Radiation and Alzheimer's Disease (AD)
Received: 30-Dec-2017 / Accepted Date: 16-Jan-2018 / Published Date: 23-Jan-2018 DOI: 10.4172/2161-0460.1000418
Alzheimer’s disease (AD) is the most common form of dementia, and accounts for 60 to 80 percent of dementia cases. While about one third of all people age 85 and older may have Alzheimer’s disease, and the number of people with the disease doubles every 5 years beyond age 65, its early onset may start from the ages of 40s and 50s. Old age and genetic factor are two most important factors for the development of Alzheimer’s disease. Epidemiological studies have suggested that excessive alcohol consumption, smoking, environmental toxin such as pesticides (Dichlorodiphenyltrichloroethane or DDT), food additives (such as nitrogen-based chemicals which are converted to toxic nitrosamines during cooking), contamination (mussels contaminated with demonic acid), food components (two amino acids in seeds of certain legumes which enhance the action of the neurotransmitter glutamate), air pollution (such as aerosolized nickel nanoparticles, a component of air pollution) may also play important roles in the development of the disease [1]. Serious head injury, heart disease, diabetes, stroke, high blood pressure and high cholesterol which damage the heart and blood vessels may be indirectly related to the development of AD due to the increase of β-amyloid (Aβ).
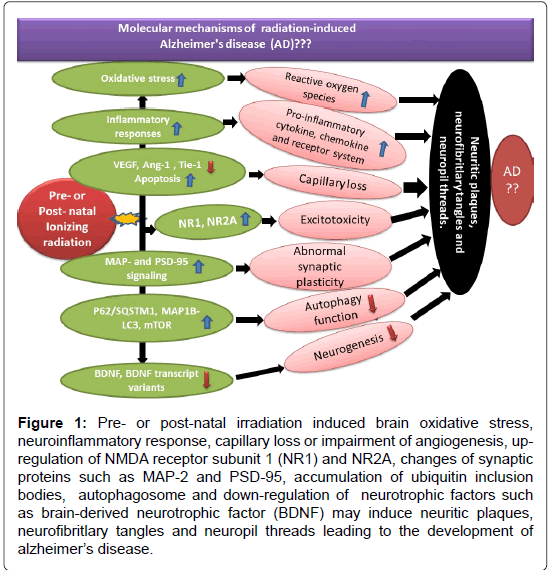
Recent systematic review of bibliographic databases from PubMed, EMBASE, Cochrane Library and Web of Science suggests that occupational exposure to extremely low frequency magnetic fields (ELF-MF) to welders, electric utility workers, train drivers and sewing machine operators may increase the risk of AD [2,3]. Experimental studies indicated that low-dose radiation exposures (10 cGy) induced genes not affected by high-dose radiation (2 Gy) and that low-dose genes were associated with unique pathways and functions. Nine neural signaling pathways had a high degree of concordance in their transcriptional response in mouse brain tissue after low-dose irradiation, in the aging human brain (unirradiated) and in brain tissue from patients with Alzheimer’s disease. Mice exposed to high-dose radiation did not show these effects and associations. It suggests that the molecular response of the mouse brain within a few hours after low-dose irradiation involves the down-regulation of neural pathways associated with cognitive dysfunctions that are also down-regulated in normal human aging and Alzheimer’s disease [4,5]. Galactic cosmic radiation consisting of high-energy, high-charged (HZE) particles such as (56) Fe at 100 mGy to 1 Gy reduced cognitive abilities. Acceleration of Aβ plaque pathology was observed in male Amyloid precursor protein (APP)/Presenilin 1 (PS1) mice. Further study suggested that (56) Fe particle-induced alterations in Aβ trafficking through the blood brain barrier might be related to plaque increase leading to AD [6]. Chronic low-dose-rate radiation exposure (1 mGy/day or 20 mGy/day) over 300 days with cumulative doses of 0.3 Gy and 6.0 Gy, respectively induced a marked alteration in the phosphoproteome and an inhibition of cAMP responsive element binding protein (CREB) signalling. Both dose rate irradiations reduced the number of activated microglia in the molecular layer of hippocampus which paralleled with decreased levels of tumor necrosis factor alpha (TNFα) expression. At the lower dose rate of 1mGy/day, alteration of Rac1-Cofilin signalling and lipid peroxidation was induced. The overlap of these changes with those of Alzheimer´s pathology suggests that low dose rate irradiation may be involved in the development of Alzheimer´s disease [7]. In the United States, analysis of AD death rates versus radon background radiation and total background radiation suggested that ionizing radiation was a risk factor for AD. Intranasal inhalation of radon gas could subject the rhinencephalon and hippocampus to damaging radiation that initiated AD [8]. The Alzheimer neurofibrillary tangle is composed of tau, which is one of the most common pathological hallmarks of AD and tau aggregation pathology at Braak stage 1 1 (out of 6 Braak stages) or beyond affects 50% of the population over the age of 45 [9-11]. Our recent review of the effect of the pre- and post-natal irradiation on animal models and human studies indicated many similarities in hippocampal neuropathology, cognitive impairment and relevant molecular mechanisms between Alzheimer’s disease and early life radiation exposure-induced neuropsychological disorders [12-16]. It suggests that irradiation of the brain in early human life may set abnormal developmental events into motion that starts from tau aggregation at the ages of 40s and 50s, leading to the development of Alzheimer’s Disease at the late stages of human life. At molecular level, pre- or post-natal irradiation induced brain oxidative stress [15], neuroinflammatory response [16,17], capillary loss or impairment of angiogenesis [18], up-regulation of NMDA receptor subunit 1 (NR1) and NR2A [19], changes of synaptic proteins such as MAP-2 and PSD-95 [20], accumulation of ubiquitin inclusion bodies, autophagosome [21] and down-regulation of neurotrophic factors such as brain derived neurotrophic factor (BDNF) [22] may induce neuritic plaques, neurofibritlary tangles and neuropil threads leading to the development of Alzheimer’s disease (Figure 1).
Figure 1: Pre- or post-natal irradiation induced brain oxidative stress, neuroinflammatory response, capillary loss or impairment of angiogenesis, upregulation of NMDA receptor subunit 1 (NR1) and NR2A, changes of synaptic proteins such as MAP-2 and PSD-95, accumulation of ubiquitin inclusion bodies, autophagosome and down-regulation of neurotrophic factors such as brain-derived neurotrophic factor (BDNF) may induce neuritic plaques, neurofibritlary tangles and neuropil threads leading to the development of alzheimer’s disease.
With increased use of X-ray Computed Tomography (CT scan) for medical diagnosis and radiotherapy, construction of more nuclear power plants worldwide and consequently potential nuclear contamination or accidents, occupational radiation exposure, frequent-flyer risks, manned space exploration and possible radiological terrorism, low dose/dose rate ionizing radiation research becomes much more imperative and urgent nowadays than ever before. Further study with lifetime monitoring of radiation effect of individuals with low dose/low dose rate exposure may still be needed for establishing the close relationship between the radiation exposure and the development of Alzheimer’s disease.
References
- Stein J, Schettler T, Rohrer B, Valenti M (2008) Environmental factors in the development of dementia focus on Alzheimer’s disease and cognitive decline in environmental threats to healthy aging, with a closer look at Alzheimer’s and Parkinson’s diseases (Nancy Myers ed). Greater Boston Physicians for Social Responsibility and Science and Environmental Health Network, pp: 97-143.
- Killin LO, Starr JM, Shiue IJ, Russ TC (2016) Environmental risk factors for dementia: A systematic review. BMC Geriatr 16: 175.
- Jalilian H, Teshnizi SH, Röösli M, Neghab M (2017) Occupational exposure to extremely low frequency magnetic field and risk of Alzheimer’s disease: A systematic review and meta-analysis. Neurotoxicology S0161-813X: 30239-30245.
- Lowe XR, Bhattacharya S, Marchetti F, Wyrobek AJ (2009) Early brain response to low-dose radiation exposure involves molecular networks and pathways associated with cognitive functions, advanced aging and Alzheimer’s disease. Radiat Res 171: 53-65.
- Lowe X, Wyrobek A (2012) Characterization of the early CNS stress biomarkers and profiles associated with neuropsychiatric diseases. Curr Genomics 13: 489-497.
- Cherry JD, Liu B, Frost JL, Lemere CA, Williams JP, et al. (2012) Galactic cosmic radiation leads to cognitive impairment and increased aß plaque accumulation in a mouse model of Alzheimer’s disease. PLoS One 7: e53275.
- Kempf SJ, Janik D, Barjaktarovic Z, Braga-Tanaka I, Tanaka S, et al. (2016) Chronic low-dose-rate ionising radiation affects the hippocampal phosphoproteome in the ApoE-/-Alzheimer's mouse model. Oncotarget 7: 71817-71832.
- Lehrer S, Rheinstein PH, Rosenzweig KE (2017) Association of radon background and total background ionizing radiation with Alzheimer’s disease Deaths in U.S. States. J Alzheimers Dis 59: 737-741.
- Braak H, Braak E (1991) Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol 82: 239-259.
- Wischik CM, Harrington CR, Storey JM (2014) Tau-aggregation inhibitor therapy for Alzheimer’s disease. Biochem Pharmacol 88: 529-539.
- Yang B, Ren BX, Tang FR (2017) Prenatal irradiation-induced brain neuropathology and cognitive impairment. Brain Dev 39: 10-22.
- Tang FR, Loke WK, Khoo BC (2017a) Postnatal irradiation-induced hippocampal neuropathology, cognitive impairment and aging. Brain Dec 39: 277-293.
- Tang FR, Loke WK, Khoo BC (2017) Low-dose or low-dose-rate ionizing radiation-induced bioeffects in animal models. J Radiat Res 58: 165-182.
- Tang FR (2017) Low dose/rate radiation exposure and human mental health. EC Psychol Psychiatry 6: 56-59.
- Tang FR, Loke WK, Yan WP, Khoo BC (2017) Radioprotective effect of ursolic acid in radiation-induced impairment of neurogenesis, learning and memory in adolescent BALB/c mouse. Physiol Behav 175: 37-46.
- Yang J, Zhang X, Yuan P, Yang J, Xu Y, et al. (2017) Oxalate-curcumin-based probe for micro- and macroimaging of reactive oxygen species in Alzheimer’s disease. Proc Natl Acad Sci U S A 114: 12384-12389.
- Minter MR, Taylor JM, Crack PJ (2016) The contribution of neuroinflammation to amyloid toxicity in Alzheimer’s disease. J Neurochem 136: 457-474.
- Kuznetsova E, Schliebs R (2013) ß-Amyloid, cholinergic transmission, and cerebrovascular system - A developmental study in a mouse model of Alzheimer’s disease. Curr Pharm Des 19: 6749-6765.
- Mishizen-Eberz AJ, Rissman RA, Carter TL, Ikonomovic MD, Wolfe BB, et al. (2004) Biochemical and molecular studies of NMDA receptor subunits NR1/2A/2B in hippocampal sub regions throughout progression of Alzheimer’s disease pathology. Neurobiol Dis 15: 80-92.
- Swatton JE, Sellers LA, Faull RL, Holland A, Iritani S, et al. (2004) Increased MAP kinase activity in Alzheimer’s and Down syndrome but not in schizophrenia human brain. Eur J Neurosci 19: 2711-2719.
- Colacurcio DJ, Pensalfini A, Jiang Y, Nixon RA (2018) Dysfunction of autophagy and endosomal-lysosomal pathways: Roles in pathogenesis of down syndrome and Alzheimer's disease. Free Radic Biol Med 14: 40-51.
- Huang R, Huang J, Cathcart H, Smith S, Poduslo SE (2007) Genetic variants in brain-derived neurotrophic factor associated with Alzheimer's disease. J Med Genet 44: e66.
Citation: Tang FR (2018) Radiation and Alzheimer’s Disease (AD). J Alzheimers Dis Parkinsonism 8: 418. DOI: 10.4172/2161-0460.1000418
Copyright: ©2018 Tang FR. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 6113
- [From(publication date): 0-2018 - Oct 10, 2025]
- Breakdown by view type
- HTML page views: 5146
- PDF downloads: 967