLQ-OV-28) [18,19] did consider ovarian cancer patients’ ratings of the relevance of each symptom or concern in the measure development phase. However, the measure was not developed specifically to reflect the concerns among women with advanced ovarian cancer and women with both early and advanced stage ovarian cancer provided relevance rankings of the symptoms/ concerns during the measure development phase [18]. Thus, although the EORTC-QLQ-OV-28 may satisfy regulatory standards for the inclusion of direct patient input in selection of items, not all of the items included may be specifically relevant to women with advanced ovarian cancer or those receiving palliative treatment.
How to interpret the NCCN-FACT symptom indexes
Interpretability constitutes an important measurement characteristic that influences both the implementation of the measure as well as the meaningfulness of its results. In the case of the NCCNFACT symptom indexes, as with all FACT/Functional Assessment of Chronic Illness Therapy (FACIT) [20] questionnaires, a total score can be obtained, and higher scores indicate better outcomes than lower ones. While this can be clinically informative at the level of the overall index, an examination of the specific subscales (e.g., disease-related, treatment side effects, and function and well-being) can yield more specific information about changes in target symptoms over time or in response to intervention. Given the NCCN-FACT indexes are new indexes, metrics to determine their meaningfulness or interpretability have not yet been established, but constitute an important area for future research. Based on the work of Yost and Eton [21], it is reasonable to anticipate that a meaningful difference for the NCCNFACT indexes would be in the range of 4-5 points. This is consistent with the previously described differences in NCCN-FACT scores between Eastern Cooperative Oncology Group (ECOG) performance status groups [22], which exceed the range of 4 to 5 points [23].
Given that many clinical providers and researchers may have used the original FACT cancer-specific measures of HRQOL, the question may arise about how to interpret the NCCN-FACT symptom index scores in the context of the original FACT measure scores. This may be of particular importance to clinicians and researchers who have used the original FACT measures longitudinally in the past, but who wish to transition to using the newer NCCN-FACT symptom indexes. Due to the fact that the newer NCCN-FACT scales include items not originally part of their original FACT counterpart, the NCCN-FACT symptom index scores cannot be directly calculated from the original FACT measures. However, comparable scores using the original FACT measures can be pro-rated to make them comparable to the NCCNFACT measures using this established formula: (number of items in NCCN-FACT measure) x [(sum of NCCN-FACT item responses)/ (number of NCCN-FACT items completed) if more than 50% of the NCCN-FACT items are completed [24].
In order to illustrate the application of the NCCN-FACT symptom indexes when evaluating the effectiveness of palliative treatment for advanced cancer, we have provided the following example related to the NCCN-FACT Ovarian Symptom Index-18 (NFOSI-18) [23] for use as a potential outcome measure in evaluating the effectiveness of chemotherapy for advanced ovarian cancer.
Applying the NFOSI-18 to Evaluate Treatment Outcomes in Advanced Ovarian Cancer
Ovarian cancer is the second most-common gynecologic cancer in the United States, as well as the most deadly [25], with nearly threefourths of women presenting with advanced stage (stage III-IV) disease [26]. The goals of treatment for ovarian cancer have historically included increasing progression-free and overall survival and minimizing symptom burden due to disease and treatment. However, recent research examining clinically-meaningful patient-centered outcomes has increasingly focused on maximizing HRQOL as an important end-point [27]. With a greater emphasis on HRQOL, research has increasingly noted the impact of disease and treatment on HRQOL. Given that certain clinical benefits of treatment for ovarian cancer may compromise HRQOL, decision-making regarding treatment often involves a consideration of the balance between efficacy and safety, or benefit and harm. Alternatively, a clinical benefit from therapy may also improve HRQOL, essentially increasing the value of that therapy beyond the clinical measures of response, disease-free survival, progression-free survival and overall survival. Consequently, ovarian cancer provides a relevant context in which to review the application of the NFOSI-18 to evaluate treatment outcomes in advanced ovarian cancer.
Overview of the NFOSI-18
The NFOSI-18 was developed as part of a larger cross-sectional study (described earlier) that developed symptom indexes for 11 different types of advanced cancer. Fifty-one women with advanced ovarian cancer rated the most important symptoms when treating advanced ovarian cancer and ten gynecologic oncologists rated whether these symptoms were predominately disease-related or treatment-related [23]. The reconciliation of the patient-rated priority symptoms with earlier published clinician-rated priority symptoms resulted in an 18-item symptom index for advanced ovarian cancer. The NFOSI-18 demonstrated good preliminary reliability, with the full scale internal consistency reliability (16 items with data) α=0.80, and subscale reliability ranging from α=0.55 (Treatment side effects) to α=0.64 (Function and Well-Being) [23]. Preliminary validity for the NFOSI-18 was also good, with significant differences in scores between performance status groups as measured by the ECOG measure of performance status [22], such that poorer performance status was associated with lower NFOSI-18 scores [23]. Although future research is needed to establish standards for clinically meaningful difference and change on the NFOSI-18, the differences in the NFOSI-18 scores between ECOG performance status groups exceeded the range of 4 to 5 points discussed in previous research to establish standards for clinically meaningful differences in measures from the FACIT measurement system [21,23].
The NFOSI-18 is highly redundant with the Functional Assessment of Cancer Therapy-Ovarian (FACT-O) [16] HRQOL measure. Prior to the production of the NFOSI-18, the most common clinical trial endpoint in advanced ovarian cancer clinical trials was the 26- item FACT-O Trial Outcome Index (TOI). Table 2 compares item content between the NFOSI-18 (built to respond to the U.S. FDA PRO Guidance on content validity), and the FACT-O TOI (built to be more inclusive of HRQOL considerations beyond the most important symptoms and concerns). Most (n=14) NFOSI-18 items are also in the TOI, suggesting that published data on the TOI would provide good and related evidence for the likely performance of the NFOSI-18 in future trials. Thus, while the NFOSI-18 offers several advantages over the FACT-O, such as its brevity, focused symptom measurement for advanced ovarian cancer, and enhanced satisfaction of regulatory guidance, its recent emergence compels us to infer much of its validity from its very similar precursor, the TOI.
| Item | Included in NFOSI-18 | Included in FACT-O TOI |
|---|---|---|
| I have a lack of energy | Yes | Yes |
| I have pain | Yes | Yes |
| I feel ill | Yes | Yes |
| I have cramps in my stomach area | Yes | Yes |
| I feel fatigued | Yes | No |
| I am bothered by constipation | Yes | No |
| I have swelling in my stomach area | Yes | Yes |
| I have control of my bowels | Yes | Yes |
| I worry that my condition will get worse | Yes | No |
| I am sleeping well | Yes | Yes |
| I have nausea | Yes | Yes |
| I am bothered by hair loss | Yes | Yes |
| I am bothered by side effects of treatment | Yes | Yes |
| I have been vomiting | Yes | Yes |
| I am bothered by skin problems | Yes | No |
| I am able to get around by myself | Yes | Yes |
| I am able to enjoy life | Yes | Yes |
| I am content with the quality of my life right now | Yes | Yes |
| Because of my physical condition, I have trouble meeting the needs of my family | No | Yes |
| I am forced to spend time in bed | No | Yes |
| I am able to work (include work at home) | No | Yes |
| My work (include work at home) is fulfilling | No | Yes |
| I have accepted my illness | No | Yes |
| I am enjoying the things I usually do for fun | No | Yes |
| I am losing weight | No | Yes |
| I have a good appetite | No | Yes |
| I like the appearance of my body | No | Yes |
| I am able to feel like a woman | No | Yes |
| I am interested in sex | No | Yes |
| I have concerns about my ability to have children | No | Yes |
Table 2: Item-level comparison of NFOSI-18 and FACT-O.
The TOI of the FACT-O includes the items on the Physical Wellbeing scale (7 items), the Functional Well-being scale (7 items), and the Ovarian Cancer Subscale (12 items). As mentioned, it is the most frequent clinical trial outcome measure in use in this setting. Its overlap with the NFOSI-18 (Table 2) enables one to inform planning for future research using the NFOSI-18 as an endpoint. An examination of published studies reporting on outcomes using the FACT-O TOI now follows.
FACT-O TOI Clinical Trial Outcomes
Combination chemotherapy
A recent prospective phase II randomized clinical trial examined HRQOL as a secondary outcome in women with recurrent platinumsensitive ovarian cancer randomized to either docetaxel in combination with carboplatin or single agent docetaxel followed sequentially by carboplatin [28,29]. Although there was no difference in overall survival, there was significantly longer progression-free survival, more neurotoxicity, and more neutropenia in the combination arm [28]. However, the sequential treatment had significantly less impact on HRQOL outcomes. Specifically, the sequential arm had less impact on the FACT-O TOI over the course of the trial, compared to the combination arm [29]. In the combination arm, the TOI decreased by 4.9 points from baseline to study end, whereas in the sequential arm, TOI increased by 1.4 points. There was however, no difference between groups in median time to TOI deterioration [29]. These findings highlight a trade-off between better progression-free survival and greater toxicity and poorer HRQOL in the combination chemotherapy arm. Given that there was no significant difference in overall survival between the combination and sequential chemotherapy strategies, the TOI findings may inform treatment decision-making. This magnitude of change in the TOI from baseline to study end in each treatment arm may help to inform planning of future clinical trials utilizing the NFOSI-18 as a HRQOL outcome measure.
Intraperitoneal chemotherapy
Intraperitoneal (IP) chemotherapy confers a survival advantage to women with advanced ovarian cancer [30-32]. Although a phase III randomized trial found that intravenous (IV) paclitaxel plus IP cisplatin and paclitaxel significantly increased progression-free and overall survival when compared to IV-onlypaclitaxel and cisplatin [30], the FACT-O TOI was significantly worse in the IP group compared with the IV group before cycle four (10 point difference) and three to six weeks after treatment (7 point difference) [33]. Patients receiving IP therapy reported significantly and clinically meaningfully more physical, functional, and ovarian cancer-specific problems during treatment and shortly after, compared to IV-treated patients [33]. Of note, with the exception of the IP group prior to cycle four, both groups reported improved TOI over time, with no differences between the IP arm and the IV arm at one year [33]. Specifically, the TOI improved from 70.0 (baseline) to 83.2 (12 months) in the IV arm and from 64.5 (baseline) to 82.2 (12 months) in the IP arm. These findings highlight the fact that discussions about treatment decision-making must balance the potential survival advantages of IP chemotherapy with the shortterm HRQOL decrements associated with it. Moreover, magnitude of differences in TOI scores between treatment arms, as well as longitudinally over time, may provide guidance when planning future clinical trials that measure HRQOL outcomes using the NFOSI-18.
Novel biologic therapies
Although improvements have been made the in surgical and chemotherapeutic treatment strategies for women with advanced ovarian cancer, the limited availability of curative treatment options has prompted a search for alternative therapeutic agents, such as novel biologic therapies. Among the novel biologic therapies under development and investigation, the selective oral epidermal growth factor receptor tyrosine kinase inhibitor ZD1839 was examined in two Phase I trials among individuals with advanced ovarian cancer, as well as other advanced solid tumors [34]. ZD1839 acts by disrupting signaling pathways that are important in tumor growth [35]. LoRusso et al. report that in both Phase I trials of ZD1839, the median TOI for participants with advanced ovarian cancer deteriorated from baseline over time, although this was not the case for other solid tumor types in the trial [34]. Specifically, in the European/Australian trial, the overall TOI median deterioration from baseline was -4.50 [34]. The small number of ovarian participants in the U.S. trial limits interpretability of TOI change over time. These findings highlight the importance of examining HRQOL in addition to safety and tolerability when examining novel therapies. The magnitude of change in the TOI among ovarian cancer participants may provide context for the future use of the NFOSI-18 as a HRQOL outcome measure in trials examining novel biologic therapies.
Relevance of FACT-O TOI Outcome Findings to the NFOSI-18
Substantial overlap in item content between the NFOSI-18 and FACT-O TOI, and anticipated similarities in the range of what constitutes a clinically meaningful difference between the NFOSI-18 and other measures in the FACIT measurement system suggests good potential for cross-walk between these two measures of ovarian-cancer HRQOL. Thus, we anticipate that the published clinical trial FACT-O TOI outcomes can play a useful role in the future application of the NFOSI-18 in several important ways. First, an examination of betweengroup and within-group differences in FACT-O TOI scores over time provides a useful metric with which to set expectations for HRQOL differences by group and time when implementing the NFOSI-18. Second, the magnitude of change observed in published trials utilizing the FACT-O- TOI can also be of assistance when addressing questions of power calculation and sample size when planning for future trials that use the NFOSI-18 to measure HRQOL outcomes. Third, the difference in NFOSI-18 scores across performance status groups provides preliminary evidence to support the NFOSI-18’s ability to detect a magnitude of change or difference consistent with published findings on FACT-O TOI outcomes. Thus, as research begins to incorporate the NFOSI-18 for use as a measure of HRQOL in advanced ovarian cancer, the relevance of the FACT-O TOI findings to the NFOSI-18 may assist clinical researchers in study design and planning involving the use of the NFOSI-18.
Conclusion
The NFOSI-18 offers clinical providers and researchers a new tool for measuring patients’ response to treatment for advanced ovarian cancer. The benefits of the NFOSI-18 include its brevity, targeted measurement of the most important symptoms specific to advanced ovarian cancer, and enhanced adherence to FDA regulatory guidance. As such, it may be particularly appealing for use in clinical settings in which the minimization of patient and provider burden is critical, as well as clinical research in which conforming to regulatory guidance is essential. Another important distinction between the NFOSI-18 and existing measures of HRQOL in ovarian cancer is its patientcenteredness, given that patients—in addition to clinicians—played a role in item development and selection by ranking the most important symptoms. The primary limitation of the NFOSI-18 is the preliminary nature of research reporting on its use. However, given its redundancy with the FACT-O, which has been used in a number of published studies, we believe that published findings using item subsets common to both the FACT-O and the NFOSI-18 can be used to assist in the development of future research utilizing the NFOSI-18 as an advanced ovarian cancer specific HRQOL measure. Future research using the NFOSI-18 will further enhance its validation and interpretability. Additionally, although the NFOSI-18 and other NCCN symptom indexes are considered brief in nature, additional investigation needs to determine whether refinement is warranted to further reduce patient burden in both clinical research and clinical treatment contexts. Presently, little information exists regarding the extent to which the NCCN symptom indexes are employed in clinical practice settings both in the U.S. as well as internationally. It is anticipated that as the awareness of these measures increases, so will their use in both clinical and research contexts. Therefore, ongoing evaluation of their responsiveness to change, generalizability to more diverse patient samples, and generalizability across administration contexts (e.g., clinical trials versus clinical practice) constitute important future steps to further develop and establish the psychometric properties of these measures. Finally, given changes in symptom profiles as new treatment and supportive care interventions emerge, it will be imperative to periodically update the scales to ensure that they continue to reflect the current priority symptoms.
Acknowledgements
Support for the study was provided by grants from the following pharmaceutical companies: Amgen, AstraZeneca, Bayer, Bristol-Myers Squibb, Centocor, Cell Therapeutics, Inc., Clovis Oncology, Genentech, GlaxoSmithKline, Eli Lilly and Company, Merck & Co., Novartis, Ortho Biotech, Pfizer, Sanofi-Aventis and Takeda Pharmaceuticals. The contents represent original work. No commercial party having a direct financial interest in the results of the research supporting this article has or will confer a benefit upon the author(s) or upon any organization with which the author(s) is/are associated.
References
- Booth CM, Eisenhauer EA (2012) Progression-free survival: meaningful or simply measurable? J Clin Oncol 30: 1030-1033.
- Eton DT, Fairclough DL, Cella D, Yount SE, Bonomi P, et al. (2003) Early change in patient-reported health during lung cancer chemotherapy predicts clinical outcomes beyond those predicted by baseline report: results from Eastern Cooperative Oncology Group Study 5592. J ClinOncol 21: 1536-1543.
- Cella D, Cappelleri JC, Bushmakin A, Charbonneau C, Li JZ, et al. (2009) Quality of life predicts progression-free survival in patients with metastatic renal cell carcinoma treated with sunitinib versus interferon alfa. J Oncol Pract 5: 66-70.
- Carey MS, Bacon M, Tu D, Butler L, Bezjak A, et al. (2008) The prognostic effects of performance status and quality of life scores on progression-free survival and overall survival in advanced ovarian cancer. Gynecol Oncol 108: 100-105.
- Reck M, Thatcher N, Smit EF, Lorigan P, Szutowicz-Zielinska E, et al. (2012) Baseline quality of life and performance status as prognostic factors in patients with extensive-stage disease small cell lung cancer treated with pemetrexed plus carboplatin vs. etoposide plus carboplatin. Lung Cancer; 78: 276-281.
- Chase DM, Huang HQ, Wenzel L, Cella D, McQuellon R, et al. (2012) Quality of life and survival in advanced cervical cancer: a Gynecologic Oncology Group study. Gynecol Oncol 125: 315-319.
- Wenzel L, Huang HQ, Monk BJ, Rose PG, Cella D (2005) Quality-of-life comparisons in a randomized trial of interval secondary cytoreduction in advanced ovarian carcinoma: a Gynecologic Oncology Group study. J Clin Olncol 23: 5605-5612.
- Maisey NR, Norman A, Watson M, Allen MJ, Hill ME, et al. (2002). Baseline quality of life predicts survival in patients with advanced colorectal cancer. Eur J Cancer 38: 1351-1357.
- Chau I, Norman AR, Cunningham D, Waters JS, Oates J, et al. (2004) Multivariate prognostic factor analysis in locally advanced and metastatic esophago-gastric cancer--pooled analysis from three multicenter, randomized, controlled trials using individual patient data. J Clin Oncol 22: 2395-2403.
- Guidance for Industry: patient-reported outcome measures: use inmedical product development to support labeling claims. U.S. Foodand Drug Administration Web site.
- Taylor KM, Macdonald KG, Bezjak A, Ng P, DePetrillo AD (1996) Physicians' perspective on quality of life: an exploratory study of oncologists. Qual Life Res 5: 5-14.
- Bezjak A, Taylor KM, Ng P, MacDonald K, DePetrillo AD (1998) Quality-of-life information and clinical practice: the oncologist's perspective. Cancer Prev Control 2: 230-235.
- Gill TM, Feinstein AR (1994) A critical appraisal of the quality of quality-of-life measurements. JAMA 272: 619-626.
- Quality Of Life Subcommittee of the Oncologic Drugs Advisory Committee, February 10, 2000. U.S. Food and Drug Administration Web site.
- Cella D, Rosenbloom SK, Beaumont JL, Yount SE, Paul D et al. (2011) Development and Validation of 11 Symptom Indexes to Evaluate Response to Chemotherapy for Advanced Cancer. J Natl Compr Canc Netw 9: 268-278.
- Basen-Engquist K, Bodurka-Bevers D, Fitzgerald MA, Webster K, Cella D, et al. (2001) Reliability and validity of the functional assessment of cancer therapy-ovarian. J Clin Oncol 19: 1809-1817.
- Ferrell B, Cullinane CA, Ervine K, Melancon C, Umman GC, et al. (2005) Perspectives on the impact of ovarian cancer: women's views of quality of life. Oncol Nursing Forum 32: 1143-1149.
- Cull A, Howat S, Greimel E, Waldenstrom AC, Arraras J, et al. (2001) Development of a European Organization for Research and Treatment of Cancer questionnaire module to assess the quality of life of ovarian cancer patients in clinical trials: a progress report. Eur J Cancer 37: 47-53.
- Greimel E, Bottomley A, Cull A, Waldenstrom AC, Arraras J, et al. (2003) An international field study of the reliability and validity of a disease-specific questionnaire module (the QLQ-OV28) in assessing the quality of life of patients with ovarian cancer. Eur J Cancer 39: 1402-1408.
- Cella D, Nowinski CJ (2002) Measuring quality of life in chronic illness: the functional assessment of chronic illness therapy measurement system. Arch Phys Med Rehab 83: S10-S17.
- Yost KJ, Eton DT (2005) Combining distribution- and anchor-based approaches to determine minimally important differences: the FACIT experience. Eval Health Prof 28: 172-191.
- Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, et al. (1982) Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am Jof ClinOncol 5: 649-655.
- Jensen SE, Rosenbloom SK, Beaumont JL, Abernethy A, Jacobsen PB, et al. (2011) A new index of priority symptoms in advanced ovarian cancer. Gynecol Oncol 120: 214-219.
- Fairclough DL, Cella DF (1996). Functional Assessment of Cancer Therapy (FACT-G): non-response to individual questions. Qual Life Res 5: 321-329.
- American Cancer Society. Cancer Facts and Figures 2012. In. Atlanta: American Cancer Society 2012.
- Benedet JL, Bender H, Jones H, Ngan HY, Pecorelli S (2000) FIGO staging classifications and clinical practice guidelines in the management of gynecologic cancers. FIGO Committee on Gynecologic Oncology. Int J Gynaecol Oncol 70: 209-262.
- Jensen SE, Cella D Quality of life in patients with ovarian cancer. In Farghaly SA (Ed). Recent advances in diagnosis and management of ovarian cancer.
- Alvarez Secord A, Berchuck A, Higgins RV, Nycum LR, Kohler MF, et al. (2012) A multicenter, randomized, phase 2 clinical trial to evaluate the efficacy and safety of combination docetaxel and carboplatin and sequential therapy with docetaxel then carboplatin in patients with recurrent platinum-sensitive ovarian cancer. Cancer 118: 3283-3293.
- Pokrzywinski R, Secord AA, Havrilesky LJ, Puls LE, Holloway RW, et al. (2011) Health-related quality of life outcomes of docetaxel/carboplatin combination therapy vs. sequential therapy with docetaxel then carboplatin in patients with relapsed, platinum-sensitive ovarian cancer: Results from a randomized clinical trial. Gynecol Oncol 123: 505-510.
- Armstrong DK, Bundy B, Wenzel L, Huang HQ, Baergen R, et al. (2006) Intraperitoneal cisplatin and paclitaxel in ovarian cancer. NEJM 354: 34-43.
- Alberts DS, Liu PY, Hannigan EV, O’Toole R, Williams SD, et al. (1996) Intraperitoneal cisplatin plus intravenous cyclophosphamide versus intravenous cisplatin plus intravenous cyclophosphamide for stage III ovarian cancer. NEJM 335: 1950-1955.
- Markman M, Bundy BN, Alberts DS, Fowler JM, Clark-Pearson DL, et al. (2011) Phase III trial of standard-dose intravenous cisplatin plus paclitaxel versus moderately high-dose carboplatin followed by intravenous paclitaxel and intraperitoneal cisplatin in small-volume stage III ovarian carcinoma: an intergroup study of the Gynecologic Oncology Group, Southwestern Oncology Group, and Eastern Cooperative Oncology Group. J Clin Oncol 19: 1001-1007.
- Wenzel LB, Huang HQ, Armstrong DK, Walker JL, Cella D et al. (2007) Health-related quality of life during and after intraperitoneal versus intravenous chemotherapy for optimally debulked ovarian cancer: a Gynecologic Oncology Group Study.J Clin Oncol 25: 437-443.
- LoRusso PM, Herbst RS, Rischin D, Ranson M, Calvert H, et al. (2003) Improvements in quality of life and disease-related symptoms in phase I trials of the selective oral epidermal growth factor receptor tyrosine kinase inhibitor ZD1839 in non-small cell lung cancer and other solid tumors. Clin Cancer Res 9: 2040-2048.
- Baselga J, Averbuch SD. (2000) ZD1839 ('Iressa') as an anticancer agent. Drugs 60 Suppl 1: 33-40.
- Cella DF, Tulsky DS, Gray G, Sarafian B, Linn E, et al. (1993) The Functional Assessment of Cancer Therapy scale: development and validation of the general measure. J Clin Oncol 11: 570-579.
- Cella D, Paul D, Yount S, Winn R, Chang CH et al. (2003). What are the most important symptom targets when treating advanced cancer? A survey of providers in the National Comprehensive Cancer Network (NCCN). Cancer Invest 21: 526-535.
- Jensen SE, Beaumont JL, Jacobsen PB, Abernethy A, Syrjala K, et al. (in press). Measuring Priority Symptoms in Advanced Bladder Cancer: Development and Initial Validation of a Brief Symptom Index. J Support Oncol.
- Garcia SF, Rosenbloom SK, Beaumont JL, Merkel D, Von Roenn JH, et al. (2012). Priority symptoms in advanced breast cancer: development and initial validation of the National Comprehensive Cancer Network-Functional Assessment of Cancer Therapy-Breast Cancer Symptom Index (NFBSI-16). Value Health 15: 183-190.
- Salsman J, Cella D, Vergo M, Yount S Multidisciplinary approach to quality of life. In: Benson AB, Colon Cancer: A Multidisciplinary Approach to Diagnosis and Management. New York: Demos Medical Publishing.
- Odom D, Barber B, Bennett, L, Peeters M, Zhao Z, et al. (2011) Health-related quality of life and colorectal cancer-specific symptoms in patients with chemotherapy-refractory metastatic disease treated with panitumumab. IntJ Colorectal Dis 26: 173-181.
- Pearman TP, Beaumont JL, Paul D, Abernethy AP, Jacobsen PB, et al. (2012). Evaluation of Treatment- and Disease-Related Symptoms in Advanced Head and Neck Cancer: Validation of the National Comprehensive Cancer Network-Functional Assessment of Cancer Therapy-Head and Neck Cancer Symptom Index-22 (NFHNSI-22). J Pain Symptom Manage.
- Butt Z, Parikh ND, Beaumont JL, Rosenbloom SK, Syrjala K, et al. (2012) Development and validation of a symptom index for advanced hepatobiliary and pancreatic cancers: The National Comprehensive Cancer Network Functional Assessment of Cancer Therapy (NCCN-FACT) Hepatobiliary-Pancreatic Symptom Index (NFHSI). Cancer 118: 5997-6004.
- Yount S, Beaumont J, Rosenbloom S, Cella D, Patel J, et al. (2012) A brief symptom index for advanced lung cancer. Clin Lung Cancer 13: 14-23.
- Victorson DE, Beaumont JL, Rosenbloom SK, Shevrin D, Cella D (2011) Efficient assessment of the most important symptoms in advanced prostate cancer: the NCCN/FACT-P Symptom Index. Psycho-Oncology 20: 977-983.
Relevant Topics
- Caregiver Support Programs
- End of Life Care
- End-of-Life Communication
- Ethics in Palliative
- Euthanasia
- Family Caregiver
- Geriatric Care
- Holistic Care
- Home Care
- Hospice Care
- Hospice Palliative Care
- Old Age Care
- Palliative Care
- Palliative Care and Euthanasia
- Palliative Care Drugs
- Palliative Care in Oncology
- Palliative Care Medications
- Palliative Care Nursing
- Palliative Medicare
- Palliative Neurology
- Palliative Oncology
- Palliative Psychology
- Palliative Sedation
- Palliative Surgery
- Palliative Treatment
- Pediatric Palliative Care
- Volunteer Palliative Care
Recommended Journals
- Journal of Cardiac and Pulmonary Rehabilitation
- Journal of Community & Public Health Nursing
- Journal of Community & Public Health Nursing
- Journal of Health Care and Prevention
- Journal of Health Care and Prevention
- Journal of Paediatric Medicine & Surgery
- Journal of Paediatric Medicine & Surgery
- Journal of Pain & Relief
- Palliative Care & Medicine
- Journal of Pain & Relief
- Journal of Pediatric Neurological Disorders
- Neonatal and Pediatric Medicine
- Neonatal and Pediatric Medicine
- Neuroscience and Psychiatry: Open Access
- OMICS Journal of Radiology
- The Psychiatrist: Clinical and Therapeutic Journal
Article Tools
Article Usage
- Total views: 15125
- [From(publication date):
June-2013 - Aug 15, 2025] - Breakdown by view type
- HTML page views : 10472
- PDF downloads : 4653