Research Article Open Access
Web-based CBT for the Treatment of Selective Mutism: Results from a Pilot Randomized Controlled Trial in Singapore
Yoon Phaik Ooi1,2, Sharon C Sung2,3, Malini Raja2, Clare Kwan2, Jessie BK Koh4 and Daniel SS Fung*2,51Clinical Psychology and Psychotherapy, Department of Psychology, University of Basel, Missionsstrasse Basel, Switzerland
2Child Guidance Clinic, 3 Second Hospital Avenue, Health Promotion Board Building #03-01, Singapore
3Clinical Sciences, DUKE-NUS Graduate Medical School Singapore, 20 College Road, Level, Singapore
4Department of Psychology, University of Alberta, Canada
5Lee Kong Chian School of Medicine, Nanyang Technological University, Singapore
- *Corresponding Author:
- Daniel Fung
Child Guidance Clinic
3 Second Hospital Avenue
Health Promotion Board Building #03-01
Singapore
Email: daniel_fung@imh.com.sg
Received date: December 12, 2015; Accepted date: May 25, 2016; Published date: May 30, 2016
Citation: Ooi YP, Sung SC, Raja M, Kwan C, Koh JBK, et al. (2016) Web-based CBT for the Treatment of Selective Mutism: Results from a Pilot Randomized Controlled Trial in Singapore. J Speech Pathol Ther 1: 112. doi: 10.4172/2472-5005.1000112
Copyright: © 2016 Ooi YP, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Speech Pathology & Therapy
Abstract
Introduction: Cognitive behavioral therapy (CBT) approaches have shown promise for some children with Selective Mutism (SM), but to date there are limited published randomized controlled trials (RCT) of CBT interventions for SM. We present findings from a pilot RCT of the Meeky Mouse program, a 14-week web-based CBT program for children with SM.
Method: A total of 21 children (6-12 years old) with a primary diagnosis of SM from a child psychiatric outpatient clinic were included in this study. They were randomly assigned to either 14 weeks of the Meeky Mouse program (n=10) or to a control condition in which they interacted with the therapist while playing computer games (n=11).
Results: No significant group differences were found on anxiety symptoms. The control group showed significant improvements from pre- to post-treatment on total frequency of speech. Children in the Meeky Mouse group showed greater improvements in clinician-rated severity of mental illness and had higher clinician-rated improvement scores relative to those in the control group.
Conclusions: Findings from our pilot RCT study suggest that the web-based CBT using anxiety management strategies may be no different from an intervention that involves regular monitoring of child’s engagement in socializing activities for improving SM symptoms. Further study is needed to determine optimal strategies for treating children with SM with the use of web-based applications.
Keywords
Anxiety; Cognitive behavioral therapy; Computerassisted therapy; Exposure therapy; Selective mutism
Abbreviations
CTONI NIQ: Comprehensive Test of Nonverbal Intelligence; CTONIPNIQ: Comprehensive Test of Pictorial Nonverbal Intelligence; BPVS: British Picture Vocabulary Scale, SMQ: Selective Mutism Questionnaire; ACAS: Asian Children’s Anxiety Scale-Caretaker Version, ACAS-C: Asian Children’s Anxiety Scale-Child; CGI: Clinical Global Improvement
Introduction
Selective mutism (SM) is a persistent childhood disorder defined as limited or a lack of speech, language, and communication in selected social settings by the Diagnostic and Statistical Manual of Mental Illness – Fifth Edition (DSM-5) [1]. Initially thought to be uncommon, with rates as low as 0.2%, more recent studies have revealed higher prevalence rates of up to 2% [2-5]. Most children often show symptoms of SM before entering school and these symptoms become more prominent upon the child's entrance into school, when there is an increased pressure to speak [6]. Children with SM tend to remain silent and are overlooked in traditional classroom situations where being meek and quiet is viewed as non-disruptive. As more schools embark on an interactive educational system, many of these children face increasing classroom demands such as speaking up in class and making group presentations.
SM has been regarded by some researchers and clinicians as a variant of Social Anxiety Disorder, and the etiology and symptomatology of both SM and Social Anxiety Disorder has been postulated to overlap [7-9]. The conceptualization of SM as an anxiety disorder is helpful in treatment of afflicted children. Hence, although there is no ‘gold standard’ of treatment for SM, treating the underlying anxiety in order to improve speech appears to be an etiologically sound option and is now supported by the reclassification of SM as an anxiety disorder in the DSM-5 [1,10,11]. Cognitive behavioral therapy (CBT) and exposure-based treatments have the strongest evidence for treating anxiety in children, with favorable long-term outcomes [12-15]. The use of cognitive strategies is usually possible in school age children with SM as they are often have an intellectual functioning that is on par with most peers of their same age group. A review of 23 studies, consisting of mainly retrospective record reviews, uncontrolled case studies, and a small number of single-participant experiments, on the treatment of SM including psychodynamic, behavioral, and cognitive behavioral approaches suggested that behavioral and cognitive behavioral techniques appeared most effective for treating children with SM [6]. Several case studies provide additional support for using CBT to treat children with SM [16-18]. For example, Fung et al. [16] presented a case study on a 7-year old Caucasian Canadian boy using the original 14-week web-based CBT program (entitled Meeky Mouse) and found improvements on anxiety symptoms and severity of SM at post-treatment. In another case study, Reuther et al. [17] found that a 8-year-old Caucasian boy with SM improved in frequency of speech and displayed lessening of anxiety symptoms and reduction in the severity of SM following 21 sessions, which included psychoeducation, exposure, cognitive restructuring, social skills, and maintenance and relapse prevention. In a more recent case series study of five children with SM, Ooi et al. [18] found that four out of five children with SM demonstrated improvements in the frequency of speech during therapy sessions at home, in school, and other social situations following 14 weeks of a modified version of the Meeky Mouse program, which was adapted for Singaporean children, in conjunction with pharmacological treatment.
In recent years, RCTs have been conducted in children with SM, providing further data to support the effectiveness of CBT treatments. Bergman et al. [19] conducted a RCT involving 21 children (4–8 years old) with SM randomized to either 24 weeks of Integrated Behavior Therapy (IBT) or 12 weeks in a waitlist control group and found that those in the IBT group showed increased speaking behavior across all raters, whereas no significant improvement was found in the waitlist group. In addition, at post-treatment, 67% of children who received IBT no longer met the criteria for SM, with improvements maintained at 3 months follow-up whereas all participants in the control group still maintained their SM diagnosis. In another study, Oerbeck et al. [20] conducted a RCT involving 24 children (3-9 years old) with SM randomized to either 12 weeks of psychosocial treatment or waitlist group and found that those receiving the psychosocial treatment significantly increased speech compared with the control group at post-treatment. Furthermore, in a follow-up study one year after the end of treatment, speech improvement was maintained and 50% of children no longer fulfilled the SM diagnosis [21]. In both studies, greater improvements were found in the younger children, indicating the importance of an early intervention [20,21]. In another more recent retrospective naturalistic study, Lang et al. [22] examined the long-term outcome of 36 children (5 to 15 years old) with SM who were treated with specifically designed modular cognitive behavioral therapy (MCBT) and found significant improvement in SM symptoms at one year follow-up; 84 % no longer met DSM-IV criteria for SM. Collectively, findings from these studies show the potential of CBT as a treatment for SM.
Despite these promising findings, there is still a lack of empirical studies substantiating the effectiveness of treatment for SM. There is a need to test the applicability of CBT for SM in diverse populations to allow examination of its generalized usage. To address this limitation, the aim of the present pilot RCT was to examine the effectiveness of the Meeky Mouse program (details available in the Method section), a 14-week web-based CBT program in children with SM using a RCT design. The computer-assisted CBT intervention may be particularly useful in treating children with SM as the use of the electronic interface could serve as a way to distract children from their anxiety to interact verbally with the therapist. In addition, the therapist directed use of games and online activities could increase their interest in learning coping strategies. Based on findings of the current literature, we hypothesized that the Meeky Mouse program would be associated with higher frequency of speaking behaviors, lower levels of anxiety, greater improvements in clinician-rated severity of mental illness, and higher clinician-rated improvement scores at post-treatment.
Method
Participants
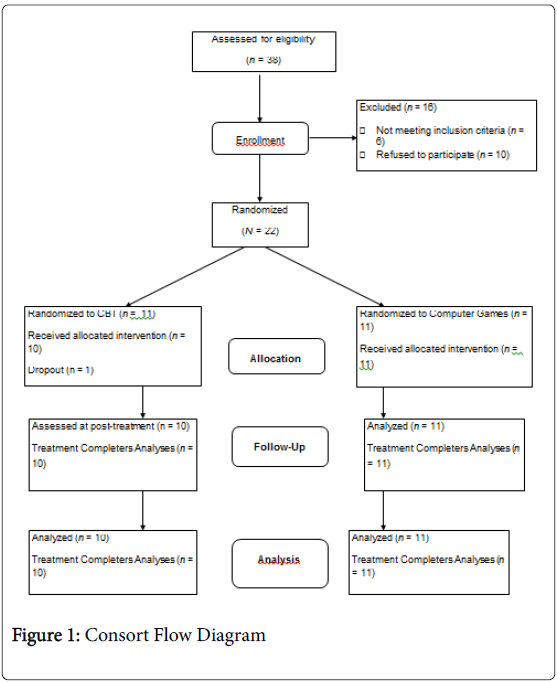
A total of 21 children referred to an outpatient child psychiatric clinic were recruited for this study. Inclusion criteria were that the child was 6-12 years old, had a primary clinical diagnosis of SM that was also confirmed by structured clinical parent interview, and was fluent in English. Exclusion criteria were below average intellectual functioning and the presence of autism and schizophrenia. Upon written parental consent and child assent, participants were randomly assigned to one of the treatment conditions: a) 14 weeks of web-based CBT (n=10) or b) 14 weeks of interaction with the therapist while playing computer games (n=11). Demographics of the participants are summarized in Table 1. Figure 1 illustrates the CONSORT flow diagram.
| Meeky Mouse Program (n=10) | Control (n=11) | Total Sample (N=21) | |
|---|---|---|---|
| Age, mean (SD) | 8.70 (1.77) | 8.55 (2.25) | 8.62 (1.99) |
| Gender, n (%) | |||
| Male | 4 | 4 | 13 (61.9) |
| Female | 6 | 7 | 8 (38.1) |
| Ethnicity, n (%) | |||
| Chinese | 9 | 9 | 18 (85.71) |
| Malay | 1 | 2 | 3 (14.29) |
| Comorbid Diagnosis, n (%) | |||
| Separation Anxiety Disorder | 1 | 1 | 2(9.5) |
| Social Anxiety Disorder | 1 | 1 | 2(9.5) |
| Specific Phobia | 4 | 1 | 5(23.81) |
| CTONI NIQ, mean (SD) | 112.20 (17.93) | 112.00 (11.73) | 112.09 (14.61) |
| CTONI PNIQ, mean (SD) | 106.10 (14.13) | 105.46 (10.29) | 105.76 (11.96) |
| CTONI GNIQ, mean (SD) | 119.20 (21.19) | 116.00 (16.21) | 117.52 (18.34) |
| BPVS, mean (SD) | 99.20 (13.44) | 96.73 (12.17) | 97.90 (12.53) |
| Note. CTONI NIQ = Comprehensive Test of Nonverbal Intelligence; CTONIPNIQ = Comprehensive Test of Pictorial Nonverbal Intelligence; CTONIGNIQ = Comprehensive Test of Geometric Nonverbal Intelligence; BPVS = British Picture Vocabulary Scale. | |||
Table 1: Demographics of Participants.
Measures
Frequency of speech: The Selective Mutism Questionnaire (SMQ) is a 17-item parentrated questionnaire that assesses a child's speaking behavior in the situations commonly associated with the failure to speak such as the home, school, and other social situations [23]. Parents rate how frequently the child speaks in each situation on a Likert scale ranging from 0 (“Never”) to 3 (“Always”). The SMQ yields a total score and three subscale scores corresponding to frequency of speech in the home, school, and public settings. Lower scores indicate lower frequency of speech (or greater SM symptom severity). Cronbach’s alpha values for the pre-treatment SMQ scores were .78 (School), .75 (Home), .25 (Other Social Situations), and .75 (Total Score).
Anxiety Symptoms: The Asian Children’s Anxiety Scale-Caretaker Version (ACAS) is a 20-item parent-rated questionnaire that evaluates the severity of child’s anxiety symptoms [24]. The ACAS-Child (ACAS-C) is a 20-item childrated questionnaire that evaluates the severity of child’s anxiety symptoms [24]. For the respective scale, parents and children rated each item on a Likert Scale ranging from 1 (Not at all like me) to 5 (Most like me). Higher scores indicate greater anxiety. Cronbach alpha values for the pre-treatment ACAS total score were .91 (ACAS) and .89 (ACAS-C).
Clinical Global Impression: The treating therapists completed the Clinical Global Impression ratings (CGI-Severity and CGI-Improvement) [25]. The CGI-Severity scores range from 1 (“Normal, not ill at all”) to 7 (“Among the most extremely ill patients). The CGI-Improvement scores range from 1 (“Very much improved”) to 7 (“Very much worse”).
Meeky Mouse Program: We modified the original web-based CBT program developed at the Hospital for Sick Children, Canada to make the situations culturally and socially appropriate for children in Singapore. For example, language, analogies, scenarios, and examples that are relevant to Singaporean children’s cultural and social context are used. In addition, the main character “Meeky”, was written based on the cultural and social experiences of a typical Singaporean child (e.g., born and/or lives in Singapore). The 14-week Meeky Mouse program consists of eight training sessions (psychoeducation and anxiety management) followed by six practice sessions (exposure using social skills training). Examples of topics within the program included recognizing feelings and bodily reactions, examining one’s thoughts, building confidence, and using the CHAT plan (Check your body’s feelings, Having bad thoughts, Attitudes and Actions that can help, Time for a reward) to improve social communication. The six practice sessions include applying the CHAT plan in increasingly difficult and anxious situations through the use of an activity (exposure) ladder. Each of the six sessions also introduces social skills that are helpful for selectively mute children such as understanding people, making and keeping friends (e.g., saying hello, introducing yourself, doing things together), dealing with unfamiliar situations.
In session 8, a “Meeky Soundpad” is introduced. This is a Microsoft Powerpoint presentation with a simple grid of six squares (per page) which is uploaded to the local personal computer (PC) from the web and used to help the child record simple messages which can be replayed during the session. If the child is mute, the therapist offers the parents two alternatives: to record the messages at home on the home computer and bring it on a memory device, or to record the messages on an audiotape, which can be transferred to the computer in the clinic. This is a treatment procedure adapted from a report using an augmentative communication device [26]. The idea is to allow the child to hear his or her own voice and thus be desensitized to speaking in various situations. Homework tasks for each session are submitted by the children weekly via the internet. Parents are encouraged to work with their children on their homework.
Exposure tasks are graduated based on the child’s comfort levels, beginning with non-verbal tasks (i.e., waving at friends, smiling nodding and shaking head) and subsequently progressing to verbal tasks, first to the “Meeky Soundpad” (i.e., saying “hello”, asking for help, buying food), and finally generalizing to real-life settings. Parents will have to accompany their children to various places (e.g., relatives’ homes, playground, shops and fast-food restaurants) to practice these exposure tasks. For more information about the Meeky Mouse program, please contact the authors.
Computer Games: Children in the control condition interacted with the therapist while playing computer games of their choice for 14 weeks. They did not receive any specific instruction on emotional regulation and problem solving techniques (i.e., CHAT plan), repeated home practice, or exposure tasks. The therapist used a warm and friendly demeanor and engaged in informal conversations with the child during the computer game sessions.
Treatment Integrity: The integrity of the Meeky Mouse program was assessed in the following ways. First, the Meeky Mouse program was delivered by Bachelor's degree psychologists with at least two years of postqualification supervised clinical experience with children. They were trained and supervised by the last author prior to conducting the Meeky Mouse program. Second, the Meeky Mouse program was manualized to increase adherence to intervention procedures. Third, specific themes to be covered for each session are documented in the web-based program. Finally, clinical supervision and feedback were provided throughout the study through direct observation, review and discussion sessions with the last author to ensure the adherence with intervention procedures and treatment fidelity.
Procedures
The present pilot RCT was approved by the Institute of Mental Health’s Clinical Research Committee (Ref: CRC 133/2005) and the National Healthcare Group’s Domain Specific Review Board (Ref: DSRB A/05/171) in Singapore. In addition, it is registered at Clinicaltrials.gov database (ID: NCT02009839). Participants were recruited over a 24-month period. The final participant completed post-treatment assessments by month 26.
Participants were referred to the present pilot RCT by their attending psychiatrist. Participation in the study was voluntary. Written informed consent to participate was obtained from the participants and their parents. Subsequently, all participants who met inclusion criteria were randomly assigned to either the treatment or control conditions. This was performed using a simple block randomization with the following treatment assignment: AB (A = Meeky Mouse and B = Computer Games). Prior to treatment, all participants were assessed using the Comprehensive Test of Nonverbal Intelligence (CTONI) [27] and the British Picture Vocabulary Scale (BPVS) [28] to establish intelligence and language abilities. In addition, each participant’s parent completed the Columbia-Diagnostic Interview Schedule for Children - Parent version (C-DISC) [29] to confirm a diagnosis of SM. All participants were required to undergo the assigned condition for 14 weeks without the prescription of medication. Each weekly session lasted for an hour. The participants, parents, and therapists completed the outcome measures at pre- and post-treatment.
Sample Size and data Analysis: To date, RCT studies involving CBT treatment in SM included sample size ranging from 21 to 25 [19,20]. Based on the number of referrals of patients with SM to our clinic, we found it likely that a minimum of 20 children would be referred within the planned recruitment time frame. Based on the projected sample size and an assumed between treatment-versus-control group standard deviation of 0.5, the current pilot RCT had 80% power to detect an effect size of . 60 at p<.05. At the end of our planned recruitment time frame, we managed to recruit 21 participants. Descriptive statistics were used to analyze the demographic characteristics of participants such as age, gender, ethnicity, diagnosis, comorbidity, and cognitive functioning. Pre-treatment group differences were assessed with chi-square tests and t-tests. Treatment effects were analyzed using 2 (Treatment: Meeky Mouse vs. Control) × 2 (Time: Pre-treatment vs. Post-treatment) repeated measures ANOVA. For significant effects, post-hoc analyses were performed in order to compare scores from pre-treatment to post-treatment. Bonferroni correction was used in these multiple comparisons. The significance level was set at p<.01 for comparisons using the SMQ and p< .025 for comparisons using the ACAS and CGI.
Results
There were no significant demographic differences between those assigned to the Meeky Mouse program and the control group. Table 2 presents the means and standard deviations of the various outcome measures used in the current study. Using the SMQ, results from 2 × 2 repeated measures ANOVA revealed no significant main effects of Treatment and Time, as well as the interaction of these two factors on all the subscales except for significant effect of Time on the following subscales of the SMQ: Other Social Situations, F (1,19)=4.63, p=.04, ηp2=.20; and Total SMQ, F(1,19)=6.95, p=.02, ηp2=.27. Follow-up analyses revealed that the control group showed significant improvements from pre- to post-treatment on total frequency of speech, t(10)=-4.25, p=.002, d=-1.38. Using the ACAS-C and ACAS-P, similar analyses also did not reveal significant main effects of Treatment and Time, as well as the interaction of these two factors. However, inspecting the means on Table 2, the Meeky Mouse group showed a non-significant increase in parent-rated anxiety symptoms at post-treatment. Using the CGI-Severity scale, similar analyses revealed significant Treatment x Time effect, F(1,19)=22.13, p=.0001, ηp2 =.54. Follow-up analyses revealed that the Meeky Mouse group improved significantly on clinician-rated severity of mental illness from pre- to post-treatment, t(9)=5.25, p=.001, d=1.66, but the control group did not, t(10)=-1.15, p=.28, d=-.36. On the CGI-Improvement scale, the Meeky Mouse group had higher clinician-rated improvement scores compared to the control group, t(19)=-5.74, p=.0001, d=-2.51.
| Meeky Mouse Program (n=10) | Control (n=11) | |||
|---|---|---|---|---|
| T1 | T2 | T1 | T2 | |
| SMQ Total | 14.10 (5.04) | 16.40 (7.81) | 13.09 (6.49) | 16.73*a (5.48) |
| School | 3.20 (2.86) | 3.40 (3.13) | 3.09 (3.14) | 4.36 (3.23) |
| Home | 9.50 (3.69) | 10.90 (5.1) | 9.45 (4.01) | 10.73 (2.41) |
| Other Situations | 1.40 (1.35) | 2.1 (2.08) | .55 (.69) | 1.64 (1.69) |
| ACAS | 40.70 (16.27) | 46.40 (14.83) | 36.45 (8.88) | 37.91 (12.79) |
| ACAS-C | 35.00 (13.01) | 33.00 (12.00) | 29.81 (9.40) | 30.55 (9.70) |
| CGI-Severity | 3.90 (.74) | 2.50*a (.71) | 4.18 (1.40) | 4.45 (1.21) |
| CGI-Improvement | - | 2.10**b (.88) | - | 4.00 (.63) |
| Note. SMQ = Selective Mutism Questionnaire; ACAS =Asian Children’s Anxiety Scale-Caretaker Version, ACAS-C = Asian Children’s Anxiety Scale-Child; CGI = Clinical Global Improvement; T1 = Pre-treatment, T2 = Post-treatment. aWithin-group comparisons, bBetween-group comparisons. *p < .01, **p < .001. |
||||
Table 2: Means and Standard Deviations of SMQ, ACAS, and CGI Scores.
Discussion
Our current study represents one of the few RCTs on the effects of a web-based CBT program for children with SM. Results from our pilot RCT indicated that the Meeky Mouse program was not superior to the control condition for improving frequency of speech. In addition, the Meeky Mouse program was not found to be more superior to the control condition for improving parent- or child-rated anxiety symptoms. These findings contradict existing literature that showed support for the use of some other CBT-based intervention programs in improving frequency of speech and reducing anxiety in children with SM [15-17,19-22].
Our Meeky Mouse program includes a combination of CBT-based components (i.e., psychoeducation, cognitive restructuring, social skills, and exposure training), which have been found to be effective in addressing anxiety in children with SM [19-22]. The combination of these components is able to address maladaptive beliefs and to increase anxiety management skills, which may enhance traditional behavioral approaches to treating SM. However, findings of our pilot RCT did not provide support for the use of these CBT-based components in treating SM for treating children with SM even though previous RCTs incorporated similar approaches such as building rapport between the therapist and child, the use of behavioral rewards, and gradual increase exposure to situations requiring verbal communication based on a predetermined hierarchy (e.g., initially speaking to the therapist with parent present, and then to the therapist alone) [19,20].
On the other hand, although parent and teacher workbooks were available on our Meeky Mouse online program, we did not include these components in our pilot RCT. Hence, our Meeky Mouse program differs from CBT-based programs from previous RCTs because we did not include parent psychoeducation and involvement of parents and teachers throughout the treatment, which may have a strong influence on child improvements [19,20]. Furthermore, although parents in the Meeky Mouse group were encouraged to help their child with homework, they were not given specific coaching how to approach the tasks. Incorporating a parent training component could have empowered the parents and allowed reinforcement of skills taught to the children in a more naturalistic home setting, thereby giving rise to generalization of skills [30].
Furthermore, it is important to note that none of the children were participating in concurrent or pharmacological treatment. Several studies and reviews have indicated that the use of fluoxetine, an antidepressant, was successful in reducing anxiety levels in children with persistent SM and comorbid anxiety disorders [18,31-33]. Hence, including pharmacological interventions in multimodal treatment packages for children with SM may provide a more holistic intervention. It is also possible that CBT is no different from an intervention that involves regular monitoring with socializing activities for improving SM symptoms. In addition, our current pilot RCT did not take into consideration parent psychopathology such as anxiety and depression. These symptoms have been found to interfere with treatment, particularly among younger children [34]. Also, it is possible that the length and duration of the exposure tasks within the Meeky Mouse program were not sufficient, and regular practice of the exposure tasks is required to increase frequency of speech and reduce anxiety symptoms in children with SM.
Another possible explanation is that although child participants and parents were not actively informed of group assignment until the end of the study, it was not possible to totally blind them on group assignment. It is possible that parents of children in the Meeky Mouse group had higher expectations of treatment effects and rated their children worse when their expected improvements were not met. This explanation may account for the non-significant increase in anxiety symptoms at post-treatment for child in the Meeky Mouse group. It may be important to include interventions that instill greater awareness and understanding of the SM condition among parents. Improvements following the Meeky Mouse program were found in clinician-rated measures, suggesting that there were improvements in the overall functioning of children in this group. However, this finding has to be interpreted with caution as the therapists were not blinded to the treatment conditions. It is possible that the therapist rated children in the Meeky Mouse group more positively than the control group due their beliefs about the effectiveness of the Meeky Mouse program.
Interestingly, the control group showed significant improvement on total frequency of speech from pre- to post-treatment. While the control condition did not incorporate CBT-based strategies, it contained several common elements to the Meeky Mouse group such as regular sessions in a structured setting, consistent therapists, and opportunities for informal social interactions. It is possible that incorporating these elements in sessions served as an effective framework for the treatment of children with SM.
Limitations of the Study
First, the study design is an important limitation. Although the current study is based on a RCT design, it may lack sufficient power to detect a statistical significance between the Meeky Mouse and control groups due to the small sample size (N=21). In addition, child participants, parents, and therapists were not blinded to the group assignment, and this may have resulted in biased behavioral ratings. Secondly, we did not incorporate a parent training component in our pilot RCT even though parents were encouraged to help children with their homework. Thirdly, when designing the study we may have overestimated the superiority of the Meeky Mouse program and underestimated the effects of the control condition, which led to an underpowered study. Findings from Bergman et al. [19] and Oerbeck et al. [20] studies revealed significant improvements in SM and anxiety symptoms following an active treatment, but these included only a wait-list control as a comparison. It is unclear if their results would have held if they used a more interactive control condition like ours. Future studies that include socializing activities such as playing games should be conducted to determine the relative impact of CBT interventions above and beyond regular informal social interaction with a therapist. Fourthly, it is possible that improvements in frequency of speech were not captured by the SMQ. Future studies may consider including a more nuanced assessment of both verbal and nonverbal communication. Finally, there was no follow-up to examine potential treatment effects. It may be possible that treatment effects for the web-based CBT intervention only emerge at follow-up.
Conclusions
In general, findings from our pilot RCT did not provide support for the use of the web-based CBT protocol for treating children with SM, suggesting that 14 weeks of CBT alone may not be sufficient. A multimodal treatment package for children with SM may need to include parent/teacher involvement and pharmacological treatment to improve SM and anxiety symptoms. Despite these limitations, the present pilot study represents one of the few RCTs that evaluated the effectiveness of a web-based CBT program in children with SM. Further RCTs to validate web-based CBT programs for treating children with SM would be needed to better understand the extent and nature of its benefits.
Acknowledgement
This research study is supported by the National Healthcare Group Cluster Research Fund Small Innovative Grant (NHG - SIG/05016). The authors declare that they have no conflict of interest. We would like to thank all those involved in the research study.
References
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders (2013). 5th edn. Washington, DC: American Psychiatric Association.
- Bergman RL, Piacentini J, McCracken JT (2002) Prevalence and description of selective mutism in a school-based sample. J Am Acad Child Adolesc Psychiatry41:938-946.
- Kumpulainen K, Rasanen E, Raaska H, Somppi V (1998) Selective mutism among second-graders in elementary school. Eur Child Adolesc Psychiatry 7: 24-29.
- Manassis K (2002) Silent suffering: understanding and treating children with selective mutism. Expert Rev Neurother 9: 235-243.
- Sharp WG, Sherman C, Gross AM (2007) Selective mutism and anxiety: a review of the current conceptualization of the disorder. J Anxiety Disord. 21:568-79.
- Cohan SL, Chavira DA, Stein MB (2006) Practitioner review: Psychosocial interventions for children with selective mutism: a critical evaluation of the literature from 1990-2005. J Child Psychol Psychiatry. 47: 1085-1097
- Bögels SM, Alden L, Beidel DC, Clark LA, Pine DS, et al. (2010) Social anxiety disorder: questions and answers for the DSM-V. Depress Anxiety [Review] 27: 168-189.
- Chavira DA, Stein MB (2005) Childhood social anxiety disorder: from understanding to treatment. Child Adolesc Psychiatr Clin N Am 14:797-718.
- Viana AG, Beidel DC, Rabian B (2009) Selective mutism: a review and integration of the last 15 years. Clin Psychol Rev 29: 57-67.
- Manassis K, Mendlowitz S, Menna R (1997) Child and parent reports of childhood anxiety: differences in coping styles. Depress Anxiety6:62-69.
- McInnes A, Manassis K (2005) When silence is not golden: an integrated approach to selective mutism. Semin Speech Lang. 26:201-210
- Chorpita BF, Daleiden EL, Ebesutani C (2011) Evidence-based treatments for children and adolescents: an updated review of indicators of efficacy and effectiveness. Clin Psychol 18:153-172.
- Albano AM, Kendall PC (2002) Cognitive behavioral therapy for children and adolescents with anxiety disorders: clinical research advances. Int Rev Psychiatry 14:129-134.
- Kendall PC, Hudson JL, Gosch E, Flannery-Schroeder E, Suveg C (2008) Cognitive-behavioral therapy for anxiety disordered youth: a randomized clinical trial evaluating child and family modalities. J Consult Clin Psychol 76: 282-297.
- Manassis K, Mendlowitz SL, Scapillato D, Avery D, Fiksenbaum L, Freire et al. (2002) Group and individual cognitive-behavioral therapy for childhood anxiety disorders: a randomized trial. J Am Acad Child Adolesc Psychiatry. 41:1423-1430.
- Fung DS, Manassis K, Kenny A, Fiksenbaum L (2002) Web-based CBT for selective mutism. J Am Acad Child Adolesc Psychiatry41:112-113.
- Reuther ET, Davis TE, Moree BN, Matson JL (2011) Treating selective mutism using modular CBT for child anxiety: a case study. J Clin Child Adolesc Psychol 40:156-163.
- Ooi YP, Raja M, Sung SC, Fung DS, Koh JB (2012) Application of a web-based cognitive-behavioral therapy program for the treatment of selective mutism in Singapore: a case series study. Singapore Med J 53:446-450.
- Bergman RL, Gonzalez A, Piacentini J, Keller ML (2013) Integrated Behavior Therapy for Selective Mutism: a randomized controlled pilot study. Behav Res Ther 51:680-689.
- Oerbeck B, Stein MB, Wentzel-Larsen T, Langsrud Ø, Kristensen H (2014) A randomized controlled trial of a home and school-based intervention for selective mutism-defocused communication and behavioral techniques. Child Adolesc Ment Health 19:192-198.
- Oerbeck B, Stein MB, Pripp AH, Kristensen H (2015) Selective mutism: follow-up study 1 year after end of treatment. Eur Child Adolesc Psychiatry24: 757-766.
- Lang C, Nir Z, Gothelf A, Domachevsky S, Ginton L (2016) The outcome of children with selective mutism following cognitive behavioral intervention: a follow-up study. Eur J Pediatr 175: 481-487.
- Bergman RL, Keller ML, Piacentini J, Bergman AJ (2008) J Clin Child Adolesc Psychol 37:456-464.
- Koh JBK, Chang WC, Fung DS, Kee CHY, Woo BSC (2002). Validation of a culturally appropriate anxiety scale for Singaporean children. Paper presented at the 49th Annual Meeting of the American Academy of Child and Adolescent Psychiatry: San Francisco, USA.
- National Institute of Mental Health (1985) Special feature: rating scales and assessment instruments for use in paediatric psychopharmacology research. Psychopharmacol Bull21:839-843.
- Kee CH, Fung DS, Ang LK (2001) An electronic communication device for selective mutism. J Am Acad Child Adolesc Psychiatry 40:389.
- Hammill DD, Pearson NA, Wiederholt JL (1997) Comprehensive test of nonverbal intelligence. Austin, TX: Pro-Edn.
- Dunn LM, Dunn LM, Whetton C, Pintillie D (1982) British picture vocabulary scale. Windsor: NFER-Nelson.
- Shaffer D, Fisher P, Lucas CP, Dulcan MK, Schwab-Stone ME (2000) NIMH Diagnostic Interview Schedule for Children Version IV (NIMH DISC-IV): description, differences from previous versions, and reliability of some common diagnoses. J Am Acad Child Adolesc Psychiatry 39:28-38.
- Khanna MS, Kendall PC (2009) Exploring the role of parent training in the treatment of childhood anxiety. J Consult Clin Psychol 77:981-986.
- Dummit ES, Klein RG, Tancer NK, Asche B, Martin J (1996) Fluoxetine treatment of children with selective mutism: an open trial. J Am Acad Child Adolesc Psychiatry 35: 615-621.
- Manassis K, Oerbeck B, Overgaard KR (2015) The use of medication in selective mutism: a systematic review. Eur Child Adolesc Psychiatry Epub ahead of print.
- Kumpulainen K (2002) Phenomenology and treatment of selective mutism. CNS Drugs 16:175-180.
- Crawford AM, Manassis K (2001) Familial predictors of treatment outcome in childhood anxiety disorders. J Am Acad Child Adolesc Psychiatry. 40:1182-1189.
Relevant Topics
- Stuttering therapy
- Active listening
- Aphasia
- Articulation disorders:
- Autism Speech Therapy
- Bilingual Speech pathology
- Clinical Linguistics
- Communicate Speech pathology
- Interventional Speech Therapy
- Late talkers
- Medical Speech pathology
- Spectrum Pathology
- Speech and Language Disorders
- Speech and Language pathology
- Speech Impediment / speech disorder
- Speech pathology
- Speech Therapy
- Speech Therapy Exercise
- Speech Therapy for Adults
- Speech Therapy for Children
- Speech Therapy Materials
Recommended Journals
Article Tools
Article Usage
- Total views: 17136
- [From(publication date):
June-2016 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 15983
- PDF downloads : 1153