Correlation Between Simultaneous Ultrasound-Guided Fine Needle Aspiration and Core Needle Biopsy Results in Thyroid Nodules of 44 Cases
Received: 30-Jan-2017 / Accepted Date: 11-Feb-2017 / Published Date: 17-Feb-2017 DOI: 10.4172/2476-2024.1000123
Abstract
Ultrasound (US)-guided fine needle aspiration biopsy (FNA) is currently the most common diagnostic method for determining surgical cases and preventing unnecessary surgery as it is easy, reliable and cost-effective. However, current multi-center studies indicate that about 5-47% of the cases that have undergone FNA are evaluated as inadequate/indeterminate for diagnosis due to factors related to the assessor, the technique and the pathologist's experience. It is reported that using US-guided percutaneous core needle biopsy (CNB) is useful, especially in cases where diagnostic surgery is planned as the cases have been reported as ‘Inadequate for diagnosis’ or ‘Atypia with unclear in significance/ Follicular lesion with unclear in significance(AUS/FLUS) after inadequate repetitive FNA results. Using FNA and CNB together has been reported to provide higher diagnostic sensitivity than either method alone in certain studies. The aim of this study is prospective evaluation of simultaneous US-guided FNA and CNB results of 44 cases who had thyroid nodules. All cases were evaluated according to the TIRADS classification used for sonographic malignancy risk categorization and recorded together with the demographic data (age/gender). FNAs were evaluated according to the Bethesda classification. We tried to adapt the results of the evaluated CNBs descriptively to the Bethesda classification to facilitate comparison of the methods. In our study, when the inadequate and indeterminate diagnosis groups were separated, all our cases diagnosed as benign and malignant with FNA and CNB were compliant. However, the ratio of insufficiency shows a significant elevation compared to CNB in our FNA procedures (4.54% vs. 43.18%). We observed that simultaneous FNA and CNB use decreased the inadequate/ indeterminate diagnosis group significantly.
Introduction
Ultrasound (US)-guided fine needle aspiration biopsy (FNA) is currently the most common diagnostic method for determining surgical cases and preventing unnecessary surgery as it is easy, reliable and cost-effective. However, current multi-center studies indicate that about 5-47% of the cases that have undergone FNA are evaluated as inadequate/indeterminate for diagnosis due to factors related to the assessor, the technique and the pathologist's experience [1,2]. Repeated FNAs are recommended to solve this problem as it is diagnostic in 13-61.8% of the cases [2-6].
It is also reported that using US-guided percutaneous core needle biopsy (CNB) is useful, especially in cases where diagnostic surgery is planned due to the fact that the cases have been reported as ‘Inadequate for diagnosis’ or ‘Atypia of uncertain significance/ Follicular lesion of uncertain significance (AUS/FLUS)’ [5-16]. Using FNA and CNB together has been reported to provide higher diagnostic sensitivity than either method alone in certain studies [9,12,17]. However, such use has not been emphasized due to the high rate of complications and difficulty in technical implementation [3]. Today, its use as an alternative method to FNA is becoming more common thanks to high-resolution US-guidance and new needles.
For us, the pathologists, the advantage of this method is the opportunity provided for immunohistochemical (IHC) and adjunct molecular studies in addition to histological examination. However, a disadvantage of the CNB histological evaluation is that standardized diagnostic criteria similar to the Bethesda criteria have not been yet identified [4].
There was only one study in which simultaneous FNA and CNB results were compared to understand usefulness of the two techniques together [13]. Our aim in this study was to evaluate the diagnostic sensitivity and consistency of simultaneous US-guided FNA and CNB results in our unit. According to the results of these kind of studies, FNA and CNB methods can be used simultaneously in our daily routine to decrease the ratio of inadequate samples.
Materials and Methods
A total of 44 cases where FNA and CNB methods had been used simultaneously between September and November 2014 were included in the study. There were no cases that were excluded. CNB method was identical to the method that was used in the current study of Guler et al. [18]. After the CNB procedure FNA was performed with 22G needles. All cases were evaluated according to the TIRADS classification used for sonographic malignancy risk categorization and recorded together with the demographic data (age/gender) [19,20]. If available, the previous FNAs of the cases were re-investigated and included in the study.
According to the radiological classification termed “Thyroid image reporting and data system (TIRADS)” used by our surgical department, TIRADS 1 identifies normal thyroid tissue, TIRADS 2 isohypoechoic and well-circumscribed nodules-Benign, TIRADS 3 hypoechoic, cystic/solid areas and well-circumscribed nodules– probably benign, TIRADS 4 a minimum of two solid components, hypoechoic, irregular borders, microcalcifications or taller than wide shape-Suspicious, TIRADS 5 a minimum of four solid components, hypoechoic, irregular borders, microcalcifications or taller than wide shape-Probably malignant, and TIRADS 6 malignant cases proven by biopsy [20].
FNAs were evaluated according to the Bethesda classification [2]. We adapted the results of the evaluated CNBs descriptively to the Bethesda classification to facilitate comparison of the two methods. Stromal or other tissue samples not containing a follicular structure were classified as inadequate. Cases having no structural or nuclear papillary carcinoma characteristics but showing nuclear coarsening of unknown significance and groove presence, or rare micro follicular architecture on a colloid-poor background and that had suspicious focal (non-membranous, luminal staining) IHC HBME-1 positivity were included in the AUS/FLUS category.
The CNB samples sent consisted of a minimum of 2 cores and were fixed with formalin (10% buffered formalin). Biopsy samples were processed with routine automatic procedures and were evaluated with serial hematoxylin-eosin (HE)-stained profiles of 2 microns thickness. All cases also underwent IHC HBME1 staining (Mesothelioma Ab-1, Mouse, ready to use, Thermo Scientific, USA) in order to investigate its benefit in the diagnosis of CNB samples. The FNA samples used after the CNB procedure were smeared onto 2-4 slides, fixed in alcohol (96%) and PAP stained.
The diagnostic correlation and the histological characteristics of the material obtained from cases that underwent resection were compared with the morphological characteristics observed in the FNA/CNB samples.
Results
The 44 cases that underwent simultaneous FNA and CNB within the scope of the study consisted of 43 females and 1 male. The age distribution was 19-76 years and the mean age was 48 years.
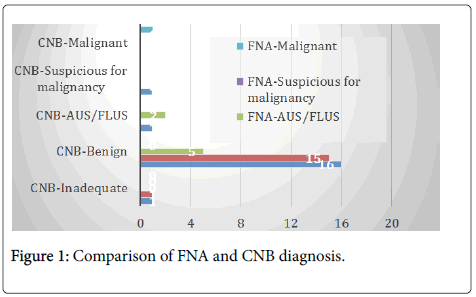
We had only 1 case diagnosed as malignant with FNA (100%) and it also received the same diagnosis with CNB. However, the resected material of this case was not referred to our department.
One out of 2 cases thought to be suspicious for malignancy with CNB was also found as suspicious for malignancy with FNA. The other case was included in the inadequate sample group and the resection result of both cases was malignant.
FNA smears that were reported as AUS/FLUS were distributes as 1 in the malignant category, 5 in the benign category, and 2 in the AUS/ FLUS category with CNB. The resection material of 1 case diagnosed with CNB as malignant, FNA-AUS/FLUS, was again diagnosed as malignant.
We had 3 cases included in the AUS/FLUS category with CNB, and of those diagnosed with FNA only 2 were AUS/FLUS.
Of the 16 cases diagnosed as benign with FNA, 15 were reported as benign and 1 as inadequate sample with CNB.
Of the 36 cases diagnosed as benign with CNB, 15 were also diagnosed as benign with FNA but 5 were interpreted in the AUS/ FLUS category and 16 as inadequate. Cases grouped in the AUS/FLUS category contained extensive blood.
CNB was able to be diagnostic in 18 of the 19 cases deemed inadequate with FNA. The diagnostic distribution of these cases was as follows: 1 suspicious for malignancy, 16 benign and 1 AUS/FLUS. One of the inadequately sampled cases was also coined inadequate in the CNB sample.
While 1 of the 2 cases in the inadequate group with CNB was also inadequate with FNA, the other was interpreted as benign on FNA.
The number of cases that were reported as inadequate when the two methods were used together was only 1 (2.2%).
The FNA and CNB diagnoses are compared in (Figure 1).
Of the 44 cases, there were 29 TIRADS 3-Probably benign, 8 TIRADS 2-Benign and 7 TIRADS 4-Suspicious cases on radiology and we then used FNA and CNB for diagnosing these samples.
Of the 7 samples sent to our department with TIRADS 4-Suspicious radiologically, 1 was inadequate, 2 were benign, 2 were AUS/FLUS, 1 was suspicious for malignancy and 1 had malignant cytology on FNA, while 4 were benign, 1 was AUS/FLUS, 1 suspicious for malignancy and 1 malignant on CNB.
Twenty-six of the cases reported as TIRADS 3 were benign on CNB. There were 2 cases of AUS/FLUS and 1 case suspicious for malignancy. When the 15 cases that were TIRADS 3-Probably benign and where FNA was inadequate were ignored, 12 of the 29 cases were considered to be benign and 2 as AUS/FLUS with FNA.
Of CNB results of 8 cases with TIRADS 2-Benign radiologically 7 were benign and 1 inadequate. There were 3 cases that were inadequate on FNA while the remaining cases consisted of 2 benign and 3 AUS/ FLUS.
TIRADS-FNA/CNB results are compared in (Table 1).
| T I R A D S |
F N A |
Malignant/ suspicious for malignancy (4.54%) |
Benign (36.36%) |
AUS/ FLUS (15.9%) |
Inadequate (43.18%) |
C N B |
Malignant/ suspicious for malignancy (6.81%) |
Benign (81.81%) |
AUS/ FLUS (6.81%) |
Inadequate (4.54%) |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 (n:0) | - | - | - | - | - | - | - | - | ||
| 2 (n:8) | - | 2 | 3 | 3 | - | 7 | - | 1 | ||
| 3 (n:29) | - | 12 | 2 | 15 | 1 | 26 | 2 | - | ||
| 4 (n:7) | 2 | 2 | 2 | 1 | 2 | 4 | 1 | - | ||
| 5 (n:0) | - | - | - | - | - | - | - | - | ||
| 6 (n:0) | - | - | - | - | - | - | - | - |
Table 1: FNA and CNB results of the cases and TIRADS correlation.
There were only 3 cases all of which we compared with FNA, CNB and permenant resection results (Table 2).
| Cases | Case 1 | Case 2 | Case 3 |
|---|---|---|---|
| CNB | Suspicious for malignancy | Suspicious for malignancy | Malignant |
| FNA | Suspicious for malignancy | Inadequate | AUS/FLUS |
| Resection | Malignant | Malignant | Malignant |
Table 2: Comparison of CNB, FNA and resection results of 3 cases.
Histologic and cytologic findings of the case which was reported as Suspicious for malignancy in both CNB and FNA samples were as follows: Thyrocytes showing nuclear coarsening and hypochromasia in a colloid-poor micro follicular and trabecular pattern were present in the CNB samples of the case thought to be suspicious for malignancy both with FNA and CNB, and the HBME-1 was positive immunohistochemically. Nuclear coarsening in the thyrocytes, hypochromasia, focal contour irregularities and nuclear crowding were found in the FNA smears of this case. An encapsulated nodule 1.5 cm in diameter consisting of thyrocytes showing a follicular pattern and papillary nuclear characteristics overlapping with the morphology seen in the CNB samples was identified in the bilateral total thyroidectomy material of the case and was reported as follicular variant papillary thyroid carcinoma-encapsulated variant. A microcarcinoma focus 0.5 cm in diameter was also found in the other lobe.
The case which was reported as AUS/FLUS on FNA and malignant on CNB had cytologic and histologic findings as follows:
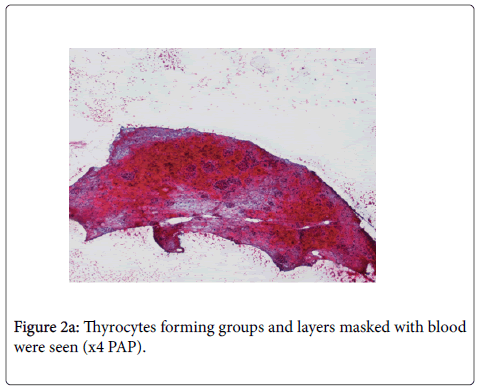
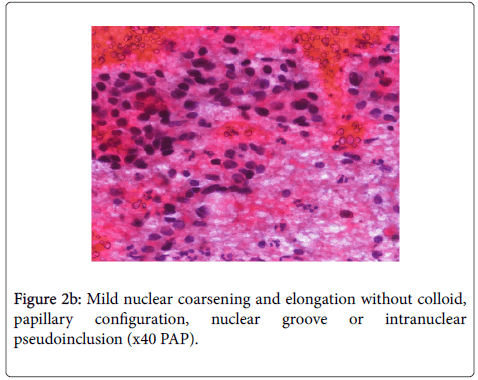
On cytologic materials, thyrocytes forming groups and layers were seen in relatively cellular smears masked with blood in some areas. Mild nuclear coarsening and oval shape was accompanied by hypochromasia focally. No colloid was observed in the smear. No papillary configuration, nuclear groove or intranuclear pseudo inclusion were seen (Figure 2a and 2b).
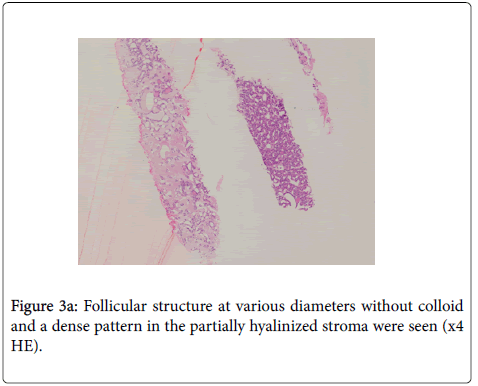
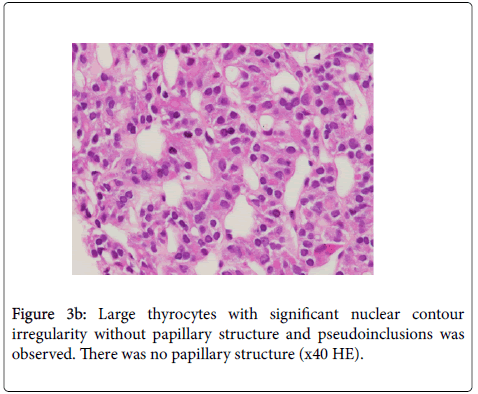
On evaluation after CNB, follicle structures without colloid that were at variable diameters and showed a dense pattern in the partially hyalinized stroma were observed. Significant nuclear contour irregularity was observed in the large thyrocytes. Mild hypochromasia and nuclear grooves were observed in some of the thyrocytes. There was no papillary structure. Pseudo inclusions were not seen (Figure 3a and 3b).
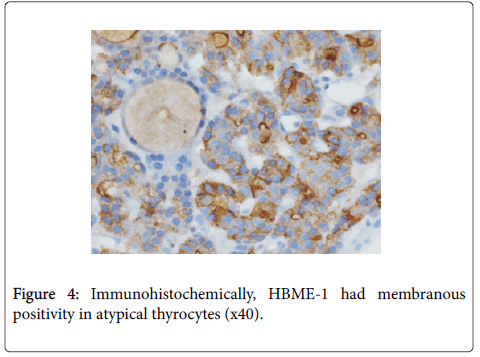
Diffuse strong membranous staining was found with IHC using HBME-1 (Figure 4).
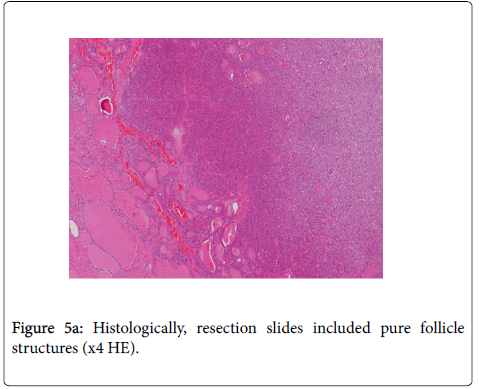
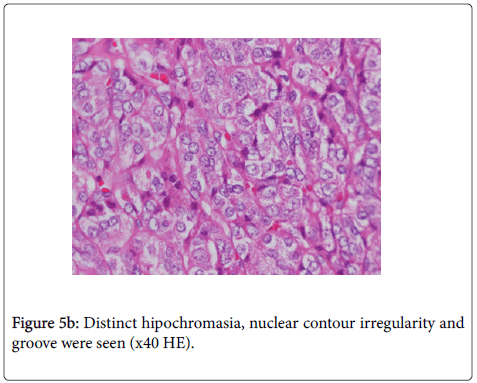
Histologically, resection included pure follicle structures consistent with follicular variant papillary carcinoma (Figure 5a and 5b).
The sizes of the nuclei were slightly larger at resection sections compared with CNB and FNA. Hypochromasia was not obvious at CNB sections in contrast to "orphan-annie" morphology of resection sections.
The common characteristic of other cases evaluated as AUS/FLUS in FNA were thyrocytes being masked with blood. Mild nuclear coarsening and hypochromasia were present in the thyrocytes with lymphocytic thyroiditis background on CNB of these cases.
Although a decreased number of follicles were seen in the CNB samples of the case that was deemed inadequate on FNA and evaluated as suspicious for malignancy on CNB, mild nuclear coarsening, hypochromasia and distinct borders with irregularity were present in the thyrocytes forming these follicles as well as membranous immunoreactivity with HBME-1. A nodule 2 cm in diameter with a dominant follicular pattern was identified in the lobe on which the biopsy was performed on the bilateral total thyroidectomy material of the case and was reported as follicular variant papillary thyroid carcinoma. A microcarcinoma focus 0.8 cm in diameter similar to the previous one was also found in the other lobe.
Discussion
The FNA procedure is a cost-effective routine diagnostic method in the evaluation of thyroid nodules and is adequate for making the diagnosis in most nodules. However, the success rate is very closely related to the experience of the person using the technique and the result can be inadequate or indeterminate in almost half of the cases according to certain studies [1,2]. Although FNA repetition according to an algorithm is preferred in these cases, diagnostic surgery is recommended for some that are suspicious on radiologic investigations [1,2]. The rate of inadequate and indeterminate diagnoses is similarly 13-61.8% in repeated FNAs [5,7,9,21-23]. CNB use is recommended as an alternative method instead of diagnostic surgery in such cases [5-16].
Studies show that the compliance between FNA and CNB results (benign/malignant) in cases that are not inadequate or indeterminate is high [6,8,9]. However, inadequate/indeterminate diagnosis with repeated FNA is also significantly higher than with CNB (30-40% versus 5.8%) [2,5,7,9,14,24,25].
In our study, when the inadequate and indeterminate diagnosis groups were excluded, all our cases diagnosed as benign and malignant with FNA and CNB were compliant. However, the ratio of insufficiency showed a significant elevation compared to CNB in our FNA procedures (4.54% vs. 43.18%). The most important reason of the high inadequate ratio found in FNA samples was the majority of the preparation being masked with blood due to the procedure being conducted after CNB.
The AUS/FLUS diagnostic group is ideally not expected to exceed 3-6% [22,25] and is used as a junk box for reasons such as defects in optimization of the sample/preparation (smear characteristics and fixation) and experience of the assessor, increasing the percentage of this group to 33% [17]. The AUS/FLUS diagnosis rate in our group was higher than the ideal at 15.9%. The most important reason was the extensive blood in the smears similar to the inadequate diagnosis group. The masking effect of blood as well as the inability to ensure optimal fixation in a bloody preparation caused an increase in the percentage of this group. The number of cases included in this group in our CNB samples was 3.5 times lower than the FNA samples. The reason could be the advantage of obtaining the histological profile as well as tissue preparation and fixation not being dependent on the relevant person. Thus, the restriction of being dependent on the evaluator can be considered excluded. On the other hand, high inadequate results on FNA procedures can be due to using the technique after CNB procedure. Since there are no standardized diagnostic criteria for cases interpreted as AUS/FLUS in CNB, a healthy interpretation can only be possible when compared with the resection material of these cases. However, we had only 3 cases (6.8%) which underwent resection. These were the limitations of this study.
Thyroid CNB samples are not clearly identified and classified with a system such as the Bethesda Classification unlike the FNA samples. Perhaps a morphologic classification is required for CNB with its increasing use for thyroid nodules. However, a classification other than the Bethesda Classification was recommended in a study conducted in a large case group (4). We used the Bethesda Classification to ensure consistency between the FNA and CNB samples and facilitate the comparison in our study.
Comparison of the radiologic TIRADS evaluation and FNA-CNB results of the cases revealed a significant correlation (77%). CNB morphologies of the cases that were considered malignant by radiology but reported benign showed that the most striking characteristic was the presence of hyalinization and calcification in the stroma in most cases. Only 1 of the TIRADS 3-probably benign cases was suspicious for malignancy with CNB and the final diagnosis was malignant after resection. The importance of working with an experienced thyroid radiology and surgery team in ensuring radiology-biopsy correlation cannot be overemphasized.
We observed that the thyrocytes were similar in size with small nuclei and there were colloid-rich macrofollicular, normofollicular and microfollicular structures in various dimensions in the thyroid tissues in most of the cases evaluated as benign. Immunohistochemical HBME-1 staining results of the cases with these characteristics were negative as expected. Metaplastic changes such as Hurthle cell metaplasia could be differentiated easily in FNA and resection material. It was possible to diagnose lymphocytic thyroiditis easier than with FNA in cases where we saw significant lymphocytic infiltration in the stroma accompanied by Hürthle cell metaplasia. IHC HBME-1 staining in the lymphocytic infiltration was a characteristic that should be taken into account and may be misleading in these cases.
Mild nuclear coarsening in the thyrocytes, mild hypochromasia and psammoma bodies in some were seen in CNB cases evaluated as AUS/ FLUS. However, IHC HBME-1 staining that could support malignancy was not present in these samples. While uncertainties such as thyrocytes covered by blood affect the evaluation according to the Bethesda Classification were placed in the AUS/FLUS category in FNA. This category was used for samples that contained features that can also be seen in benign nodules such as reactive changes like hypochromasia and enlarged nuclei that are one step behind the samples reported as suspicious for malignancy. Considering that lobectomy is recommended for cases diagnosed as AUS/FLUS in repetitive FNA and/or when no significant result is obtained with molecular studies according to the new ATA criteria, it would be interesting to find out the malignancy rate in the resection material of these cases that have an AUS/FLUS diagnosis with CNB and better defined characteristics than with FNA according to our results [1,26-29]. However, since resection was not performed in any of the cases we evaluated as AUS/FLUS in CNB and/or FNA, our experience on this subject is inadequate.
The morphologic characteristics of 2 cases that were evaluated as suspicious for malignancy in CNB overlapped, while the FNA of one was suspicious for malignancy and the other inadequate. The reason of insufficiency is the samples covered by blood or diluted with blood as seen in all other FNA samples. FNA could have been performed first to avoid this situation but it was not attempted and we do not know what degree of limitation it could cause in the CNB samples performed afterwards.
In one of the cases that underwent resection with a result of suspicious for malignancy had a dominant microfollicular pattern, more significant in CNB and the resection material of the case was reported as follicular variant papillary thyroid carcinoma, consistent with CNB. This was because microfollicular structures of the same case in FNA samples were not obvious or the evaluating pathologist, not being a cytopathologist, may have missed this feature. According to this result, CNB can be thought to be superior to FNA in determining the variants of clinical importance prior to resection, regardless of the assessor's experience.
Conclusion
It was observed that using simultaneous FNA and CNB methods decreased the inadequate/indeterminate diagnosis group significantly in our study. Although the presence of smears covered by blood and/or diluted with blood developed due to the use of FNA following CNB and greatly limited the diagnostic accuracy of FNA, the simplicity of morphological and IHC evaluation in CNB favors it as the preferred diagnostic method. However, comparison of the diagnostic success of FNA and CNB results is not possible due to the inadequate number of cases that underwent resection in the sample group we evaluated. Our experience in the pathological evaluation of CNB and FNA samples is growing with the gradually increasing number of cases in thyroid radiology as well as in surgery. We need to widen our case group to be able to obtain more definite results and make better-informed comments.
References
- Haugen BR, Alexander EK, Bible KC, Doherty G, Mandel SJ, et al. (2016) American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 26: 1-133.
- Edmund S. Cibas ES, Syed Z. Ali (2009) The Bethesda System for Reporting Thyroid Cytopathology. Am J Clin Pathol 132: 658-665.
- Samir AE, Vij A, Seale MK, Desai G, Halpern E, et al. (2012) Ultrasound-guided percutaneous thyroid nodule core biopsy: Clinical utility in patients with prior non diagnostic fine-needle aspirate. Thyroid 22: 461-467.
- Jung CK, Min HS, Park HJ, Song DE, Kim JH, et al. (2015) Pathology Reporting of Thyroid Core Needle Biopsy: A Proposal of the Korean Endocrine Pathology Thyroid Core Needle Biopsy Study Group. J Patho Transl Med 49: 288-299.
- Choi SH, Baek JH, Lee JH, Choi YJ, Hong MJ, et al. (2014) Thyroid nodules with initially non-diagnostic, fine-needle aspiration results: Comparison of core-needle biopsy and repeated fine-needle aspiration. Eur Radiol 24: 2819-2826.
- Na DG, Kim J, Sung JY, Baek JH, Jung KC, et al. (2012) Core-Needle Biopsy Is More Useful Than Repeat Fine-Needle Aspiration in Thyroid Nodules Read as Nondiagnostic or Atypia of Undetermined Significance by the Bethesda System for Reporting Thyroid Cytopathology. Thyroid 22: 468-475.
- Paja M, Del Cura JL, Zabala R, Corta I, Lizarraga A, et al. (2015) Ultrasound-guided core-needle biopsy in thyroid nodules. A study of 676 consecutive cases with surgical correlation. Eur Radiol 26: 1-8.
- Trimboli P, Nasrollah N, Guidobaldi L, Taccogna S, CicciarellaModica DD, et al. (2014) The use of core needle biopsy as first-line in diagnosis of thyroid nodules reduces false negative and inconclusive data reported by fine-needle aspiration. World J Surg Oncol 12: 61.
- Yi KS, Kim JH, Na DG, Seo H, Min HS, et al. (2015) Usefulness of core needle biopsy for thyroid nodules with macrocalcifications: Comparison with fine-needle aspiration. Thyroid 25: 657-664.
- López JI, Zabala R, Del Cura JL (2013) Histological Diagnosis of Thyroid Disease Using Ultrasound-Guided Core Biopsies. Eur Thyroid J 2: 29-36.
- Renshaw AA, Pinnar N (2007) Comparison of thyroid fine-needle aspiration and core needle biopsy. Am J Clin Pathol 128: 370-374.
- Hakala T, Kholova I, Sand J, Saaristo R, Kellokumpu-Lehtinen P, et al. (2013) A core needle biopsy provides more malignancy-specific results than fine needle aspiration biopsy in thyroid nodules suspicious for malignancy. J Clin Pathol 66: 1046-1050.
- Strauss EB, Iovino A, Upender S (2008) Simultaneous fine-needle aspiration and core biopsy of thyroid nodules and other superficial head and neck masses using sonographic guidance. Am J Roentgenol 190: 1697-2709.
- Lee SH, Kim MH, Bae JS, Lim DJ, Jung SL, et al. (2014) Clinical outcomes in patients with non-diagnostic thyroid fine needle aspiration cytology: Usefulness of the thyroid core needle biopsy. Ann Surg Oncol 21: 1870-1877.
- Min HS, Kim JH, Ryoo I, Jung SL, Jung CK, et al. (2014) The role of core needle biopsy in the preoperative diagnosis of follicular neoplasm of the thyroid. APMIS 122: 993-1000.
- Sung JY, Na DG, Kim KS, Yoo H, Lee H, et al. (2012) Diagnostic accuracy of fine-needle aspiration versus core-needle biopsy for the diagnosis of thyroid malignancy in a clinical cohort. Eur Radiol 22: 1564-1572.
- Yoon JH, Kim EK, Kwak JY, Moon HJ (2015) Effectiveness and limitations of core needle biopsy in the diagnosis of thyroid nodules: Review of current literature. J Pathol Transl Med 49: 230-235.
- Guler B, Kiran T, Arici DS, Aysan E, Sönmez FC, et al. (2016) Should core needle biopsy be used in the evaluation of thyroid nodules. Endocr Pathol 27: 352-358.
- Kwak JY, Han KH, Yoon JH, Moon HJ, Son EJ, et al. (2011) Thyroid Imaging Reporting and Data System for US Features of Nodules: A Step in Establishing Better Stratification of Cancer Risk. Radiology 260: 892-899.
- Gyu ND, Hwan BJ, Yong SJ, Ji-Hoon K, Kyun KJ, et al. (2016) Thyroid Imaging Reporting and Data System Risk Stratification of Thyroid Nodules: Categorization Based on Solidity and Echogenicity. Thyroid 26: 562-572.
- Gharib H, Papini E, Paschke R, Duick DS, Valcavi R, et al. (2010) American Association of Clinical Endocrinologists, Associazione Medici Endocrinologi, and European Thyroid Association Medical guidelines for clinical practice for the diagnosis and management of thyroid nodules: executive summary of recommendations. AACE/AME/ETA Task Force on Thyroid Nodules. Endocr Pract 16: 468-475.
- Yassa L, Cibas ES, Benson CB, Frates MC, Doubilet PM, et al. (2007) Long-term assessment of a multidisciplinary approach to thyroid nodule diagnostic evaluation. Cancer 111: 508-516.
- Cooper DS, Doherty GM, Haugen BR, Kloos RT, Lee SL, et al. (2009) Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid 19: 1167-1214.
- Orija IB, Piñeyro M, Biscotti C, Reddy SS, Hamrahian AH, et al. (2007) Value of repeating a nondiagnostic thyroid fine-needle aspiration biopsy. Endocr Pract 13: 735-742.
- Yang J, Schnadig V, Logrono R, Wasserman PG (2007) Fine-needle aspiration of thyroid nodules: A study of 47003 patients with histologic and clinical correlations. Cancer 111: 306-315.
- Kim SK, Hwang TS, Yoo YB, Han HS, Kim DL, et al. (2011) Surgical results of thyroid nodules according to a management guideline based on the BRAF (V600E) mutation status. J Clin Endocrinol Metab 96: 658-664.
- Adeniran AJ, Hui P, Chhieng DC, Prasad ML, Schofield K, et al. (2011) BRAF mutation testing of thyroid fine-needle aspiration specimens enhances the predictability of malignancy in thyroid follicular lesions of undetermined significance. Acta Cytol 55: 570-575.
- Nikiforov YE, Ohori NP, Hodak SP, Carty SE, LeBeau SO, et al. (2011) Impact of mutational testing on the diagnosis and management of patients with cytologically indeterminate thyroid nodules: A prospective analysis of 1056 FNA samples. J Clin Endocrinol Metab 96: 3390-3397.
- Nikiforov YE, Steward DL, Robinson-Smith TM, Haugen BR, Klopper JP, et al. (2009) Molecular testing for mutations in improving the fine-needle aspiration diagnosis of thyroid nodules. J Clin Endocrinol Metab 94: 2092-2098.
Citation: Kiran T, Guler B, Aysan E, Arici DS (2017) Correlation Between Simultaneous Ultrasound-Guided Fine Needle Aspiration and Core Needle Biopsy Results in Thyroid Nodules of 44 Cases. Diagn Pathol Open 2: 123. DOI: 10.4172/2476-2024.1000123
Copyright: © 2017 Kiran T, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Open Access Journals
Article Tools
Article Usage
- Total views: 5099
- [From(publication date): 0-2017 - Jul 01, 2025]
- Breakdown by view type
- HTML page views: 4140
- PDF downloads: 959