Research Article Open Access
Association between Postpartum Depression and GnΒ3 C825T Polymorphism
Ri-hua Xie1,2, Hai-yan Xie3, Daniel Krewski2, Mark Walker4,5, Shi Wu Wen4,5
1School of Nursing, Hunan University of Medicine, Huaihua, P.R.China
2McLaughlin Centre for Population Health Risk Assessment, Institute of Population Health, University of Ottawa, Ottawa, Canada
3Department of Nursing, Social Work College, Changsha, P.R.China
4Department of Obstetrics and Gynecology, Faculty of Medicine, University of Ottawa, Canada
5Clinical Epidemiology Program, Ottawa Hospital Research Institute, Ottawa, Canada
- *Corresponding Author:
- E-mail: swwen@ohri.ca
Visit for more related articles at International Journal of Emergency Mental Health and Human Resilience
Abstract
Background: Postpartum depression (PPD), affecting mother’s and baby’s health and wellbeing, is a subtype of major depression with onset within 4 weeks after childbirth. G-protein β3 subunit gene C825T polymorphism has been shown to be associated with major adult depression. This study aims to investigate whether PPD is associated with GNβ3 C825T polymorphism in Chinese Han women. Methods: This was a case control study, nested to a cohort that was established in three hospitals in Changsha, Hunan, China between February and September 2007. PPD was measured using the Chinese version of Edinburgh Postnatal Depression Scale (EPDS) at two weeks postpartum with a score of 13 or higher as the cut-off. GNB3 gene Rs5443 (C/T) was determined with ligase detection reaction (LDR). Genotype and allele frequency of GNB3 gene Rs5443 polymorphism were compared between women with PPD and hospital and age-matched controls. Results: A total of 43 PPD cases and 86 matched controls were included in the final analysis. GNβ3 Rs5443SNP genotypes were CC = 23.30%, C/T = 51.2%, TT = 25.6%, C = 48.84%, and T = 51.16%, respectively, in PPD cases, and corresponding figures were CC = 30.20%, C/T = 39.50%, TT = 30.20%, C = 50.00%, and T = 50.00%, respectively, in matched controls (P>0.05). For PPD subjects, the genotypes were not associated with the clinical phenotype: EPDS total score and/or subunit scores (P>0.05). Conclusion: No association between GNβ3 rs5443SNP polymorphism and PPD is observed in the pilot data from a small sample of Chinese Han women. Replications of our finding from a large sample are needed.
Keywords
Postpartum depression, GNβ3, gene, polymorphism
Introduction
Postpartum depression (PPD) is a subtype of major depression with onset within 4 weeks after childbirth (American Psychiatric Association, 2000). Prevalence of PPD ranges from 10% - 20% worldwide, including those reported in China (Xie et al., 2007; 2009; 2011). Previous studies have identified several environmental risk factors of PPD (Glasser et al., 2000; O’Hara, Schlechte, Lewis, & Varner, 1991; Robertson, Grace, Wallington, & Stewart, 2004; Xie et al., 2007; 2009; 2011). Genetic factors may also contribute to the development of PPD (Comasco et al., 2012; Fasching et al., 2012; Mehta et al., 2012; Skalkidou et al., 2012).
Heterotrimeric guanine nucleotide-binding proteins (G proteins) play important roles in transducing transmembrane signal and regulating secondary messenger pathways for a variety of membrance-bound receptors. G-protein complex is composed of α,β,γ subunits. On receptor activation, G proteins dissociate into free G α and G β γ subunits that can modulate various intracellular effectors. Shiffert et al., (1998) described a single-nucleotide polymorphism (SNP) of C825T in exon 10 of the gene encoding the β3 subunit of heterotrimeric G- proteins (GNβ3 or rs5443), which is located on chromosome 12p13, about 7.5 kilobase (kb) long with11 exons and 10 introns (Siffert et al., 1998). The T allele of this SNP is related to the occurrence of a splice variant (Gβ3s), which causes the deletion of 41 amino acids. The splice variant Gβ3s resulted in an increased signal transduction (Siffert et al., 1998) and an increased risk of affective disorders (Avissar & Schreiber, 1992; Avissar et al., 1996; 1999). Recently, a number of studies found that GNβ3 C825T was associated with disease development and the response to antidepressant treatment for major depressive disorders (Zill et al., 2000; Joyce et al., 2003; Serretti et al., 2003; Lee et al., 2004; Wilkie et al., 2007; Cao, Hu, Zhang, & Xia, 2007). The results of these studies showed that the frequency of T alleles was significantly higher in depressive patients than that in healthy controls. In addition, they also revealed that depressive patients with T allele had severe symptoms and a better response to antidepressant treatment (Zill et al., 2000; Joyce et al., 2003; Serretti et al., 2003; Lee et al., 2004; Wilkie et al., 2007; Cao, Hu, Zhang, & Xia, 2007). A meta-analysis by Lopez-Leon et al., (2008) found statistically significant association of GNB3 825T (OR 1.38) with major depressive disorder. In another meta-analysis, Hu et al., (2014) concluded that the GNB3 C825T polymorphism was significantly correlated with a higher response rate to antidepressants in major depressive disorder, and ethnicitystratified analysis indicated that GNB3 C825T polymorphisms may be strongly related to the efficacy of antidepressants in the treatment of major depressive disorder among Asians than in Caucasians. On the other hand, a meta-analysis by Niitsu et al., (2013) suggested that no major effect of any single gene variant on antidepressants efficacy. The aim of the present study is to assess whether PPD is associated with GNβ3 rs5443SNP in a pilot study of Chinese Han women.
Methods
Study Subjects
This was a case control study, nested to a prospective cohort study conducted in Changsha, Hunan, China, from February to September 2007. Subjects were recruited in Hunan Maternal and Infant Hospital and the First Affiliated and the Third Affiliated Hospitals of the Central South University in Changsha, Hunan, People’s Republic of China, during their prenatal visits at 30 to 32 weeks of gestation. Primiparous and married women aged 20-45 years presenting in the three above-mentioned participating hospitals for prenatal care and planning to stay in Changsha city during the postpartum period were invited to for this study. Demographic data, including women’s ethnic background, were obtained by structured interview by research nurses of the study.
The Chinese version of Edinburgh Postnatal Depression Scale (EPDS), a PPD screening tool originally developed and validated in English environment and was later translated and validated in Chinese environment (Cox & Holden, 1987; Lee, Yip, & Chiu, 1998), was used at two weeks postpartum to assess PPD status with a cut-off score over or equal to 13. Participating women with history of bipolar disorder, schizophrenia or other mental illnesses, a major chronic disease, or obstetric and pregnancy complication (severe preeclampsia/eclampsia, placenta previa, placental abruption, major postpartum infection, still birth, major birth defects, or birth weight less than 1,500 g) as recorded in medical charts were excluded. All participants were Han Chinese in origin and signed the informed consent. A sample was randomly selected from PPD cases. For each PPD case, two women with no PPD, matched by hospital for childbirth (i.e., who gave birth at the same hospital as the corresponding PPD case) and age (± 5 y). Approval for this study was obtained from the Research Ethics Board of Central South University.
DNA Extraction
Approximately 3 ml of peripheral blood was collected from the study participants at two weeks postpartum using EDTA anticoagulant venous blood tubes. Genomic DNAs were isolated from peripheral blood with the phenol-chloroform extraction method.
Genotyping
The genetic polymorphism (Genbank dbSNP: rs5443) was typed by Multiplex Polymerase chain reaction (PCR) and multiple Ligase Detection Reaction (LDR). The genotyping experiment was conducted by Shanghai Bio Wing Applied Biotechnology Company (http://www.biowing.com.cn/) using ligase detection reactions (LDR). The target DNA sequences were amplified using a multiplex PCR method. After the completion of the amplification, 1 ul of Proteinase K (20 mg/ml) was added, then heated at 70°C for 10 min and quenched at 94°C for 15 min. The LDR was performed in a solution (10ul) containing 1ul Tris-HCl (pH 8.5), 25 mM potassium acetate, 10 mM magnesium acetate, 10 mMDTT, 1 mM NAD, 0.1% Triton X-100, 0.5 pm of each LDR probe, 1ul PCR products, and 0.05 ul of 40 U/ ul Taq DNA ligase (New England Biolabs, USA).
Statistical Analysis
Socio-demography, perinatal characteristics were compared using Chi-square test (or Fisher’ Exact test if needed). Whether GNβ3 rs5443SNP genotype distribution was in equilibrium was investigated using the Hardy-Weinberg test. Independent association between PPD with genotype and allele frequencies were examined using multiple logistic regression analysis, with odds ratio (OR) and 95% confidence interval (CI) expressed as the effect measures. Potential confounding variables adjusted in the logistic regression analysis included maternal education, income, and mode of delivery. ANOVA was used to assess the associations between EPDS score and genotypes in PPD cases. All analyses were performed using SPSS for windows (Version 13.0; SPSS, Chicago, IL, USA).
Results
Between February and September 2007, a total of 666 women were invited to participate in the study and 634 women agreed and completed the prenatal survey. At 2 weeks postpartum survey, of them, 24 women withdrew, 25 lost to follow up, and 10 had missing information over 20% of the variables, and 41 women were excluded because of recorded major psychiatric disorders and severe obstetric and/or pregnancy complications. Thus, a total of 534 (86.8%) women were included in this cohort study. Of the 534 women, 103 women (19.3%) had PPD. Because of budget restriction, we selected only 43 PPD cases (randomly) from all 103 PPD cases for genetic testing. For each selected women with PPD, two women with no PPD, matched by hospital for childbirth (same hospital) and age (± 5 y) for genetic testing. As a result, a total of 43 PPD cases and 86 controls were tested for genetic polymorphism.
A genetic testing was done in 43 samples per batch. To reduce lab cost of the study and to maintain a reasonable study power, we elected to use a 1:2 case control ratio with 43 samples per batch.
Socio-demographic and perinatal aspects of PPD cases and controls are shown in Table 1.
| Characteristics | PPD group (n=43) | Control group (n=86) | χ2 | P |
|---|---|---|---|---|
Age (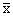 ± ï½�?) ± ï½�?) |
27.53 ± 3.58 | 28.15 ± 3.40 | t =0.95 | 0.34 |
| Education | ||||
| University or higher | 7 | 37 | 9.72 | 0.008** |
| College | 14 | 23 | ||
| High School or lower | 22 | 26 | ||
| Occupation | ||||
| Public servant, Professional | 12 | 32 | 5.26 | 0.15 |
| Worker, Clerk | 11 | 29 | ||
| Farmer | 13 | 20 | ||
| Others | 7 | 5 | ||
| Income (per month per member) | ||||
| ≥2000Yuan | 14 | 56 | 12.26 | 0.002** |
| 1000~2000Yuan | 18 | 19 | ||
| ≤1000Yuan | 11 | 11 | ||
| Housing | ||||
| Satisfactory | 38 | 78 | 0.17 | 0.68 |
| Unsatisfactory | 5 | 8 | ||
| Planned pregnancy | ||||
| Yes | 31 | 68 | 0.78 | 0.38 |
| No | 12 | 18 | ||
| Gravidity | ||||
| 1 | 18 | 41 | 2.80 | 0.25 |
| 2 | 7 | 21 | ||
| ≥3 | 18 | 24 | ||
| Use of pain relief | ||||
| Yes | 38 | 72 | 0.49 | 0.48 |
| No | 5 | 14 | ||
| Mode of delivery | ||||
| Vaginal | 4 | 22 | 0.63 | 0.04* |
| Caesarean | 39 | 64 | ||
Birth weight (g) (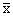 ± ï½�?) ± ï½�?) |
3216.74 ± 542.49 | 3325.58 ± 428.11 | 0.10 | 0.92 |
| Fetal gender | ||||
| Male | 26 | 56 | 0.27 | 0.61 |
| Female | 17 | 30 |
Table 1: Comparison of socio-demographic and perinatal characteristics between PPD group and control group
The mean (SD) of age was 27.53 (± 3.58) years in PPD cases and 28.15 (± 3.40) years in controls. Education level and income in PPD case were much lower than those in controls. For mode of delivery, there was a significant difference (p = 0.04), 4 were vaginal delivery and 39 PPD case were caesarean delivery in PPD cases, while vaginal delivery were 22 and 64 caesarean section in controls. There were no differences in other socio-demographic and perinatal aspects of the two groups.
The GNβ3 rs5443SNP genotype distribution in our samples did not deviate significantly from the Hardy-Weinberg equilibrium (χ2 = 3.44, P = 0.18)
Genotype and allele frequencies of the polymorphism in rs5443SNP in PPD cases and controls are shown in Table 2. There were no associations of PPD with GNβ3 rs5443SNP genotypes and allele frequencies. Adjustment for maternal education, income, and mode of delivery did not change the associations.
| genotype frequency�?%�? | Allele frequency�?%�? | ||||
|---|---|---|---|---|---|
| CC | C/T | TT | C | T | |
| PPD group (n=43) | 10 (23.30) | 22 (51.20) | 11 (25.60) | 42 (48.84) | 44 (51.16) |
| Control group (n=86) | 26 (30.20) | 34 (39.50) | 26 (30.20) | 86 (50.00) | 86 (50.00) |
| Unadjusted OR (95% CI) | 0.91 (0.33, 2.51) | 1.53 (0.63, 3.71) | Reference | 0.96 (0.57,1.60) | Reference |
| Adjusted OR (95% CI)* | 0.85 (0.29, 2.51) | 1.53 (0.60, 3.95) | Reference | 0.92 (0.53, 1.61) | Reference |
Table 2: Comparisons of genotype and allele frequencies of the polymorphism in rs5443SNP between PPD group and control group
Associations of clinical characteristics with genotype in rs5443SNP in PPD cases are shown in Table 3. The clinical phenotypes were not associated with age or EPDS total score or subunit scores (P>0.05).
| Phenotype | CC (n=10) | C/T (n=22) | TT (n=11) | χ2/F | p |
|---|---|---|---|---|---|
| Age | 28.90 ± 4.75 | 27.14 ± 2.55 | 27.09 ± 4.18 | 0.95 | 0.38 |
| EPDS | 15.10 ± 2.42 | 15.45 ± 2.46 | 15.82 ± 3.37 | 0.18 | 0.83 |
| Item 1 | 1.10 ± 1.10 | 1.18 ± 0.73 | 1.18 ± 0.87 | 0.03 | 0.97 |
| Item 2 | 0.90 ± 0.74 | 1.32 ± 0.95 | 1.00 ± 1.18 | 0.79 | 0.46 |
| Item 3 | 2.10 ± 0.74 | 2.09 ± 0.75 | 2.27 ± 0.65 | 0.25 | 0.78 |
| Item 4 | 2.20 ± 0.42 | 2.00 ± 0.44 | 1.91 ± 0.30 | 1.44 | 0.25 |
| Item 5 | 1.80 ± 0.63 | 1.95 ± 0.65 | 1.64 ± 0.50 | 1.00 | 0.37 |
| Item 6 | 2.00 ± 0.94 | 1.59 ± 0.67 | 1.64 ± 0.67 | 1.10 | 0.34 |
| Item 7 | 2.00 ± 0.82 | 1.77 ± 0.81 | 1.82 ± 0.75 | 0.28 | 0.76 |
| Item 8 | 1.10 ± 0.57 | 1.32 ± 0.65 | 1.27 ± 0.65 | 0.42 | 0.66 |
| Item 9 | 1.00 ± 0.47 | 1.09 ± 0.43 | 1.45 ± 0.69 | 2.49 | 0.10 |
| Item 10 | 0.90 ± 0.74 | 1.09 ± 0.75 | 1.64 ± 0.67 | 3.06 | 0.06 |
*Item 1: I have been able to laugh and see the funny side of things; Item 2: I have looked forward with enjoyment to things; Item 3: I have blamed myself unnecessarily when things went wrong; Item 4: I have been anxious or worried for no good reason; Item 5: I have felt scared or panicky for no very good reason; Item 6: Things have been getting on top of me; Item 7: I have been so unhappy that I have had difficulty sleeping; Item 8: I have felt sad or miserable; Item 9: I have been so unhappy that I have been crying; Item 10: The thought of harming myself has occurred to me.
Table 3: Associations between clinical phenotype and genotype in GNβ3 gene rs5443SNP in PPD cases (n = 43)
Discussion
In this case control study nested to a cohort of 534 Chinese Han women, we found that there were no significant difference in the frequencies of the allele (C, T) and the rs5443SNP polymorphism gene between PPD cases and controls. Among PPD cases, clinical phenotypes were not associated with age, and EPDS total score or subunit scores. To our knowledge, very few studies have assessed the association between rs5443SNP polymorphism gene and PPD. Our earlier studies of the same cohort of Chinese Han women identified several environmental risk factors of PPD, such as cesarean delivery (Xie et al., 2011) and lack of social support (Xie et al., 2007; 2009), which were consistent with previous studies on environmental risk factors of PPD, such as perinatal events and lack of social support (Glasser et al., 2000; O’Hara et al., 1991; Robertson, Grace, Wallington, & Stewart, 2004).
On the other hand, previous studies on major adult depression suggested that rs5443SNP polymorphism gene play an important role in the pathogenesis of adult major depression (Zill et al., 2000; Joyce et al., 2003; Serretti et al., 2003; Lee et al., 2004; Wilkie et al., 2007, Cao, Hu, Zhang, & Xia, 2007; Lopez-Léon et al., 2008; Niitsu, Fabbri, Bentini, & Serrett, 2013; Hu et al., 2014). Zill et al., (2000) investigated whether this GNβ3 polymorphism was associated with affective disorders or with the response to antidepressant treatment in 88 depressive patients (10 bipolar disorder, 78 major depression) compared with 68 schizophrenic patients and 111 healthy controls. The results showed that there was a significantly higher frequency of the T allele in depressive patients than that in healthy controls and schizophrenic patients. Lee et al., (2004) conducted a similar study in 106 major depressive disorders and 133 healthy controls (hypertensive subjects were excluded from the study because association between GNβ3 variants). The study showed significantly more carriers of the 825T allele in major depressive disorders patients than those in normal controls ((P = 0.012; OR = 2.19, 95% CI 1.18-4.05). The T-allele carriers showed higher scores than those with the CC genotype in some subcategories of Hamilton Depression Rating Scale (P<0.05. A study in a Chinese population (Xiao, Yao, & Guo, 2003) indicated that there was a higher frequency of the TT fragment in depressive patients group than that in controls, and the depressive patients with TT fragment had significantly decrease in total score of Hamilton Depression Rating Scale after four weeks antidepressant treatment.
In addition, a body of studies also identified several other genes that may associate with PPD. In a cohort study involving 361 Caucasian women, Fasching et al., assessed the severity of depression prospectively during pregnancy (third trimester) and postpartum period (2-3 days and 6-8 months) using EPDS and analyzed the association of EPDS values over time with haplotype block in the promoter region of TPH2 (Fasching et al., 2012). The results revealed significant associations of TPH2 with depression values (measured by EPDS) during pregnancy and 6-8 months afterwards. The effect of TPH2 haplotypes on EPDS values was strongest during pregnancy and 6 months after birth, with a low depression rating in the first few days after delivery (Fasching et al., 2012). In a case control study comprised of 275 women from a population-based cohort of delivering women in Sweden who completed a questionnaire containing EPDS at 6 weeks and 6 months postpartum, Comasco et al., (2012) examined whether functional polymorphic variants, BDNF Val66Met, 5-HTTLPR, or PER2 SNP 10870, were associated with PPD symptoms and whether these genetic polymorphisms interact with season in predicting PPD symptoms. A significant association between BDNF Met66 carrier status and development of PPD symptoms at 6 weeks postpartum, even when controlling for prenatal and postpartum environmental risk factors, was observed among mothers who delivered during autumn/winter (Comasco et al., 2012). In a non-psychiatric cohort of 419 Caucasians, Mehta et al assessed the association between 5-HTTLPR S-allele carrier statuses with severity of depression during pregnancy (3rd trimester) and the postpartum period (2-3 days and 6-8 months) (Mehta et al., 2012). They found that 5-HTTLPR S-allele carrier status predicted late postpartum depressive symptom severity only in the presence of negative life events (Mehta et al., 2012).
There are several limitations in our study. First, the sample size was small, with limited power (<50%) to detect the association (if any). Second, DNA extraction process and genotyping were operator dependent and were subject to human errors. Although laboratory procedures were blinded (operators were not aware of the PPD status of the subjects), random error may further lower the study power. Third, EPDS is a screening tool, not a diagnostic tool.
To summarize, our pilot study in a small sample of Chinese Han women did not find an association between GNβ3 gene polymorphism and PPD. Replications of the study findings of this pilot study with a larger sample size and with the recently updated diagnostic tool (DSM-V; American Psychiatric Association, 2013) for PPD are needed. Such an exercise would be helpful not only for the advancement in the science of depression, but for the development of prevention strategies.
Acknowledgements
This study was supported by grants from Hunan Provincial Natural Science Foundation (06JJ4055), Hunan Ministry of Science and Technology (06FJ4103), and Hunan Ministry of Education (06C072) of the People’s Republic of China. We thank the pregnant women and the staff of the participating hospitals in Changsha, China for their support.
References
- American Psychiatric Association. (2000). Diagnostic and Statistical Manual of Mental Disorders (DSM), 4th Edn. Text revision. Washington DC: American Psychiatric Association
- American Psychiatric Association. (2013). Diagnostic and Statistical Manual of Mental Disorders (DSM), 5th Edn. Text revision. Washington DC: American Psychiatric Association
- Avissar, S., Barki-Harrington, L., Nechamkin, Y., Roitman, G., & Schreiber, G. (1996). Reduced beta-adrenergic receptor-coupled Gs protein function and Gs alpha immunoreactivity in mononuclear leukocytes of patients with depression. Biological Psychiatry, 39, 755-760
- Avissar, S., & Schreiber, G. (1992). The involvement of guanine nucleotide binding proteins in the pathogenesis and treatment of affective disorders. Biological Psychiatry, 31, 435-459
- Avissar, S., Schreiber, G., Nechamkin, Y., Neuhaus, I., Lam, G.K., Schwartz, P., et al. (1999). The effects of seasons and light therapy on G protein levels in mononuclear leukocytes of patients with seasonal affective disorder. Archives of General Psychiatry, 56, 178-183
- Cao, M.Q., Hu, S.Y., Zhang, C.H., & Xia, D.S. (2007). Study on the interrelationship between 5-HTTLPR/G-protein beta3 subunit (C825T) polymorphisms and depressive disorder. Psychiatric Genetics, 17(4), 233-238
- Comasco, E., Sylvén, S.M., Papadopoulos, F.C., Oreland, L., Sundström-Poromaa, I., & Skalkidou, A. (2011). Postpartum depressive symptoms and the BDNF Val66Met functional polymorphism:effect of season of delivery. Archives of Women's Mental Health, 14(6), 453-463
- Cox, J., & Holden, J. (1987). Detection of postnatal depression-Development of the 10-item Edinburgh Postnatal Depression Scale. British Journalof Psychiatry, 150, 782-786
- Fasching, P.A., Faschingbauer, F., Goecke, T.W., Engel, A., Häberle, L., et al. (2012). Genetic variants in the tryptophan hydroxylase 2 gene (TPH2) and depression during and after pregnancy. Journal of Psychiatric Research, 46(9), 1109-1117
- Glasser, S., Barell, V., Boyko, V., Ziv, A., & Lusky, A. (2000). Postpartum depression in an Israeli cohort:demographic, psychosocial and medical risk factors. Journal of psychosomatic obstetrics and gynaecology, 21, 99-108
- Hu, Q., Zhang, S.Y., Liu, F., Zhang, X.J., Cui, G.C., Yu, E.Q., et al. (2014). Influence of GNB3 C825T polymorphism on the efficacy of antidepressants in the treatment of major depressive disorder: A meta-analysis. Journal of Affective Disorders. 72, 103-109
- Joyce, P.R., Mulder, R.T., Luty, S.E., McKenzie, J.M., Miller, A.L., Rogers, G.R., et al. (2003). Age-dependent antide- pressant pharmacogenomics: polymorphisms of the serotonin transporter and G protein beta3 subunit as predictors of response to fluoxetine and nortriptyline. International Journal of Neuropsychopharmacology, 6, 339-346
- Lee, D.T.S., Yip, S.K., & Chiu, H.F.K. (1998). Detecting postnatal depression in Chinese women. British Journal of Psychiatry, 172(5), 433-437
- Lee, H.J., Cha, J.H., Ham, B.J., Han, C.S., Kim, Y.K., Lee, S.H., et al. (2004). Association between a G- protein beta 3 subunit gene polymorphism and the symptomatology and treatment responses of major depressive disorders. Pharmacogenomics Journal, 4, 29-33
- López-León, S., Janssens, A.C., González-Zuloeta, L.A.M., Del-Favero, J., Claes, S.J., Oostra, B.A., et al. (2008). Meta-analyses of genetic studies on major depressive disorder. Molecular Psychiatry, 13(8), 772-785
- Mehta, D., Quast, C., Fasching, P.A., Seifert, A., Voigt, F., Beckmann, M.W., et al. (2012). The 5-HTTLPR polymorphism modulates the influence on environmental stressors on peripartum depression symptoms. Journal of Affective Disorders, 136(3), 1192-1197
- Niitsu, T., Fabbri, C., Bentini, F., & Serretti, A. (2013). Pharmacogenetics in major depression: a comprehensive meta-analysis. Progress in Neuro-Psychopharmacology & Biological Psychiatry, 45, 183-194
- O’Hara, M.W., Schlechte, J.A., Lewis, D.A., Varner, M.W. (1991). Controlled prospective study of postpartum mood disorders: psychological, environmental, and hormonal variables. Journal of Abnormal Psychology, 100, 63-73
- Robertson, E., Grace, S., Wallington, T., Stewart, D.E. (2004). Antenatal risk factors for postpartum depression: a synthesis of recent literature. General Hospital Psychiatry, 26, 289-295
- Serretti, A., Lorenzi, C., Cusin, C., Zanardi, R., Lattuada, E., Rossini, D., et al. (2003). SSRIs antidepressant activity is influenced by G beta 3 variants. European Neuropsychopharmacology, 13, 117-122
- Skalkidou, A., Hellgren, C., Comasco, E., Sylvén, S., & Sundström P.I., (2012). Biological aspects of postpartum depression. Womens Health (Lond Engl), 8(6), 659-672
- Siffert, W., Rosskopf, D., Siffert, G., Busch, S., Moritz, A., Erbel, R. et al. (1998). Association of a human G-protein beta3 subunit variant with hypertension. Nature Genetics, 18, 45-48
- Wilkie, M.J., Smith, D., Reid, I.C., Day, R.K., Matthews, K., Wolf, C.R., et al. (2007). A splice site polymor- phism in the G-protein beta subunit influences antidepressant efficacy in depression. Pharmacogenetics and Genomics, 17, 207-215
- Xiao, H., Yao, H., & Guo, S.H. (2003).The Gβ3 gene C825T polymorphism of and response to antidepressant treatment. Chinese Journal of Clinical Pharmacy, 12(2), 65-68
- Xie, R.H., He, G., Koszycki, D., Walker, M., & Wen, S.W. (2009). Prenatal social support, postnatal social support, and postpartum depression. Annals of Epidemiology, 19(9), 637-643
- Xie, R.H., He, G., Liu, A., Bradwejn, J., Walker, M., & Wen, S.W. (2007). Fetal gender and postpartum depression in a cohort of Chinese women. Social Science & Medicine, 65, 680-684
- Xie, R.H., Lei, J., Wang, S., Xie, H., Walker, M., & Wen, S.W. (2011). Cesarean section and postpartum depression in a cohort of Chinese women with a high cesarean delivery rate. Journal of Womens Health, 20(12), 1881-1886
- Zill, P., Baghai, T.C., Zwanzger, P., Schüle, C., Minov, C., Riedel, M., et al. (2000). Evidence for an association between a G-protein beta3-gene variant with depression and response to antidepressant treatment. Neuroreport, 11, 1893-1897.
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 14234
- [From(publication date):
September-2015 - Aug 15, 2025] - Breakdown by view type
- HTML page views : 9532
- PDF downloads : 4702
