Research Article Open Access
The Acute Effects Different Quantities of Branched-Chain Amino AcidsHave On Recovery of Muscle Function
Harry F Dorrell* and Thomas I Gee
School of Sport and Exercise Science, University of Lincoln, Lincoln, UK
- *Corresponding Author:
- Harry F Dorrell
School of Sport and Exercise Science
College of Social Sciences
University of Lincoln
Lincoln, England, UK
Tel: +44(0)1522 887308
Fax: +44(0)1522 886026
E-mail: hdorrell@lincoln.ac.uk
Received Date: October 13, 2016; Accepted Date: November 21, 2016; Published Date: December 02, 2016
Citation: Dorrell H, Gee T (2016) The Acute Effects Different Quantities of Branched-Chain Amino Acids Have On Recovery of Muscle Function. Sports Nutr Ther 1: 115. doi: 10.4172/2473-6449.1000115
Copyright: © 2016 Dorrell H, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Nutrition Science Research
Abstract
The purpose of the study was to investigate the acute effects of two different quantities of branched-chain amino acids (BCAA) on the recovery of muscle function following high-intensity resistance training in trained individuals. Five resistance-trained males were recruited and performed baseline assessments of, countermovement (CMJ) and squat jump (SJ), peak (PP) and mean (MP) power (6s Cycle Sprint) and perceived muscle soreness (RMS). In a counterbalanced, single-blind fashion, participants were provided with either, placebo or 6 g or 18 g BCAA 20 min prior to, and immediately after completion of a high-intensity strength session (ST). At 24 h post ST, all assessments were completed under all three experimental conditions. Following ST, there were significant decrements in all measures of muscle function across conditions when compared to baseline values (P
Keywords
Amino Acid; Recovery; Strength Training; Muscle Function
Introduction
Resistance training is regarded as integral to many athletes’ periodised regimes. Adaptations including increased muscular hypertrophy, improved glycolytic capacity and increased neuromuscular power have been well established [1]. However, it is widely acknowledged that intensive resistance training leads to exercise-induced muscle damage (EIMD) that can last several days [2]. Symptoms of EIMD include decreased muscle function, reduced range of functional movement [3,4], increased muscle soreness and swelling [5], and increased serum intramuscular proteins [6]. Consequently EIMD can have an abstruse effect on the ability to complete successive bouts of physical training, thus directly impacting consistency within a given training programme [7]. Concomitantly, contemporary research has investigated a wide variety of interventions with the objective being to decrease or remove the negative effects associated with EIMD, these include, compression clothing [8], cryotherapy [9], and non-steroidal anti-inflammatory drugs [10]. Although all of these methods have theoretical foundations, they have generally produced varied results. However, one such nutritional intervention; branched-chain aminoacids (BCAA) supplementation, has repeatedly produced statistically positive results with regards to alleviating the symptoms associated with EIMD, and attenuating performance decrements commonly witnessed [11].
Supplementation with BCAA (a combination of leucine, valine and isoleucine) both pre- and post-training has been shown to alleviate some of the symptoms most commonly associated with EIMD, including, serum CK levels [12], immediate-onset muscle soreness [13], reductions in muscle strength [14], as well as muscle catabolism [11]. Statistically significant results have been reported when supplementing varied dosages of BCAA alongside resistance training, from 3 g pre and post exercise to 20 g pre and post exercise [2,13,15]. Although this data has shown that BCAA can be used to reduce the effects of EIMD following resistance training, to date, no scientific research has explored whether a lower dose of BCAA has the same magnitude of effect as a higher dose.
The purpose of the study was to investigate the acute effects of two different quantities of BCAA on the recovery of muscle function following high-intensity resistance training in trained individuals. The main aim being to examine whether a dose-response effect existed of using a higher dosage of BCAA on recovery when compared to a lower dosage. It was hypothesised that supplementation with 18 g BCAA pre and post training will significantly attenuate reductions in muscle function over 6 g BCAA pre and post training, with both dose conditions eliciting greater recovery of muscle function than placebo.
Materials and Methods
Participants
In a randomised fashion five resistance-trained males were recruited (mean ± SD, age: 21.8 ± 0.8 years, stature: 1.81 ± 0.57 m, body mass: 83.3 ± 7.2 kg). Following a health-screening questionnaire all participants provided written informed consent to participate in the study, which was approved by the local ethics committee in line with the Helsinki Declarations for research with human volunteers.
Procedures
The study followed a single-blind, counterbalanced design. Prior to testing, participants completed a 5 min warm-up on a cycle ergometer (60 rpm, 60 W). Following this, participants completed a battery of experimental tests which consisted the baseline trial; including countermovement jump, squat jump, seated medicine-ball chest throw, 6 s Cycle sprint and rating of perceived muscle soreness.
Participants were blinded and provided with either 6 or 18 g of BCAA or a placebo in a counterbalanced manner 20 min before and immediately after completion of a strength training session. Following 24 h rest, participants were required to return and recomplete the experimental tests, under the same conditions as described for baseline measures. Dietary and physical-activity patterns were documented over a 24 h period prior to the participant’s first bout of testing. Participants were then instructed to follow the same dietary and activity pattern, in preparation for successive assessments. Furthermore, participants were advised to abstain from taking additional supplements throughout the testing period, with particular emphasis placed upon protein-based products. Following a further 120 h washout period, participants repeated training / testing under alternative condition in the aforementioned counterbalanced manner.
Supplement protocol
The supplement, Optimum Nutrition BCAA 1000 (Optimum Nutrition, Cheshire, UK), contained a ratio of 2:1:1 leucine, isoleucine and valine respectively and was provided in powered form. Participants ingested either 6 g or 18 g of BCAA or a placebo (artificial sweetener) both 20 min before and immediately after completion of the resistance session. Both the supplement and placebo were pre-mixed with 300 ml of zero-calorie fruit squash with only the researcher knowing which drink contained which ingredient.
Strength training session (ST)
The resistance session consisted of a series of multi-joint barbell exercises selected based on their replication of the structural lifts often used as the basis of athletic strength training programmes [16]. The following exercises were performed; 1.Back Squat 2.Bench Press 3.Deadlift 4.Military Press, all performed for 4 sets of 8 repetitions at 75% of one-repetition maximum (1-RM), two-min rest was allocated between each set. Individual loading of exercises was determined from 1-RM assessment, established prior to the experimental protocol.
Experimental tests
Countermovement jump (CMJ) and Squat jump (SJ): The Just Jump measurement system (Just Jump, Probotics, Huntsville, AL, USA) was used to calculate vertical-jumping ability due to high levels of reliability (r=0.97) [17]. For the CMJ, participants stood fully erect in the middle of the contact mat with hands placed on iliac crest. Participants then squatted to their perceived optimum depth, before immediately driving upwards fully utilising the stretchshortening- cycle, with the aim of attaining maximum height. For the SJ, participants began in the same position as for CMJ. From standing, participants squatted to achieve a 90° angle at the knees which was held for 5 s, before explosively rising upwards into a vertical jump. Research suggests that both CMJ (r=0.93) and SJ (r=0.88) should be used assess muscle function as they evaluate different elements of lower bod power [18]. Each test was carried out on three separate occasions, interspaced with 30 s recovery.
Seated medicine-ball chest throw (MBT): Subjects were instructed to sit on the floor; legs straight out in front of them, with back fully erect and pressed against a wall. The medicine-ball (3 kg) was held with both hands behind the ball, at chest height. From this position, participants pushed the ball forward, as forcefully as possible, the distance was measured in meters from the participant’s chest to the landing point. This test has shown high levels of reliability (r=0.98) as a valid measure of upper body power [19].
6s Cycle sprint test: The 6 s cycle sprint test was used to assess any changes in peak (PP) and mean power (MP) following ST. Participants completed the test on a Monark 874E cycle ergometer (Monark Exercise AB, Vansbro, Dalarna, Sweden). Resistance was calculated as 7.5% of body mass as recommended in literature [20]. Both PP and MP have shown high levels of reliability [Typical error (%) 1.8 and 2.2% respectively] [21]. Due to the level of intensity, this test was only completed once during each testing period.
Rating of perceived muscle soreness (RMS): Participant’s RMS was assessed via a 200 mm visual analogue scale, as used previously [2]. While performing a static wall squat, with knees at 90°, participants were instructed to make a line on the scale, depicting their RMS. The scale is labelled ‘No pain or discomfort’ (0 mm) to ‘Extreme pain and/ or constant discomfort’ (200 mm). The distance between the 0 mm point and the participant’s mark was then measured in mm and used to represent perceived soreness [2].
Data analysis: All collected data is expressed as mean ± standard deviation (SD) unless otherwise stated. The best score (highest: CMJ, SJ, MBT, 6 s Cycle, lowest: RMS) was used for data analysis, as this has shown to significantly increase intersession reliability as opposed to means [22]. Changes were assessed across all conditions using a repeated measures ANOVA through SPSS for Windows, release 22.0 (SPSS, IBM Corporation, Armonk, NY, USA). If a significant maineffect across time was apparent, interactions were followed up using LSD post-hoc pairwise comparisons. The alpha level for significance was set at P<0.05 for all data, with trends showing P=0.051<0.1 acknowledged.
Results
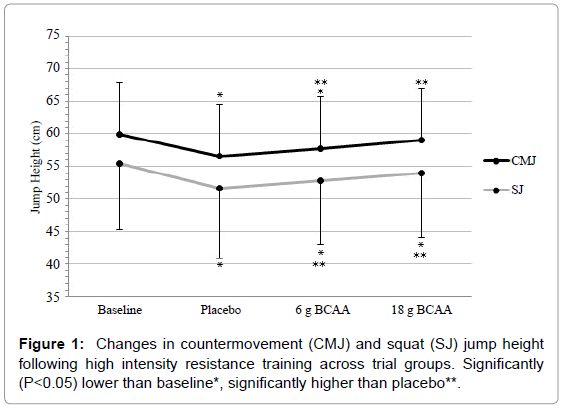
There were significant main-effects across conditions for CMJ (F=25.70, P<0.01) and SJ (F=20.55, P<0.01) (Figure 1). Following ST, significant decrements in CMJ and SJ performance were witnessed across all supplementation conditions in comparison to baseline measures (P<0.05). However, ingestion of both 6 g and 18 g of BCAA significantly (P<0.05) reduced these decrements for CMJ (57.7 ± 8.0 cm vs. 59.1 ± 7.9 cm vs. 56.6 ± 7.9 cm respectively) and SJ (52.8 ± 9.9 cm vs. 54.0 ± 9.9 cm vs. 51.7 ± 10.7 cm respectively) in comparison to the placebo trial. Furthermore, a significant effects were witnessed when comparing effects of 18 g to 6 g (CMJ: P=0.022, SJ: P=0.008).
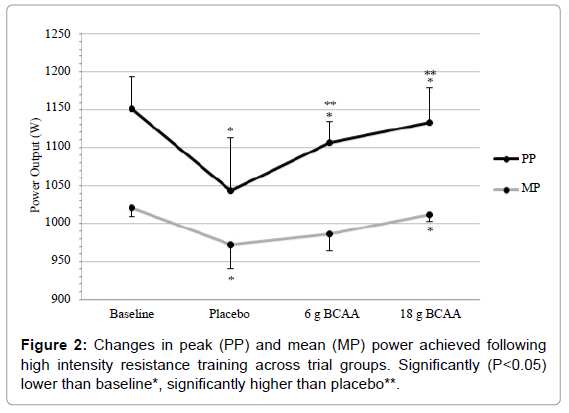
There was a significant main-effect across conditions for 6 s Cycle PP (F=16.49, P<0.01) and MP (F=7.06, P=0.005) (Figure 2). Following ST, significant decrements in PP and MP performance were witnessed across all supplementation conditions in comparison to baseline values (P<0.05). However, ingestion of both 6 g and 18 g of BCAA significantly (P<0.05) reduced the decrements in PP in comparison to the placebo trial (1107 ± 27 W vs. 1133 ± 46 W vs. 1044 ± 69 W respectively). Whilst ingestion of 6 g and 18 g BCAA showed trends approaching significance for MP (P=0.067 and P=0.062 respectively) in comparison to placebo (986 ± 21 W vs. 1011 ± 9 W vs. 972 ± 32 W respectively). Furthermore, trends were witnessed following ST when comparing effects of 18 g to 6 g for PP (P=0.090) and MP (P=0.076).
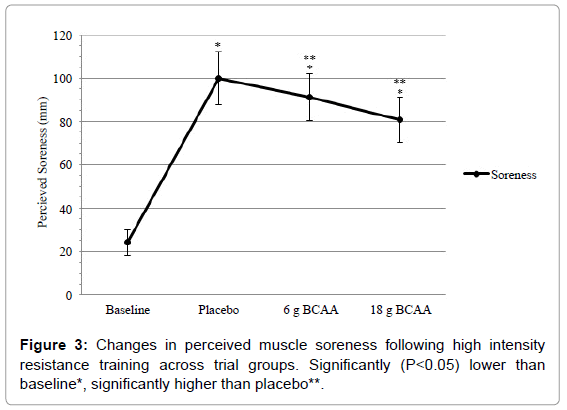
There was no significant main-effect across conditions for MBT (F=2.17, P=0.144). Following ST, no significant effect was witnessed for the placebo trial in comparison to baseline measures (P>0.05), however 18 g BCAA showed significance (P=0.004), with 6 g BCAA showing a trend approaching significance (P=0.085). There was a significant main-effect across conditions for RMS (F=167.49, P<0.01) (Figure 3). Following ST, significant increases in RMS scores were witnessed across all groups in comparison to baseline values (P<0.05). However, ingestion of both 6 g and 18 g of BCAA significantly (P<0.05) reduced these increases in comparison to the placebo trial (91 ± 11 mm vs. 81 ± 11 mm vs. 100 ± 12 mm respectively). Furthermore, a significant effect was witnessed when comparing effects of 18 g to 6 g (P=0.025).
Discussion
This was the first empirical study with the aim of investigating the acute effects of two varying doses of BCAA have on recovery of muscle function following conventionally practised strength training. The principle findings showed that both 6 g and 18 g BCAA, administered pre and post intensive resistance training, attenuate the decrease commonly witnessed in muscle function, while relieving symptoms of muscle soreness in resistance trained males. Furthermore, the findings have highlighted that the magnitude of effect is significantly increased (P<0.05) following the higher dosage when compared to both the lower dose and placebo. These effects were in accordance with the experimental hypotheses. The findings support previous literature suggesting BCAA can be supplemented to relieve the negative effects of muscle damaging exercise [13,23] and more specifically damaging resistance exercise [24-26].
Following ST, decrements in CMJ and SJ were recorded in the placebo group at ~6 and 7% respectively when compared to baseline values. Previous authors have recorded comparable decrements in jump performance, when participants have followed similar resistance protocols, at 24 h post-exercise [27,28]. However, ingestion of BCAA was shown to significantly attenuate these performance decrements, with 6 g BCAA resulting in ~2% increase and 18 g BCAA resulting in ~4% increase across both CMJ and SJ when compared to the placebo trials. Additional significant decrements were recorded in both PP and MP of the 6 s Cycle during the placebo trial in comparison to baseline values following completion of the ST. These decrements, ~8% (PP) and ~5% (MP), were reduced following ingestion of BCAA at both doses. When comparing the 6 g trials to the PP and MP placebo values, increases of ~6 and 2% respectively were witnessed. Likewise, with regards to the 18 g trials, increases of ~9 and 4% respectively were recorded in comparison to placebo values. Furthermore, RMS values rose significantly following completion of the ST in all groups, with the placebo group increasing by ~300% when compared to baseline values. This vast increment was significantly decreased following both 6 g BCAA (~9%) and 18 g BCAA (~19%) when compared to placebo values.
In the present study, BCAA ingestion has been shown to alleviate the decrements reported in muscle function (~2-8%), as well as the increments in RMS following high intensity resistance training. The attenuations witnessed after ingestion of BCAA were slightly less prominent than those reported by Howatson et al., [2] (~9%) following repetitive concentrically loaded, eccentric muscle actions. However, this could be attributed to the more ‘representative’ nature of the ST in this study, as opposed to protocol completed by Howatson et al., [2]. The ST was designed to replicate the demands of traditionally practiced athletic strength sessions, rather than predominantly focus on inducing extreme levels of EIMD and subsequent decrements in functional performance and elevations in markers of muscle damage [16,27]. Repetitive high force, eccentric muscle actions, as used by Howatson et al., [2], result in amplified EIMD, as witnessed by the vast decrements in maximal voluntary contraction (~20-27%) [5,26] and increments in RMS across groups [2,29]. This is relevant, as contemporary research has shown that perceptible effects on muscle function, created through prophylactic interventions such as BCAA ingestion, are likely to be more noticeable under conditions of greater muscle damage [30]. Furthermore, evaluation of Howatson et al., [2] highlights the vast increase in BCAA dosage given to participants throughout the study period in comparison to the present study. The total dose administered to each participant by Howatson et al., [2] was 260 g (7 day loading of 20 g/day, 20 g pre and post exercise, 20 g/day for 96 h following exercise) as opposed to either 12 g (6 g BCAA pre and post) or 36 g (18 g BCAA pre and post) in the present study.
Research has highlighted two main mechanisms related to BCAA ingestion and the subsequent effects on muscle function following exercise, including increased stimulation of protein synthesis and suppression of skeletal muscle protein proteolysis [2,31,32]. Matsumoto et al., [33] reported that a relatively small dose of BCAA (2 g) taken pre cycling exercise provided protection against skeletal muscle proteolysis in healthy participants. This information becomes relevant when considering work by Koba et al., [34] where it was reported that muscle proteolysis during exercise was directly related to subsequent muscle damage. In the present study, significantly greater doses (6 g or 18 g) were administered to the participants pre and post resistance exercise, and thus is it reasonable to advocate that the anti-catabolic properties of BCAA proposed by Matsumoto et al., [33] directly influenced the observed recovery of muscle function in comparison to the placebo group.
Furthermore, ingestion of BCAA-rich supplements including essential amino-acids, whey protein hydrolysate and whey protein isolate post-resistance training, has been reported to positively augment muscle protein synthesis as well as aid recovery [35,36]. Since BCAA ingestion has been shown to replicate the effect of a complete mixture of amino-acids in the stimulation of protein synthesis [37], it would seem rational that BCAA supplementation, following resistance training, would yield similar benefits to those listed above. Indeed, BCAA supplementation, alongside exercise, has been shown to stimulate mTOR and p70-S6 kinase, both of which are key regulators in cell growth, transcription and protein synthesis [38,39]. Moreover, contemporary research has revealed that under inflammatory conditions, such as those experienced after intense exercise, BCAA can be transaminated to glutamine, a substrate highly consumed by inflammatory cells and linked to attenuation of strength decrements and DOMS following resistance exercise [31,40].
One of the main limitations from the present study was the lack of physiological muscle damage markers taken such as serum levels of creatine kinase (CK), lactate dehydrogenase (LDH), myoglobin and myosin heavy chain fragments (MHC). Assessment of these markers, through venous blood samples, would have allowed for the detection of muscle proteins present in the blood following completion of the ST [28,41]. Collection of such data would enable physiological evidence to support the decrements commonly witnessed in functional testing following damaging protocols. A further limitation was the lack of specific dietary control across participants. This may have lead to inconsistencies in caloric, and more specifically protein ingestion directly impacting upon results. Participants were required to record a 24 h food diary and mimic this on training days, thus increasing consistency independently, however no specific intervention was in place between participants. Potential differences in protein intake would affect the bioavailability of substrate and thus directly influence protein turnover and fundamentally affect the data collected [42,43].
Further research should look to incorporate a longer, and more specific, dietary analysis to enable greater insight into both macro and micronutrient consumption, thus removing, or at least acknowledging, potential difference across trials and/or participants.
Conclusion
This was the first study with the aim of investigating the acute effects two varying doses of BCAA have on recovery of muscle function following conventionally practised strength training in resistance trained athletes. This study found that both 6 g and 18 g BCAA, administered pre and post intensive resistance training, attenuate the decrease commonly witnessed in muscle function, while relieving symptoms of muscle soreness. Furthermore, the findings have highlighted that the magnitude of effect is significantly increased following the higher dosage when compared to both the lower dose and placebo. The ergogenic benefit of BCAA is likely due to a synergistic combination of increased stimulation of protein synthesis and suppression of skeletal muscle protein proteolysis. Athletes whose performance and training regimen requires repetitive, dynamic, highpowered muscle actions, should consider ingestion of 18 g BCAA prior to and immediately following intensive strength training bouts. Thereby, acting as a means to initiate augmented muscle recovery and concomitant maintenance of quality training output.
References
- Cormie P, McGuigan M, Newton R (2011) Developing maximal neuromuscular power. Sports Med 41: 17-38.
- Howatson G, Hoad M, Goodall S, Tallent J, Bell P, French D (2012) Exercise-induced muscle damage is reduced in resistance-trained males by branched chain amino acids: a randomized, double-blind, placebo controlled study. J Int Soc Sports Nutr 9: 20-27.
- Ingham S, van Someren K, Howatson G (2010) Effect of a concentric warm-up exercise on eccentrically induced soreness and loss of function of the elbow flexor muscles. J Sports Sci28: 1377-1383.
- Skurvydas A, Brazaitis M, Venckusas T, Kamandulis S, Stanislovatis A, et al (2011) The effect of sports specialization on musculus quadriceps function after exercise-induced muscle damage. Appl Physiol Nutr Metab 36: 873-881.
- Howatson G, van Someren K (2008) The Prevention and Treatment of Exercise-Induced Muscle Damage. Sports Med38: 483-503.
- Fernandez-Gonzalo R, Lundberg T, Alvarez L, Paz J (2014) Muscle damage responses and adaptations to eccentric-overload resistance exercise in men and women. European J Appl Phy114: 1075-1084.
- Beneka A, Malliou P, Missailidou V, Chatzinikolaou A, Fatouros I, Gourgoulis V, Georgiadis E (2013) Muscle performance following an acute bout of plyometric training combined with low or high intensity weight exercise. J Sports Sci 31: 335-344.
- Hill J, Howatson G, van Someren K, Leeder J, Pedlar C (2014) Compression garments and recovery from exercise-induced muscle damage: a meta-analysis. Br J Sports Med48: 1340-1347.
- Burgess T, Lambert M (2010) The efficacy of cryotherapy on recovery following exercise-induced muscle damage. Int SportMed J11: 258-278.
- Schoenfeld B (2012) The use of nonsteroidal anti-inflammatory drugs for exercise-induced muscle damage: Implications for skeletal muscle development. Sports Med42: 1017-1029.
- Marangon A, Lacerda V, Correa R (2010) Effect of supplementation of branched chain amino acids in muscle damage induced by resistance training. J Int Soc Sports Nutr 7: 3-10.
- Nosaka K, Sacco P, Mawatari K (2006) Effects of amino acid supplementation on muscle soreness and damage. Int J Sport Nutr Exerc Metab 16: 620-636.
- Ishikura K, Miyazaki T, Ra S, Ohmori H (2014) The Ameliorating Effect of Branched-chain Amino Acids Ingestion on Different Types of Muscle Soreness after Swimming and Full-marathon Running. Adv Exercise and Sports Phy20: 9-18.
- Kraemer W, Ratamess N, Volek J, Hakkinen K, Rubin M, et al (2006) The effects of amino acid supplementation on hormonal responses to resistance training over reaching. Metabolism55: 282-291.
- Shimomura Y, Kobayashi H, Mawatari K, Akita K, Inaguma A, Watanabe S, Bajotto G, Sato J (2009) Effects of squat exercise and branched-chain amino acid supplementation on plasma free amino acid concentrations in young women. J Nutr Sci Vitaminol55: 288-291.
- Gee T, Olsen P, Berger N, Golby J, Thompson K (2011) Strength and conditioning practices in rowing. J strength cond Res 25: 668-682.
- Leard J, Cirillo M, Katsnelson E, Kimiatek D, Miller T, Trebincevic K, Garbalosa J (2007) Validty of two alternative systems for measuring vertical jump height. The J Strength and Cond Res 21: 1296-1299.
- Slinde F, Suber C, Suber L, Edwen C, Svantesson U (2008) Test-retest reliability of three different countermovement jumping tests. The J Strength Cond Res 22: 640-644.
- Harris C, Wattles A, DeBeliso M, Sevene-Adams P, Berning J, Adams K (2011) The seated medicine ball throw as a test of upper body power in older adults. J Strength and Cond Res 25: 2344-2348.
- Lopez E, Smoliga J, Zavorsky G (2014) The Effect of Passive Versus Active Recovery on Power Output Over Six Repeated Wingate Sprints. Res Q Exerc Sport 85: 519-527.
- Mendez-Villanueva A, Bishop D, Hamer, P (2007) Reproducibility of a 6-s maximal cycling sprint test. J Sci Med Sport 10: 323-326.
- Moir G, Shastri P, Connaboy C (2008) Intersession reliability of vertical jump height in women and men. J Strength Cond Res 22: 1779-1784.
- Kim D, Kim S, Jeong W, Lee H (2013) Effect of BCAA intake during endurance exercises on fatigue substances, muscle damage substances, and energy metabolism substances. J Exerc Nutrition Biochem 17: 169-180.
- Atashak S, Baturak K (2012) The Effect of BCAA Supplementation on Serum C - Reactive Protein and Creatine Kinase after Acute Resistance Exercise in Soccer Players. Annals Bio Res3: 1569-1576.
- Ra S, Miyazaki T, Ishikura K, Nagayama H, Komine S, et al (2013) Combined effect of branched-chain amino acids and taurine supplementation on delayed onset muscle soreness and muscle damage in high-intensity eccentric exercise. J Int Soc Sports Nutr 10: 51-62.
- Shimomura Y, Inaguma A, Watanabe S, Yamamoto Y, Muramatsu Y, et al (2010) Branched-Chain Amino Acid Supplementation Before Squat Exercise and Delayed-Onset Muscle Soreness. Int J Sport Nutr Exerc Metab 20: 236-244.
- Gee T, French D, Howatson G, Payton S, Berger N, et al (2011) Does a bout of strength training affect 2,000 m rowing ergometer performance and rowing-specific maximal power 24 h later? Eur J Appl Physiol 111: 2653-2662.
- Gee T, Olsen P, Fritzdorf S, White D, Golby J, et al (2012) Recovery of rowing sprint performance after high intensity strength training. Int J Sports Sci Coach 7: 109-120.
- Jackman S, Witard O, Jeukendrup A, Tipton K (2010) Branched-chain amino acid ingestion can ameliorate soreness from eccentric exercise. Med Sci Sports Exerc 42: 962-971.
- da Luz C, Nicastro H, Zanchi N, Chaves D, Lancha Jr A (2011) Potential therapeutic effects of branched-chain amino acids supplementation on resistance exercise-based muscle damage in humans. J Int Soc Sports Nutr 8: 23-27.
- Nicastro H, da Luz C, Chaves D, Bechara L, Voltarelli V, et al (2012) Does branched-chain amino acids supplementation modulate skeletal muscle remodeling through inflammation modulation? Possible mechanisms of action. J Nutr Metab 20: 10-19.
- Zanchi NE, Nicastro H, Lancha A Jr (2008) Potential antiproteolytic effects of L-leucine: observations of in vitro and in vivo studies. Nutr Metab 5: 20.
- Matsumoto K, Mizuno M, Mizuno T, Dilling-Hansen B, Lahoz A, et al (2007) Branched-chain amino acids and arginine supplementation attenuates skeletal muscle proteolysis induced by moderate exercise in young individuals. Int J Sports Med 28: 531-538.
- Koba T, Hamada K, Sakurai M, Matsumoto K, Hayase H, et al (2007) Branched-chain amino acids supplementation attenuates the accumulation of blood lactate dehydrogenase during distance running. J Sports Med Phys Fitness 47: 316-322.
- Dreyer H, Drummond M, Pennings B, Fujita S, Glynn E, et al (2008) Leucine-enriched essential amino acid and carbohydrate ingestion following resistance exercise enhances mTOR signaling and protein synthesis in human muscle.Am J Physiol Endocrinol Metab 294: 392-400.
- Tang J, Moore D, Kujbida G, Tarnopolsky M, Phillips S (2009) Ingestion of whey hydrolysate, casein, or soy protein isolate: effects on mixed muscle protein synthesis at rest and following resistance exercise in young men. J Appl Physiol 107: 987-992.
- Kimball S, Jefferson L (2006) Signalling pathways and molecular mechanisms through which branched-chain amino acids mediate translational control of protein synthesis. J Nutr 136: 227-231.
- Melnik B, Schmitz G, John S, Carrera-Bastos P, Lindeberg S, et al (2013) Metabolic effects of milk protein intake strongly depend on pre-existing metabolic and exercise status. Nutr Metab 10: 60-66.
- Neishabouri S, Hutson S, Davoodi J (2015) Chronic activation of mTOR complex 1 by branched chain amino acids and organ hypertrophy. Amino Acids 2: 1-16.
- Street B, Bryne C, Eston R (2011) Glutamine supplementation in recovery from eccentric exercise attenuates strength loss and muscle soreness. J Exercise Sci Fit 9: 116-122.
- Clarkson P, Hubal J (2002) Exercise-induced muscle damage in humans. Am J Phys Med Rehabil 81: 52-69.
- Churchward-Venne T, Burd N, Phillips M (2012) Nutritional regulation of muscle protein synthesis with resistance exercise: strategies to enhance anabolism. Nutr Metab 9: 40-48.
- Moore R, Camera M, Areta L, Hawley A (2014). Beyond muscle hypertrophy: why dietary protein is important for endurance athletes. Appl Physiol Nutr Metab 39: 987-997.
Relevant Topics
- Aminoacid Suppliments
- Bodybuilding Nutrition
- Clinical Sports Nutrition
- Creatine Sports Nutrition
- Diet
- Fitness Nutrition
- Food and Nutrition
- Gym Suppliments
- Herbal Suppliments
- Micronutrients
- Natural Suppliments
- Nutrition Sport Fitness
- Nutritional Health
- Protein Diet
- Protein Suppliments
- Sports Nutrition Suppliments
- Vitamin Supplement
Recommended Journals
Article Tools
Article Usage
- Total views: 5514
- [From(publication date):
December-2016 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 4457
- PDF downloads : 1057