Research Article Open Access
Regeneration of Pulp/Dentin-Like Tissue in Immature Necrotic Permanent Dog Teeth Using Adipose Tissue-Derived Mesenchymal Stem Cells
Najlaa MA1*, Eman AEA1,2, Ibrahim MM3, Hazem MA4, Khaled HEA5 and Douaa AED1,61Pediatric Dentistry Department, Faculty of Dentistry, King Abdulaziz University, Jeddah, Kingdom of Saudi Arabia
2Pedodontic Department, Faculty of Dental Medicine (Girls), El Azhar University, Cairo, Egypt
3Department of Orthodontics, Ibrahim Masoud Dental Speciality Clinic, Jeddah, Kingdom of Saudi Arabia
4Clinical Biochemistry Department, Faculty of Medicine, Rabigh Branch, King Abdulaziz University, Jeddah, Kingdom of Saudi Arabia and Medical Biochemistry, Faculty of Medicine, Cairo University, Cairo, Egypt
5Oral Radiology Department, Faculty of Dentistry, Umm Al Qura- University, Kingdom of Saudi Arabia, and Ain Shams University, Cairo, Egypt
6Community Medicine and Public Health Department, Cairo University, Faculty of Medicine, Cairo, Egypt
- *Corresponding Author:
- Najlaa MA
Professor, Pediatric Dentistry Department
Faculty of Dentistry, King Abdulaziz University
Jeddah, Kingdom of Saudi Arabia
Tel: +6401000 (22026-20388-22264)
E-mail: nalamoudi2011@gmail.com
Received date: January 05, 2017; Accepted date: March 05, 2017; Published date: March 13, 2017
Citation: Najlaa MA, Eman AEA, Ibrahim MM, Hazem MA, Khaled HEA, et al. (2017) Regeneration of Pulp/Dentin-Like Tissue in Immature Necrotic Permanent Dog Teeth Using Adipose Tissue-Derived Mesenchymal Stem Cells. J Oral Hyg Health 5:217. doi: 10.4172/2332-0702.1000217
Copyright: © 2017 Najlaa MA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Oral Hygiene & Health
Abstract
Objective: Our aim was to evaluate the potential of adipose tissue-derived mesenchymal stem cells (AD-MSCs) in the regeneration of canal pulp/dentin-like tissue and completion of root maturation of immature necrotic permanent dog teeth with apical periodontitis. Materials and methods: This study is a collaborative work between King Abdulaziz University with, Cairo University, Egypt. A split-mouth design was used in 6 healthy male dogs. Four teeth were selected per dog (total, 24 teeth). The immature upper left second and third permanent incisors were designated as group A (experimental group), and contralateral teeth were designated as group B (control group). Periapical periodontitis was induced in both groups. After disinfection, group A received AD-MSCs and growth factors within a chitosan scaffold, whereas group B received growth factors within a chitosan scaffold. Periradicular healing, root-wall thickening and lengthening, and apical closure were blindly monitored radiographically for 4 months. The animals were euthanized and the teeth examined histologically for healthy pulp/dentin complex-like tissue. Results: Group A showed significantly higher increase in radicular thickening, root lengthening, and apical closure than group B. However, there was no significant difference in apical healing. Histologically, the teeth of group A, but not group B, showed regeneration of pulp/dentin-like tissue. Conclusion: AD-MSCs in conjunction with growth factors incorporated into a chitosan hydrogel scaffold regenerated pulp/dentin-like tissue and facilitated complete root maturation of non-vital immature permanent dog teeth with apical periodontitis.
Keywords
Adipose tissue-derived mesenchymal stem cells; Necrotic immature permanent teeth; Tissue engineering
Introduction
Conventional treatment of immature necrotic permanent teeth, resulting from trauma or infection, does not support the regeneration and tooth maturation. This condition presents numerous challenges in root canal debridement and obturation and poses an increased risk of any slight trauma fracturing the thin dentinal walls. Furthermore, regeneration of new dentin and pulp tissue cannot be achieved with conventional root canal treatment [1,2]. The use of a calcium hydroxide and mineral trioxide aggregate (MTA) through the root canal for an apexification procedure can result in the formation of a calcific bridge at the root apex [3,4]. In addition, trials using composite resin with internal bonding have been shown to improve the fracture resistance of the root canal procedure and decrease treatment time; however, no method has shown apical maturation [5,6].
The basic principle of tissue engineering involves the recognition of the appropriate cells to yield the desired tissue, structures, and function; development of conducive scaffolds; and regulation of the process by biologically active substances, most notably, growth factors. Generally, stem cells are the preferred source of differentiated cells for tissue generation during development and for the potential regeneration of tissue and construction following disease or injury [7].
Advancements in tissue engineering and stem cell biology, including the separation and characterization of dental pulp stem cells (DPSCs), allow the regeneration [8-11] and reconstruction of pulp/dentin-like tissue. However, because the amount of available autologous pulp tissue decreases with age, alternative sources of mesenchymal stem/ progenitor cells (MSCs) have been sought [12,13].
Stem cells are easily accessed and isolated from harvested subcutaneous adipose tissues [14,15]. Furthermore, adipose tissuederived MSCs (AD-MSCs) demonstrate stable growth and propagation dynamics, extensive self-regenerative capacity, and ability to differentiate into various cell lineages, similar to bone marrow-derived MSCs [16-18]. Adipose tissue plays a major role in enhancing wound healing. This tissue secretes angiogenic growth factors, including vascular endothelial growth factor (VEGF), basic fibroblast growth factor (bFGF), platelet-derived growth factor (PDGF), and angiogenin, all of which are capable of supporting endothelial differentiation. ADMSCs can be isolated with minimally invasive procedures, while large quantities of AD-MSCs can be retrieved under local anesthesia [15,19-21]. Few studies have used ADMSCs in pulp regeneration in immature non-vital permanent teeth with open apices. We therefore aimed to evaluate the potential of AD-MSCs in the regeneration of canal pulp/ dentin-like tissue and completion of root maturation of immature necrotic permanent teeth with apical periodontitis in dogs (Figure 1).
Materials and Methods
Experimental animals
This study was carried out from September 2013 till January 2015 in collaboration between King Abdulaziz University and the Faculty of Medicine, Cairo University, Egypt. Ethical approval was obtained from King Abdulaziz University (number 026-12). We adopted the NIH guidelines for animal care, as well as the institutional animal care guidelines for animal care and use of both King Abdulaziz University and the Faculty of Medicine, Cairo University.
Sample size was calculated according to a published “resource equation” method [22]. “E,” is the degree of freedom in the analysis of variance, was measured by the following formula: E = total number of animals − total number of groups. The value of E should lie between 10 and 20. The calculation was based on the fact that we used the splitmouth technique, with 2 teeth used on each side (2 sides × 2 teeth=4 study units). The calculation was E=[(6 animals × 2 sides × 2 teeth) – 2]=22; therefore, 6 animals were sufficient to satisfy the sample number [22].
The study was conducted on 24 teeth from 6-month-old male mongrel dogs (6 dogs, 4 teeth each). The dogs had immature upper permanent incisors (with open apices) and were orally and systemically healthy. The experiment was performed using a split-mouth design. In each of the dogs, 4 teeth, 2 on each side, were selected for the study procedure. The immature upper second and third permanent incisors on the left were designated as group A (experimental group), and those on the right were designated as group B (control group). In group A, the pulp chamber and root canals were injected with AD-MSCs and growth factors incorporated into a chitosan hydrogel scaffold, whereas the root canals of group B teeth received the chitosan hydrogel scaffold with only growth factors. All the procedures were performed under aseptic conditions. General anesthesia was induced by intravenous administration of 13.5 mg/kg pentothal (Abbott Laboratories, North Chicago, IL, USA). The dogs were intubated, and anesthesia was maintained with isofluorane (Halocarbon Laboratories, River Edge, NJ, USA) supplemented with local anesthesia (0.5% bupivacaine, Abbott Laboratories, North Chicago, IL, USA). Analgesics (Torbugesic® (butorphanol tartrate) 0.2 mg/kg, Fort Dodge Animal Health, Fort Dodge, IA, USA) were administered for pain relief after each procedure. All operative and postoperative procedures were monitored by the staff of the Department of Laboratory and Animal Medicine. The clinical study was implemented in 7 phases, described in the following subsections.
Phase I: isolation of MSCs from adipose tissue
We followed the procedure of Tomiyama [23] under general anesthesia, adipose tissue was removed from the omentum and inguinal pads of each dog and. The fat isolated from each dog was marked and kept separately then used to generate autologous AD-MSCs. The isolated adipose tissue was rinsed and placed in a sterile tube containing 15 mL phosphate-buffered saline (PBS; Gibco/Invitrogen, Grand Island, NY, USA). The tissues were digested with 0.075% collagenase II (Serva Electrophoresis GmbH, Mannheim, Germany) and dissolved in Hank’s balanced salt solution for 60 min at 37°C under continuous shaking. This was followed by the addition of erythrocyte lysis buffer and removal of debris by mild centrifugation. The remaining cell suspension was then transferred to tissue culture flasks containing Dulbecco’s modified Eagle medium (DMEM; Gibco/BRL Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS; Gibco/ BRL, Carlsbad, CA, USA). The flasks were incubated at 37°C in 5% CO2 for 24 h. The non-adherent cells were removed, and the adherent cells were cultured in DMEM supplemented with 10% FBS, 100 U/ mL penicillin, 100 μg/mL streptomycin (Gibco BRL, Carlsbad, CA, USA 1.25 mg/L amphotericin B (Gibco/BRL). When the cultured cells attained 80–90% confluence, the cells were washed twice with PBS and trypsinized using 0.25% trypsin in 1 mM ethylenediaminetetraacetic acid – EDTA(Gibco BRL, Carlsbad, CA, USA) for 5 min at 37°C with intermit shaking. The trypsinized cells were centrifuged at 2000 rpm for 10 min and then resuspended in serum-supplemented medium and incubated in 2.5 × 10.5 cm2 culture flasks (Falcon, BD Biosciences, Two Oak Park, Bedford, MA, USA). This first-passage culture was expanded in vitro until the third passage.
Phase II: characterization of AD-MSCs
Morphological characterization: The identity of the AD-MSCs was confirmed by their fusiform shape and adherence power.
In vitro differentiation: Their propensity to differentiate into osteocytes was evaluated by growth in osteo-inductive Eagle’s minimum essential medium with α-modification (α-MEM) supplemented with 50 mg/ mL ascorbic acid 2-phosphate, 10 nM dexamethasone, 10 mM β-glycerophosphate (Sigma-Aldrich, St. Louis, MO, USA), and 10% FBS 3 times a week. The cultures were subsequently stained with alizarin red [24]. In addition, the ability of the MSCs to differentiate into chondrocytes was determined by culturing in chondro-inductive medium containing 500 mg/mL bone morphogenetic protein-2 (BMP- 2; R&D Systems, Minneapolis, MN, USA) and 10 ng/mL transforming growth factor β3 (TGFβ3; Peprotech, London, UK) for 3 weeks, followed by the detection of chondrocyte cells by alcian blue staining [25,26].
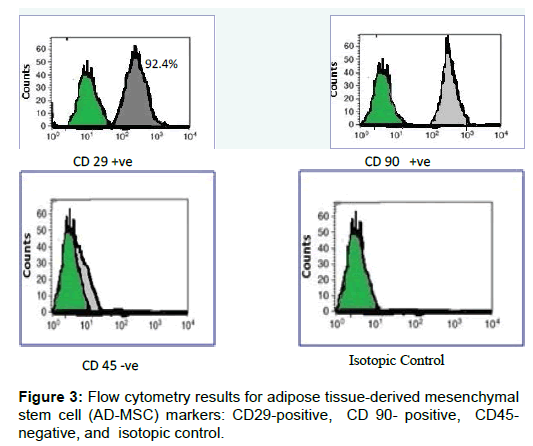
Immunophenotypic characteristics by flow cytometry analysis: At the end of the 3rd passage adherent AD-MSCs cells were treated with trypsin solution. Next, 1 × 105 cells/ml were incubated in phosphatebuffered saline (PBS), 2% fetal bovina solution (FBS) and 10 μL of monoclonal antibodies - CD (cluster of differentiation): CD 90, CD29 CD 45 monoclonal antibodies (Santa Cruz Biotechnology, Dallas, TX, USA) for 45 min at 4°C in the dark. The same species isotypes served as negative controls. After 20 min of incubation, 2 mL of PBS containing 2% FBS solution was added to each tube of monoclonal treated cells. The mixtures were then centrifuged for 5 min at 2500 rpm. Next, the supernatant was discarded and the cells were re-suspended in 500 μL PBS containing 2% FBS. Cells were then incubated with an antimouse immunoglobulin G fluorescein-conjugated secondary antibody (Millipore Corp, Temecula, CA, USA) for 45 min on ice, after which the cell suspensions were washed twice and analyzed using the FACS Calibur flow cytometry system using CXP Software version 2.2. (BD Biosciences NJ, USA)[27].
RT-PCR and primer sequences
To confirm the mesenchymal lineage of undifferentiated cells and the osteogenic and chondrogenic potential of the differentiated ADMSCs, the gene expression of osteocalcin and type 2 collagen was assessed. Cultured cells were disrupted by Trizol reagent (Invitrogen, Life Technologies, Carlsbad, CA, USA), and total cellular RNA was extracted and precipitated by centrifugation (15,000 ×g for 20 min at 4°C). The cell pellets were washed with 70% ethanol, air dried, and dissolved in 0.1% v/v ribonuclease-free water. Reverse transcription was carried out using Moloney murine leukemia virus reverse transcriptase. For type 2 collagen, the forward and reverse primer sequences were 5ʹ-AGT GCTGTC CCA TCT GCT CA-3ʹ and 5ʹ-GCC TTC TCA TCA AAT CCT CCA-3ʹ, respectively. For osteocalcin, the forward and reverse primers were 5ʹ-AGT GCT GTC CCA TCT GCT CA- 3ʹ and 5ʹ-GCC TTC TCA TCA AAT CCT CCA-3ʹ, respectively. For glyceraldehyde-3-phosphate dehydrogenase (GAPDH), the forward and reverse primers were 5ʹ-GGG GCC ATC CAC AGT CTT CT- 3ʹ and 5ʹ-GCC AAA AGG GTC ATC ATC TC-3ʹ, respectively [28]. PCR was performed in a total volume of 25 μL containing 10 mM Tris-hydrochloride (Tris-HCl, pH 8.3), 1.5 mM magnesium chloride (MgCl2), 10 mM deoxynucleotide triphosphates (dNTPs), 0.8 M of each primer, and 2.5 U Taq DNA polymerase (Qiagen, Venlo, The Netherlands). PCR was carried out for 30 cycles of denaturation (94°C, 1 min), annealing (56°C, 45 s), and extension (72°C, 40 s). The products were analyzed by 2% agarose gel electrophoresis and visualized with ethidium bromide staining. GAPDH was used as the housekeeping gene, serving as a positive amplification control [29].
Phase III: induction of periapical periodontitis
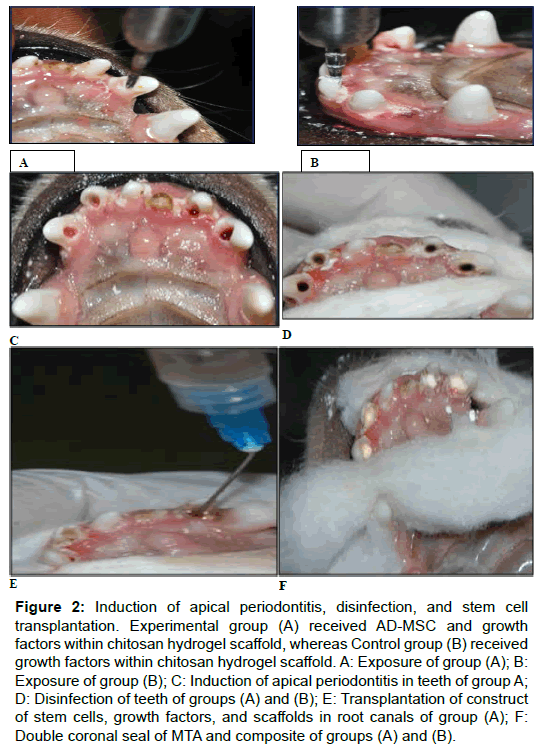
In groups A and B, mechanical tooth exposure was performed using a #2 round carbide burr. Pulp tissue in the canal spaces was interrupted endodontically using a sterile #20 stainless steel hand file, and the plaque from the supragingival area was scaled and mixed with sterile saline (0.9% NaCl, Hospira Inc., Lake Forest, IL, USA). The suspension was then introduced into the canal chamber using a sterile sponge and sealed with temporary intermediate restorative material (IRM; Dentsply Corporate, York, PA, USA). Analgesics were administered to the animals postoperatively as previously described (Figures 1A-1C).
Radiographs were obtained using a parallel technique for teeth in both groups, and the same procedure was applied for subsequent follow-up radiographs to ensure the same angle and magnification. Radiographic monitoring was performed every 3 weeks until apical periodontitis was evident.
Phase IV: disinfection
Under general anesthesia the previously infected dog’s teeth in both groups (A and B) were aseptically re-penetrated using rubber dam isolation with retractors, and then the area was disinfected with 0.12% chlorhexidine and tincture of iodine (Humco, Texarkana, TX, USA). After removal of the IRM and sponge each canal was irrigated with 10 mL 1.25% NaOCl (Clorox, Oakland, CA, USA) and rinsed with 10 mL of sterile saline (0.9% NaCl, Hospira Inc.) without using a mechanical instrumentation.
A paste of equal quantities of metronidazole, minocycline, and ciprofloxacin with saline was introduced into the canal after drying with sterile paper points (Dentsply Maillefer, Tulsa, OK, USA) [30]. Using a Lentulo spiral (Dentsply Maillefer, Johnson City, TN, USA), the paste was then applied in the canal space with a slow-speed hand-piece. The teeth were closed temporarily with sterile sponges, IRM, and glass ionomers for 2 weeks to permit complete disinfection. After 2 weeks of the aseptic condition, the canals were re-penetrated and irrigated with NaOCl and saline, and the teeth were then ready for stem cell transplantation (Figure 1D) [5].
Phase V: transplantation of stem cells, growth factors, and scaffold into root canals
Each root canal of group A teeth received an aliquot of 0.2 mL containing expanded AD-MSCs at a dose of 5 × 105 cells/mL [31] and the growth factors VEGF-2 (10 ng/mL), bFGF (100 ng/mL), PDGF (10 ng/mL), nerve growth factor (NGF; 50 ng/mL, R&D, Minneapolis, MN, USA), and BMP-7 (100 ng/mL). The growth factors were adsorbed on a chitosan hydrogel scaffold seeded with the differentiated AD-MSCs [31]. The chitosan hydrogel scaffold was fabricated by dissolving 1 g chitosan (deacylation degree, 77%, high-molecular weight; Sigma- Aldrich) in a 2% solution of acetic acid with agitation. Then, sodium alginate powder (Sigma-Aldrich) was dissolved in 25 mL distilled water, and the dissolved chitosan was added and mixed to achieve a homogenous mixture. The pulp chambers and root canals of group A teeth were injected with the combination of AD-MSCs and growth factors in the chitosan hydrogel scaffold, whereas the root canals of group B were injected with the chitosan hydrogel scaffold with growth factors alone. Finally, the teeth of both groups were sealed with a double coronal seal of white MTA (Dentsply Tulsa Dental, Johnson City, TN, USA) and composite (P60; 3M Dental Products, St Paul, MN, USA) (Figures 1E, 1F and 2).
Figure 2: Induction of apical periodontitis, disinfection, and stem cell transplantation. Experimental group (A) received AD-MSC and growth factors within chitosan hydrogel scaffold, whereas Control group (B) received growth factors within chitosan hydrogel scaffold. A: Exposure of group (A); B: Exposure of group (B); C: Induction of apical periodontitis in teeth of group A; D: Disinfection of teeth of groups (A) and (B); E: Transplantation of construct of stem cells, growth factors, and scaffolds in root canals of group (A); F: Double coronal seal of MTA and composite of groups (A) and (B).
Phase VI: radiographic analysis
The investigated teeth of groups A and B were blindly monitored radiographically monthly for 4 months to determine the presence or absence of healing of the periradicular lesion, root wall thickening, and apical lengthening and closure. The radiographs obtained at the time of disinfection were compared with the follow-up radiographs obtained after treatment procedures. All images were taken using a storage phosphor plate (size 2) covering the experimental (group A) and control sites (group B) of each dog, using the parallel technique with an Xtention Con Paralleling Instrument (XCP) receptor holding device (Dentsply Rinn, Elgin, III Santa Monica, CA, USA). For standardization, custom bite registration material (Regisil; Dentsply Caulk, Milford, DE, USA) using additional silicone impression material (ExpressTM VPS Impression Material, 3M ESPE, USA) was applied to the biting surface of the XCP holder. These radiographic aids were used in the same position of the bite every time the animal was radiographed. The exposure was performed at 70 kV and 8 mA for 0.04 s (Orix-70; ARDET, Italy) [5].
Phase VII: histopathological study
The animals were euthanized under general anesthesia, and the jaws with the manipulated teeth were resected and placed in formaldehyde (Fisher Scientific, Pittsburgh, PA, USA). After soft tissue and excess hard tissue were removed from the specimens, the teeth were decalcified by first placing them in Formical (Decal Chemical Corporation, Tallman, NY, USA) for 6 days and then in Immunocal (Decal Chemical Corporation) for 2 months. The teeth were then removed from the jaws, embedded in paraffin, sectioned longitudinally (labiopalatally) every 5 μm through the apical foramen of the roots, stained with hematoxylin and eosin, and was evaluated blindly under light microscopy for the presence or absence of healthy, vital, and newly formed mineralized tissue (pulp/dentin-like tissue).
Statistical analysis
Statistical analyses were performed using the statistical package for the social sciences (SPSS) version 18.0 for Windows (SPSS Inc., Chicago, IL, USA). The comparison between the groups was performed using the chi-square test; statistical significance was set at P<0.05.
Results
Characterization of cultured AD-MSCs
The AD-MSCs isolated from adipose tissue had a fibroblastic spindle shape and attained 80% confluence. After the third passage, Immunophenotypic characteristics results for adipose tissue-derived mesenchymal stem cell by flow cytometric analysis (Figures 3A-3D) revealed that a homogenous population of AD-MSCs was obtained. The cells were CD29-positive (Figure 3A), CD90-positive (Figure 3B), CD45- negative (Figures 3C and 3D), isotopic control (Figure 3D).
Differentiation potential of AD-MSCs
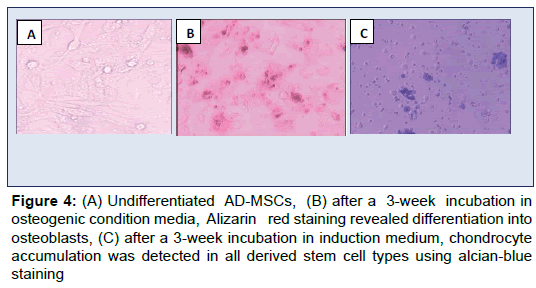
Representative images of the differentiated AD-MSCs (Figure 4A) show that the cells grown in osteogenic inductive medium for 21 days were positive for alizarin red staining (Figure 4B). The cells were also positive for alcian blue staining, demonstrating the ability to differentiate into chondrocytes (Figure 4C).
Figure 4: (A) Undifferentiated AD-MSCs, (B) after a 3-week incubation in osteogenic condition media, Alizarin red staining revealed differentiation into osteoblasts, (C) after a 3-week incubation in induction medium, chondrocyte accumulation was detected in all derived stem cell types using alcian-blue staining
Expression of stem cell markers of AD-MSCs using RT-PCR
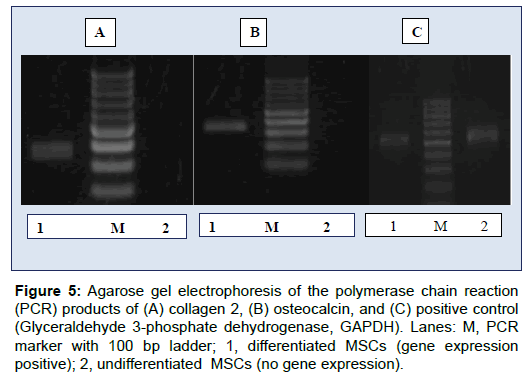
Both the undifferentiated and differentiated AD-MSCs were assessed for the expression of osteogenic markers (osteocalcin) and chondrogenic markers (collagen type 2) using the expression of GAPDH as the positive control. All the AD-MSCs expressed these markers (Figures 5A-5C).
Figure 5: Agarose gel electrophoresis of the polymerase chain reaction (PCR) products of (A) collagen 2, (B) osteocalcin, and (C) positive control (Glyceraldehyde 3-phosphate dehydrogenase, GAPDH). Lanes: M, PCR marker with 100 bp ladder; 1, differentiated MSCs (gene expression positive); 2, undifferentiated MSCs (no gene expression).
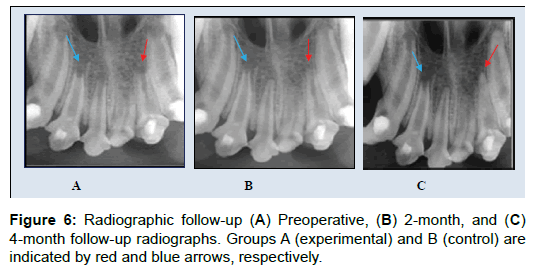
Radiographic findings
Chi-square test analysis of the radiographic results showed a statistically significant increase in radicular thickening, root lengthening, and apical closure after treatment (P=0.003) in group A compared to group B. However, there was no statistically significant difference between the groups for degree of apical radiolucency (P=1.000) (Table 1 and Figure 6).
| Group | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Tested | A (N=12) | B (N=12) | P-value | ||||||
| characteristics | YES | NO | YES | NO | |||||
| n (%) | n (%) | n (%) | n (%) | ||||||
| Radiographic Healing of Periapical Radiolucency | 11 | (91.67) | 1 | (8.33) | 10 | (8.33) | 2 | (16.67) | 1 |
| Radicular thickening | 11 | (91.67) | 1 | (8.33) | 3 | (8.33) | 9 | (75) | 0.003* |
| Radicular lengthening | 9 | (75) | 3 | (25) | 1 | (25) | 11 | (91.67) | 0.003* |
| Apical closure | 10 | (83.33) | 2 | (16.67) | 2 | (16.67) | 10 | (83.33) | 0.003* |
| *P < 0.05 | |||||||||
Table 1: Comparison of radiographic analysis outcomes for the two study groups.
Histological findings
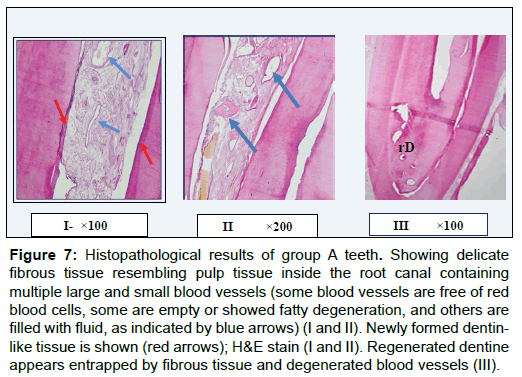
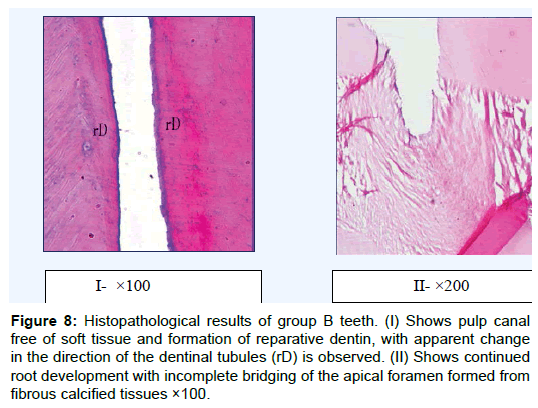
The histopathological analysis of the teeth of the experimental group (Figure 7) transplanted with the chitosan scaffold enriched with AD-MSCs and growth factors showed that the previously empty canals were filled with newly generated pulp-like tissue. Fibrous tissue with a detached pulp stone was also detected. There were multiple large and small blood vessels; some were empty, while others showed fatty degeneration or contained fluid. The new pulp-like tissue resembled natural pulp. In contrast, no soft tissue formed in the pulp canals of the control teeth (Figure 8).
Figure 7: Histopathological results of group A teeth. Showing delicate fibrous tissue resembling pulp tissue inside the root canal containing multiple large and small blood vessels (some blood vessels are free of red blood cells, some are empty or showed fatty degeneration, and others are filled with fluid, as indicated by blue arrows) (I and II). Newly formed dentinlike tissue is shown (red arrows); H&E stain (I and II). Regenerated dentine appears entrapped by fibrous tissue and degenerated blood vessels (III).
Figure 8: Histopathological results of group B teeth. (I) Shows pulp canal free of soft tissue and formation of reparative dentin, with apparent change in the direction of the dentinal tubules (rD) is observed. (II) Shows continued root development with incomplete bridging of the apical foramen formed from fibrous calcified tissues ×100.
A new layer of generative dentin-like tissue was formed on the canal walls of both groups. There were some apparent gaps between the new layer of deposited dentin-like tissue and the original dentin because of histologic processing artifacts. The regenerated dentinlike tissue showed a few regions where the odontoblasts were highly aligned. Some fibrous tissue and degenerated blood vessels were seen to be entrapped in the regenerated dentine. Apical closure was observed in 10 out of 12 in group A teeth, whereas 2 out of 12 in group B teeth showed this feature (Table 1).
Discussion
The present study was an in vivo experimental study implemented in the canine model to evaluate the role of regeneration of dental pulpal tissue in necrotic immature permanent teeth. The study involved the isolation, propagation, and characterization of MSCs from the adipose tissue of dogs. We induced periapical periodontitis in necrotic non-vital immature permanent teeth of dogs and studied the effect of AD-MSCs on radicular thickening, root lengthening, and apical closure clinically, histologically, and radiologically. The isolated cells showed the morphological characteristics of MSCs; differentiated into osteoblasts, chondrocytes, and adipocytes; and expressed MSC markers. In line with our findings, Guercio [32] isolated and characterized MSCs from the adipose tissue of adult dogs and confirmed their ability to differentiate into different phenotypes and produce pluripotency markers, which are usually produced by embryonic stem cells. The MSCs were used to treat osteoarthritis of the humeroradial joints. Furthermore, Reich [33]. Isolated MSCs from the intra-abdominal fat of dogs and reported that the cells showed rapid population doubling and chondrogenic differentiation potential.
In our study, radiographic analysis of the experimental group, which received AD-MSCs, showed a statistically significant increase in radicular thickening, root lengthening, and apical closure compared to the control group, which received growth factors but no MSCs. Histological investigation showed pulp-like tissue regeneration in the root canals of group A teeth but not group B teeth, which showed no soft tissue formation. Group A teeth also contained a layer of tissue with dental structure and dentinal tubules. The disorganized arrangement of this newly formed dentin-like tissue and the arrangement of the odontoblast-like cells might be attributable to the replacement of the dental tubules by the newly differentiated tissue. Ishizaka [31] evaluated the adipose tissue and bone marrow of the same individual as alternate sources of pulp stem/progenitor cells for the induction of complete pulp regeneration. They demonstrated that adipose CD31-SP transplantation resulted in the formation of similar amounts of regenerative tissue to the pulp stem/progenitor cells. They also reported that transplanted bone marrow CD31-SP cells produced the least amount of regenerated tissue.
The combination of growth factors and stem cells has been reported to regenerate dental pulp [34]. Autologous pulp stem cells were transplanted with granulocyte-colony stimulating factor (G-CSF) in a pulpectomized tooth of a dog. Full regeneration of pulp tissue and dentin was achieved, filling the defect up to the coronal part, and microleakage was prevented for up to 180 days. The use of G-CSF in concert with stem cells yielded a significantly larger amount of dentin/ pulp tissue compared with the transplantation of either agent alone. Histological examination showed a reduction in inflammatory cells and apoptotic cells without toxicity or adverse events. In another study [13], autologous transplantation of CD31-/CD146- SP cells plus stromal cell-derived factor-1 (SDF-1) into root canals after whole-pulp removal of mature teeth resulted in complete regeneration of pulp, replete with nerves and vasculature, by day 14, followed by dentin formation along the dentinal wall by day 35.
In tissue engineering, scaffolds provide the three-dimensional environment for cell attachment and growth. Injectable hydrogels are an appealing scaffold because they are easy to prepare and have homogenous cell distribution, structurally similar to the extracellular matrix of many tissues. In the present study, we used chitosan hydrogel as a scaffold owing to the ease of preparation and biocompatibility, solubility, biodegradability, reactivity, and lack of toxicity. Injectable chitosan hydrogels can be used as a delivery system, cell carriers, and scaffolds for tissue engineering. Furthermore, they can be processed under relatively mild conditions and delivered in a minimally invasive manner. Chitosan also inhibits the growth of many types of fungi, yeasts, and bacteria [35-38]. All these features make it an attractive option for several applications, such as in food and nutrition, medicine, microbiology, immunology, agriculture, and veterinary medicine.
Yamada [39] obtained MSCs from dental tissue stem cells (pDTSCs), parent canine DPSCs (cDPSCs), and canine bone marrow mesenchymal stem cells (cMSCs) extracted from deciduous teeth from puppies. These MSCs were grafted into a defect in a parent canine mandible as an allograft using platelet-rich plasma (PRP) as a scaffold. Bone regeneration in the defect was evaluated by histomorphometric analysis. The groups that received the cMSCs/PRP, cDPSCs/PRP, and pDTSCs/PRP had well-formed mature bone and neovascularization compared with the control (defect only) and PRP groups, and the mineralized tissues in cMSCs/PRP, cDPSCs/PRP, and pDTSCs/PRP specimens were positive for osteocalcin at 8 weeks. These results demonstrated that stem cells mixed with PRP scaffold have the ability to form dentine like tissue. A recent systematic review was reported by Kabir R [40]. They indicated that dental pulp stem cells (DPSCs) appear to be a promising innovation for the re-growth of tissues in the regeneration of the pulpal tissue. However, they mentioned that this merits further research. Though this research deals with the role of ADMSCs, the authors of the present study are already investigating the potential of DPSCs, in the regeneration of pulpal tissue and the results are being prepared for publications.
In our study, building fully functional pulp and teeth was an important goal. Our results indicated that a chitosan scaffold enriched with growth factors and AD-MSCs facilitated radicular thickening and generation of pulp/dentin-like tissue closely resembling normal tooth structure. Nakashima and Iohara [13] used SDF-1, a stem cell chemoattractant, while Iohara [33] used G-CSF, which is mainly associated with inflammation. In our study, a full panel of cytokines specific to nerve generation (NGF), blood vessels (VEGF), and connective and bone tissue (BMP-7, PDGF, and b-FGF) was used to regenerate dental pulp.
Some of the limitations of our study were the loss and replacement of some of the dogs because of their young age. Furthermore, working with dogs in Jeddah is not feasible, which is why we had to collaborate with Cairo University. In addition, this is an animal study and might not necessarily reflect the actual human response. Nevertheless, several studies have been performed using dog models. The authors encourage further researches in this field before applying this technique in humans.
Although this study was performed in an experimental animal model, we characterized the stem cells using different techniques, which adds to the strength of the research. Moreover, the use of AD-MSCs in young permanent teeth has never been attempted, and previous studies have only been performed in completely formed roots. This work points to the necessity of optimizing tissue engineering techniques to create low-cost biocompatible scaffolds enriched with suitable growth factors and autologous stem cells that are suitable for the regeneration of pulp/dentine-like tissue.
Conclusion
Painful and unpleasant dental problems such as pulp infection and periapical periodontitis can lead to difficulties in maintaining good oral hygiene, which further affects eating, drinking, taste, breathing, and well speech. These conditions can severely affect one’s quality life and well-being. Here, we demonstrated that implanting chitosan hydrogel scaffolds enriched with autologous AD-MSCs and growth factors might be useful in the regeneration of new pulp/dentin-like tissues in necrotic immature permanent teeth with apical periodontitis. AD-MSCs have the potential to survive well and generate new pulp/dentin-like tissues. The relative ease of obtaining subcutaneous fat in the clinical setting makes this technique very important for future applications in humans. The optimization of the type of scaffolds used and the growth factor type and amount warrants future investigation.While further research is needed before applying this technique in humans, we believe that our findings lay the foundation for a novel approach to treat necrotic permanent teeth resulting from trauma or infection.
Acknowledgment
This project was funded by the Deanship of Scientific Research (DSR) at King Abdulaziz University (Jeddah, Saudi Arabia) under grant Number 106-165/1432. The authors acknowledge with thanks the Deanship of Scientific Research DSR for their technical and financial support.
References
- Ingle JI, Bakland LK, Baumgartner JC (2008) Ingle’s endodontics (6thedn). BC Decker Inc., pp. 1330-1357.
- Yang M (2013) Regenerative endodontics: A new treatment modality for pulp regeneration. JSM Dent 1: 1011.
- Andreasen JO, Farik B, Munksgaard EC (2002) Long-term calcium hydroxide as root canal dressing may increase risk of root fracture. Dent Traumatol 18: 134-137.
- Giuliani V, Baccetti T, Pace R, Pagavino G (2002) The use of MTA in teeth with necrotic pulp and open apices. Dent Traumatol 18: 217-221.
- Thibodeau B, Teixeira F, Yamauchi M, Caplan DJ, Trope M (2007) Pulp revascularization of immature dog teeth with apical periodontitis. J Endod 33: 680-689.
- El-Backly RM, Massoud AG, El-Badry AM, Sherif RA, Marei MK (2008) Regeneration of dentine/pulp-like tissue using a dental pulp stem cell/poly (lactic-co-glycolic) acid scaffold construct in New Zealand white rabbits. Aust Endod J 34: 52-67.
- Casagrande L, Cordeiro MM, Nör SA,Nör JE (2011) Dental pulp stem cells in regenerative dentistry. Odontology 99: 1-7.
- Salem AM, Ahmed HH, Atta H (2011) Potential of bone marrow mesenchymal stem cells in management of Alzheimer’s disease in female rats. Cell Biol Int 38: 1367-1383.
- Sloan AJ, Smith AJ (2007) Stem cells and the dental pulp: Potential roles in dentine regeneration and repair. Oral Dis 13: 151-157.
- Gronthos S, Mankani M, Brahim J, Robey PG, Shi S (2000) Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proc Nat Acad Sci USA 97: 13625-13630.
- Miura M, Gronthos S, Zhao M, Lu B, Fisher LW, et al. (2003) SHED: Stem cells from human exfoliated deciduous teeth. Proc Nat Acad Sci U S A 100: 5807-5812.
- Iohara K, Imabayashi K, Ishizaka R, Watanabe A, Nabekura J, et al. (2011) Complete pulp regeneration after pulpectomy by transplantation of CD105 stem cells with stromal cell-derived factor-1. Tissue Eng Part A 17: 1911-1920.
- Nakashima M, Iohara K (2011) Regeneration of dental pulp by stem cells. Adv Dent Res 23: 313-319.
- Alamoudi NM, El Ashiry EA, Farsi NM, Douaa A, El Derwi, et al. (2014) Atta treatment of oral ulcers in dogs using adipose tissue-derived mesenchymal stem cells. J Clin Pediatr Dent 38: 215-222.
- Schaffler A, Büchler C (2007) Concise review: Adipose tissue-derived stromal cells-basic and clinical implications for novel cell-based therapies. Stem Cells 25: 818-827.
- Romanov YA, Darevskaya AN, Merzlikina NV, Buravkova LB (2005) Mesenchymal stem cells from human bone marrow and adipose tissue: isolation, characterization and differentiation potentialities. Bull Exp Biol Med 40: 138-143.
- Izadpanah R, Trygg C, Patel B, Kriedt C, Dufour J, et al. (2006) Biologic properties of mesenchymal stem cells derived from bone marrow and adipose tissue. J Cell Bioche 99: 1285-1297.
- Liu J, Yu F, Sun Y Jiang B, Zhang W, et al. (2015) Concise reviews: Characteristics and potential applications of human dental tissue-derived mesenchymal stem cells. Stem Cells 33: 627-638.
- Locke M, Feisst V, Dunbar PR (2011) Concise review: Human adipose-derived stem cells: separating promise from clinical need. Stem Cell 29: 404-411.
- Choudhery MS, Badowski M, Muise A, Harris DT (2013) Comparison of human adipose and cord tissue derived mesenchymal stem cells. Cytotherapy 15: 330-343.
- Aust L, Devlin B, Foster SJ, Halvorsen YD, Hicok K, et al. (2010) Yield of human adipose-derived adult stem cells from liposuction aspirates. Cytotherapy 46: 7-14.
- Charan J, Kantharia ND (2013) How to calculate sample size in animal studies? J Pharmacol Pharmacother 4: 303-306.
- Tomiyama K, Murase N, Stolz DB, Toyokawa H, O'Donnell DR, et al. (2008) Characterization of transplanted green fluorescent protein+ bone marrow cells into adipose tissue. Stem Cells 26: 330-338.
- Jaiswal N, Haynesworth SE, Caplan AI, Bruder SP (1997) Osteogenic differentiation of purified, culture-expanded human mesenchymal stem cells in vitro. J Cell Biochem 64: 295-312.
- Mokbel A, El-Tookhy O, Shamaa AA, Sabry D, Rashed L (2011) Homing and efficacy of intra-articular injection of autologous mesenchymal stem cells in experimental chondral defects in dogs. Clin Exp Rheumatol 29: 275-284.
- Abdel Aziz MT, El Asmar MF, Atta HM, Mahfouz S, Fouad HH, et al. (2011) Efficacy of mesenchymal stem cells in suppression of hepatocarcinorigenesis in rats: Possible role of Wnt signaling. J Exp Clin Cancer Res 30: 49-57.
- Qu H, Liu X, Ni Y, Zheng C (2014) Laminin 411 acts as a potent inducer of umbilical cord mesenchymal stem cell differentiation into insulin-producing cells. J Transl Med 12: 135.
- El-Menoufy H, Aly LA, Aziz MT, Roshdy NK, Rashed LA, et al. (2010) The role of bone marrow-derived mesenchymal stem cells in treating formocresol induced oral ulcers in dogs. J Oral Pathol Med 39: 281-289.
- Grone A, Fonfara S, Markus S, Baumgärtner W (1999) RT-PCR amplification of various canine cytokines and so-called house-keeping genes in a species-specific macrophage cell line (DH82) and canine peripheral blood leukocytes. Zentralbl Veterinarmed B 46: 301-310.
- Adl A, Shojaee NS, Motamedifar M (2012) A Comparison between the antimicrobial effects of triple antibiotic paste and calcium hydroxide against Enterococcus faecalis. Iran Endod J 7: 149-155.
- Ishizaka R, Iohara K, Murakami M, Fukuta O, Nakashima M (2012) Regeneration of dental pulp following pulpectomy by fractionated stem/progenitor cells from bone marrow and adipose tissue. Biomaterials 33: 2109-2118.
- Guercio A, Di Marco P, Casella S, Cannella V, Russotto L, et al. (2012) Production of canine mesenchymal stem cells from adipose tissue and their application in dogs with chronic osteoarthritis of the humeroradial joints. Cell Biol Int 36: 189-194.
- Reich CM, Raabe O, Wenisch S, Bridger PS, Kramer M, et al. (2012) Isolation, culture and chondrogenic differentiation of canine adipose tissue- and bone marrow-derived mesenchymal stem cells—a comparative study. Vet Res Commun 36: 139-148.
- Iohara K, Murakami M, Takeuchi N, Osako Y, Ito M, et al. (2013) A novel combinatorial therapy with pulp stem cells and granulocyte colony-stimulating factor for total pulp regeneration. Stem Cells Transl Med 2: 521-533.
- Drury JL, Mooney DJ (2003) Hydrogels for tissue engineering: Scaffold design variables and applications. Biomaterials 24: 4337-4351.
- Tan H, Ramirez CM, Miljkovic N, Li H, Rubin JP, et al. (2009) Thermosensitive injectable hyaluronic acid hydrogel for adipose tissue engineering. Biomaterials 30: 6844-6853.
- Tan H, Marra KG (2010) Injectable, biodegradable hydrogels for tissue engineering applications. Materials 3: 1746-1767.
- Croisier F, Jérôme C (2013) Chitosan-based biomaterials for tissue engineering. Eur Polym J 49: 780-792.
- Yamada Y, Ito K, Nakamura S, Ueda M, Nagasaka T, et al. (2011) Promising cell-based therapy for bone regeneration using stem cells from deciduous teeth, dental pulp, and bone marrow. Cell Transplant 20: 1003-1013.
- Kabir R, Gupta M, Aggarwal A, Sharma D, Sarin A, et al. (2014) Imperative role of dental pulp stem cells in regenerative therapies: A systematic review. Niger J Surg 20: 1-8.
Relevant Topics
- Advanced Bleeding Gums
- Advanced Receeding Gums
- Bleeding Gums
- Children’s Oral Health
- Coronal Fracture
- Dental Anestheia and Sedation
- Dental Plaque
- Dental Radiology
- Dentistry and Diabetes
- Fluoride Treatments
- Gum Cancer
- Gum Infection
- Occlusal Splint
- Oral and Maxillofacial Pathology
- Oral Hygiene
- Oral Hygiene Blogs
- Oral Hygiene Case Reports
- Oral Hygiene Practice
- Oral Leukoplakia
- Oral Microbiome
- Oral Rehydration
- Oral Surgery Special Issue
- Orthodontistry
- Periodontal Disease Management
- Periodontistry
- Root Canal Treatment
- Tele-Dentistry
Recommended Journals
Article Tools
Article Usage
- Total views: 5374
- [From(publication date):
July-2017 - Sep 03, 2025] - Breakdown by view type
- HTML page views : 4334
- PDF downloads : 1040