Case Report Open Access
Use of Human Embryonic Stem Cells in the Treatment of Parkinsons Disease: A Case Report
Geeta Shroff1*, Petra Hopf-Seidel2
1Director, Nutech Mediworld, New Delhi, India
2Neurologist and psychiatrist, expert for Lyme Disease, Ansbach, Germany
Visit for more related articles at International Journal of Emergency Mental Health and Human Resilience
Abstract
Parkinson’s disease (PD), a progressive disorder of the central nervous system, affects every 1 in 100 persons, aged around 70 in Europe. PD is characterized by abnormalities affecting both the motor system and the non-motor systems that include tremor, gait disorder, bradykinesia, depression, cognitive impairments, dysautonomia and sleep disturbances. Both surgical and drug treatments are available for the treatment of PD, but none has been able to halt the progression of the degenerative disease. We present the case of 65-year old male patient with PD who was treated with human embryonic stem cells (hESCs). The patient showed significant improvement in his health following the treatment; such as reduction in tremors, bradykinesia, muscle rigidity, pain and stiffness in the neck, shoulder and low back and improvement in the range of movements of the neck. The patient was able to balance himself while walking, had reduction in numbness in his left hand and both of his legs. He was able to speak louder and had improved writing skills. No adverse event or teratoma formation was observed. hESC therapy in our study showed beneficial effects in the patient with PD, but future clinical trials are needed to gather more evidence to support our findings.
Keywords
Human embryonic stem cells, neurodegeneration, stem cell therapy
Abbreviations
Parkinson’s disease (PD), Dopamenergic Neurons (DA), Substantia Nigria (SN), Deep Brain Stimulation (DBS), Quality of Life of Patients (QoL), Induced Pluripotent Stem Cells (iPSCs), Mesenchymal Stem Cells (MSCs), Human Embryonic Stem Cells (hESCs), Single-photon Emission Computed Tomography (SPECT) Scan, Magnetic Resonance Imaging (MRI)
Introduction
Parkinson’s disease (PD) is defined as a progressive abnormality of the central nervous system which affects every 1 in 100 persons of age around 70 years in Europe (de Rijk et al., 2000). PD may arise either due to several environmental factors or due to a genetic defect (Bonifati et al., 2003; Polymeropoulos et al., 1997; Priyadarshi, Khuder, Schaub, & Priyadarshi, 2001; Valente et al., 2004). Clinically, PD is characterized by abnormalities of the motor system such as tremor, bradykinesia, gait disorder and muscle rigidity mostly accompanied by the non-motor system abnormalities including depression and cognitive impairment, dysautonomia (especially of the bladder function) and sleep disturbances (Ehrt & Aarsland, 2005; Fahn, 2003; Savitt, Dawson, & Dawson, 2006). Women with PD have a longer life expectancy than men, but the reasons for this difference are unknown (Diamond et al., 1990). Two major pathological manifestations of this condition include degeneration with the loss of dopamenergic neurons (DA) in the substantia nigria (SN) and the presence of biomarkers such as α-synuclein positive Lewy bodies in several regions of the brain (Braak et al., 2003; Marsden, 1983).
Both surgical and drug treatments are available for the treatment of PD. Deep brain stimulation (DBS) has been demonstrated to improve the quality of life of patients (QoL) with PD (Tykocki et al., 2013). There are numerous anti-parkinson drugs available which help to lessen the symptoms of the disease for quite a while. But, none of the treatment has been able to permanently improve the QoL of the patients, because of the neurodegenerative and progressive nature of the disease (Savitt, Dawson, & Dawson, 2006). Currently, a new therapeutic option, cell replacement therapy for PD is aimed at replacing the degenerated DA neuron cells or providing a cell source that can differentiate into DA neurons (Wijeyekoon & Barker, 2009). Cells that have been considered for replacement therapy in PD include human neuronal tissue and stem cells including induced pluripotent stem cells (iPSCs), mesenchymal stem cells (MSCs) and human embryonic stem cells> (hESCs) (Ghosh, Zhang, & Smith, 2014).
In our previous studies, we have shown an improvement in the condition of patients suffering from spinal cord injury, cerebral palsy or/and cortical visual impairment, spinocerebellar ataxia, Friedreich ataxia after being treated with hESC therapy (Shroff, 2015a, 2015b; Shroff & Das, 2015; Shroff, Gupta, & Barthakur, 2014; Shroff & Gupta, 2015). Here, we used hESCs in a patient with PD.
Methodology
hESCs used in the treatment are cultured and maintained as per our proprietary in-house patented technology (Patent-WO 2007/141657A PCT/1B 2007 Published 13 Dec 2007) in a Good Manufacturing Practices (GMP), Good Laboratory Practices (GLP) and Good Tissue Practices (GTP) compliant laboratory. The evidence for the use of hESCs at our facility has been submitted and accepted at the House of Lords, Regenerative Medicine, Science and Technology Committee ("House of Lords SATSC. Available online: http://www. parliament.uk/documents/lords-committees/science-technology/ RegenerativeMedicine/RegenMed.pdf (Accessed 23 September 2014)."). The cell lines are free of any animal or microbial product and are chromosomally stable. The derivation and characterization of hESCs has been presented previously (Shroff, Gupta, & Barthakur, 2014). The safety of hESCs in the treatment of various terminal conditions has also been established (Shroff & Bharthakur, 2015).
The treatment plan consisted of treatment phases (T1, T2 and so on) with gap periods in between. A gap phase allows the injected cells to develop into mature cells and to regenerate the affected area. In the first phase, T1 (8-week to 12-week), 0.25 ml (<4 million cells) hESCs were administered intramuscularly, twice daily to “prime” the body, 1 ml hESCs (<16 million cells) were administered twice every 7 days intravenously to “home in” to the required area and 1 to 5 ml hESCs were administered every 7 days by any of the supplemental routes including brachial plexus block, epidural cathetar caudal, intrathecal, epidural and popliteal block and/or deep spinal muscle. Nasal sprays of hESCs (3.5 million/ml) were given twice a week. Nasal spray results in a quick absorption of hESCs to the brain via nervus olfactorius. Subsequent to a gap period of 4-8 months, the successive phases like T2 (4 to 6 weeks) and T3 (4 to 6 weeks) also incorporated the same dosage regime as T1.
Independent Ethics Committee (IEC) approved the study. The IEC of Nutech Mediworld, New Delhi, India reports clinical study to the National Apex Committee for Stem Cell Research and Therapy (NAC-SCRT). The patient provided written and videotaped informed consent prior to start of the treatment. The condition of the patient was videographed regularly during the treatment. The radiological investigations including single-photon emission computed tomography (SPECT) scan and magnetic resonance imaging (MRI) were done prior to the treatment and then at the end of each treatment session. The eventual occurrence of an antigenic or anaphylactic response in the patient was monitored by the in-house physicians and nurses during the therapy.
Case
A 65-year old male was admitted to our facility on 19 November 2012 with complaints of resting tremors, back and neck stiffness, which occurred mainly in the morning and causing restricted movements. Furthermore, he suffered from bradykinesia, muscle rigidity, unclear speech, micrographia, imbalanced walking and urinary urgency with incomplete voiding. In 2012, the patient was diagnosed with PD elsewhere and was since then on anti-parkinson drugs.
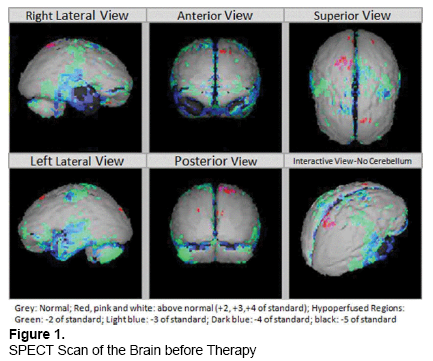
On 20 November 2012, SPECT scan of the brain revealed moderate hypoperfusion in bilateral parieto-temporal regions and moderate to severe hypoperfusion in bilateral basal ganglia and bilateral cerebellar regions (Figure 1).
MRI of the brain showed generalized mild cerebral and cerebellar atrophy with periventricular small vessel type, ischemic changes in frontal and parietal lobes bilaterally with few small focal hyperintense areas involving bilateral white matter of frontal lobe, also likely representing ischemic lesions. No intracranial hemorrhage or other acute abnormalities were seen.
At our facility, the patient primarily received four sessions of hESC therapy along with physiotherapy and occupational therapy. During the hESC therapy, the patient also continued on his anti-parkinson medications that he had been receiving previously. The medications included Rasagilin, an irreversible monoamine oxidase B inhibitor, 1 mg once a day (OD; Taj Pharmaceuticals Ltd.), Pramipexole, a dopamine agonist, 1 mg OD; (Taj Pharmaceuticals Ltd.), Meloxicam for the arthritis pain management 7.5 mg OD (Taj Pharmaceuticals Ltd.), Cymbalta, an antidepressant, 30 mg OD; (Eli Lilly and Co. Pvt. Ltd.), and Amlodipine for hypertension management, 5 mg OD; (Pfizer Inc.).
Results
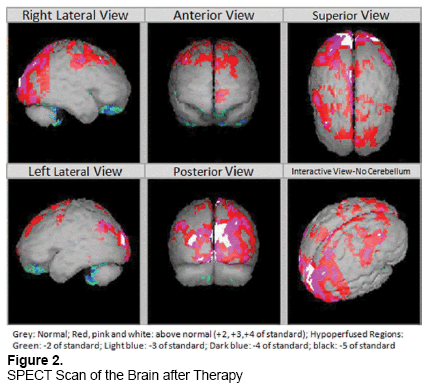
Following the hESC treatment, significant improvements were observed such as reduction in tremors, in muscle rigidity, in bradykinesia and an improvement in speaking and writing skills. The patient was also able to balance himself again while walking and a shuffling gait was no more present. He had less pain and stiffness in the neck and a better ability to turn it around. There was a reduction in pain in his shoulder pain. The patient had reduced numbness in his left (lt) hand and in both legs. The improvement in the condition of the patient following hESC therapy is stable. Following the hESC treatment, the SPECT scan of 7 July, 2014 showed normal hypoperfusion in the cerebral region and a significant improvement (>60%) occurred in the degree of perfusion in the cerebellar regions (Figure 2). He is currently off all anti-parkinson medications and has a mild resting tremor in his right hand as the only persisting symptom. No adverse events (AEs) or teratoma formation was reported in our patient.
Discussion
This study presents the clinical outcome in a patient with PD treated with hESC therapy. The patient demonstrated an improvement in his overall condition and symptoms related to PD. SPECT scan after the therapy also showed significant reduction (>60%) in hypoperfusion in cerebral and cerebellar regions of the brain.
Some of already available treatments such as levodopa, MAOB inhibitors, dopamine agonists and DBS are known only to suppress the parkinson symptoms and so can better the QoL of the patients. But sometimes these drugs are associated with complications too (Fytagoridis & Blomstedt, 2010; Pinsker et al., 2013; Savitt, Dawson, & Dawson, 2006). Use of neuroprotective agents (e.g. such as selegiline and coenzyme Q10) in PD is also very much in the focus of today`s research. Although some of the studies have shown beneficial results of these agents,the evidence is not yet enough to put them in the first line of anti-parkinson drugs (Kouti & Noroozian, 2013; Savitt, Dawson, & Dawson, 2006).
The steadily growing possibilities for cell replacement therapies have attracted researchers to explore its use in the treatment of PD. Neuronal tissue transplantation from human embryo or fetus and direct stem cells transplantation has also been studied in the patients with PD (Ghosh, Zhang, & Smith, 2014).
Roy et al., (2006) conducted a trial in which they implanted DA neurons derived from hESC into parkinsonian rats. Implanted neurons resulted in partial function recovery in the disease models (Roy et al., 2006). Kang et al., (2014) showed that primitive neural stem cells transplanted in vivo released dopamine and were responsible in reducing PD-like behavior (Kang et al., 2014). Freed and colleagues (2001) carried out a clinical trial in which human embryonic DA neurons were transplanted into 40 patients with PD. The results showed clinical benefits especially for the younger patients (Freed et al., 2001). Feldman et al conducted a phase I trial to assess the safety of using hESC in the treatment of amyotrophic lateral sclerosis, another neurodegenerative disease like PD. The study observed that hESC therapy did not show any AEs in the patients and also inferred that this therapy will show a potential therapeutic effect in the treatment of PD, multiple sclerosis and Alzheimer’s disease ("Research on treatment for ALS aided by stem cells. Available online http://www.michigandaily.com/news/new-treatments-als-reach-phase-ii-trial (accessed on 31 March 2015)."). Clinical trials involving the transplantation of DA neurons from the fetal brain have been done and showed mixed results. It was found that patients who responded well had at least 100,000 transplanted dopamine-producing neurons and three fetuses are needed to have this quantity of cells (Abbott, 2014). Use of fetal tissue may pose several ethical concerns too. hESCs or iPSCs can thus be a better option Ghosh, Zhang, & Smith, 2014). A study by Han and colleagues in the 6-hydroxydopamine (OHDA)-induced PD rats model demonstrated that neural stem cells (NSCs) derived from induced pluripotent stem cells can differentiate into dopamine neurons (Han et al., 2015). Clinical trial has been conducted using MSCs and showed them to be safe in patients with PD (Venkataramana et al., 2010). Grafted MSCs release several immunomodulatory, anti-inflammatory, neurotrophic, anti-apoptotic and angiogenic growth factors promoting recovery of the compromised DA neurons in the host (Glavaski-Joksimovic & Bohn, 2013). We used hESCs in our patient with PD and they might have followed the same mechanism of action as MSCs. In our study, successive treatment phases were followed by a gap phase in between. The purpose behind adding a gap phase was to provide hESCs enough time to grow and differentiate similar to the time required for organogenesis in human fetus (14-16 weeks) ("MedlinePlus. Available online http://www.nlm.nih.gov/medlineplus/ency/article/002398.htm (Accessed on 30 September 2014)."). We administered more than 20 million hESC cells in our patient in a single treatment phase. We used hESCs of size less than 1 μm, because these small sized cells were able to cross the blood brain barrier and thus repair and regenerate the injured cells in the basal ganglia. This is also reflected in the SPECT scan (>60% improvement) performed after hESC therapy. The safety of using hESCs in vivo is being questioned because of the AEs such as immune rejection or teratoma formation which has been reported to occur in previous studies (Wijeyekoon & Barker, 2009). But in our study, no AEs or teratomas were observed.
Though the use of hESCs poses an ethical dilemma, no one can deny the huge potential that these cells have in curing the worst afflictions. The use of these cells is highly controversial because they are derived from human preâ�?implantation embryos that has to be destroyed for deriving cells. Most of the cell lines are derived from the inner cell mass (ICM) of human embryo. However, hESCs used in our study have been derived from 2-cell staged fertilized spare ovum obtained after a regular IVF procedure. The world is still evolving in hESC therapy with UK being the first to pass a law governing the use of human embryos for stem cell research("Regulation of Stem Cell Research in the United Kingdom. Available online http://www.eurostemcell.org/regulations/regulation-stem-cell-research-united-kingdom (accessed on 1 September 2015),"). In Europe, a committee to make an inventory of the positions taken by governments of the countries within Europe regarding stem cell research has been established by the European Science Foundation (European Science Foundation, 2001) (de Wert & Mummery, 2003). Recently, FDA has also approved a clinical trial with hESCs for patients with spinal cord injury ("Treatment for Spinal Cord Injury To Start Clinical Trial Funded by California’s Stem Cell Agency. Available online: https://www.cirm.ca.gov/about-cirm/newsroom/press-releases/08262014/treatment-spinal-cord-injury-start-clinical-trial-funded (Accessed on 1 September 2015),")
This study presents the clinical outcome in a patient with PD treated with hESC therapy. The patient demonstrated an improvement in his overall condition and symptoms related to PD.
Conclusion
Results of our study on a single patient are very encouraging but not enough to prove the clinical efficacy and safety of hESCs in general. Therefore further studies are needed to prove this new treatment chance as really effective and to understand how hESC therapy in patients with PD works.
Acknowledgements
The authors acknowledge all the doctors, all staff and patients of the Nutech Mediworld. The authors also acknowledge Knowledge Isotopes Pvt. Ltd. (http://www.knowledgeisotopes.com) for the medical writing assistance
References
- Abbott, A. (2014). Fetal-cell revival for Parkinson's. (Historical Article News). Nature, 510(7504), 195-196
- Bonifati, V., Rizzu, P., van Baren, M.J., Schaap, O., Breedveld, G.J., Krieger, E., et al. (2003). Mutations in the DJ-1 gene associated with autosomal recessive early-onset parkinsonism. Science, 299(5604), 256-259
- Braak, H., Del Tredici, K., Rub, U., de Vos, R.A., Jansen Steur, E.N., & Braak, E. (2003). Staging of brain pathology related to sporadic Parkinson's disease. Neurobiology of Aging, 24(2), 197-211.
- de Rijk, M.C., Launer, L.J., Berger, K., Breteler, M.M., Dartigues, J.F., Baldereschi, M., et al. (2000). Prevalence of Parkinson's disease in Europe: A collaborative study of population-based cohorts. Neurologic Diseases in the Elderly Research Group. Neurology, 54(11), S21-23.
- de Wert, G., & Mummery, C. (2003). Human embryonic stem cells: research, ethics and policy. Human Reproduction, 18(4), 672-682.
- Diamond, S.G., Markham, C.H., Hoehn, M.M., McDowell, F.H., & Muenter, M.D. (1990). An examination of male-female differences in progression and mortality of Parkinson's disease. Neurology, 40(5), 763-766.
- Ehrt, U., & Aarsland, D. (2005). Psychiatric aspects of Parkinson's disease. Current opinion in psychiatry, 18(3), 335-341.
- Fahn, S. (2003). Description of Parkinson's disease as a clinical syndrome. Annals of the New York Academy of Sciences, 991, 1-14.
- Freed, C.R., Greene, P.E., Breeze, R.E., Tsai, W.Y., DuMouchel, W., Kao, R., et al. (2001). Transplantation of embryonic dopamine neurons for severe Parkinson's disease. New England Journal of Medicine, 344(10), 710-719.
- Fytagoridis, A., & Blomstedt, P. (2010). Complications and side effects of deep brain stimulation in the posterior subthalamic area. Stereotactic and Functional Neurosurgery, 88(2), 88-93
- Ghosh, B., Zhang, C., & Smith, G.M. (2014). Bridging between transplantation therapy and neurotrophic factors in Parkinson's disease. Frontiers in Bioscience, 6, 225-235.
- Glavaski-Joksimovic, A., & Bohn, M.C. (2013). Mesenchymal stem cells and neuroregeneration in Parkinson's disease. Experimental Neurology, 247, 25-38
- Han, F., Wang, W., Chen, B., Chen, C., Li, S., Lu, X., et al. (2015). Human induced pluripotent stem cell-derived neurons improve motor asymmetry in a 6-hydroxydopamine-induced rat model of Parkinson's disease. Cytotherapy. 17(5), 665-679
- House of Lords SATSC. Available online: http://www.parliament.uk/documents/lords-committees/science-technology/RegenerativeMedicine/RegenMed.pdf (accessed 23 September 2014).
- Kang, X., Xu, H., Teng, S., Zhang, X., Deng, Z., Zhou, L., et al. (2014). Dopamine release from transplanted neural stem cells in Parkinsonian rat striatum in vivo. Proceedings of the National Academy of Sciences, USA. 111(44), 15804-15809
- L Kouti, K.E., Noroozian, M. (2013). Neuroprotective Agents for Management of Parkinson’s disease. Journal of Pharmaceutical Care, 1(1), 33-37.
- Marsden, C.D. (1983). Neuromelanin and Parkinson's disease. Journal of Neural Transmission, 19, 121-141.
- MedlinePlus. Available online http://www.nlm.nih.gov/medlineplus/ency/article/002398.htm (Accessed on 30 September 2014).
- Pinsker, M., Amtage, F., Berger, M., Nikkhah, G., & van Elst, L.T. (2013). Psychiatric side-effects of bilateral deep brain stimulation for movement disorders. Acta Neurochirurgica, 117, 47-51
- Polymeropoulos, M.H., Lavedan, C., Leroy, E., Ide, S.E., Dehejia, A., Dutra, A., et al. (1997). Mutation in the alpha-synuclein gene identified in families with Parkinson's disease. Science, 276(5321), 2045-2047.
- Priyadarshi, A., Khuder, S.A., Schaub, E.A., & Priyadarshi, S.S. (2001). Environmental risk factors and Parkinson's disease: a metaanalysis. Environmental Research, 86(2), 122-127
- Regulation of Stem Cell Research in the United Kingdom. Available online http://www.eurostemcell.org/regulations/regulation-stem-cell-research-united-kingdom (Accessed on 1 September 2015).
- Research on treatment for ALS aided by stem cells. Available online http://www.michigandaily.com/news/new-treatments-als-reach-phase-ii-trial (Accessed on 31 March 2015).
- Roy, N.S., Cleren, C., Singh, S.K., Yang, L., Beal, M.F., & Goldman, S.A. (2006). Functional engraftment of human ES cell-derived dopaminergic neurons enriched by coculture with telomerase-immortalized midbrain astrocytes. Nature Medicine, 12(11), 1259-1268.
- Savitt, J. M., Dawson, V.L., & Dawson, T.M. (2006). Diagnosis and treatment of Parkinson disease: molecules to medicine. Journal of Clinical Investigation, 116(7), 1744-1754.
- Shroff, G. (2015a). Human Embryonic Stem Cells in the Treatment of Spinocerebellar Ataxia: A Case Series. Clinical Case Reports, 5, 1.
- Shroff, G. (2015b). A novel approach of human embryonic stem cells therapy in treatment of Friedrich’s Ataxia. International Journal of Case Reports and Images.
- Shroff, G., & Bharthakur, J.K. (2015). Safety of Human Embryonic Stem Cells in Patients with Terminal Conditions. In Press. Annals of Neurosciences.
- Shroff, G., & Das, L. (2015). Human Embryonic Stem Cell Therapy in Cerebral Palsy Children with Cortical Visual Impairment: A Case Series of 40 Patients (In Press). Journal of Cell Science and Therapy, 5, 6.
- Shroff, G., & Gupta, R. (2015). Human Embryonic Stem Cells in the Treatment of Patients with Spinal Cord Injury. In Press. Annals of Neurosciences.
- Shroff, G., Gupta, A., & Barthakur, J.K. (2014). Therapeutic potential of human embryonic stem cell transplantation in patients with cerebral palsy. Journal of Translational Medicine, 12(1), 318.
- Treatment for Spinal Cord Injury To Start Clinical Trial Funded by California’s Stem Cell Agency. Available online: https://www.cirm.ca.gov/about-cirm/newsroom/press-releases/08262014/treatment-spinal-cord-injury-start-clinical-trial-funded (Accessed on 1 September 2015).
- Tykocki, T., Szalecki, K., Koziara, H., Nauman, P., & Mandat, T. (2013). Quality of life and depressive symptoms in Parkinson's disease after subthalamic deep brain stimulation: a 2-year follow-up study. Turkish Neurosurgery, 23(3), 379-384
- Valente, E.M., Abou-Sleiman, P.M., Caputo, V., Muqit, M.M., Harvey, K., Gispert, S., et al. (2004). Hereditary early-onset Parkinson's disease caused by mutations in PINK1. Science, 304(5674), 1158-1160.
- Venkataramana, N.K., Kumar, S.K., Balaraju, S., Radhakrishnan, R.C., Bansal, A., Dixit, A., et al. (2010). Open-labeled study of unilateral autologous bone-marrow-derived mesenchymal stem cell transplantation in Parkinson's disease. Translational Research, 155(2), 62-70
- Wijeyekoon, R., & Barker, R.A. (2009). Cell replacement therapy for Parkinson's disease. Biochimica et Biophysica Acta, 7(1792), 688-702.
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 18481
- [From(publication date):
September-2015 - Aug 30, 2025] - Breakdown by view type
- HTML page views : 13546
- PDF downloads : 4935