Research Article Open Access
The Apparent Diffusion Coefficient in Glial Tumors
Tan FC1*, Gokoglu A2, Tucer B3 and Caner Y11Department of Biophysics, Erciyes University, Kayseri, Turkey
2Department of Neurosurgery, Kayseri Education and Research Hospital, Kayseri, Turkey
3Department of Neurosurgery, Acıbadem Hospital, Kayseri, Turkey
- *Corresponding Author:
- Fazile Canturk Tan
Department of Biophysics
Faculty of Medicine, Erciyes University
Kayseri, Turkey
Tel: +90-505-657 5327
E-mail: fcanturk@erciyes.edu.tr
Received Date: February 16, 2017; Accepted Date: March 13, 2017; Published Date: March 20, 2017
Citation: Tan FC, Gokoglu A, Tucer B, Caner Y (2017) The Apparent Diffusion Coefficient in Glial Tumors. OMICS J Radiol 6:256. doi: 10.4172/2167-7964.1000256
Copyright: © 2017 Tan FC, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Radiology
Abstract
Background: Diffusion-Weighted Imaging (DWI) is one of the fast developing techniques in the field of MRI. This method uses the random diffusion motion of water molecule depending on physiological and anatomical characteristics of living organisms. Aim: To calculate the apparent diffusion coefficient and diffusion values in order to determine the degree in the glial tumors. Patients and methods: In the study, cases were selected among those who presented with a diagnosis of intracranial mass to Neurosurgery Department of Erciyes University, Medicine School. Overall, 20 patients were included to the study: 7 patients with Glioblastoma Multiforme (GBM), 4 patients with anaplastic astrocytoma (AA), 4 patients with diffuse astrocytoma, 3 patients with ependymoma and 2 patients with low-grade astrocytoma. The apparent diffusion coefficient (ADC) images were acquired from echo-planar diffusion-weighted images (DWIs). For each tumor, the mean ADC values and the mean diffusion value were measured at a large region of interest (ROI) defined within tumor and at symmetrical normal brain tissue. Statistical analyses were performed by using SPSS 15.0 Statistical Package Program. Results: We calculated mean ADC values (2.91 × 10-3 mm2/s, 0.69 × 10-3 mm2/s, 3.13 × 10-3 mm2/s, 2.41 × 10-3 mm2/s and 0.70 × 10-3 mm2/s) and mean diffusion values (0.11 × 10-3 mm2/s, 1.6 × 10-3 mm2/s, 0.10 × 10-3 mm2/s, 1.58 × 10-3 mm2/s, 0.90 × 10-3 mm2/s) for GBM, AA, diffuse astrocytoma, low degree astrocytoma and ependymoma, respectively. The diffusion value of normal brain was calculated to be 0.85 × 10-3 mm2/s. A P-value <0.05 was considered statistically significant. We estimated a significant difference between GBM and AA (p=0.001). Although there were marked differences among diffuse astrocytomas, low degree astrocytoma and ependymoma, the difference did not reach statistical significance. Conclusion: The mean ADC value, the mean diffusion value and intensity region may provide additional information in determining tumor degree in malignant glial tumors.
Keywords
Brain tumor; Apparent Diffusion Coefficient (ADC); Diffusion Weighted Imaging (DWI); Region of Interest (ROI)
Introduction
The primary malignant central nervous system tumors account for about 2% of all cancers. In United States, 43.800 new cases are diagnosed as benign and malignant brain tumors annually. Of these, 3410 cases are children and adolescents while about 12,760 deaths occur among these patients. The malignant central nervous system tumors in children are leading cause of deaths from solid tumors whereas it is third leading cause of death among adolescents and adults aged 15-34 years [1]. Glial tumors are the largest group among central nervous system tumors. The average incidence is 4/100.000 [2]. Highgrade glial tumors are the most common and most infiltrative ones among primary brain tumors. They arise from astrocytes and oligodendrocytes and display varying malignancy degree in the spectrum. Astrocytic tumors are classified as astrocytoma, anaplastic astrocytoma and glioblastoma multiforme according to triple grade system. The grading is performed based on pathological characteristics (endothelial proliferation, cellular pleomorphism and mitosis). The presence of necrosis is diagnostic for glioblastoma multiforme.
In 1953, The World Health Organization (WHO) suggested that grade I CNS tumors are less aggressive and that grade IV CNS tumors are more aggressive. If it is failed to grade a tumor by a pathologist, the WHO grading system is used for prognosis and grading purposes [3]. According to current WHO classification, anaplastic (malignant) astrocytoma (AA), a high-grade glial tumor, is classified as grade III whereas Glioblastoma Multiforme (GBM) is classified as grade IV. GBMs are seen four-folds more common than AAs. These are highly malignant and infiltrate pathways along white matter; thus, they may reach large sizes before onset of symptoms [4]. High-grade gliomas include glioblastoma, anaplastic astrocytoma and anaplastic oligodendroglioma. These are highly invasive tumors. Tumor cells can be found 4 cm away from primary tumor mass. In the low grade glial tumors, the factors indicating good prognosis are as follows: Younger age, large resection performed, high Karnofsky performance score, contrast enhancement on Computed Tomography (CT) and magnetic resonance imaging (MRI), prolonged symptoms before surgery. In low grade glial tumors, mean survival is 7.5 years whereas 5-years and 10- years survival rates are 60% and 40%, respectively [5,6].
Diffusion-Weighted Imaging (DWI) is one of the fast developing techniques in the field of MRI. This method uses the random diffusion motion of water molecule depending on physiological and anatomical characteristics of living organisms. This technique is based on measurement of the Brownian motion of molecules. The Brownian motion is defined as heat-dependent free motion of molecules on three-dimensional media. It was first identified by Scottish Naturalist Robert Brown in 1827 [7] and it was measured by Einstein in 1905. DWI was firstly discovered in 1986 [8]. However, DWI was available for clinical routine in the mid-1980s [8]. In recent years, development of high-performance gradient has accelerated the use of DWI.
Materials and Methods
The study cases were selected among those who presented with a diagnosis of intracranial mass to Neurosurgery Department of Erciyes University, Medicine School. The study was approved by Local Ethics Committee of Erciyes University (Kayseri, Turkey) and exempted from informed consent. The cases underwent surgery previously or any kind of treatment was excluded. In all cases, glial tumors were confirmed by histopathological examination. Overall, 20 patients were included to the study: 7 patients with glioblastoma multiforme (GBM), 4 patients with Anaplastic Astrocytoma (AA), 4 patients with diffuse astrocytoma, 3 patients with ependymoma and 2 patients with low-grade astrocytoma.
MR imaging protocol
The imaging studies were performed by using a quadrature head coil by 1.5 Tesla MRI (Philips Intera Achieva Nova, Netherland) at Radiology Department of Erciyes University, Medicine School. In all patients, T1W-SE, T2W-TSE and DWI-EPI images were obtained according to following protocols:
T1W-SE: TR/TE, 623/15; 23 slice; slice thickness, 5 mm; FOV, 260 × 80; matrix, 256 × 256; flip angle, 70 degrees; gap, 1 mm; total time 1 min 40 s.
T2W-TSE: TR/TE, 4618/100; 23 slice; slice thickness, 5 mm; FOV, 260 × 80; matrix, 256 × 256; flip angle, 90 degrees; gap, 1 mm; total time 1 min 55 s.
The multi-slice, single-shot, axial echo planar spin echo sequences were for DWI. In this sequence, the technical parameters used were:
DWI-EPI: TR/TE, 2609/89; 18 slice; slice thickness, 2.5 mm; FOV, 230 × 230; matrix 256 × 256; gap, 1 mm; total time 2 min 4 s.
Diffusion gradients were sequentially applied throughout 3 main vectorial axes (slice selection, readout, and phase) with a b value of 1000. Then, apparent diffusion coefficient (ADC) was calculated from DWI. ADC map images were transferred to DTI studio for post-processing.
Post-processing
The mathematical ADC values are measured according to two main methods in the echo-planar DWI. The first method is Stejskal-Tanner formula while second method is direct measurement from ADC map. In both, primary ROIs (Regions of Interest) or pixel lens measurements were taken in desired region or regions. ROI can be large as well as desired and it can be applied as circular, rectangular or irregular. The pixel lens can compass 1 to16 pixels. Following measurements, ADC value are calculated from Stejskal-Tanner formula or pixel value on the ADC map.
Stejskal–Taner formula: ADC=-1/b In (S/So)
Here: So is the pixel value T (T=Trace) in b=0 or b=50. S is the pixel value in b=1000 s/mm2. In is the natural logarithm and its value is b=1000 at 1/b. This calculation can be made by using scientific calculator in any computer. The second method involving the direct calculation from pixel value on the ADC map is much easier and more reliable. The reliability is provided by automatic ADC maps.
The image post-processing was achieved by using pixel-by-pixel calculations produced on DWI and ADC maps via freeware (www.mristudio.org/DTIstudio). On DTI studio, the mean diffusion value of ADC maps were measured manually with elliptically placed wide-ROIs for tumoral areas and normal brain tissue symmetrical and/or neighbour to tumor at b=1000 value
Data analysis and statistical calculations
The mean diffusion value of ADC maps were calculated for both tumor area and normal brain tissue from DWI. The statistical analysis was performed by using SPSS 15.0 Statistical Package Program. Normal distribution was tested by using Kolmogorov Smirnov test. Independent two samples t-test was used to compare groups. A P-value <0.05 was considered statistically significant.
Results
We calculated that mean ADC value was 2.91 (± 0.04 SD) × 10-3 mm2/s for GMB, 0. 69 (± 0.03 SD) × 10-3 mm2/s for AA, 3.13 (± 0.07 SD) × 10-3 mm2/s for diffuse astrocytoma, 2.41 × 10-3 mm2/s for low-grade astrocytoma and 0.70 (± 0.03 SD) × 10-3 mm2/s for ependymoma. A P-value <0.05 was considered statistically significant (Table 1).
| DWI (mm2/ s) | ADC (mm2/ s) | NWMNT (mm2/ s) | |
|---|---|---|---|
| GBM | 0.11 × 10-3 | 2.91 × 10-3 | 1.3 × 10-3 |
| AA | 1.16 × 10-3 | 0.69 × 10-3 | 1.29 × 10-3 |
| Diffuse astrocytoma | 0.10 × 10-3 | 3.13 × 10-3 | 1.36 × 10-3 |
| Low-degree astrocytoma | 1.58 × 10-3 | 2.41 × 10-3 | 1.37 × 10-3 |
| Ependymoma | 0.90 × 10-3 | 0.70 × 10-3 | 1.10 × 10-3 |
| Normal brain | - | 0.85 × 10-3 | - |
Table 1: The mean diffusion value of glial tumors. GBM: Glia Blastome Multiforme, AA: Anaplastic Astrocytoma; DWI: Diffusion Weighted Image; ADC: Apparent Diffusion Coefficient; NWMNT: Normal White Matter Neighbour to Tumor.
We found a significant difference in mean ADC values between GBM and AA (p=0.001). Although there were marked differences among diffuse astrocytomas, low degree astrocytoma and ependymoma, the difference didn't reach statistical significance.
The diffusion values were calculated from the normal brain areas symmetrical to tumor on ADC map (Table 2).
| ADC value of normal brain to tumor symmetry (mm2/s) | |
|---|---|
| GBM | 0.87 × 10-3 |
| AA | 0.87 × 10-3 |
| Diffuse astrocytoma | 0.84 × 10-3 |
| Low-degree astrocytoma | 0.85 × 10-3 |
| Ependymoma | 0.85 × 10-3 |
Table 2: The diffusion values were calculated from the normal brain to tumor symmetry on ADC map. GBM: Glia Blastome Multiforme; AA: Anaplastic Astrocytoma.
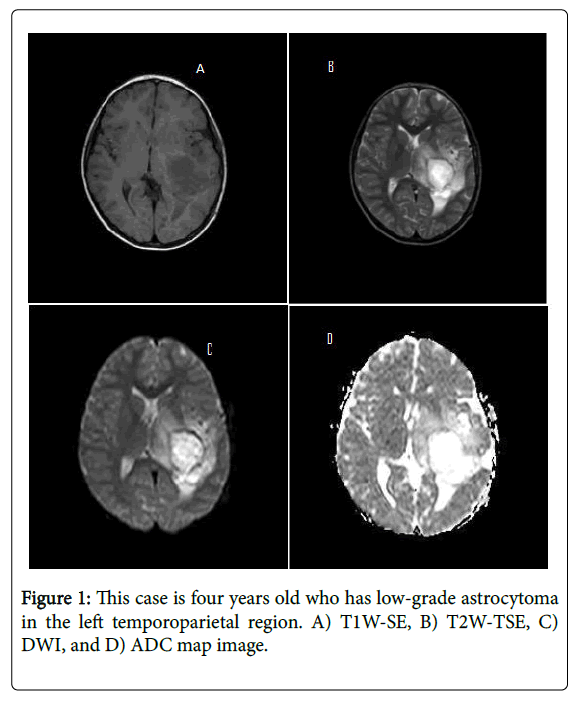
Intensity characteristics for normal brain parenchyma and tumoral regions were evaluated in each case due to TIW-SE, T2W-TSE, DWI ve ADC maps imaging’s (Table 3 and Figure 1).
| Brain Parenchyma | Tumor Area | |||||||
|---|---|---|---|---|---|---|---|---|
| T1W-SE | T2W-TSE | DWI | ADC map | T1W-SE | T2W-TSE | DWI | ADC map | |
| GBM | Hipointense | Hipointense | Hiperintense | Hipointense | Hiperintense | Hiperintense | Hipointense | Hiperintense |
| AA | Hiperintense | Hipointense | Hiperintense | Hipointense | Hipointense | Hiperintense | Hipointense | Hipointense |
| Diffuse A | Hiperintense | Hipointense | Hipointense | Hipointense | Hipointense | Hiperintense | Hiperintense | Hiperintense |
| Low-degreeA | Hipointense | Hipointense | Hipointense | Hipointense | Hiperintense | Hiperintense | Hiperintense | Hiperintense |
| Ependymoma | Hipointense | Hipointense | Hipointense | Hipointense | Hiperintense | Hiperintense | Hiperintense | Hipointense |
Table 3: Comparison of brain parenchyma and tumor area. GBM: Glia Blastome Multiforme; AA: Anaplastic Astrocytoma; Diffuse A: Diffuse Astrocytoma.
In GBM cases imaging studies revealed normal brain parenchyma T1W-SE, T2W-TSE and ADC maps hypointense and tumoral regions hyperintense, while DWI studies revealed normal brain parenchyma hyperintense and tumoral regions hypointense.
Imaging studies of AA cases revealed normal brain parenchyma T1W-SE, T2W-TSE and ADC maps hyperintense and tumoral regions were hypointense.
T2W-TSE and ADC maps imaging of normal brain parenchyma revealed hypointense while tumoral regions were T2W-TSE hyperintense and hypointense in ADC maps.
In diffuse astrocytoma cases imaging studies resulted normal brain parenchyma as T1W-SE hyperintense, tumoral regios as hypointense while in T2W-TSE, DWI ve ADC maps imaging studies revealed as normal brain parenchyma as hypointense and tumoral regions were hyperintense.
Imaging studies of ependymoma cases revealed normal brain parenchyma as hypointense in T1W-SE, T2W-TSE, DWI and ADC maps. Tumoral regions were hyperintense in T1W-SE, T2W-TSE and DWI imaging’s and hypointense in ADC maps.
In low-grade astrocytomas T1W-SE, T2W-TSE, DWI and ADC maps imaging studies revealed as hyperintense and hypointense for normal parenchyma differently.
Discussion and Conclusion
Nowadays, other MRI methods as well as conventional MRI have entered into routine use. One of these applications is Diffusion Weighted Imaging (DWI). Although a large part of using of DWI is restricted with brain, it is a powerful technique for the evaluation of different diseases. This method uses the random diffusion motion of water molecule depending on physiological and anatomical characteristics of living organisms. DWI can disclose pathology of non-attention cases in the conventional MRI.
DWI can reveal pathologies in cases where conventional MRI is failed to establish abnormality. DWI has high sensitivity in the early detection of acute cerebral ischemia, and seems promising in the evaluation of traumatic brain injury. DWI can discriminate lesions by increased and decreased diffusion. In addition, full-tensor DWI can evaluate microscopic architecture of the brain and partially white matter pathways by measuring spatial distribution and degree of anisotropic diffusion in the brain. In addition to acute ischemic stroke, it is used to discriminate arachnoid cyst of dermoid/epidermoid cysts from brain abscesses and brain tumors, to demonstrate normal brain myelination in new-borns and to identify ischemic-non ischemic central nervous system disorder. It is also used to detect and characterize multiple sclerosis-related tissue changes in the Multiple Sclerosis (MS) patients. Moreover, it was reported that it can be used for spinal cord injuries, bone marrow changes in vertebras and differential diagnosis of malignant vertebral fractures from acute benign osteoporotic vertebral compression.
The ADC map is another parameter that eliminates T2 effect in the mathematical calculation and provides important data for quantitative measurement. The small lesions could not be readily seen on the DWI because of low spatial resolution but they are easily recognized on the ADC maps since ADC map provides a marked difference between parenchyma and signal lesion [9]. The DWI together with the ADC value measurements is a functional imaging method that can provide a significant contribution to the diagnosis and differential diagnosis. Lack of need for contrast material and fast and simple imaging capturing within seconds are major advantages [9]. Illustration of water diffusion values in a tissue in a mathematical manner by measuring directly on the ADC maps is another great advantage compared to other functional imaging methods [9]. The tissue with restricted diffusion appears as low signal on ADC while tissue with fast as high signal with high ADC value [10].
The DWI is useful in the assessment traumatic brain injury, demyelinating diseases, and Creutzfeld-Jakob disease as well as differential diagnosis of necrotic tumors and abscesses [11-13].
Necrotic or cystic tumors display signal intensity similar to CSF with calculated ADC values ranging from (0.3-2.7) (1.69 ± 0.9) × 10-3 mm2/s and (1.7-3.8) (2.2 ± 0.9) × 10-3 mm2/s [14]. However, abscesses display different findings and are hyperintense with calculated ADC value of (0.21-0.34) × 10-3 mm2/s [11]. Cystic or necrotic tumors differ from acute ischemic lesions as being hyperintense on the DWI (similar to an abscess) with ADC value of (0.29-0.33) × 10-3 mm2/s.
Ischemic lesions are evaluated within 8 h after onset of lesion and ADC value is 0.48 (± 0.05) × 10-3 mm2/s. It is difficult to recognize small lesions on DWI due to low spatial resolution; however, these lesions are easily recognized on the ADC maps since ADC map provides a detectable difference between parenchyma and lesion signal. These lesions appear to be hyperintense on the DWI [15]. In the intracranial epidermoid tumors, DWI confirms total removal of the contents, but postoperative DWI always showed bright signal in patients [16].
In this study, mean ADC value was found as 2.91 (± 0.04 SD) × 10-3 mm2/s for GBM, 0. 69 (± 0.03 SD) × 10-3 mm2/s for AA, 3.13 (± 0.07 SD) × 10-3 mm2/s for diffuse astrocytoma, 2.41 × 10-3 mm2/s for lowgrade astrocytoma and 0.70 (± 0.03 SD) × 10-3 mm2/s for ependymoma. A p-value <0.05 was considered statistically significant. We found a significant difference in mean ADC values between GBM and AA (p=0.001). Although there were marked differences among diffuse astrocytoma, low degree astrocytoma and ependymoma, the difference didn't reach statistical significance.
The mean ADC value was greater in GBM (2.91 × 10-3 mm2/s) than AA (0.69 × 10-3 mm2/s). The mean ADC value was greater in diffuse astrocytoma (3.13 × 10-3 mm2/s) than GBM (2.91 × 10-3 mm2/s). The mean ADC value of low- degree astrocytoma (2.41 × 10-3 mm2/s) was greater than mean ADC value of ependymoma (0.70 × 10-3 mm2/s). The ADC value of symmetrical normal brain area was calculated 0.85 × 10-3 mm2/s. The ADC value of normal white matter was 0.84 × 10-3 mm2/s [17].
Functionally, values <0.60 × 10-3 mm2/s indicate restricted of movement of water molecule such as cytotoxic edema (acute ischemia and acute infarction).
The values >1.05 × 10-3 mm2/s means increased movement of water molecule compared to normal brain parenchyma, indicating tissue distortion or loosening such as vasogenic edema. On the other hand, water molecules freely move in the cerebrospinal fluid and ADC value varies from 2.40 to 4.40 × 10-3 mm2/s [18]. The GBM has ADC value of 2.71 (± 0.24) × 10-3 mm2/s [19]. The movement of water molecules has increased compared to normal brain parenchyma in the GBM, diffuse astrocytoma, and low-grade astrocytoma. This indicates that there is loosening or distortion in tissue integration. Restricted diffusion was seen in the ependymoma and AA. The calculated ADC value >1.05 × 10-3 mm2/s indicates restricted of movement of water molecule [4]. Although there were marked differences among diffuse astrocytomas, low degree astrocytoma and ependymoma, the difference did not reach statistical significance.
The DWI with ADC value measurements is a functional imaging modality that can provide a significant contribution to the diagnosis and differential diagnosis. It doesn't require contrast agent or ionizing radiation, and is a non-invasive technique. It can be performed quickly within seconds.
As a result, the mean ADC value, the mean diffusion value and intensity is a simple technique that could be used to determine tumor degree in the glial tumors.
References
- Jemal A, Siegal R, Ward E, Murray T, Xu J, et al. (2007) Cancer statistics 2007. CA Cancer J Clin 57: 43-66.
- Berens ME, Rutka JT, Rosenblum ML (1990) Brain tumor epidemiology, growth and invasion. Neurosurg Clin North Am 1: 1-18.
- Fritz A, Percy C, Jack A, Sobin L, Parkin M, et al. (2000) International Classification of Diseases for Oncology (ICD-O-3), Geneva, Switzerland, WHO.
- Yılmazlar S, Aksoy K (2004) Yüksek gradeli hemisferik glial tümörler. Türkiye Klinikleri J Neur 2: 24-29.
- Adilay HU (2006) Glioblastoma multiforme ve benign meningiom olgularında tümör dokusunda ölçülen çinko, demir ve kadmiyum düzeylerinin kar�?ıla�?tırılması (Uzmanlık tezi). İstanbul: Bakırköy Ruh Sa�?lı�?ı ve Sinir Hastalıkları E�?itim ve Ara�?tırma Hastanesi pp: 1-5.
- Burger PC, Vogel F, Green SB, Strike TA (1985) Glioblastoma multiforme and anaplastic astrocytoma pathologic criteria and prognostic implications. Cancer 56: 1106-1111.
- Brown R (1828) A brief account of microscopical observations made in the months of June, July and August 1827 on the particles contained in the pollen of plants and on the general existence of active molecules in organic and inorganic bodies. Philosoph Mag 4: 161-173.
- Le Bihan D, Breton E, Lallemand D, Grenier P, Cabanis E, et al. (1986) MR imaging of intravoxel incoherent motions: application to diffusion and perfusion in neurologic disorders. Radiology 161: 401-407.
- �?ener RN (2001) Diffusion MRI: apparent diffusion coefficient (ADC) values in the normal brain and a classification of brain disorders based on ADC values. Comput Med Imaging Graph 25: 299-326.
- Bergui M, Zhong J, Bradac GB, Sales S (2001) Diffusion-weighted images of intracranial cyst-like lesions. Neuroradiology 43: 824-829.
- Desprechins B, Stadnik T, Koerts G, Shabana W, Breucq C, et al. (1999) Use of diffusion-weighted MR imaging in differential diagnosis between intracerebral necrotic tumors and cerebral abscesses. Am J Neuroradiol 20: 1252-1257.
- Ebisu T, Tanaka C, Umeda M, Kitamura M, Naruse S, et al. (1996) Discrimination of brain abscess from necrotic or cystic tumors by diffusion-weighted echo planar imaging. Mag Res Imag 14: 1113-1116.
- Schaefer PW, Grant PE, Gonzalez RG (2000) Diffusion-weighted MR imaging of the brain. Radiology 217: 331-345.
- Tien RD, Felsberg GJ, Friedman H, Brown M, MacFall J (1994) MR imaging of high-grade cerebral gliomas: value of diffusion-weighted echoplanar pulse sequences. AJR 162: 671-677.
- Raffin LS, Bachesch LA, Machado LR, Nóbrega JP, Coelho C, et al. (2001) Diffusion-weighted MR imagıng of cystic lesions of neurocysticercosis: a preliminary study. Arq Neuropsiquiatr 59: 839-842.
- Aboud E, Abolfotoh M, Pravdenkova S, Gokoglu A, Gokden M, et al. (2015) Giant intracranial epidermoids: is total removal feasible? J Neurosurg 122: 743-756.
- �?ener RN (2001) The "apperent diffusion coefficient" (ADC) values �??�??on diffusion MRI. Tanısal ve Giri�?imsel Radyoloji 7: 460-463.
- �?ener RN (2002) Tuberous sclerosis: diffusion MRI findings in the brain. Eur Radiol 12: 138-143.
- Güney B, Kiti�? Ö, Çallı C, Yünten N (2003) İntrakranial kistik lezyonlarda difüzyon-a�?ırlıklı MR görüntüleme. Ege Tıp Dergisi 42: 107-111.
Relevant Topics
- Abdominal Radiology
- AI in Radiology
- Breast Imaging
- Cardiovascular Radiology
- Chest Radiology
- Clinical Radiology
- CT Imaging
- Diagnostic Radiology
- Emergency Radiology
- Fluoroscopy Radiology
- General Radiology
- Genitourinary Radiology
- Interventional Radiology Techniques
- Mammography
- Minimal Invasive surgery
- Musculoskeletal Radiology
- Neuroradiology
- Neuroradiology Advances
- Oral and Maxillofacial Radiology
- Radiography
- Radiology Imaging
- Surgical Radiology
- Tele Radiology
- Therapeutic Radiology
Recommended Journals
Article Tools
Article Usage
- Total views: 4476
- [From(publication date):
April-2017 - Aug 31, 2025] - Breakdown by view type
- HTML page views : 3488
- PDF downloads : 988